Abelmoschus Esculentus (L.) Moench’s Peel Powder Improves High-Fat-Diet-Induced Cognitive Impairment in C57BL/6J Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of the Plant Material
2.2. Feed Formulations and Preparation
2.3. Animal Studies
2.4. Morris Water Maze (MWM) Test
2.5. Serum Lipid Profile Assessment
2.6. Statistical Analysis
3. Results
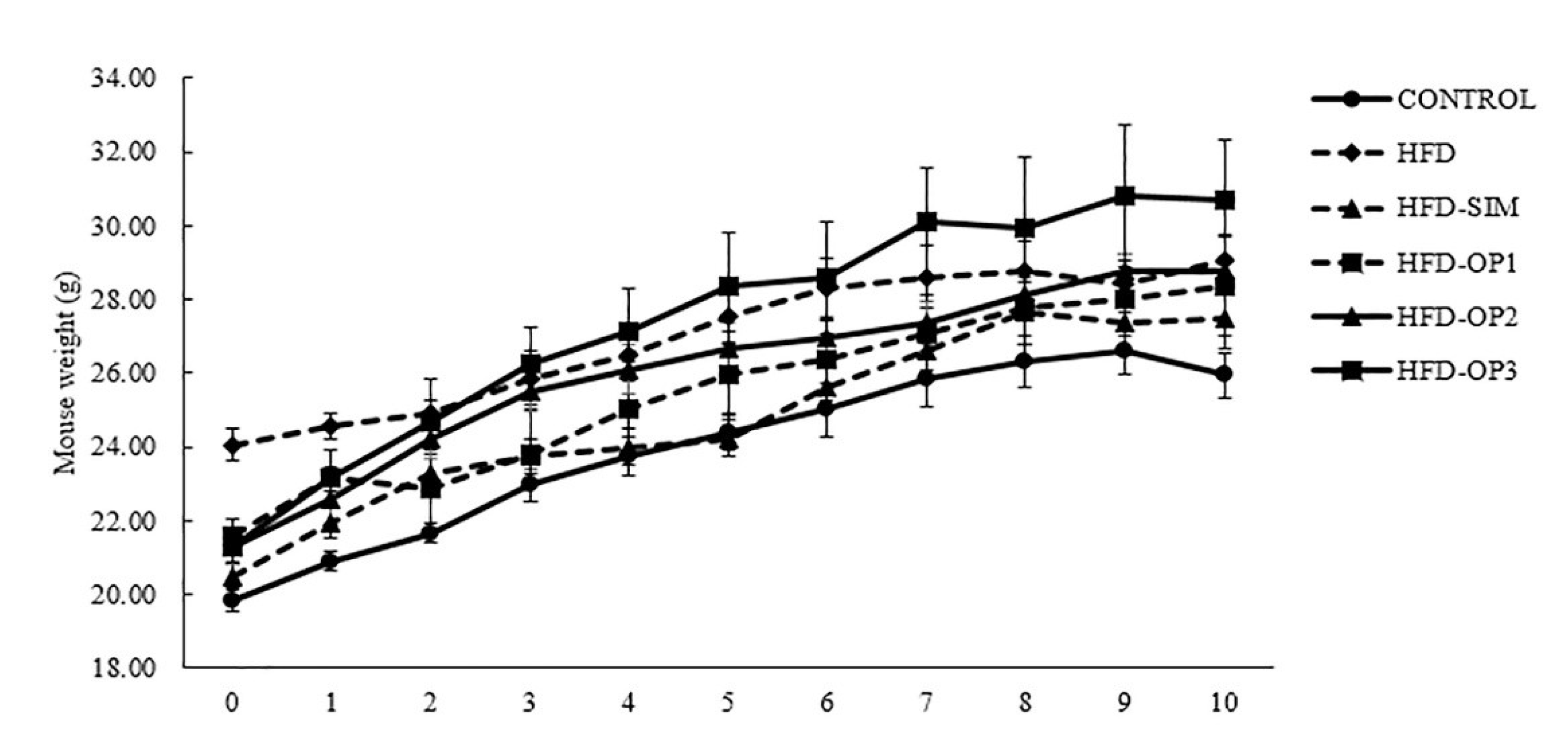
3.1. Body Weight Changes
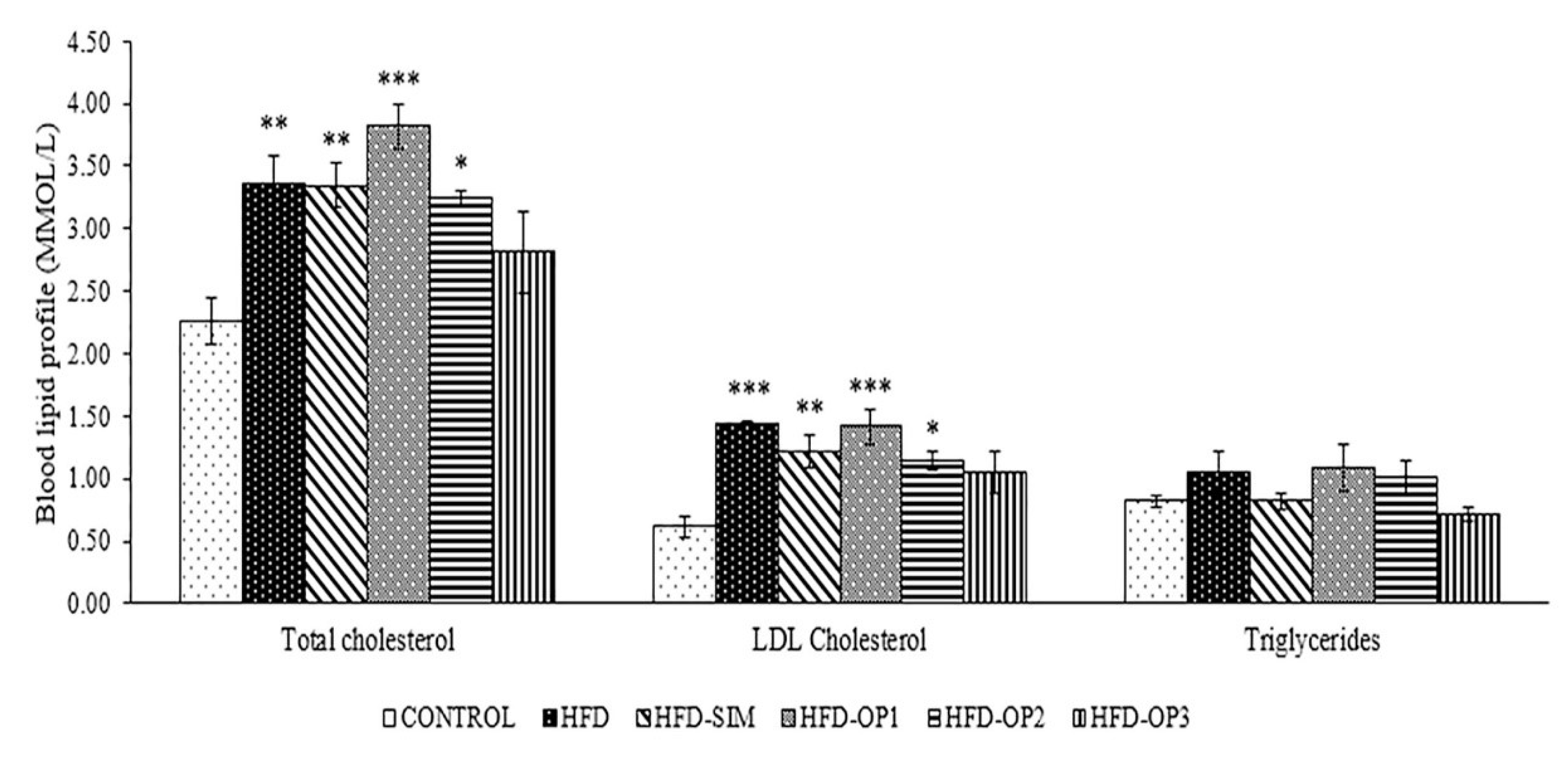
3.2. Effects on Blood Lipid Profiles
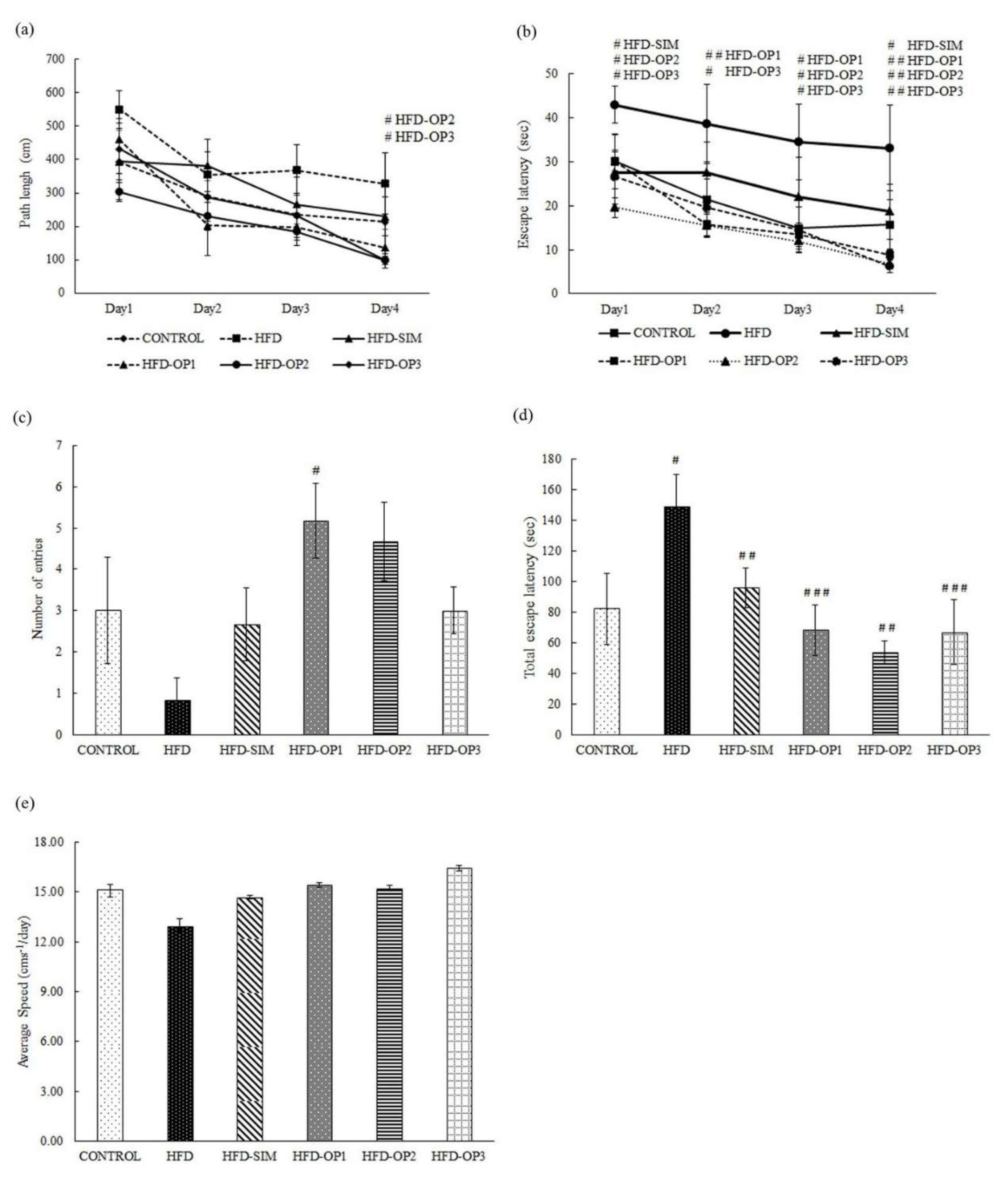
3.3. Morris Water Maze Test
4. Discussion
5. Limitations and Future Perspectives
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chooi, Y.C.; Ding, C.; Magkos, F. The epidemiology of obesity. Metabolism 2019, 92, 6–10. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Obesity and Overweight. 2016. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/ (accessed on 20 April 2020).
- Klop, B.; Elte, J.W.F.; Cabezas, M.C. Dyslipidemia in Obesity: Mechanisms and Potential Targets. Nutrients 2013, 5, 1218–1240. [Google Scholar] [CrossRef] [PubMed]
- Jung, U.J.; Choi, M.-S. Obesity and its metabolic complications: The role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia and nonalcoholic fatty liver disease. Int. J. Mol. Sci. 2014, 15, 6184–6223. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.M.; Danaei, G.; Farzadfar, F.; Stevens, G.A.; Woodward, M.; Wormser, D.; Kaptoge, S.; Whitlock, G.; Qiao, Q.; Lewington, S.; et al. The age-specific quantitative effects of metabolic risk factors on cardiovascular diseases and diabetes: A pooled analysis. PLoS ONE 2013, 8, e65174. [Google Scholar] [CrossRef] [PubMed]
- Czernichow, S.; Kengne, A.-P.; Stamatakis, E.; Hamer, M.; Batty, G.D. Body mass index, waist circumference and waist-hip ratio: Which is the better discriminator of cardiovascular disease mortality risk?: Evidence from an individual-participant meta-analysis of 82 864 participants from nine cohort studies. Obes. Rev. 2011, 12, 680–687. [Google Scholar] [CrossRef] [PubMed]
- Lauby-Secretan, B.; Scoccianti, C.; Loomis, D.; Grosse, Y.; Bianchini, F.; Straif, K. Body Fatness and Cancer—Viewpoint of the IARC Working Group. N. Engl. J. Med. 2016, 375, 794–798. [Google Scholar] [CrossRef]
- Anstey, K.J.; Cherbuin, N.; Budge, M.; Young, J. Body mass index in midlife and late-life as a risk factor for dementia: A meta-analysis of prospective studies. Obes. Rev. 2011, 12, e426–e437. [Google Scholar] [CrossRef]
- Saiyasit, N.; Chunchai, T.; Apaijai, N.; Pratchayasakul, W.; Sripetchwandee, J.; Chattipakorn, N.; Chattipakorn, S.C. Chronic high-fat diet consumption induces an alteration in plasma/brain neurotensin signaling, metabolic disturbance, systemic inflammation/oxidative stress, brain apoptosis, and dendritic spine loss. Neuropeptides 2020, 102047. [Google Scholar] [CrossRef]
- Robison, L.S.; Albert, N.M.; Camargo, L.A.; Anderson, B.M.; Salinero, A.E.; Riccio, D.A.; Abi-Ghanem, C.; Gannon, O.J.; Zuloaga, K.L. High-fat diet-induced obesity causes sex-specific deficits in adult hippocampal neurogenesis in mice. eNeuro 2020, 7. [Google Scholar] [CrossRef]
- Csonka, C.; Sárközy, M.; Pipicz, M.; Dux, L.; Csont, T. Modulation of hypercholesterolemia-induced oxidative/nitrative stress in the heart. Oxidative Med. Cell. Longev. 2015, 2016, 3863726. [Google Scholar] [CrossRef]
- E Petro, A.; Cotter, J.; A Cooper, D.; Peters, J.C.; Surwit, S.J.; Surwit, R.S. Fat, carbohydrate, and calories in the development of diabetes and obesity in the C57BL/6J mouse. Metabolism 2004, 53, 454–457. [Google Scholar] [CrossRef] [PubMed]
- Kang, E.; Koo, J.; Jang, Y.; Yang, C.; Lee, Y.; Cosio-Lima, L.M.; Cho, J. Neuroprotective effects of endurance exercise against high-fat diet-induced hippocampal neuroinflammation. J. Neuroendocr. 2016, 28. [Google Scholar] [CrossRef] [PubMed]
- Bhat, N.R.; Thirumangalakudi, L. Increased tau phosphorylation and impaired brain insulin/igf signaling in mice fed a high fat/high cholesterol diet. J. Alzheimer’s Dis. 2013, 36, 781–789. [Google Scholar] [CrossRef] [PubMed]
- You, S.; Jang, M.; Kim, G.-H. Mori Cortex Radicis Attenuates High Fat Diet-Induced Cognitive Impairment via an IRS/Akt Signaling Pathway. Nutrients 2020, 12, 1851. [Google Scholar] [CrossRef]
- Wu, M.; Liao, M.; Huang, R.; Chen, C.; Tian, T.; Wang, H.; Li, J.; Li, J.; Sun, Y.; Wu, C.; et al. Hippocampal overexpression of TREM2 ameliorates high fat diet induced cognitive impairment and modulates phenotypic polarization of the microglia. Genes Dis. 2020. [Google Scholar] [CrossRef]
- Lu, J.; Wu, D.M.; Zheng, Y.L.; Hu, B.; Cheng, W.; Zhang, Z.F.; Shan, Q. Ursolic acid improves high fat diet-induced cognitive impairments by blocking endoplasmic reticulum stress and IκB kinase β/nuclear factor-κB-mediated inflammatory pathways in mice. Brain Behav. Immun. 2011, 25, 1658–1667. [Google Scholar] [CrossRef]
- Batista, Â.G.; Mendonça, M.C.P.; Soares, E.S.; Da Silva-Maia, J.K.; Dionísio, A.P.; Sartori, C.R.; Da Cruz-Höfling, M.A.; Júnior, M.R.M. Syzygium malaccense fruit supplementation protects mice brain against high-fat diet impairment and improves cognitive functions. J. Funct. Foods 2020, 65, 103745. [Google Scholar] [CrossRef]
- Kim, T.-W.; Baek, K.-W.; Yu, H.S.; Ko, I.-G.; Hwang, L.; Park, J.-J. High-intensity exercise improves cognitive function and hippocampal brain-derived neurotrophic factor expression in obese mice maintained on high-fat diet. J. Exerc. Rehabil. 2020, 16, 124–131. [Google Scholar] [CrossRef]
- Wang, Z.; Ge, Q.; Wu, Y.; Zhang, J.; Gu, Q.; Han, J. Impairment of long-term memory by a short-term high-fat diet via hippocampal oxidative stress and alterations in synaptic plasticity. Neuroscience 2020, 424, 24–33. [Google Scholar] [CrossRef]
- Heyward, F.D.; Walton, R.G.; Carle, M.S.; Coleman, M.A.; Garvey, W.T.; Sweatt, J.D. Adult mice maintained on a high-fat diet exhibit object location memory deficits and reduced hippocampal SIRT1 gene expression. Neurobiol. Learn. Mem. 2012, 98, 25–32. [Google Scholar] [CrossRef]
- Kaczmarczyk, M.M.; Machaj, A.S.; Chiu, G.S.; Lawson, M.A.; Gainey, S.J.; York, J.M.; Meling, D.D.; Martin, S.A.; Kwakwa, K.A.; Newman, A.F.; et al. Methylphenidate prevents high-fat diet (HFD)-induced learning/memory impairment in juvenile mice. Psychoneuroendocrinol 2013, 38, 1553–1564. [Google Scholar] [CrossRef] [PubMed]
- Richter-Levin, G.; Errington, M.; Maegawa, H.; Bliss, T. Activation of metabotropic glutamate receptors is necessary for long-term potentiation in the dentate gyrus and for spatial learning. Neuropharmacology 1994, 33, 853–857. [Google Scholar] [CrossRef]
- Knowlton, B.J.; Fanselow, M.S. The hippocampus, consolidation and on-line memory. Curr. Opin. Neurobiol. 1998, 8, 293–296. [Google Scholar] [CrossRef]
- Kumar, J.; Teoh, S.L.; Das, S.; Mahakknaukrauh, P. Oxidative stress in oral diseases: Understanding its relation with other systemic diseases. Front. Physiol. 2017, 8, 693. [Google Scholar] [CrossRef] [PubMed]
- Kamil, K.; Yazid, M.D.; Idrus, R.B.H.; Das, S.; Kumar, J. Peripheral Demyelinating Diseases: From Biology to Translational Medicine. Front. Neurol. 2019, 10, 87. [Google Scholar] [CrossRef] [PubMed]
- Lasker, S.; Rahman, M.; Parvez, F.; Zamila, M.; Miah, P.; Nahar, K.; Kabir, F.; Sharmin, S.B.; Subhan, N.; Ahsan, G.U.; et al. High-fat diet-induced metabolic syndrome and oxidative stress in obese rats are ameliorated by yogurt supplementation. Sci. Rep. 2019, 9, 20026. [Google Scholar] [CrossRef]
- Alagumanivasagam, G.; Veeramani, p. A review on medicinal plants with hypolipidemic activity. Int. J. Pharm. Anal. Res. 2015, 4, 129–134. [Google Scholar]
- Kim, B.; Cho, B.O.; Jang, S.I. Muscat Bailey A grape stalk extract ameliorates high-fat diet-induced obesity by downregulating PPARγ and C/EPBα in mice. Int. J. Mol. Med. 2018, 43. [Google Scholar] [CrossRef]
- Kamil, K.; Kumar, J.; Yazid, M.D.; Idrus, R.H. Olive and its phenolic compound as the promising neuroprotective agent. Sains Malays. 2018, 47, 2811–2820. [Google Scholar] [CrossRef]
- Kamil, K.; Yazid, M.D.; Ruszymah, B.H.I.; Kumar, J. Hydroxytyrosol promotes proliferation of human schwann cells: An in vitro study. Int. J. Environ. Res. Public Heath 2020, 17, 4404. [Google Scholar] [CrossRef]
- Arapitsas, P. Identification and quantification of polyphenolic compounds from okra seeds and skins. Food Chem. 2008, 110, 1041–1045. [Google Scholar] [CrossRef] [PubMed]
- Dubey, P.; Mishra, S. A review on: Diabetes and okra (Abelmoschus esculentus). J. Med. Plants Stud. 2017, 5, 23–26. [Google Scholar]
- Dubey, P.; Mishra, S. Effect of okra seed in reduction of cholesterol. J. Entomol. Zool. Stud. 2017, 5, 94–97. [Google Scholar]
- Fan, S.; Zhang, Y.; Sun, Q.; Yu, L.; Li, M.; Zheng, B.; Wu, X.; Yang, B.; Li, Y.; Huang, C. Extract of okra lowers blood glucose and serum lipids in high-fat diet-induced obese C57BL/6 mice. J. Nutr. Biochem. 2014, 25, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Tongjaroenbuangam, W.; Ruksee, N.; Chantiratikul, P.; Pakdeenarong, N.; Kongbuntad, W.; Govitrapong, P. Neuroprotective effects of quercetin, rutin and okra (Abelmoschus esculentus Linn.) in dexamethasone-treated mice. Neurochem. Int. 2011, 59, 677–685. [Google Scholar] [CrossRef]
- Tripathi, K.; Govila, O.; Warrier, R.; Ahuja, V. Biology of Abelmoschus esculentus L.(okra); Govt of India, Department of Biotechnology: New Delhi, India, 2011. [Google Scholar]
- Jain, N.; Jain, R.; Jain, V.; Jain, S. A review on: Abelmoschus esculentus. Pharmacia 2012, 1, 84–89. [Google Scholar]
- Ngoc, T.H.; Ngoc, N.Q.; Van, A.T.T.; Phung, N.V. Hypolipidemic effect of extracts from Abelmoschus esculentus L. (Malvaceae) on tyloxapol-induced hyperlipidemia in mice. J. Pharm. Sci. 2008, 35, 42–46. [Google Scholar]
- Yan, T.; Nian, T.; Liao, Z.; Xiao, F.; Wu, B.; Bi, K.; He, B.; Jia, Y. Antidepressant effects of a polysaccharide from okra (Abelmoschus esculentus (L) Moench) by anti-inflammation and rebalancing the gut microbiota. Int. J. Boil. Macromol. 2020, 144, 427–440. [Google Scholar] [CrossRef]
- Xia, F.; Li, C.; Li, M.; Liao, Y.; Liu, X.; Si, J.; Chang, Q.; Pan, R. Antidepressant activity of an aqueous extract from okra seeds. RSC Adv. 2018, 8, 32814–32822. [Google Scholar] [CrossRef]
- Yan, T.; Nian, T.; Wu, B.; Xiao, F.; He, B.; Bi, K.; Jia, Y. Okra polysaccharides can reverse the metabolic disorder induced by high-fat diet and cognitive function injury in Aβ1–42 mice. Exp. Gerontol. 2020, 130, 110802. [Google Scholar] [CrossRef]
- Ramachandran, S.; Naveen, K.R.; Panneerselvam, K.; Sabitha, V. Investigation of in vivo antioxidant property of Abelmoschus esculentus (L) moench. fruit seed and peel powders in streptozotocin-induced diabetic rats. J. Ayurveda Integr. Med. 2012, 3, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; Lucarini, M.; Novellino, E.; Da Silva, C.F.; Daliu, P.; Souto, E.B. Abelmoschus esculentus (L.): Bioactive components’ beneficial properties—Focused on Antidiabetic Role—For Sustainable Health Applications. Molecules 2018, 24, 38. [Google Scholar] [CrossRef] [PubMed]
- Xia, F.; Zhong, Y.; Li, M.; Chang, Q.; Liao, Y.; Liu, X.; Pan, R.-L. Antioxidant and Anti-Fatigue Constituents of Okra. Nutrients 2015, 7, 8846–8858. [Google Scholar] [CrossRef] [PubMed]
- Gemede, H.F.; Ratta, N.; Haki, G.D.; Woldegiorgis, A.Z.; Gemede, H.F. Nutritional quality and health benefits of okra (Abelmoschus esculentus): A review. J. Food Process. Technol. 2015, 6, 2. [Google Scholar] [CrossRef]
- Khomsug, P.; Thongjaroe, W.; Pakdeenaro, N.; Suttajit, M.; Chantirati, P. Antioxidative activities and phenolic content of extracts from okra (Abelmoschus esculentus L.). Res. J. Boil. Sci. 2010, 5, 310–313. [Google Scholar] [CrossRef]
- Bromley-Brits, K.; Deng, Y.; Song, W. Morris water maze test for learning and memory deficits in Alzheimer’s disease model mice. J. Vis. Exp. 2011, 53, e2920. [Google Scholar] [CrossRef]
- Vorhees, C.V.; Williams, M. Morris water maze: Procedures for assessing spatial and related forms of learning and memory. Nat. Protoc. 2006, 1, 848–858. [Google Scholar] [CrossRef]
- Fan, S.; Guo, L.; Zhang, Y.; Sun, Q.; Yang, B.; Huang, C. Okra polysaccharide improves metabolic disorders in high-fat diet-induced obese C57BL/6 mice. Mol. Nutr. Food Res. 2013, 57, 2075–2078. [Google Scholar] [CrossRef]
- Sparrow, C.P.; Burton, C.A.; Hernandez, M.; Mundt, S.; Hassing, H.; Patel, S.; Rosa, R.; Hermanowski-Vosatka, A.; Wang, P.-R.; Zhang, N.; et al. Simvastatin has anti-inflammatory and antiatherosclerotic activities independent of plasma cholesterol lowering. Arter. Thromb. Vasc. Boil. 2001, 21, 115–121. [Google Scholar] [CrossRef]
- Ouweneel, A.; Van Der Sluis, R.J.; Nahon, J.E.; Van Eck, M.; Hoekstra, M. Simvastatin treatment aggravates the glucocorticoid insufficiency associated with hypocholesterolemia in mice. Atherosclerosis 2017, 261, 99–104. [Google Scholar] [CrossRef]
- Kita, T.; Brown, M.S.; Goldstein, J.L. Feedback regulation of 3-hydroxy-3-methylglutaryl coenzyme a reductase in livers of mice treated with mevinolin, a competitive inhibitor of the reductase. J. Clin. Investig. 1980, 66, 1094–1100. [Google Scholar] [CrossRef]
- Majd, N.E.; Azizian, H.; Tabandeh, M.R.; Shahriari, A. Effect of Abelmoschus esculentus powder on ovarian histology, expression of apoptotic genes and oxidative stress in diabetic rats fed with high fat diet. Iran. J. Pharm. Res. 2019, 18, 369–382. [Google Scholar]
- De Francesco, P.N.; Cornejo, M.P.; Barrile, F.; Romero, G.G.; Valdivia, S.; Andreoli, M.F.; Perello, M. Inter-individual variability for high fat diet consumption in inbred C57BL/6 mice. Front. Nutr. 2019, 6, 67. [Google Scholar] [CrossRef]
- Kim, D.-H.; Ryu, J.-H. Differential Effects of Scopolamine on Memory Processes in the Object Recognition Test and the Morris Water Maze Test in Mice. Biomol. Ther. 2008, 16, 173–178. [Google Scholar] [CrossRef]
- Park, H.R.; Park, M.; Choi, J.; Park, K.-Y.; Chung, H.Y.; Lee, J. A high-fat diet impairs neurogenesis: Involvement of lipid peroxidation and brain-derived neurotrophic factor. Neurosci. Lett. 2010, 482, 235–239. [Google Scholar] [CrossRef]
- Rojas-Fernandez, C.H.; Cameron, J.-C.F. Is Statin-Associated Cognitive Impairment Clinically Relevant? A Narrative Review and Clinical Recommendations. Ann. Pharmacother. 2012, 46, 549–557. [Google Scholar] [CrossRef]
- Zissimopoulos, J.M.; Barthold, D.; Brinton, R.D.; Joyce, G. Sex and Race Differences in the Association Between Statin Use and the Incidence of Alzheimer Disease. JAMA Neurol. 2017, 74, 225–232. [Google Scholar] [CrossRef]
| Part of Okra | Chemical Constituents | Bioactivities | References |
|---|---|---|---|
| Pods (fruits) | Polysaccharides, phenolic, mucilage, fibers, flavonoids, quercetin 3-O-gentiobioside, Uronic acid | Decrease glucose and lipid serum, antioxidant, antidiabetic, neuroprotective | [33,35,36,38,39,43,44,45,46] |
| Peel (skin of fruits) | Phenolic, quercetin, rutin, polysaccharide, quercetin-3-O-gentiobiose | Antioxidant, decrease glucose, anti-hyperlipidemic, antidiabetic | [32,33,38,43,44,45,46] |
| Leaves | Mucilage, phenolic | Anti-inflammatory, antioxidant | [37,38,39,42,45,46] |
| Flowers | Phenolic tannins, flavonol glycosides | Anti-inflammatory, antioxidant | [45,46] |
| Roots | Mucilage, flavonol glycosides | Antioxidant, anti-hyperlipidemic | [39,45,46] |
| Seeds | Linoleic acid, palmitic acid, oleic acid, catechin, epicatechin, quercetin, rutin, quercetin-3-O-gentiobiose | Antispasmodic, antioxidant, antidiabetic, anti-hyperlipidemic neuroprotective | [32,33,34,36,37,38,41,43,44,45,46,47] |
| On a Caloric Basis (in 100 g) | CONTROL Diet with w/10% Energy from Fat (C 1090–10) | HFD with w/60% Energy from Fat (C 1090–60) |
|---|---|---|
| Moisture | 7.9% | 2.9% |
| Crude Ash | 4.3% | 3.2% |
| Crude Fiber | 3.1% | 4.7% |
| Crude Fat | 4.0% | 35.0% |
| Crude Protein | 20.7% | 21.0% |
| Nitrogen free extractives | 60.0% | 33.2% |
| Total calories | 351 kcal | 523 kcal |
| Group | Food Intake (g/day) |
|---|---|
| Control | 3.67 ± 0.11 |
| HFD | 2.9 ± 0.13 ** |
| HFD-SIM | 2.99 ± 0.04 ** |
| HFD-OP1 | 3.22 ± 0.21 |
| HFD-OP2 | 3.03 ± 0.07 * |
| HFD-OP3 | 3.33 ± 0.11 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prom-in, S.; Kaewsrichan, J.; Wangpradit, N.; Kien Hui, C.; Yahaya, M.F.; Kamisah, Y.; Kumar, J. Abelmoschus Esculentus (L.) Moench’s Peel Powder Improves High-Fat-Diet-Induced Cognitive Impairment in C57BL/6J Mice. Int. J. Environ. Res. Public Health 2020, 17, 5513. https://doi.org/10.3390/ijerph17155513
Prom-in S, Kaewsrichan J, Wangpradit N, Kien Hui C, Yahaya MF, Kamisah Y, Kumar J. Abelmoschus Esculentus (L.) Moench’s Peel Powder Improves High-Fat-Diet-Induced Cognitive Impairment in C57BL/6J Mice. International Journal of Environmental Research and Public Health. 2020; 17(15):5513. https://doi.org/10.3390/ijerph17155513
Chicago/Turabian StyleProm-in, Supattra, Jasadee Kaewsrichan, Nuntika Wangpradit, Chua Kien Hui, Mohamad Fairuz Yahaya, Yusof Kamisah, and Jaya Kumar. 2020. "Abelmoschus Esculentus (L.) Moench’s Peel Powder Improves High-Fat-Diet-Induced Cognitive Impairment in C57BL/6J Mice" International Journal of Environmental Research and Public Health 17, no. 15: 5513. https://doi.org/10.3390/ijerph17155513
APA StyleProm-in, S., Kaewsrichan, J., Wangpradit, N., Kien Hui, C., Yahaya, M. F., Kamisah, Y., & Kumar, J. (2020). Abelmoschus Esculentus (L.) Moench’s Peel Powder Improves High-Fat-Diet-Induced Cognitive Impairment in C57BL/6J Mice. International Journal of Environmental Research and Public Health, 17(15), 5513. https://doi.org/10.3390/ijerph17155513






