A Focused Review on the Maximal Exercise Responses in Hypo- and Normobaric Hypoxia: Divergent Oxygen Uptake and Ventilation Responses
Abstract
1. Introduction
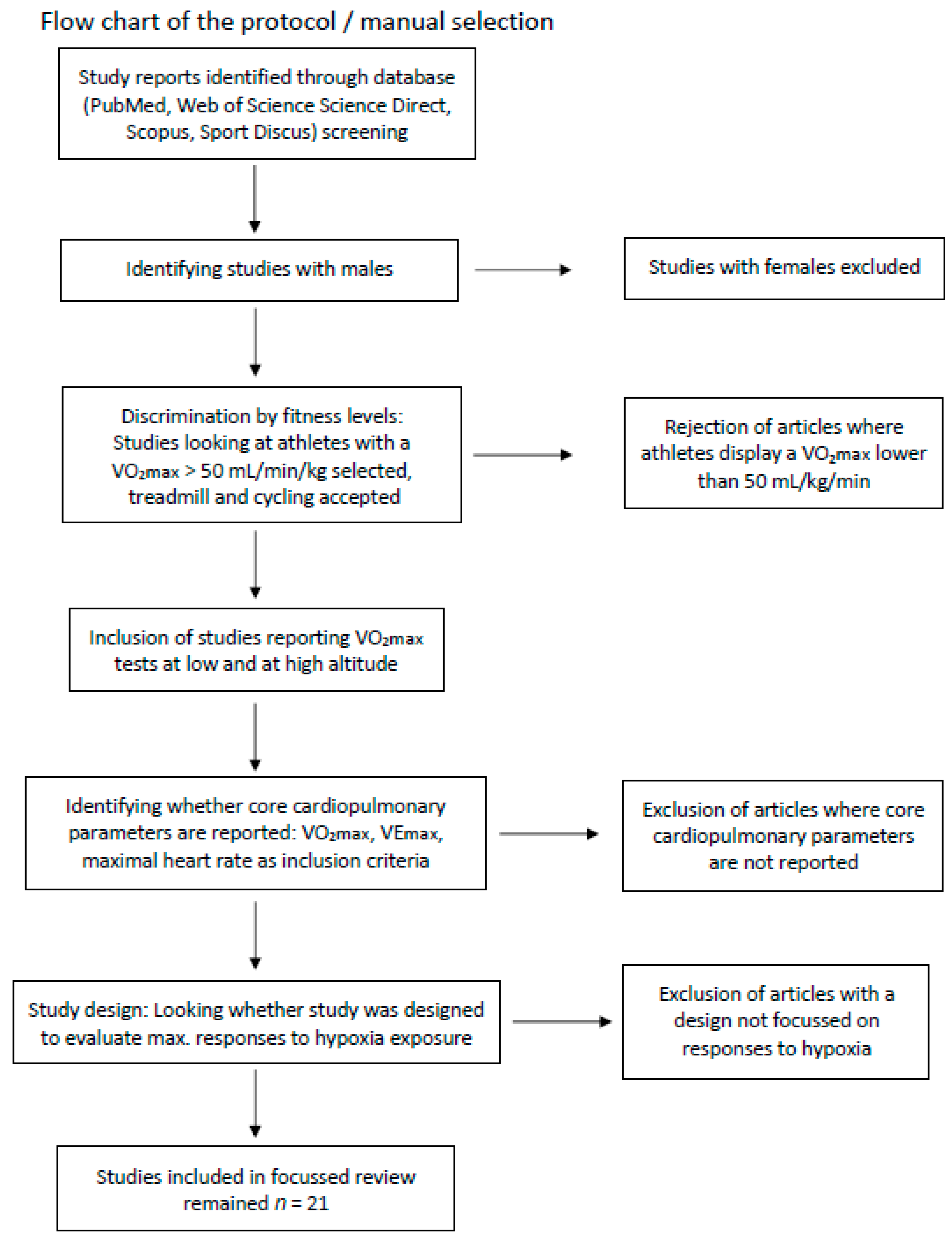
2. Materials and Methods
Statistics
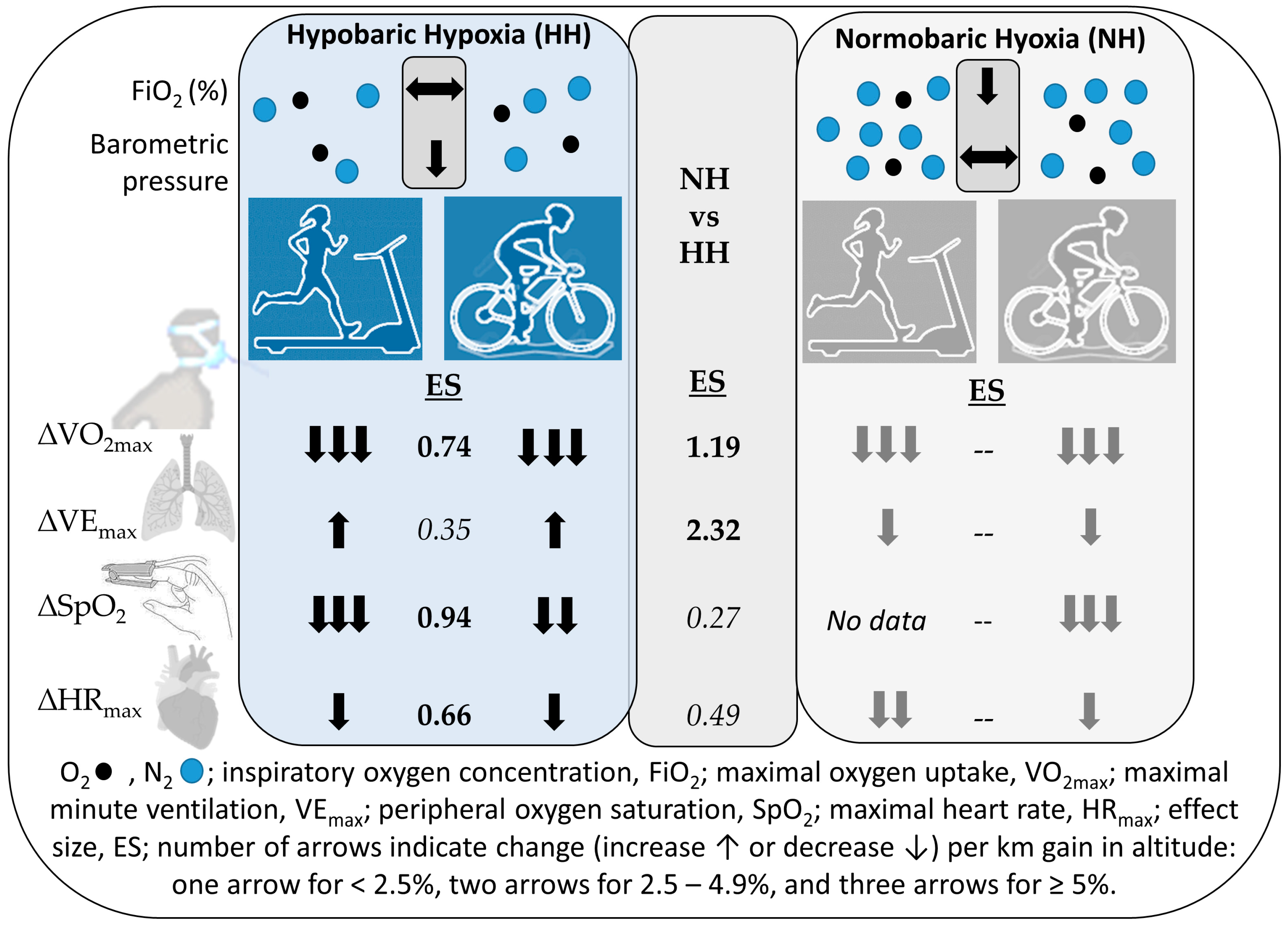
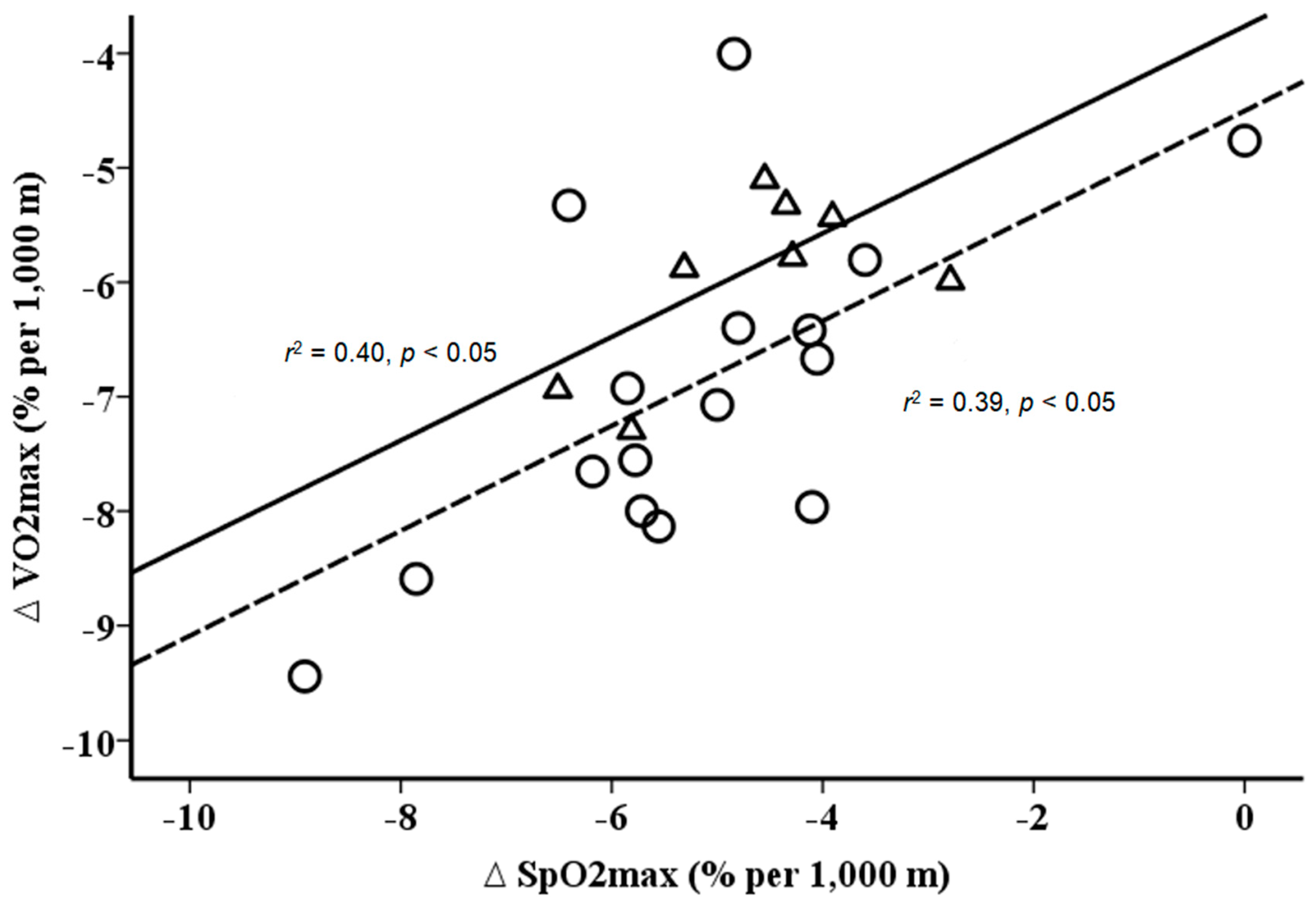
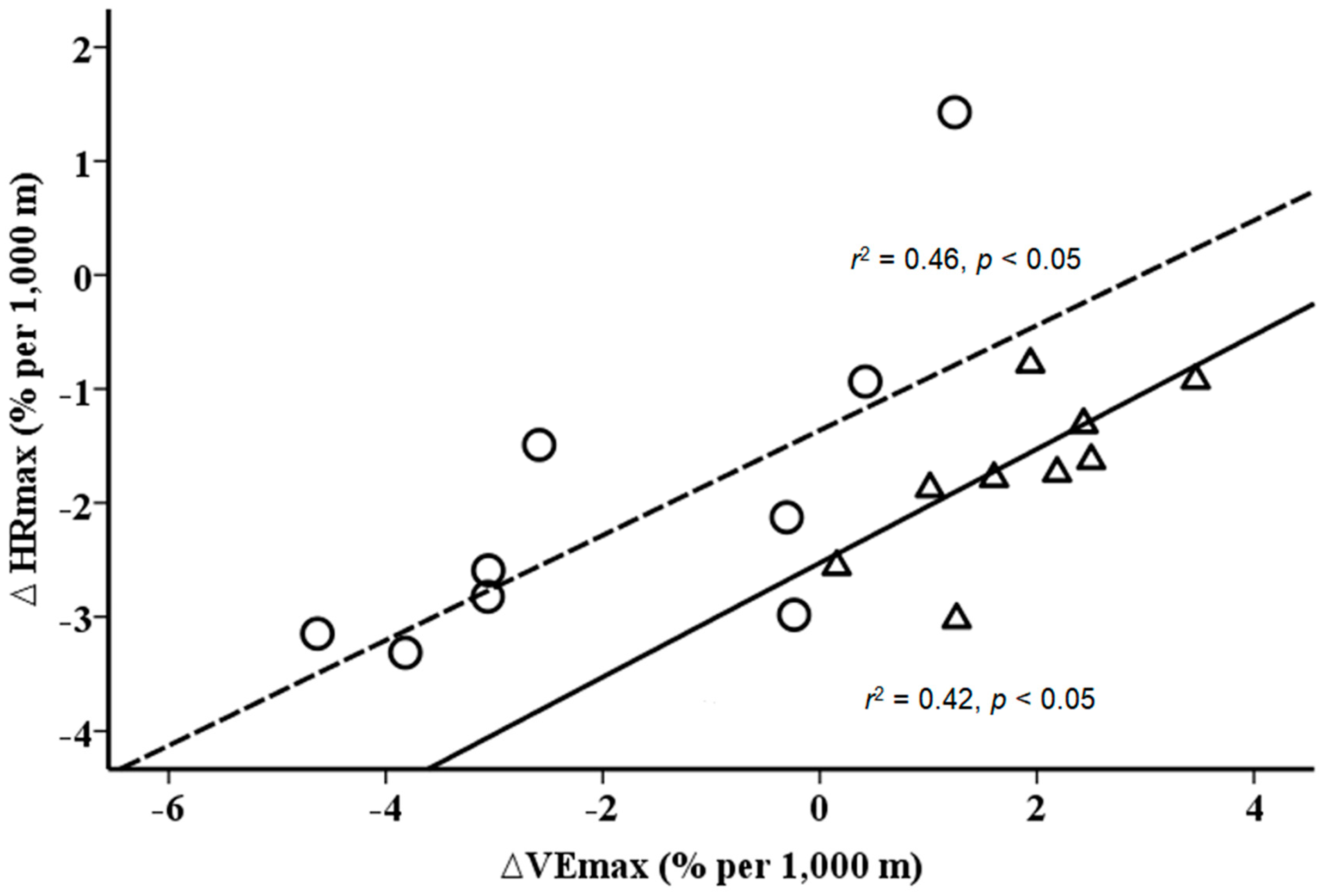
3. Results
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pugh, L.G. Athletes at altitude. J. Physiol. 1967, 192, 619–646. [Google Scholar] [CrossRef] [PubMed]
- Daniels, J.; Oldridge, N. The effects of alternate exposure to altitude and sea level on world-class middle-distance runners. Med. Sci. Sports 1970, 2, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Mellerowicz, H.; Meller, W.; Wowerier, J.; Zerdick, J.; Ketusinh, O.; Kral, B.; Heepe, W. Comparative studies on the effect of high altitude training on permanent performance at lower altitudes. Schweiz. Z. Sportmed. 1971, (Suppl. 5–17). [Google Scholar] [PubMed]
- Hammond, M.D.; Gale, G.E.; Kapitan, K.S.; Ries, A.; Wagner, P.D. Pulmonary gas exchange in humans during normobaric hypoxic exercise. J. Appl. Physiol. 1986, 61, 1749–1757. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, W.; Eckardt, K.U.; Hilgendorf, A.; Strauch, S.; Bauer, C. Effects of maximal and submaximal exercise under normoxic and hypoxic conditions on serum erythropoietin level. Int. J. Sports Med. 1991, 12, 457–461. [Google Scholar] [CrossRef] [PubMed]
- Basualto-Alarcón, C.; Rodas, G.; Galilea, P.A.; Riera, J.; Pagés, T.; Ricart, A.; Torrella, J.R.; Behn, C.; Viscor, G. Cardiorespiratory parameters during submaximal exercise under acute exposure to normobaric and hypobaric hypoxia. Apunts. Med. Esport 2012, 47, 65–72. [Google Scholar]
- Richard, N.A.; Sahota, I.S.; Widmer, N.; Ferguson, S.; Sheel, A.W.; Koehle, M.S. Acute mountain sickness, chemosensitivity, and cardiorespiratory responses in humans exposed to hypobaric and normobaric hypoxia. J. Appl. Physiol. 2014, 116, 945–952. [Google Scholar] [CrossRef]
- Woods, D.R.; O’Hara, J.P.; Boos, C.J.; Hodkinson, P.D.; Tsakirides, C.; Hill, N.E.; Jose, D.; Hawkins, A.; Phillipson, K.; Hazlerigg, A.; et al. Markers of physiological stress during exercise under conditions of normoxia, normobaric hypoxia, hypobaric hypoxia, and genuine high altitude. Eur. J. Appl. Physiol. 2017, 117, 893–900. [Google Scholar] [CrossRef]
- Horstman, D.; Weiskopf, R.; Jackson, R.E. Work capacity during 3-wk sojourn at 4300 m: Effects of relative polycythemia. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1980, 49, 311–318. [Google Scholar] [PubMed]
- Burtscher, M.; Niedermeier, M.; Burtscher, J.; Pesta, D.; Suchy, J.; Strasser, B. Preparation for Endurance Competitions at Altitude: Physiological, Psychological, Dietary and Coaching Aspects. A Narrative Review. Front. Physiol. 2018, 9, 1504. [Google Scholar] [CrossRef]
- Coppel, J.; Hennis, P.; Gilbert-Kawai, E.; Grocott, M.P. The physiological effects of hypobaric hypoxia versus normobaric hypoxia: A systematic review of crossover trials. Extrem. Physiol. Med. 2015, 4, 2. [Google Scholar] [CrossRef] [PubMed]
- Boos, C.J.; O’Hara, J.P.; Mellor, A.; Hodkinson, P.D.; Tsakirides, C.; Reeve, N.; Gallagher, L.; Green, N.D.; Woods, D.R. A Four-Way Comparison of Cardiac Function with Normobaric Normoxia, Normobaric Hypoxia, Hypobaric Hypoxia and Genuine High Altitude. PLoS ONE 2016, 11, e0152868. [Google Scholar] [CrossRef] [PubMed]
- Viscor, G.; Torrella, J.R.; Corral, L.; Ricart, A.; Javierre, C.; Pages, T.; Ventura, J.L. Physiological and Biological Responses to Short-Term Intermittent Hypobaric Hypoxia Exposure: From Sports and Mountain Medicine to New Biomedical Applications. Front. Physiol. 2018, 9, 339. [Google Scholar] [CrossRef] [PubMed]
- Debevec, T.; Millet, G.P. Discerning normobaric and hypobaric hypoxia: Significance of exposure duration. J. Appl. Physiol. 2014, 116, 1255. [Google Scholar] [CrossRef][Green Version]
- Beidleman, B.A.; Fulco, C.S.; Staab, J.E.; Andrew, S.P.; Muza, S.R. Cycling performance decrement is greater in hypobaric versus normobaric hypoxia. Extrem. Physiol. Med. 2014, 3, 8. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Beidleman, B.A.; Muza, S.R.; Rock, P.B.; Fulco, C.S.; Lyons, T.P.; Hoyt, R.W.; Cymerman, A. Exercise responses after altitude acclimatization are retained during reintroduction to altitude. Med. Sci. Sports Exerc. 1997, 29, 1588–1595. [Google Scholar] [CrossRef]
- Fagraeus, L.; Karlsson, J.; Linnarsson, D.; Saltin, B. Oxygen uptake during maximal work at lowered and raised ambient air pressures. Acta Physiol. Scand. 1973, 87, 411–421. [Google Scholar] [CrossRef]
- Fulco, C.S.; Rock, P.B.; Trad, L.; Forte, V.; Cymerman, A. Maximal cardiorespiratory responses to one- and two-legged cycling during acute and long-term exposure to 4300 meters altitude. Eur. J. Appl. Physiol. Occup. Physiol. 1988, 57, 761–766. [Google Scholar] [CrossRef]
- Koistinen, P.; Takala, T.; Martikkala, V.; Leppäluoto, J. Aerobic fitness influences the response of maximal oxygen uptake and lactate threshold in acute hypobaric hypoxia. Int. J. Sports Med. 1995, 16, 78–81. [Google Scholar] [CrossRef]
- Squires, R.W.; Buskirk, E.R. Aerobic capacity during acute exposure to simulated altitude, 914 to 2286 meters. Med. Sci. Sports Exerc. 1982, 14, 36–40. [Google Scholar] [CrossRef]
- Wehrlin, J.P.; Hallen, J. Linear decrease in VO2max and performance with increasing altitude in endurance athletes. Eur. J. Appl. Physiol. 2006, 96, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Robergs, R.A.; Quintana, R.; Parker, D.L.; Frankel, C.C. Multiple variables explain the variability in the decrement in VO2max during acute hypobaric hypoxia. Med. Sci. Sports Exerc. 1998, 30, 869–879. [Google Scholar] [PubMed]
- Benoit, H.; Busso, T.; Castells, J.; Geyssant, A.; Denis, C. Decrease in peak heart rate with acute hypoxia in relation to sea level VO(2max). Eur. J. Appl. Physiol. 2003, 90, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, G.; Moia, C.; Thomet, J.M.; Kayser, B. The decrease of maximal oxygen consumption during hypoxia in man: A mirror image of the oxygen equilibrium curve. J. Physiol. 1997, 498, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Friedmann, B.; Frese, F.; Menold, E.; Bärtsch, P. Effects of acute moderate hypoxia on anaerobic capacity in endurance-trained runners. Eur. J. Appl. Physiol. 2007, 101, 67–73. [Google Scholar] [CrossRef]
- Gavin, T.P.; Derchak, P.A.; Stager, J.M. Ventilation’s role in the decline in VO2max and SaO2 in acute hypoxic exercise. Med. Sci. Sports Exerc. 1998, 30, 195–199. [Google Scholar] [CrossRef]
- Heubert, R.A.; Quaresima, V.; Laffite, L.P.; Koralsztein, J.P.; Billat, V.L. Acute moderate hypoxia affects the oxygen desaturation and the performance but not the oxygen uptake response. Int. J. Sports Med. 2005, 26, 542–551. [Google Scholar] [CrossRef]
- Lawler, J.; Powers, S.K.; Thompson, D. Linear relationship between VO2max and VO2max decrement during exposure to acute hypoxia. J. Appl. Physiol. 1988, 64, 1486–1492. [Google Scholar] [CrossRef]
- Martin, D.; O’Kroy, J. Effects of acute hypoxia on the VO2 max of trained and untrained subjects. J. Sports Sci. 1993, 11, 37–42. [Google Scholar] [CrossRef]
- Mollard, P.; Woorons, X.; Letournel, M.; Lamberto, C.; Favret, F.; Pichon, A.; Beaudry, M.; Richalet, J.P. Determinants of maximal oxygen uptake in moderate acute hypoxia in endurance athletes. Eur. J. Appl. Physiol. 2007, 100, 663–673. [Google Scholar] [CrossRef]
- Ofner, M.; Wonisch, M.; Frei, M.; Tschakert, G.; Domej, W.; Kröpfl, J.M.; Hofmann, P. Influence of acute normobaric hypoxia on physiological variables and lactate turn point determination in trained men. J. Sports Sci. Med. 2014, 13, 774–781. [Google Scholar] [PubMed]
- Peltonen, J.E.; Tikkanen, H.O.; Ritola, J.J.; Ahotupa, M.; Rusko, H.K. Oxygen uptake response during maximal cycling in hyperoxia, normoxia and hypoxia. Aviat. Space Environ. Med. 2001, 72, 904–911. [Google Scholar] [PubMed]
- Robach, P.; Calbet, J.A.; Thomsen, J.J.; Boushel, R.; Mollard, P.; Rasmussen, P.; Lundby, C. The ergogenic effect of recombinant human erythropoietin on VO2max depends on the severity of arterial hypoxemia. PLoS ONE 2008, 3, e2996. [Google Scholar] [CrossRef] [PubMed]
- Pugh, L.G.C.E. Man at High Altitude. J. R. Coll. Physic. Lond. 1969, 3, 385–397. [Google Scholar]
- Lenfant, C.; Sullivan, K. Adaptation to high altitude. N. Engl. J. Med. 1971, 284, 1298–1309. [Google Scholar] [CrossRef]
- Cerretelli, P. Energy sources for muscular exercise. Int. J. Sports Med. 1992, 13 (Suppl. 1), S106–S110. [Google Scholar] [CrossRef]
- Friedmann, B. Training in hypoxia: Detrimental for muscular aerobic capacity? Acta Physiol. 2007, 190, 177. [Google Scholar] [CrossRef]
- Mollard, P.; Woorons, X.; Letournel, M.; Lamberto, C.; Favret, F.; Pichon, A.; Beaudry, M.; Richalet, J.P. Determinant factors of the decrease in aerobic performance in moderate acute hypoxia in women endurance athletes. Respir. Physiol. Neurobiol. 2007, 159, 178–186. [Google Scholar] [CrossRef]
- Millet, G.P.; Debevec, T. CrossTalk proposal: Barometric pressure, independent of PO2, is the forgotten parameter in altitude physiology and mountain medicine. J. Physiol. 2020, 598, 893–896. [Google Scholar] [CrossRef]
- Saugy, J.J.; Rupp, T.; Faiss, R.; Lamon, A.; Bourdillon, N.; Millet, G.P. Cycling Time Trial Is More Altered in Hypobaric than Normobaric Hypoxia. Med. Sci. Sports Exerc. 2016, 48, 680–688. [Google Scholar] [CrossRef]
- Ogawa, T.; Fujii, N.; Kurimoto, Y.; Nishiyasu, T. Effect of hypobaria on maximal ventilation, oxygen uptake, and exercise performance during running under hypobaric normoxic conditions. Physiol. Rep. 2019, 7, e14002. [Google Scholar] [CrossRef]
- Richard, N.A.; Koehle, M.S. Differences in cardio-ventilatory responses to hypobaric and normobaric hypoxia: A review. Aviat. Space Environ. Med. 2012, 83, 677–684. [Google Scholar] [CrossRef] [PubMed]
- Savourey, G.; Launay, J.C.; Besnard, Y.; Guinet, A.; Travers, S. Normo-and hypobaric hypoxia: Are there any physiological differences? Eur. J. Appl. Physiol. 2003, 89, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Faiss, R.; Pialoux, V.; Sartori, C.; Faes, C.; Deriaz, O.; Millet, G.P. Ventilation, Oxidative Stress, and Nitric Oxide in Hypobaric versus Normobaric Hypoxia. Med. Sci. Sports Exerc. 2013, 45, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Amann, M.; Dempsey, J.A. Ensemble Input of Group III/IV Muscle Afferents to CNS: A Limiting Factor of Central Motor Drive During Endurance Exercise from Normoxia to Moderate Hypoxia. Adv. Exp. Med. Biol. 2016, 903, 325–342. [Google Scholar] [PubMed]
- Mourot, L. Limitation of maximal heart rate in hypoxia: Mechanisms and clinical importance. Front. Physiol. 2018, 9, 972. [Google Scholar] [CrossRef] [PubMed]
- Mourot, L.; Millet, G.P. Is Maximal Heart Rate Decrease Similar Between Normobaric Versus Hypobaric Hypoxia in Trained and Untrained Subjects? High. Alt. Med. Biol. 2019, 20, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Siebenmann, C.; Rasmussen, P.; Sorensen, H.; Bonne, T.C.; Zaar, M.; Aachmann-Andersen, N.J.; Nordsborg, N.B.; Secher, N.H.; Lundby, C. Hypoxia increases exercise heart rate despite combined inhibition of beta-adrenergic and muscarinic receptors. Am. J. Physiol. Heart Circ. Physiol. 2015, 308, H1540–H1546. [Google Scholar] [CrossRef][Green Version]
- Gavin, T.P.; Stager, J.M. The effect of exercise modality on exercise-induced hypoxemia. Respir. Physiol. 1999, 115, 317–323. [Google Scholar] [CrossRef]
- Kalsås, K.; Thorsen, E. Breathing patterns during progressive incremental cycle and treadmill exercise are different. Clin. Physiol. Funct. Imaging 2009, 29, 335–338. [Google Scholar] [CrossRef]
- Millet, G.P.; Vleck, V.E.; Bentley, D.J. Physiological differences between cycling and running: Lessons from triathletes. Sports Med. 2009, 39, 179–206. [Google Scholar] [CrossRef] [PubMed]
- Netzer, N.C.; Rausch, L.; Eliasson, A.H.; Gatterer, H.; Friess, M.; Burtscher, M.; Pramsohler, S. SpO2 and Heart Rate During a Real Hike at Altitude Are Significantly Different than at Its Simulation in Normobaric Hypoxia. Front. Physiol. 2017, 8, 81. [Google Scholar] [CrossRef] [PubMed]
- Richalet, J.P. CrossTalk opposing view: Barometric pressure, independent of PO2, is not the forgotten parameter in altitude physiology and mountain medicine. J. Physiol. 2020, 598, 897–899. [Google Scholar] [CrossRef] [PubMed]
| Reference | Participants | VO2max at Sea Level | Altitude or Equivalent | Hypoxia Condition | Baro-Metric Pressure | FiO2 | Exercise Mode | VO2max at Altitude |
|---|---|---|---|---|---|---|---|---|
| (number) | (mL/min/kg) | (m) | (mmHg) | (%) | (mL/min/kg) | |||
| Beidleman et al. (1997) [17] | 6 | 57.0 | 4300 | HH (HC) | 446 | treadmill | 40.0 | |
| Benoit et al. (2003) [24] | 12 | 64.2 | 5400 | NH | 10.4 | cycling | 36.0 | |
| Benoit et al. (2003) [24] | 17 | 50.8 | 5400 | NH | 10.4 | cycling | 31.4 | |
| Fagraeus et al. (1973) [18] | 11 | 50.1 | 3250 | HH (HC) | 520 | cycling | 43.5 | |
| Ferretti et al. (1997) [25] | 5 | 62.1 | 2182 | NH | 16.0 | cycling | 53.4 | |
| Ferretti et al. (1997) [25] | 5 | 62.1 | 4966 | NH | 11.0 | cycling | 35.6 | |
| Friedmann et al. (2007) [26] | 20 | 68.0 | 2682 | NH | 15.0 | treadmill | 53.1 | |
| Fulco et al. (1988) [19] | 7 | 50.1 | 4300 | HH (HC) | 446 | cycling | 37.2 | |
| Gavin et al. (1998) [27] | 7 | 60.4 | 3591 | NH | 13.3 | cycling | 43.8 | |
| Gavin et al. (1998) [27] | 6 | 63.7 | 3591 | NH | 13.3 | cycling | 42.1 | |
| Heubert et al. (2005) [28] | 9 | 62.7 | 2500 | NH | 16.0 | cycling | 53.6 | |
| Horstman et al. (1980) [9] | 9 | 51.0 | 4300 | HH (TA) | 460 | treadmill | 35.0 | |
| Koistinen et al. (1995) [20] | 12 | 57.4 | 3200 | HH (HC) | 520 | cycling | 46.6 | |
| Lawler et al. (1988) [29] | 7 | 65.0 | 3207 | NH | 14.0 | cycling | 51.1 | |
| Martin et al. (1993) [30] | 8 | 67.2 | 3760 | NH | 13.0 | cycling | 49.7 | |
| Mollard et al. (2007) [31] | 8 | 65.5 | 2479 | NH | 15.4 | cycling | 57.7 | |
| Mollard et al. (2007) [31] | 8 | 65.5 | 4527 | NH | 11.7 | cycling | 46.4 | |
| Ofner et al. (2014) [32] | 10 | 51.0 | 3500 | NH | 14.0 | cycling | 42.5 | |
| Peltonen et al. (2001) [33] | 6 | 61.8 | 2682 | NH | 15.0 | cycling | 48.6 | |
| Robach et al. (2008) [34] | 7 | 50.0 | 2500 | NH | 15.3 | cycling | 42.0 | |
| Robach et al. (2008) [34] | 7 | 50.0 | 3500 | NH | 13.4 | cycling | 36.0 | |
| Robach et al. (2008) [34] | 7 | 50.0 | 4500 | NH | 11.5 | cycling | 33.0 | |
| Robergs et al. (1998) [23] | 14 | 60.4 | 2559 | HH (HC) | 566 | cycling | 52.0 | |
| Squires et al. (1982) [21] | 12 | 60.0 | 2286 | HH (HC) | 574 | treadmill | 53.0 | |
| Wehrlin et al. (2006) [22] | 8 | 66.1 | 2300 | HH (HC) | 573 | treadmill | 58.0 | |
| Wehrlin et al. (2006) [22] | 8 | 66.1 | 2800 | HH (HC) | 573 | treadmill | 55.4 |
| All | Divided by Exercise Mode | ||||||
|---|---|---|---|---|---|---|---|
| Hypobaric Hypoxia Studies | |||||||
| Variable | n | n | Cycling Exercise | n | Treadmill Running | ||
| Age (yr) | 90 | 25.7 ± 3.7 | 47 | 26.1 ± 4.3 | 43 | 25.3 ± 3.0 | |
| Weight (kg) | 90 | 73.6 ± 2.5 | 47 | 73.5 ± 1.2 | 43 | 73.7 ± 3.4 | |
| Height (cm) | 90 | 178.8 ± 2.0 | 47 | 178.1 ± 2.1 | 43 | 179.5 ± 1.5 | |
| Altitude level (m) | 90 | 3117 ± 755 | 47 | 3144 ± 577 | 43 | 3087 ± 918 | |
| Normobaric Hypoxia Studies | |||||||
| Age (yr) | 147 | 25.2 ± 3.4 | 129 | 26.0 ± 2.9 | 18 | 24.1 ± 4.1 * | |
| Weight (kg) | 147 | 72.6 ± 5.2 | 129 | 72.9 ± 5.5 | 18 | 71.2 ± 6.5 | |
| Height (cm) | 147 | 178.7 ± 2.7 | 129 | 178.2 ± 2.5 | 18 | 182.1 ± 5.9 | |
| Altitude level (m) | 147 | 3678 ± 1095 | 129 | 3830 ± 1099 | 18 | 2682 | |
| HH | NH | HH | NH | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | n | Cycling + Treadmill | n | Cycling + Treadmill | n | Cycling Exercise | n | Treadmill Running | n | Cycling Exercise | n | Treadmill Running |
| ∆VO2max (%/km) | 9 90 | −5.6 ± 0.9 | 18 151 | −7.0 ± 1.4 a | 4 47 | −5.3 ± 0.7 | 5 43 | −5.9 ± 0.9 b | 17 131 | −6.8 ± 1.4 | 1 20 | −8.1 ± 0.0 |
| ∆VEmax (%/km) | 9 90 | 1.9 ± 0.9 | 15 132 | −1.4 ± 1.8 a | 4 47 | 1.8 ± 1.1 | 5 43 | 2.1 ± 0.5 c | 14 112 | −1.6 ± 1.9 | 1 20 | −0.2 ± 0.0 |
| ∆SpO2 (%/km) | 8 79 | −4.6 ± 1.0 | 17 131 | −5.0 ± 1.8 c | 3 36 | −4.2 ± 0.9 | 5 43 | −5.0 ± 0.8 a | 17 131 | −5.0 ± 1.9 | / | / |
| ∆HRmax (%/km) | 9 90 | −1.6 ± 0.6 | 13 117 | −2.1 ± 1.3 c | 4 47 | −1.4 ± 0.7 | 5 43 | −1.8 ± 0.5 b | 12 97 | −2.0 ± 1.3 | 1 20 | −3.0 ± 0.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Treml, B.; Gatterer, H.; Burtscher, J.; Kleinsasser, A.; Burtscher, M. A Focused Review on the Maximal Exercise Responses in Hypo- and Normobaric Hypoxia: Divergent Oxygen Uptake and Ventilation Responses. Int. J. Environ. Res. Public Health 2020, 17, 5239. https://doi.org/10.3390/ijerph17145239
Treml B, Gatterer H, Burtscher J, Kleinsasser A, Burtscher M. A Focused Review on the Maximal Exercise Responses in Hypo- and Normobaric Hypoxia: Divergent Oxygen Uptake and Ventilation Responses. International Journal of Environmental Research and Public Health. 2020; 17(14):5239. https://doi.org/10.3390/ijerph17145239
Chicago/Turabian StyleTreml, Benedikt, Hannes Gatterer, Johannes Burtscher, Axel Kleinsasser, and Martin Burtscher. 2020. "A Focused Review on the Maximal Exercise Responses in Hypo- and Normobaric Hypoxia: Divergent Oxygen Uptake and Ventilation Responses" International Journal of Environmental Research and Public Health 17, no. 14: 5239. https://doi.org/10.3390/ijerph17145239
APA StyleTreml, B., Gatterer, H., Burtscher, J., Kleinsasser, A., & Burtscher, M. (2020). A Focused Review on the Maximal Exercise Responses in Hypo- and Normobaric Hypoxia: Divergent Oxygen Uptake and Ventilation Responses. International Journal of Environmental Research and Public Health, 17(14), 5239. https://doi.org/10.3390/ijerph17145239






