Impact of Whole Body Electromyostimulation on Velocity, Power and Body Composition in Postmenopausal Women: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Approach
2.2. Participants
2.3. Interventions
2.4. Outcomes
2.4.1. Primary Outcomes: Body Composition
2.4.2. Secondary Outcomes: Power and Velocity
2.5. Sample Size
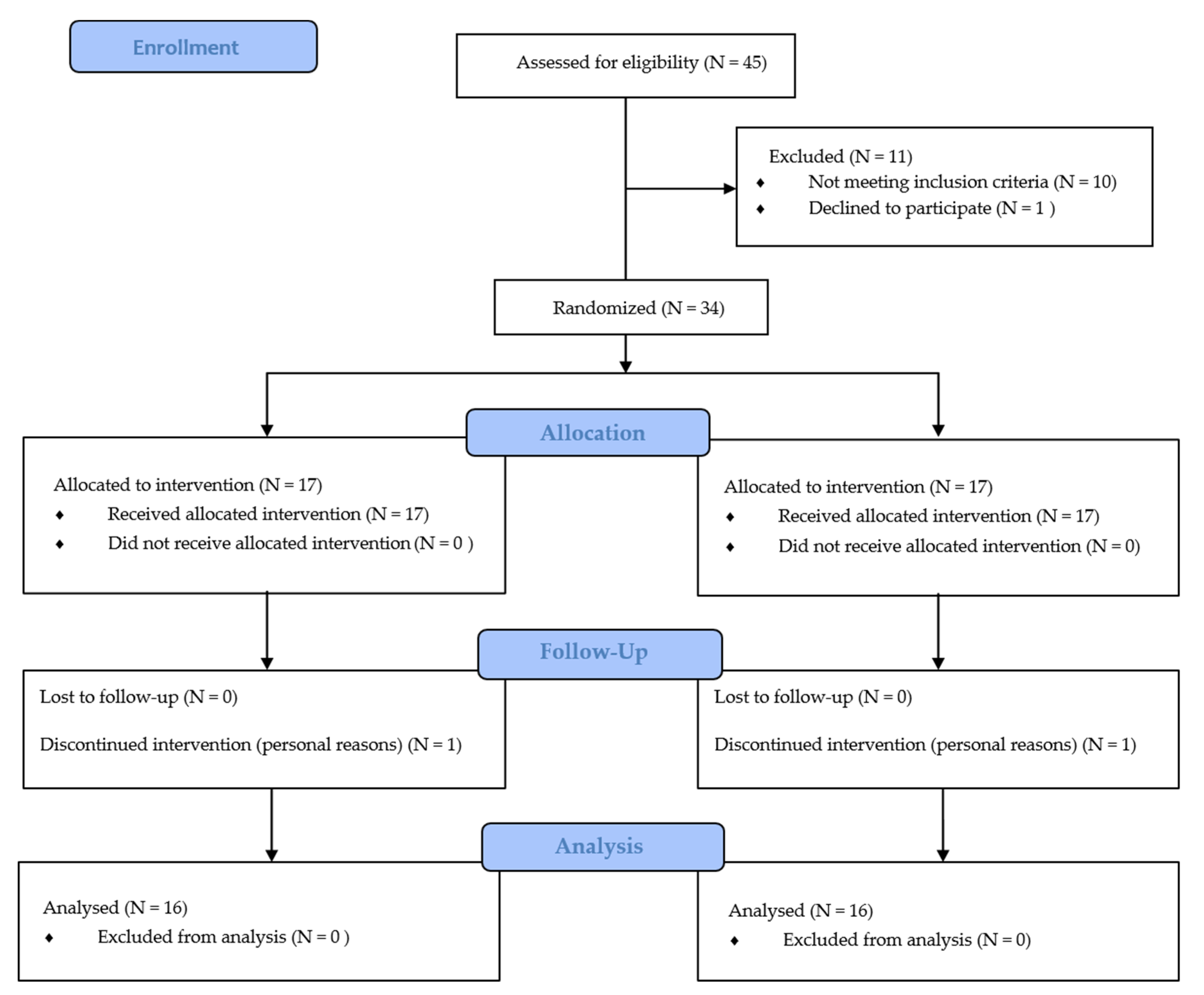
2.6. Randomization
2.7. Statistical Procedures
3. Results
3.1. Sample Characteristics
3.2. Body Composition
3.3. Power and Velocity
4. Discussion
4.1. Body Composition
4.2. Power and Velocity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Trombetti, A.; Reid, K.F.; Hars, M.; Herrmann, F.R.; Pasha, E.; Phillips, E.M.; Physiology, E.; Human, J.M. Age-associated declines in muscle mass, strength, power, and physical performance: Impact on fear of falling and quality of life. Osteoporos. Int. 2016, 27, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Bloom, D.E.; Chatterji, S.; Kowal, P.; Lloyd-sherlock, P.; Mckee, M.; Rosenberg, L.; Smith, J.P. Macroeconomic implications of population ageing and selected policy responses. Lancet 2016, 385, 649–657. [Google Scholar] [CrossRef]
- Milanović, Z.; Pantelić, S.; Trajković, N.; Sporiš, G.; Kostić, R.; James, N. Age-related decrease in physical activity and functional fitness among elderly men and women. Clin. Interv. Aging 2013, 8, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Dakkak W, T.A. Pathogenesis and Management of Sarcopenia. Physiol. Behav. 2017, 176, 139–148. [Google Scholar]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.-P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef]
- Goodpaster, B.H.; Park, S.W.; Harris, T.B.; Kritchevsky, S.B.; Nevitt, M.; Schwartz, A.V.; Simonsick, E.M.; Tylavsky, F.A.; Visser, M.; Newman, A.B. The loss of skeletal muscle strength, mass, and quality in older adults: The health, aging and body composition study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2006, 61, 1059–1064. [Google Scholar] [CrossRef]
- Janssen, I.; Heymsfield, S.B.; Ross, R. Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J. Am. Geriatr. Soc. 2002, 50, 889–896. [Google Scholar] [CrossRef]
- Dmitruk, A.; Czeczelewski, J.; Czeczelewska, E.; Golach, J.; Parnicka, U. Body composition and fatty tissue distribution in women with various menstrual status. Rocz. Panstw. Zakl. Hig. 2018, 69, 95–101. [Google Scholar]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A. Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef]
- Hirvensalo, M.; Rantanen, T.; Heikkinen, E. Mobility difficulties and physical activity as predictors of mortality and loss of independence in the community-living older population. J. Am. Geriatr. Soc. 2000, 48, 493–498. [Google Scholar] [CrossRef]
- Orkaby, A.R.; Forman, D.E. Physical activity and CVD in older adults: An expert’s perspective. Expert Rev. Cardiovasc. Ther. 2018, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wullems, J.A.; Verschueren, S.M.P.; Degens, H.; Morse, C.I.; Onambélé, G.L. A review of the assessment and prevalence of sedentarism in older adults, its physiology/health impact and non-exercise mobility counter-measures. Biogerontology 2016, 17, 547–565. [Google Scholar] [CrossRef] [PubMed]
- Etnier, J.L.; Karper, W.B.; Park, S.Y.; Shih, C.H.; Piepmeier, A.T.; Wideman, L. Motivating mature adults to be physically active. J. Aging Phys. Act. 2017, 25, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Lohne-Seiler, H.; Torstveit, M.K.; Anderssen, S.A. Training: Effects on Muscle Strength and Power in the Elderly. J. Aging Phys. Act. 2013, 21, 51–70. [Google Scholar] [CrossRef]
- Vechin, F.C.; Libardi, C.A.; Conceicao, M.S.; Damas, F.R.; Lixandrao, M.E.; Berton, R.P.B.; Tricoli, V.A.A.; Roschel, H.A.; Cavaglieri, C.R.; Chacon-Mikahil, M.P.T.; et al. Comparisons between low-intensity resistance training with blood flow restriction and high-intensity resistance training on quadriceps muscle mass and strength in elderly. J. Strength Cond. Res. 2015, 29, 1071–1076. [Google Scholar] [CrossRef]
- Narici, M.V.; Reeves, N.D.; Morse, C.I.; Maganaris, C.N. Muscular adaptations to resistance exercise in the elderly. J. Musculoskelet. Neuronal Interact. 2004, 4, 161–164. [Google Scholar]
- Rice, J.; Keogh, J.W.L. Power Training: Can it Improve Functional Performance in Older Adults? A Systematic Review. Int. J. Exerc. Sci. 2009, 2, 131–151. [Google Scholar]
- Winett, R.A.; Ogletree, A.M. Evidence-Based, High-Intensity Exercise and Physical Activity for Compressing Morbidity in Older Adults: A Narrative Review. Innov. Aging 2019, 3, igz020. [Google Scholar] [CrossRef]
- Sogaard, D.; Lund, M.T.; Scheuer, C.M.; Dehlbaek, M.S.; Dideriksen, S.G.; Abildskov, C.V.; Christensen, K.K.; Dohlmann, T.L.; Larsen, S.; Vigelso, A.H.; et al. High-intensity interval training improves insulin sensitivity in older individuals. Acta Physiol. 2018, 222, e13009. [Google Scholar] [CrossRef]
- Garcia-Pinillos, F.; Laredo-Aguilera, J.A.; Munoz-Jimenez, M.; Latorre-Roman, P.A. Effects of 12-Week Concurrent High-Intensity Interval Strength and Endurance Training Program on Physical Performance in Healthy Older People. J. Strength Cond. Res. 2019, 33, 1445–1452. [Google Scholar] [CrossRef]
- Kemmler, W.; Bebenek, M.; Engelke, K.; von Stengel, S. Impact of whole-body electromyostimulation on body composition in elderly women at risk for sarcopenia: The Training and ElectroStimulation Trial (TEST-III). Age 2014, 36, 395–406. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; Teschler, M.; Weissenfels, A.; Bebenek, M.; Frohlich, M.; Kohl, M.; von Stengel, S. Effects of Whole-Body Electromyostimulation versus High-Intensity Resistance Exercise on Body Composition and Strength: A Randomized Controlled Study. Evid. Based Complement. Alternat. Med. 2016, 2016, 9236809. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; Schliffka, R.; Mayhew, J.L.; von Stengel, S. Effects of whole-body electromyostimulation on resting metabolic rate, body composition, and maximum strength in postmenopausal women: The Training and ElectroStimulation Trial. J. Strength Cond. Res. 2010, 24, 1880–1887. [Google Scholar] [CrossRef] [PubMed]
- Von Stengel, S.; Bebenek, M.; Engelke, K.; Kemmler, W. Whole-body electromyostimulation to fight osteopenia in elderly females: The randomized controlled training and electrostimulation trial (TEST-III). J. Osteoporos. 2015, 2015, 643520. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; Teschler, M.; Weissenfels, A.; Bebenek, M.; von Stengel, S.; Kohl, M.; Freiberger, E.; Goisser, S.; Jakob, F.; Sieber, C.; et al. Whole-body electromyostimulation to fight sarcopenic obesity in community-dwelling older women at risk. Resultsof the randomized controlled FORMOsA-sarcopenic obesity study. Osteoporos. Int. 2016, 27, 3261–3270. [Google Scholar] [CrossRef] [PubMed]
- Schink, K.; Herrmann, H.J.; Schwappacher, R.; Meyer, J.; Orlemann, T.; Waldmann, E.; Wullich, B.; Kahlmeyer, A.; Fietkau, R.; Lubgan, D.; et al. Effects of whole-body electromyostimulation combined with individualized nutritional support on body composition in patients with advanced cancer: A controlled pilot trial. BMC Cancer 2018, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; Grimm, A.; Bebenek, M.; Kohl, M.; von Stengel, S.; Weissenfels, A.; Teschler, M.; Willert, S.; Bebenek, M.; Shojaa, M.; et al. Effects of Combined Whole-Body Electromyostimulation and Protein Supplementation on Local and Overall Muscle/Fat Distribution in Older Men with Sarcopenic Obesity: The Randomized Controlled Franconia Sarcopenic Obesity (FranSO) Study. Calcif. Tissue Int. 2018, 103, 266–277. [Google Scholar] [CrossRef]
- Kemmler, W.; Weissenfels, A.; Teschler, M.; Willert, S.; Bebenek, M.; Shojaa, M.; Kohl, M.; Freiberger, E.; Sieber, C.; von Stengel, S. Whole-body electromyostimulation and protein supplementation favorably affect sarcopenic obesity in community-dwelling older men at risk: The randomized controlled FranSO study. Clin. Interv. Aging 2017, 12, 1503–1513. [Google Scholar] [CrossRef]
- Kemmler, W.; von Stengel, S. Whole-body electromyostimulation as a means to impact muscle mass and abdominal body fat in lean, sedentary, older female adults: Subanalysis of the TEST-III trial. Clin. Interv. Aging 2013, 8, 1353–1364. [Google Scholar] [CrossRef]
- Amaro-Gahete, F.J.; De-la-O, A.; Sanchez-Delgado, G.; Robles-Gonzalez, L.; Jurado-Fasoli, L.; Ruiz, J.R.; Gutierrez, A. Functional Exercise Training and Undulating Periodization Enhances the Effect of Whole-Body Electromyostimulation Training on Running Performance. Front. Physiol. 2018, 9, 720. [Google Scholar] [CrossRef]
- von Stengel, S.; Kemmler, W. Trainability of leg strength by whole-body electromyostimulation during adult lifespan: A study with male cohorts. Clin. Interv. Aging 2018, 13, 2495–2502. [Google Scholar] [CrossRef] [PubMed]
- Pano-Rodriguez, A.; Beltran-Garrido, J.V.; Hernandez-Gonzalez, V.; Reverter-Masia, J. Effects of whole-body electromyostimulation on health and performance: A systematic review. BMC Complement. Altern. Med. 2019, 19, 87. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340. [Google Scholar] [CrossRef]
- Pano-Rodriguez, A.; Beltran-Garrido, J.V.; Hernandez-Gonzalez, V.; Reverter-Masia, J. Effects of Whole-Body Electromyostimulation on Physical Fitness in Postmenopausal Women: A Randomized Controlled Trial. Sensors 2020, 20, 1482. [Google Scholar] [CrossRef] [PubMed]
- Sjöström, M.; Oja, P.; Hagströmer, M.; Smith, B.J.; Bauman, A. Health-enhancing physical activity across European Union countries: The Eurobarometer study. J. Public Health (Bangkok) 2006, 14, 291–300. [Google Scholar] [CrossRef]
- Kovanen, V.; Aukee, P.; Kokko, K.; Finni, T.; Tarkka, I.M.; Tammelin, T.; Kujala, U.M.; Sipilä, S.; Laakkonen, E.K. Design and protocol of Estrogenic Regulation of Muscle Apoptosis (ERMA) study with 47 to 55-year-old women’s cohort: Novel results show menopause-related differences in blood count. Menopause 2018, 25, 1020–1032. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; Froehlich, M.; von Stengel, S.; Kleinöder, H. Whole-Body Electromyostimulation – The Need for Common Sense! Rationale and Guideline for a Safe and Effective Training. Dtsch. Z. Sportmed. 2016, 2016, 218–221. [Google Scholar] [CrossRef]
- Aragão-Santos, J.C.; de Resende-Neto, A.G.; Costa Nogueira, A.; Feitosa-Neta, M.L.; Albuquerque Brandão, L.H.; da Silva Chaves, L.M.; da Silva-Grigoletto, M.E. The effects of functional and traditional strength training on different parameters of strength elderly women: A trial randomized and controlled. J. Sports Med. Phys. Fit. 2018, 59, 380–386. [Google Scholar] [CrossRef]
- Gonzalez-Badillo, J.J.; Sanchez-Medina, L. Movement velocity as a measure of loading intensity in resistance training. Int. J. Sports Med. 2010, 31, 347–352. [Google Scholar] [CrossRef]
- Reed, J.L.; Pipe, A.L. The talk test: A useful tool for prescribing and monitoring exercise intensity. Curr. Opin. Cardiol. 2014, 29, 475–480. [Google Scholar] [CrossRef]
- Wirtz, N.; Wahl, P.; Kleinöder, H.; Wechsler, K.; Achtzehn, S.; Mester, J.; Kleinder, H.; Wechsler, K.; Achtzehn, S.; Mester, J. Acute metabolic, hormonal, and psychological responses to strength training with superimposed EMS at the beginning and the end of a 6 week training period. J. Musculoskelet. Neuronal Interact. 2015, 15, 325–332. [Google Scholar] [PubMed]
- Borg, G. Perceived exertion as an indicator of somatic stress. Scand. J. Rehabil. Med. 1970, 2, 92–98. [Google Scholar]
- Electroestimulación Deportiva Inalámbrica EMS—Wiemspro. Available online: http://wiemspro.com/es/ (accessed on 10 August 2017).
- Petrofsky, J. The effect of the subcutaneous fat on the transfer of current through skin and into muscle. Med. Eng. Phys. 2008, 30, 1168–1176. [Google Scholar] [CrossRef] [PubMed]
- Dörmann, U. Isometrische und Isoinertiale Parameter in der Kraftdiagnostik: Reliabilitätsprüfung und Evaluation von Effek-ten mechanischer und Elektrischer Krafttrainingsreize; German Sport University Cologne: Cologne, Germany, 2011. [Google Scholar]
- Filipovic, A.; Klein Der, H.; Rmann, U.D.; Mester, J. Electromyostimulation—a Systematic Review of the Influence of Training Regimens and Stimulation Parameters on Effectiveness in Electromyostimulation Training of Selected Strength Parameters. J. Strength Cond. Res. 2011. [Google Scholar] [CrossRef] [PubMed]
- Doucet, B.M.; Lam, A.; Griffin, L. Neuromuscular electrical stimulation for skeletal muscle function. Yale J. Biol. Med. 2012, 85, 201–215. [Google Scholar] [PubMed]
- A Guide to the Use of Electrical Stimulation in Pediatric Nurodisbility / Association of Paediatric Chartered Physiotherapists. Available online: https://apcp.csp.org.uk/content/guide-use-electrical-stimulation-paedaitric-neurodisability (accessed on 19 December 2019).
- Deley, G.; Babault, N. Could low-frequency electromyostimulation training be an effective alternative to endurance training? An overview in one adult. J. Sports Sci. Med. 2014, 13, 444–450. [Google Scholar]
- Amaro-Gahete, F.J.; de la O, A.; Jurado-Fasoli, L.; Ruiz, J.R.; Gutiérrez, Á. Could superimposed electromyostimulation be an effective training to improve aerobic and anaerobic capacity? Methodological considerations for its development. Eur. J. Appl. Physiol. 2017, 117, 1513–1515. [Google Scholar] [CrossRef]
- Alvero Cruz, J.E. Al Protocolo de valoración de la composición corporal para el reconocimiento médico-deportivo. Documento de Consenso del Grupo Español de Cineantropometría de la Federacion Española de Medicina del Deporte. Arch. Med. Deport. 2009, 26, 166–179. [Google Scholar]
- Lee, R.C.; Wang, Z.; Heo, M.; Ross, R.; Janssen, I.; Heymsfield, S.B. Total-body skeletal muscle mass: Development and cross-validation of anthropometric prediction models. Am. J. Clin. Nutr. 2000, 72, 796–803. [Google Scholar] [CrossRef]
- International Society for Advancement of Kinanthropometry. International Strandars for Anthropometric Assessment: ISAK; International Society for the Advancement of Kinanthropometry: Potchefstroom, South Africa, 2001; ISBN 0868037125. [Google Scholar]
- Fernando; Jiménez Gutiérrez, A.; Alvar, B.A.; Peterson, M.D. Assessing strength and power in resistance training. J. Hum. Sport Exerc. Off. J. Area Phys. Educ. Sport Fac. Educ. Univ. Alicant. Spain 2009, 4. [Google Scholar]
- Lamboley, C.R.; Wyckelsma, V.L.; Dutka, T.L.; McKenna, M.J.; Murphy, R.M.; Lamb, G.D. Contractile properties and sarcoplasmic reticulum calcium content in type I and type II skeletal muscle fibres in active aged humans. J. Physiol. 2015, 593, 2499–2514. [Google Scholar] [CrossRef]
- Morales, J.; Sobonya, S. Use of Submaximal Repetition Tests for Predicting 1-RM Strength in Class Athletes. J. Strength Cond. Res. 1996, 10, 186–189. [Google Scholar]
- Vivancos, A.; Zambudio, A.; Ramírez, F.; Del Águila, A.; Castrillón, F.; Pardo, P. OC14 Reliability and validity of a linear position transducer for strength assessment. Br. J. Sports Med. 2014, 48, A5.2-A5. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Beck, T.W. The importance of a priori sample size estimation in strength and conditioning research. J. Strength Cond. Res. 2013, 27, 2323–2337. [Google Scholar] [CrossRef] [PubMed]
- Moniker Privacy Services Random Team Generator—Split a List into Random Groups. Available online: https://www.randomlists.com/team-generator (accessed on 26 November 2019).
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. Sports Exerc. 2009, 41, 3–12. [Google Scholar] [CrossRef]
- Field, A. Discovering Statistics Using IBM SPSS Statistics; SAGE: London, UK, 2017; ISBN 9781526419521. [Google Scholar]
- Vincent, W.J.; Weir, J.P. Statistics in Kinesiology; Human Kinetics: Champaign, IL, USA, 2012; ISBN 9781450402545. [Google Scholar]
- Boutcher, Y.N.; Boutcher, S.H.; Yoo, H.Y.; Meerkin, J.D. The Effect of Sprint Interval Training on Body Composition of Postmenopausal Women. Med. Sci. Sports Exerc. 2019, 51, 1413–1419. [Google Scholar] [CrossRef]
- Falsarella, G.R.; Gasparotto, L.P.R.; Barcelos, C.C.; Coimbra, I.B.; Moretto, M.C.; Pascoa, M.A.; Ferreira, T.C.B.R.; Coimbra, A.M.V. Body composition as a frailty marker for the elderly community. Clin. Interv. Aging 2015, 10, 1661–1666. [Google Scholar] [CrossRef]
- Kemmler, W.; Birlauf, A. von Stengel Einfluss eines Elektromyostimulations-trainings auf die körperzusammensetzung bei älteren männern mit metabolischem syndrom. die tEst-II-studie. Dtsch. Z. Sportmed. 2010, 61, 117–123. [Google Scholar]
- Wittmann, K.; Sieber, C.; Von Stengel, S.; Kohl, M.; Freiberger, E.; Jakob, F.; Lell, M.; Engelke, K.; Kemmler, W. Impact of whole body electromyostimulation on cardiometabolic risk factors in older women with sarcopenic obesity: The randomized controlled FORMOsA-sarcopenic obesity study. Clin. Interv. Aging 2016, 11, 1697–1706. [Google Scholar] [CrossRef]
- Kemmler, W.; Grimm, A.; Bebenek, M.; Kohl, M.; von Stengel, S.; Weissenfels, A.; Teschler, M.; Willert, S.; Bebenek, M.; Shojaa, M.; et al. Effect of whole-body electromyostimulation and / or protein supplementation on obesity and cardiometabolic risk in older men with sarcopenic obesity: The randomized controlled FranSO trial. BMC Geriatr. 2018, 18, 70. [Google Scholar] [CrossRef] [PubMed]
- Schink, K.; Reljic, D.; Herrmann, H.J.; Meyer, J.; Mackensen, A.; Neurath, M.F.; Zopf, Y. Whole-Body Electromyostimulation Combined With Individualized Nutritional Support Improves Body Composition in Patients With Hematological Malignancies – A Pilot Study. Front. Physiol. 2018, 9, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Filipovic, A.; Grau, M.; Kleinoeder, H.; Zimmer, P.; Hollmann, W.; Bloch, W. Effects of a Whole-Body Electrostimulation Program on Strength, Sprinting, Jumping, and Kicking Capacity in Elite Soccer Players. J. Sports Sci. Med. 2016, 15, 639–648. [Google Scholar]
- Filipovic, A.; Kleinoder, H.; Pluck, D.; Hollmann, W.; Bloch, W.; Grau, M. Influence of Whole-Body Electrostimulation on Human Red Blood Cell Deformability. J. Strength Cond. Res. 2015, 29, 2570–2578. [Google Scholar] [CrossRef] [PubMed]
- Wirtz, N.; Zinner, C.; Doermann, U.; Kleinoeder, H.; Mester, J. Effects of Loaded Squat Exercise with and without Application of Superimposed EMS on Physical Performance. J. Sports Sci. Med. 2016, 15, 26–33. [Google Scholar]
- Filipovic, A.; DeMarees, M.; Grau, M.; Hollinger, A.; Seeger, B.; Schiffer, T.; Bloch, W.; Gehlert, S. Superimposed Whole-Body Electrostimulation Augments Strength Adaptations and Type II Myofiber Growth in Soccer Players During a Competitive Season. Front. Physiol. 2019, 10, 1187. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; Von Stengel, S.; Schwarz, J.; Mayhew, J.L. Effect of whole-body electromyostimulation on energy expenditure during exercise. J. Strength Cond. Res. 2012, 26, 240–245. [Google Scholar] [CrossRef]
- McKinnon, N.B.; Connelly, D.M.; Rice, C.L.; Hunter, S.W.; Doherty, T.J. Neuromuscular contributions to the age-related reduction in muscle power: Mechanisms and potential role of high velocity power training. Ageing Res. Rev. 2017, 35, 147–154. [Google Scholar] [CrossRef]
- Amaro-gahete, F.J.; De-la-o, A.; Jurado-fasoli, L.; Dote-montero, M.; Gutiérrez, Á.; Ruiz, J.R.; Castillo, M.J. Changes in Physical Fitness After 12 Weeks of Structured Concurrent Exercise Training, High Intensity Interval Training, or Whole-Body Electromyostimulation Training in Sedentary Middle-Aged Adults: A Randomized Controlled Trial. Front. Physiol. 2019, 10, 1–15. [Google Scholar] [CrossRef]
- Bassey, E.J.; Fiatarone, M.A.; O’Neill, E.F.; Kelly, M.; Evans, W.J.; Lipsitz, L.A. Leg extensor power and functional performance in very old men and women. Clin. Sci. 1992, 82, 321–327. [Google Scholar] [CrossRef]
- Hakkinen, K.; Kraemer, W.J.; Kallinen, M.; Linnamo, V.; Pastinen, U.M.; Newton, R.U. Bilateral and unilateral neuromuscular function and muscle cross-sectional area in middle-aged and elderly men and women. J. Gerontol. A Biol. Sci. Med. Sci. 1996, 51, B21–B29. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, M.; Aguado, X.; Gonzalez, R.; Lopez, J.L.; Hakkinen, K. Maximal and explosive force production capacity and balance performance in men of different ages. Eur. J. Appl. Physiol. Occup. Physiol. 1999, 79, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Skelton, D.A.; Greig, C.A.; Davies, J.M.; Young, A. Strength, power and related functional ability of healthy people aged 65-89 years. Age Ageing 1994, 23, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Nilwik, R.; Snijders, T.; Leenders, M.; Groen, B.B.L.; van Kranenburg, J.; Verdijk, L.B.; van Loon, L.J.C. The decline in skeletal muscle mass with aging is mainly attributed to a reduction in type II muscle fiber size. Exp. Gerontol. 2013, 48, 492–498. [Google Scholar] [CrossRef]
- Verdijk, L.B.; Snijders, T.; Drost, M.; Delhaas, T.; Kadi, F.; van Loon, L.J.C. Satellite cells in human skeletal muscle; from birth to old age. Age 2014, 36, 545–547. [Google Scholar] [CrossRef]
- Snijders, T.; Verdijk, L.B.; Smeets, J.S.J.; McKay, B.R.; Senden, J.M.G.; Hartgens, F.; Parise, G.; Greenhaff, P.; van Loon, L.J.C. The skeletal muscle satellite cell response to a single bout of resistance-type exercise is delayed with aging in men. Age 2014, 36, 9699. [Google Scholar] [CrossRef]
- Skelton, D.A.; Kennedy, J.; Rutherford, O.M. Explosive power and asymmetry in leg muscle function in frequent fallers and non-fallers aged over 65. Age Ageing 2002, 31, 119–125. [Google Scholar] [CrossRef]
- Micke, F.; Kleinöder, H.; Dörmann, U.; Wirtz, N.; Donath, L. Effects of an Eight-Week Superimposed Submaximal Dynamic Whole-Body Electromyostimulation Training on Strength and Power Parameters of the Leg Muscles: A Randomized Controlled Intervention Study. Front. Physiol. 2018, 9, 1719. [Google Scholar] [CrossRef]
- Colson, S.; Martin, A.; Van Hoecke, J. Re-examination of training effects by electrostimulation in the human elbow musculoskeletal system. Int. J. Sports Med. 2000, 21, 281–288. [Google Scholar] [CrossRef]
- Bezerra, P.; Zhou, S.; Crowley, Z.; Brooks, L.; Hooper, A. Effects of unilateral electromyostimulation superimposed on voluntary training on strength and cross-sectional area. Muscle Nerve 2009, 40, 430–437. [Google Scholar] [CrossRef]
- Gondin, J.; Duclay, J.; Martin, A. Neural drive preservation after detraining following neuromuscular electrical stimulation training. Neurosci. Lett. 2006, 409, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Gondin, J.; Duclay, J.; Martin, A. Soleus- and gastrocnemii-evoked V-wave responses increase after neuromuscular electrical stimulation training. J. Neurophysiol. 2006, 95, 3328–3335. [Google Scholar] [CrossRef] [PubMed]
- Gregory, C.M.; Bickel, C.S. Recruitment patterns in human skeletal muscle during electrical stimulation. Phys. Ther. 2005, 85, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Grill, W.M.; Mortimer, J.T. Mortimer Stimulus waveforms for selective neural stimulation. IEEE Eng. Med. Biol. Mag. 1995, 14, 375–385. [Google Scholar]
- Bickel, C.S.; Gregory, C.M.; Dean, J.C. Motor unit recruitment during neuromuscular electrical stimulation: A critical appraisal. Eur. J. Appl. Physiol. 2011, 111, 2399–2407. [Google Scholar] [CrossRef]
- Alghannam, A.F.; Gonzalez, J.T.; Betts, J.A. Restoration of muscle glycogen and functional capacity: Role of post-exercise carbohydrate and protein co-ingestion. Nutrients 2018, 10, 253. [Google Scholar] [CrossRef]
- Borges, N.; Reaburn, P.; Driller, M.; Argus, C. Age-Related Changes in Performance and Recovery Kinetics in Masters Athletes: A Narrative Review. J. Aging Phys. Act. 2016, 24, 149–157. [Google Scholar] [CrossRef]

| Variable | Total (n = 32) | EX + WB-EMS (n = 16) | EX (n = 16) | p-Value |
|---|---|---|---|---|
| Age (years) | 61.38 ± 3.95 | 63.06 ± 3.42 | 59.71 ± 3.82 | 0.011 |
| Body mass (kg) | 67.44 ± 10.84 | 67.11 ± 10.84 | 67.78 ± 10.12 | 0.866 |
| Height (cm) | 158.32 ± 5.28 | 156.69 ± 5.02 | 159.94 ± 5.18 | 0.081 |
| Body mass index (BMI, kg/m2) | 26.91 ± 4.11 | 27.28 ± 4.24 | 26.54 ± 4.08 | 0.620 |
| Variable | Group | Baseline | Post | % Change | MD [95% CI] | d |
|---|---|---|---|---|---|---|
| Weight (kg) | EX + WB-EMS | 67.11 ± 11.84 | 66.82 ± 12.10 | −0.43 | −0.29 [−1.38, 0.80] | −0.19 [−0.68, 0.31] |
| EX | 67.78 ± 10.12 | 67.49 ± 10.27 | −0.43 | −0.28 [−1.38, 0.80] | −0.19 [−0.68, 0.31] | |
| BMI (kg/m2) | EX + WB-EMS | 27.27 ± 4.24 | 27.16 ± 4.28 | −0.40 | −0.11 [−0.56, 0.34] | −0.18 [−0.67, 0.32] |
| EX | 26.54 ± 4.08 | 26.46 ± 4.15 | −0.30 | −0.08 [−0.54, 0.37] | −0.13 [−0.62, 0.36] | |
| Body fat () | EX + WB-EMS | 35.80 ± 5.75 | 35.99 ± 5.48 | 0.53 | 0.19 [−1.13, 1.51] | 0.10 [−0.39, 0.59] |
| EX | 36.61 ± 5.15 | 35.85 ± 5.39 | −2.08 | −0.76 [−2.08, 0.56] | −0.41 [−0.91, 0.11] | |
| Fat mass (kg) | EX + WB-EMS | 24.56 ± 8.15 | 24.73 ± 8.38 | 0.69 | 0.18 [−2.17, 2.52] | 0.05 [−0.44, 0.54] |
| EX | 26.27 ± 7.79 | 24.65 ± 7.72 | −6.17 | −1.61 [−1.96, 0.73] | −0.49 [−100, 0.04] | |
| Lean mass (kg) | EX + WB-EMS | 42.57 ± 4.80 | 42.11 ± 4.37 | −1.08 | −0.46 [−2.21, 1.30] | −0.18 [−0.68, 0.31] |
| EX | 41.80 ± 5.42 | 42.84 ± 3.20 | 2.49 | 1.04 [−0.71, 2.80] | 0.42 [−0.10, 0.93] | |
| Visceral fat (kg) | EX + WB-EMS | 9.06 ± 2.29 | 9.13 ± 2.36 | 0.77 | 0.06 [−2.71, 2.84] | 0.02 [−0.47, 0.51] |
| EX | 10.01 ± 6.07 | 8.38 ± 2.16 | −16.28 | −1.64 [−4.41, 1.14] | −0.42 [−0.92, 0.10] | |
| Abdominal fold (mm) | EX + WB-EMS | 26.43 ± 9.98 | 23.44 ± 6.43 | −11.31 | −2.99 [−6.57, 0.59] | −0.59 [−1.12, −0.05] |
| EX | 27.85 ± 10.61 | 27.13 ± 10.86 | −2.59 | −0.73 [−4.19, 2.74] | −0.15 [−0.64, 0.35] | |
| Waist to hip ratio | EX + WB-EMS | 0.83 ± 0.08 | 0.80 ± 0.06 | −3.61 | −0.03 [−0.14, 0.09] | −0.16 [−0.65, 0.34] |
| EX | 0.76 ± 0.22 | 0.83 ± 0.07 | 9.21 | 0.07 [−0.04, 0.18] | 0.45 [−0.07, 0.96] | |
| 6-fold (mm) | EX + WB-EMS | 132.48 ± 35.39 | 126.06 ± 22.50 | −4.85 | −6.41 [−19.20, 6.37] | 0.35 [−0.85, 0.16] |
| EX | 130.74 ± 37.62 | 133.13 ± 37.12 | 1.83 | 2.39 [−10.40, 15.17] | 0.13 [−0.36, 0.62] | |
| Waist (cm) | EX + WB-EMS | 84.13 ± 10.88 | 83.47 ± 10.11 | −0.78 | −0.66 [−9.81, 8.48] | −0.05 [−0.54, 0.44] |
| EX | 79.90 ± 21.64 | 84.24 ± 11.19 | 5.43 | 4.34 [−4.80, 13.48 | 0.34 [−0.17, 0.83] | |
| Hip (cm) | EX + WB-EMS | 102.21 ± 8.28 | 99.08 ± 14.79 | −3.06 | −3.14 [−12.22, 5.95] | −0.24 [−0.74, 0.26] |
| EX | 97.42 ± 14.54 | 101.37 ± 7.78 | 4.05 | 3.95 [−5.13, 13.03] | 0.31 [−0.20, 0.80] |
| Variable | Group | Baseline | Post | % Change | MD [95% CI] | d |
|---|---|---|---|---|---|---|
| Squat | ||||||
| Velocity (m·s−1) | EX + WB-EMS | 0.48 ± 0.10 | 0,75 ± 0.10 | 56.25 | 0.27 [0.20, 0.33] *** | 2.85 [1.72, 3.96] ^ |
| EX | 0.50 ± 0.05 | 0.66 ± 0.10 | 32.00 | 0.15 [0.09, 0.22] *** | 1.63 [0.86, 2.37] # | |
| Power (W) | EX + WB-EMS | 478.87 ± 143.32 | 782.18 ± 194.52 | 63.34 | 303.31 [223.18, 383.44] *** | 2.67 [1.60, 3.73] ^ |
| EX | 548.26 ± 100.43 | 730.571 ± 160.14 | 33.25 | 182.31 [102.18, 262.44] *** | 1.61 [0.84, 2.35] # | |
| Bench press | ||||||
| Velocity (m·s−1) | EX + WB-EMS | 0.50 ± 0.13 | 0.81 ± 0.08 | 62.00 | 0.39 [0.22, 0.39] *** | 2.48 [1.46, 3.47] ^ |
| EX | 0.58 ± 0.07 | 0.59 ± 0.07 | 1.72 | 0.01 [−0.08, 0.09] | 0.07 [−0.42, 0.56] | |
| Power (W) | EX + WB-EMS | 47.17 ± 14.35 | 78.24 ± 14.02 | 65.87 | 31.08 [22.63, 39.51] *** | 2.61 [1.55, 3.64] ^ |
| EX | 52.45 ± 53.41 | 53.41 ± 7.00 | 2.22 | 0.96 [−7.21, 9.13] | 0.08 [−0.41, 0.57] |
| Variable | ∆ EX + WB-EMS | ∆ EX | MD [95% CI] | d |
|---|---|---|---|---|
| Squat | ||||
| Velocity (m·s−1) | 0.11 ± 0.01 | 0.06 ± 0.01 | 0.04 [0.01, 0.08] * | 0.98 [0.23, 1.71] $ |
| Power (w) | 99.51 ± 11.96 | 60.83 ± 11.96 | 38.69 [1.75, 75.62] * | 0.81 [0.08, 1.52] $ |
| Bench press | ||||
| Velocity (m·s−1) | 0.13 ± 0.01 | 0.03 ± 0.01 | 0.10 [0.06, 0.14] *** | 1.90 [1.11, 2.82] # |
| Power (w) | 28.77 ± 2.69 | 3.12 ± 2.59 | 25.64 [17.48, 33.82] *** | 2.39 [1.49, 3.34] ^ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pano-Rodriguez, A.; Beltran-Garrido, J.V.; Hernandez-Gonzalez, V.; Nasarre-Nacenta, N.; Reverter-Masia, J. Impact of Whole Body Electromyostimulation on Velocity, Power and Body Composition in Postmenopausal Women: A Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2020, 17, 4982. https://doi.org/10.3390/ijerph17144982
Pano-Rodriguez A, Beltran-Garrido JV, Hernandez-Gonzalez V, Nasarre-Nacenta N, Reverter-Masia J. Impact of Whole Body Electromyostimulation on Velocity, Power and Body Composition in Postmenopausal Women: A Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2020; 17(14):4982. https://doi.org/10.3390/ijerph17144982
Chicago/Turabian StylePano-Rodriguez, Alvaro, Jose Vicente Beltran-Garrido, Vicenç Hernandez-Gonzalez, Natalia Nasarre-Nacenta, and Joaquin Reverter-Masia. 2020. "Impact of Whole Body Electromyostimulation on Velocity, Power and Body Composition in Postmenopausal Women: A Randomized Controlled Trial" International Journal of Environmental Research and Public Health 17, no. 14: 4982. https://doi.org/10.3390/ijerph17144982
APA StylePano-Rodriguez, A., Beltran-Garrido, J. V., Hernandez-Gonzalez, V., Nasarre-Nacenta, N., & Reverter-Masia, J. (2020). Impact of Whole Body Electromyostimulation on Velocity, Power and Body Composition in Postmenopausal Women: A Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 17(14), 4982. https://doi.org/10.3390/ijerph17144982








