Clinical Presentation of COVID-19: Case Series and Review of the Literature
Abstract
1. Introduction
2. Methods
3. Statistical Analysis
4. Case Series
5. Review of Literature
5.1. Clinical Presentation of COVID-19
5.1.1. Typical Clinical Manifestations
5.1.2. Atypical Clinical Manifestations
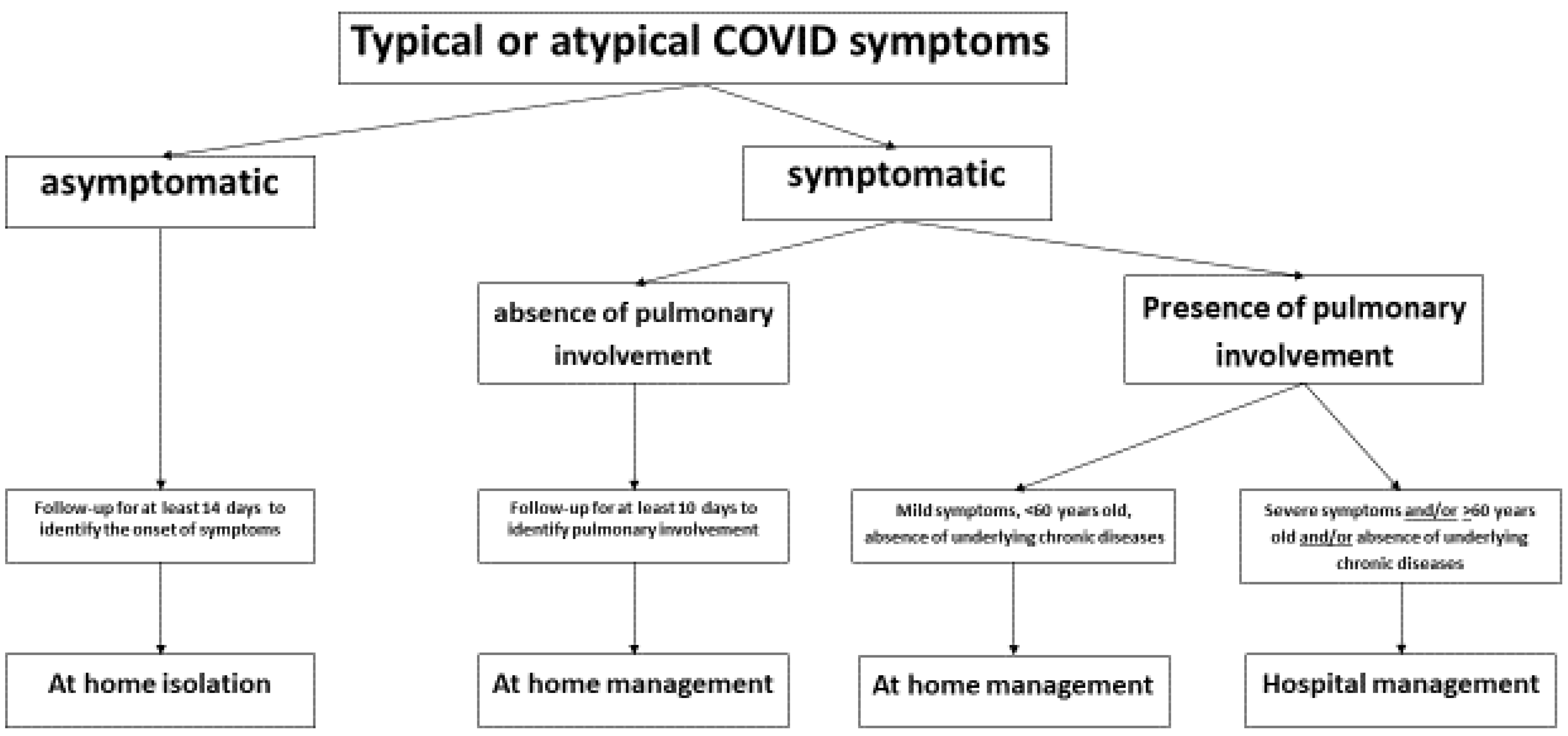
5.2. Correlation between Clinical Presentation and Clinical Evolution
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
References
- Russo, A.; Minichini, C.; Starace, M.; Astorri, R.; Calò, F.; Coppola, N.; Vanvitelli COVID-19 Group. Current status of laboratory diagnosis for COVID-19: A narrative review. Infect. Drug Resist. 2020. In press. [Google Scholar]
- De Groot, R.J.; Baker, S.C.; Baric, R.S.; Brown, C.S.; Drosten, C.; Enjuanes, L.; Fouchier, R.A.M.; Galiano, M.; Gorbalenya, A.E.; Memish, Z.A.; et al. Middle East respiratory syndrome coronavirus (MERS-CoV): Announcement of the Coronavirus Study Group. J. Virol. 2013, 87, 7790. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Middle East Respiratory Syndrome Coronavirus (MERS-CoV). 2019. Available online: https://www.who.int/emergencies/mers-cov/en/ (accessed on 29 June 2020).
- World Health Organization (WHO). Coronavirus Disease (COVID-19) Outbreak China. 2020. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019?gclid=EAIaIQobChMI_Pryo4Sn6gIVnIBQBh3lLwlpEAAYASAAEgJ_2vD_BwEat (accessed on 29 June 2020).
- Kobayashi, T.; Jung, S.M.; Linton, N.M.; Kinoshita, R.; Hayashi, K.; Miyama, T.; Anza, A.; Yang, Y.; Yuan, B.; Akhmetzhanov, A.R.; et al. Communicating the Risk of Death from Novel Coronavirus Disease (COVID-19). J. Clin. Med. 2020, 9, 580. [Google Scholar] [CrossRef] [PubMed]
- Cucinotta, D.; Vanelli, M. WHO Declares COVID-19 a Pandemic. Acta. Biomed. 2020, 91, 157–160. [Google Scholar] [CrossRef]
- Saglietto, A.; D’Ascenzo, F.; Zoccai, G.B.; De Ferrari, G.M. COVID-19 in Europe: The Italian lesson. Lancet 2020, 395, 1110–1111. [Google Scholar] [CrossRef]
- Sanders, J.M.; Monogue, M.L.; Jodlowski, T.Z.; Cutrell, J.B. Pharmacologic Treatments for Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Chen, Y.; Qin, Q.J. Unique epidemiological and clinical features of the emerging 2019 novel coronavirus pneumonia (COVID-19) implicate special control measures. Med. Virol. 2020, 92, 568–576. [Google Scholar] [CrossRef]
- Singhal, T. A Review of Coronavirus Disease-2019 (COVID-19). Indian J. Pediatr. 2020, 87, 281–286. [Google Scholar] [CrossRef]
- Sica, A.; Casale, D.; Rossi, G.; Casale, B.; Ciccozzi, M.; Fasano, M.; Ciotti, M.; Sagnelli, E.; Papa, A.; Sagnelli, C. The impact of the SARS-CoV-2 infection, with special reference to the haematological setting. J. Med. Virol. 2020. [Google Scholar] [CrossRef]
- Linton, N.M.; Kobayashi, T.; Yang, Y.; Hayashi, K.; Akhmetzhanov, A.R.; Jung, S.M.; Yuan, B.; Kinoshita, R.; Nishiura, H. Incubation Period and Other Epidemiological Characteristics of 2019 Novel Coronavirus Infections with Right Truncation: A Statistical Analysis of Publicly Available Case Data. J. Clin. Med. 2020, 9, 538. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Rayner, S.; Luo, M.H. Does SARS-CoV-2 has a longer incubation period than SARS and MERS? J. Med. Virol. 2020, 92, 476–478. [Google Scholar] [CrossRef] [PubMed]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.S.C.; et al. China Medical Treatment Expert Group for Covid-19. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China. JAMA 2020. [Google Scholar] [CrossRef]
- Li, R.; Pei, S.; Chen, B.; Song, Y.; Zhang, T.; Yang, W.; Shaman, J. Substantial undocumented infection facilitates the rapid dissemination of novel coronavirus (SARS-CoV2). Science 2020, 368, 489–493. [Google Scholar] [CrossRef]
- Bai, Y.; Yao, L.; Wei, T.; Tian, F.; Jih, D.Y.; Chen, L.; Wang, M. Presumed asymptomatic carrier transmission of COVID-19. J. Am. Med. Assoc. 2020, 323, 1406–1407. [Google Scholar] [CrossRef]
- Yu, P.; Zhu, J.; Zhang, Z.; Han, Y.; Huang, L. A Familial Cluster of Infection Associated With the 2019 Novel Coronavirus Indicating Possible Person-to-Person Transmission During the Incubation Period. J. Infect. Dis. 2020, 221, 1757–1761. [Google Scholar] [CrossRef]
- Liu, Y.C.; Liao, C.H.; Chang, C.F.; Chou, C.C.; Lin, Y.R. A locally transmitted case of SARS-CoV-2 infection in Taiwan. N. Engl. J. Med. 2020, 382, 1070–1072. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Laing, W.H.; Ou, C.Q.; He, J.X. Clinical characteristics of 2019 novel coronavirus infection in China. MedRxiv 2020. [Google Scholar] [CrossRef]
- Liu, Z.; Jin, C.; Wu, C.C.; Liang, T.; Zhao, H.; Wang, Y.; Wang, Z.; Li, F.; Zhou, J.; Cai, S.; et al. Association between Initial Chest CT or Clinical Features and Clinical Course in Patients with Coronavirus Disease 2019 Pneumonia. Korean J. Radiol. 2020, 21, 736–745. [Google Scholar] [CrossRef]
- Kim, G.U.; Kim, M.J.; Ra, S.H.; Lee, J.; Bae, S.; Jung, J.; Kim, S.H. Clinical characteristics of asymptomatic and symptomatic patients with mild COVID-19. Clin. Microbiol. Infect. 2020, 26, 948.e1–948.e3. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.; Lin, M.; Wei, L.; Xie, L.; Zhu, G.; Dela Cruz, C.S.; Sharma, L. Epidemiologic and clinical characteristics of novel coronavirus infections involving 13 patients outside Wuhan, China. JAMA 2020, 323, 1092–1093. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiologicaland clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, e497–e506. [Google Scholar] [CrossRef]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern. Med. 2020, e200994. [Google Scholar] [CrossRef] [PubMed]
- Sagnelli, C.; Gentile, V.; Tirri, R.; Macera, M.; Cappabianca, S.; Ciccia, F.; Coppola, N. Chronic conventional disease-modifying anti-rheumatic drugs masking severe SARS-CoV-2 manifestations in an elderly rheumatic patient. J. Infect. 2020, 29. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, Y.; Zhang, C.; Huang, F.; Wang, F.; Yuan, J.; Wang, Z.; Li, J.; Li, J.; Feng, C.; et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci. China Life Sci. 2020, 63, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.W.; Wu, X.X.; Jiang, X.G.; Xu, X.W.; Wu, X.X.; Jiang, X.G.; Xu, K.J.; Ying, L.J.; Ma, C.L.; Sho Bi, L.; et al. Clinical findings in a group of patients infected with the 2019 novel coronavirus (SARS-Cov-2) outside of Wuhan, China: Retrospective case series. BMJ 2020, 368, m606. [Google Scholar] [CrossRef]
- Pan, L.; Mu, M.; Yang, P.; Sun, Y.; Wang, R.; Yan, J.; Li, P.; Hu, B.; Wang, J.; Hu, C.; et al. Clinical characteristics of COVID-19 patients with digestive symptoms in Hubei, China: A descriptive, cross sectional, multicenter study. Am. J. Gastroenterol. 2020, 115, 766–773. [Google Scholar] [CrossRef]
- Lippi, G.; Plebani, M. Laboratory abnormalities in patients with COVID-2019 infection. Clin. Chem. Lab. Med. 2020. [Google Scholar] [CrossRef]
- Mann, D.L. Innate immunity and the failing heart: The cytokine hypothesis revisited. Circ. Res. 2015, 116, 1254–1268. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.H.; Liu, Y.X.; Yuan, J.; Wang, F.X.; Wu, W.B.; Li, J.X.; Wang, L.F. First case of COVID-19 complicated with fulminant myocarditis: A case report and insights. Infection 2020, 1–5. [Google Scholar] [CrossRef]
- Chen, C.; Zhou, Y.; Wang, D.W. SARS-CoV-2: A potential novel etiology of fulminant myocarditis. Herz 2020, 45, 230–232. [Google Scholar] [CrossRef] [PubMed]
- Inciardi, R.M.; Lupi, L.; Zaccone, G.; Italia, L.; Raffo, M.; Tomasoni, D.; Cani, D.S.; Cerini, M.; Farina, D.; Gavazzi, E.; et al. Cardiac involvement in a patient with coronavirus disease 2019 (COVID-19). JAMA Cardiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Hu, B.; Hu, C.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. J. Am. Med. Assoc. 2020, 323, 1061–1069. [Google Scholar] [CrossRef]
- Zhang, L.; Yan, X.; Fan, Q.; Liu, H.; Liu, X.; Liu, Z.; Zhang, Z. D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. J. Thromb. Haemost. 2020, 18, 1324–1329. [Google Scholar] [CrossRef]
- Han, H.; Yang, L.; Liu, R.; Yang, L.; Liu, R.; Liu, F.; Wu, K.L.; Li, J.; Liu, X.H.; Zhu, C.L. Prominent changes in blood coagulation of patients with SARS-CoV-2 infection. Clin. Chem. Lab. Med. 2020. [Google Scholar] [CrossRef]
- Recalcati, S. Cutaneous manifestations in COVId19: A first perspective. J. Eur. Acad. Dermatol. Venereol. 2020, 34, e212–e213. [Google Scholar] [CrossRef]
- Ahmad, I.; Rathore, F.A. Neurological manifestations and complications of COVID-19: A literature review. J. Clin. Neurosci. 2020. [Google Scholar] [CrossRef]
- Mao, L.; Jin, H.; Wang, M.; Hu, Y.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D.; et al. Neurologic Manifestations of Hospitalized Patients With Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020. [Google Scholar] [CrossRef]
- Filatov, A.; Sharma, P.; Hindi, F.; Espinosa, P.S. Neurological Complications of Coronavirus Disease (COVID-19): Encephalopathy. Cureus March 2020, 12, e7352. [Google Scholar] [CrossRef] [PubMed]
- Helms, J.; Kremer, S.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Kummerlen, C.; Collange, O.; Boulay, C.; Fafi-Kremer, S.; Ohana, M.; et al. Neurologic Features in Severe SARS-CoV-2 Infection. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Baig, A.M.; Khaleeq, A.; Ali, U.; Syeda, H. Evidence of the COVID-19 Virus Targeting the CNS: Tissue Distribution, Host Virus Interaction, and Proposed Neurotropic Mechanisms. ACS Chem. Neurosci. 2020, 11, 995–998. [Google Scholar] [CrossRef] [PubMed]
- Netland, J.; Meyerholz, D.K.; Moore, S.; Cassell, M.; Perlman, S. Severe acute respiratory syndrome coronavirus infection causes neuronal death in the absence of encephalitis in mice transgenic for human ACE2. J. Virol. 2008, 82, 7264–7275. [Google Scholar] [CrossRef]
- The Novel Coronavirus Pneumonia Emergency Response Epidemiology Team. The epidemiological characteristics of an outbreak of 2019 novel coronavirus disease (COVID-19) e China. China CDC Wkly. 2020, 41, 145. Available online: https://www.cdc.gov/mmwr/Novel_Coronavirus_Reports.html (accessed on 29 June 2020).
- Henry, B.M.; de Oliveira, M.H.S.; Benoit, S.; Plebani, M.; Lippi, G. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): A meta-analysis. Clin. Chem. Lab. Med. 2020, 58, 1021–1028. [Google Scholar] [CrossRef]
- Fu, L.; Wang, B.; Yuan, T.; Chen, X.; Ao, Y.; Fitzpatrick, T.; Li, P.; Zhou, Y.; Lin, Y.F.; Duan, Q.; et al. Clinical characteristics of coronavirus disease 2019 (COVID-19) in China: A systematic review and meta-analysis. J. Infect. 2020, 80, 656–665. [Google Scholar] [CrossRef]
- Sardu, C.; D’Onofrio, N.; Balestrieri, M.L.; Barbieri, M.; Rizzo, M.R.; Messina, V.; Maggi, P.; Coppola, N.; Paolisso, G.; Marfella, R. Outcomes in Patients With Hyperglycemia Affected by COVID-19: Can We Do More on Glycemic Control? Diabetes Care 2020, 43, 1408–1415. [Google Scholar] [CrossRef]
- Marfella, R.; Paolisso, P.; Sardu, C.; Bergamaschi, L.; D’Angelo, E.C.; Barbieri, M.; Rizzo, M.R.; Messina, V.; Maggi, P.; Coppola, N.; et al. Negative impact of hyperglycaemia on tocilizumab therapy in Covid-19 patients. Diabetes Metab. 2020, 21. [Google Scholar] [CrossRef]
- Behzad, S.; Aghaghazvini, L.; Radmard, A.R.; Gholamrezanezhad, A. Extrapulmonary manifestations of COVID-19: Radiologic and clinical overview. Clin. Imaging 2020, 66, 35–41, Online ahead of print. [Google Scholar] [CrossRef]
- Xiong, Y.; Sun, D.; Liu, Y.; Fan, Y.; Zhao, L.; Li, X.; Zhu, W.I. Clinical and High-Resolution CT Features of the COVID-19 Infection: Comparison of the Initial and Follow-up Changes. Invest. Radiol. 2020, 55, 332–339. [Google Scholar] [CrossRef] [PubMed]
| All | In Home Isolation (a) | Hospitalized (b) | p-Value a vs. b | |
|---|---|---|---|---|
| N° of patients | 40 | 24 | 16 | |
| N° (%) of patients in class: | ||||
| 18–29 | 4 (10) | 3 (13) | 1 (6) | 0.63 |
| 30–39 | 2 (5) | 2 (8) | 0 | 0.50 |
| 40–49 | 11 (28) | 7 (29) | 4 (25) | 1.0 |
| 50–59 | 8 (20) | 6 (25) | 2 (13) | 0.43 |
| 60–69 | 8 (20) | 6 (25) | 2 (13) | 0.43 |
| >70 | 7 (17) | 0 | 7 (44) | 0.0006 |
| Age, years, median (IQR) | 52 (41.25–65.75) | 43.5 (39.75–55.25) | 69 (48.5–80.25) | 0.0017 |
| N° (%) of males | 20 (50%) | 14 (58) | 6 (38) | 0.33 |
| N° (%) of patients with comorbidity: | 22 (55) | 10 (42) | 12 (75) | 0.054 |
| Arterial Hypertension | 17 (42) | 5 (21) | 12 (75) | 0.0011 |
| Diabetes Mellitus | 4 (10) | 1 (4) | 3 (19) | 0.28 |
| Malignancy | 4 (10) | 0 | 4 (25) | 0.019 |
| Chronic Respiratory Disease | 4 (10) | 1 (4) | 3 (29) | 0.28 |
| Cardiovascular Disease | 8 (20) | 4 (17) | 4 (25) | 0.69 |
| Renal Insufficiency | 2 (5) | 1 (4) | 1 (6) | 1.0 |
| Symptoms, N° (%) of subjects: | 37 (92.5) | 21 (88) | 16 (100) | 1.0 |
| Fever | 31 (77) | 17 (71) | 14 (88) | 0.27 |
| Cough | 15 (37) | 6 (25) | 9 (56) | 0.093 |
| Dyspnea | 5 (13) | 0 | 5 (33) | 0.0066 |
| Anosmia | 12 (30) | 6 (25) | 6 (38) | 0.48 |
| Ageusia/Dysgeusia | 13 (33) | 8 (33) | 5 (32) | 1.0 |
| Diarrhea | 8 (20) | 2 (8) | 6 (38) | 0.042 |
| Nausea | 3 (8) | 1 (4) | 2 (13) | 0.55 |
| Lack of appetite | 23 (58) | 15 (63) | 8 (50) | 0.52 |
| Fatigue | 24 (60) | 15 (63) | 9 (56) | 0.75 |
| Myalgia | 23 (58) | 15 (63) | 8 (50) | 0.52 |
| Rhinorrea | 2 (5) | 1 (4) | 1 (6) | 1.0 |
| Conjunctivitis | 2 (5) | 0 | 2 (13) | 0.15 |
| Skin lesions | 2 (5) | 1 (4) | 1 (6) | 1.0 |
| N° (%) of patients with CT evidence of interstitial pneumonia | 14 (35) | 0 | 14 (88) | <0.00001 |
| Time, days elapsed from the first positive swab to negative swab, median (IQR) | 22.5 (13.75–32) | 22.0 (12.2–32.0) | 22.5 (17.5–32.7) | 0.75 |
| Author [Ref.] | Country | N° Patients | Males N° (%) | Age, Years Median (Range) | Evidence |
|---|---|---|---|---|---|
| Gastro-intestinal manifestations | |||||
| Guan WJ, et al. [21] | China | 1099 | 640 (58.1%) | 47 (35–58) | 55 (5.0%) nausea or vomiting 42 (3.8%) diarrhea |
| Chen N, et al. [25] | China | 99 | 67 (68%) | 55 (21–82) | 43 (43%)liver function abnormality |
| Huang C, et al. [26] | China | 41 | 30 (73%) | 49 (IQR 41–58) | 1 (3%) diarrhea |
| Pan L, et al. [31] | China | 204 | 107 (52%) | 52.9 ± 16 | 103 (50.5%) digestive symptom 81 (78.6%) lack of appetite35 (34%) diarrhea 4 (3.9%) vomiting 2 (1.9%) abdominal pain |
| Cardio-vascular manifestations | |||||
| Chen C, et al. [35] | China | 41 | 30 (73%) | N/A | 5 (12%) acute cardiac injury |
| Wang D, et al. [36] | China | 138 | 75 (54.3%) | 56 (22–92) | 10 (7.2%) acute cardiac injury |
| Zhang L, et al. [38] | China | 343 | 169 (49.7%) | 68 (18–92) | 67 (19%) D-dimer levels over 2.0 µg/mL |
| Han H, et al. [39] | China | 94 cases 40 controls | 48 (51%) cases 28 (70%) in the control group | N/A | D-dimer (10.36 vs. 0.26 ng/L; p < 0.001), and FDP (33.83 vs. 1.55 mg/L; p < 0.001) were higher in case than in control group |
| Dermatological manifestations | |||||
| Recalcati S, et al. [40] | Italy | 88 | N/A | N/A | 18 (20%) cutaneous manifestation 14 (77%) erythematous rash 3 (16%) widespread urticaria 1 (5.5%) chickenpox-like vesicles |
| Neurological manifestations | |||||
| Mao L, et al. [42] | China | 214 | 87 (40.7%) | 52.7 (SD 15.5) | 78 (36.4%) neurologic symptoms (more common in patients with severe infection (45.5%) 36 (16.8%) dizziness 28 (13.1%) headache 12 (5.6%) taste impairment11 (5.1%) smell impairment |
| Helms J, et al. [44] | France | 58 | N/A | 63 (IQR, 37–65) | 47(81%) neurologic findings |
| Author [Ref.] | Country | N° Patients | N° (%) of Males | Age, Years Median (Range) | N° (%) of Severe Forms | N° (%) of Deaths | Factors Associated with Severe Forms |
|---|---|---|---|---|---|---|---|
| Guan W, et al. [15] | China | 1099 | 640 (58.1%) | 47 (35–58) Median (IQR) | 173 (15.7%) | 15 (1.4%) | Age, presence of any coexisting illness, laboratory abnormalities |
| Wang D, et al. [37] | China | 138 | 75 (54.3%) | 56 (22–92) | 36 (26.1%) | 6 (4.3%) | Age, comorbidities, pharyngeal pain, dyspnea, dizziness, abdominal pain, anorexia, higher levels of D-dimer, creatine kinase, and creatine |
| Chen N, et al. [25] | China | 99 | 67 (68%) | 55 (21–82) | 23 (23%) [ICU] | 11 (11%) | Age, smoking, lymphopenia, bilateral pneumonia, hypertension |
| Huang C, et al. [26] | China | 41 | 30 (73%) | 49 (41–58) Median (IQR) | 13 (32%) [ICU] | 6 (15%) | Higher plasma levels of IL2, IL7, IL10, GSCF, IP10, MCP1, MIP1A, and TNFα, higher prothrombin time and D-dimer level |
| The Novel Coronavirus Pneumonia Emergency Response Epidemiology Team [47] | China | 44,672 | 22,981 (51.4%) | (30–79) | 6168 (13.8%), severe 2087 (4.7%), critical | 1023 (2.3%) | Age, male, comorbidities |
| Fu L, et al. [49] | China | 3600 (from 43 studies) | 56.50% (from 42 studies) | 41 (39–72) | 25.6% (from 21 studies) | 3.60% | Age, laboratory abnormalities, comorbidities |
| Liu Z, et al. [22] | China | 72 | 39 (54.2%) | 46.2 ± 5.9 (M ± SD) | 8 (11.1%) | 0 | Age, higher lung severity score, lymphopenia |
| Zhang L, et al. [38] | China | 343 | 169 (49.7%) | 68 (18–92) | N/A | 13 (3.8%) | Higher D-dimer level |
| Mao L, et al. [42] | China | 214 | 87 (40.7%) | 52.7 ± 15.5 (M ± SD) | 88 (41.1%) | N/A | Age, comorbidities (especially hypertension), neurologic manifestations, increased inflammatory response, including higher white blood cell counts, neutrophil counts, lower lymphocyte counts, increased C-reactive protein levels, higher D-dimer level, and multiple organ involvement |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Macera, M.; De Angelis, G.; Sagnelli, C.; Coppola, N.; Vanvitelli COVID-19 Group. Clinical Presentation of COVID-19: Case Series and Review of the Literature. Int. J. Environ. Res. Public Health 2020, 17, 5062. https://doi.org/10.3390/ijerph17145062
Macera M, De Angelis G, Sagnelli C, Coppola N, Vanvitelli COVID-19 Group. Clinical Presentation of COVID-19: Case Series and Review of the Literature. International Journal of Environmental Research and Public Health. 2020; 17(14):5062. https://doi.org/10.3390/ijerph17145062
Chicago/Turabian StyleMacera, Margherita, Giulia De Angelis, Caterina Sagnelli, Nicola Coppola, and Vanvitelli COVID-19 Group. 2020. "Clinical Presentation of COVID-19: Case Series and Review of the Literature" International Journal of Environmental Research and Public Health 17, no. 14: 5062. https://doi.org/10.3390/ijerph17145062
APA StyleMacera, M., De Angelis, G., Sagnelli, C., Coppola, N., & Vanvitelli COVID-19 Group. (2020). Clinical Presentation of COVID-19: Case Series and Review of the Literature. International Journal of Environmental Research and Public Health, 17(14), 5062. https://doi.org/10.3390/ijerph17145062







