The Role of Electrocardiography in Occupational Medicine, from Einthoven’s Invention to the Digital Era of Wearable Devices
Abstract
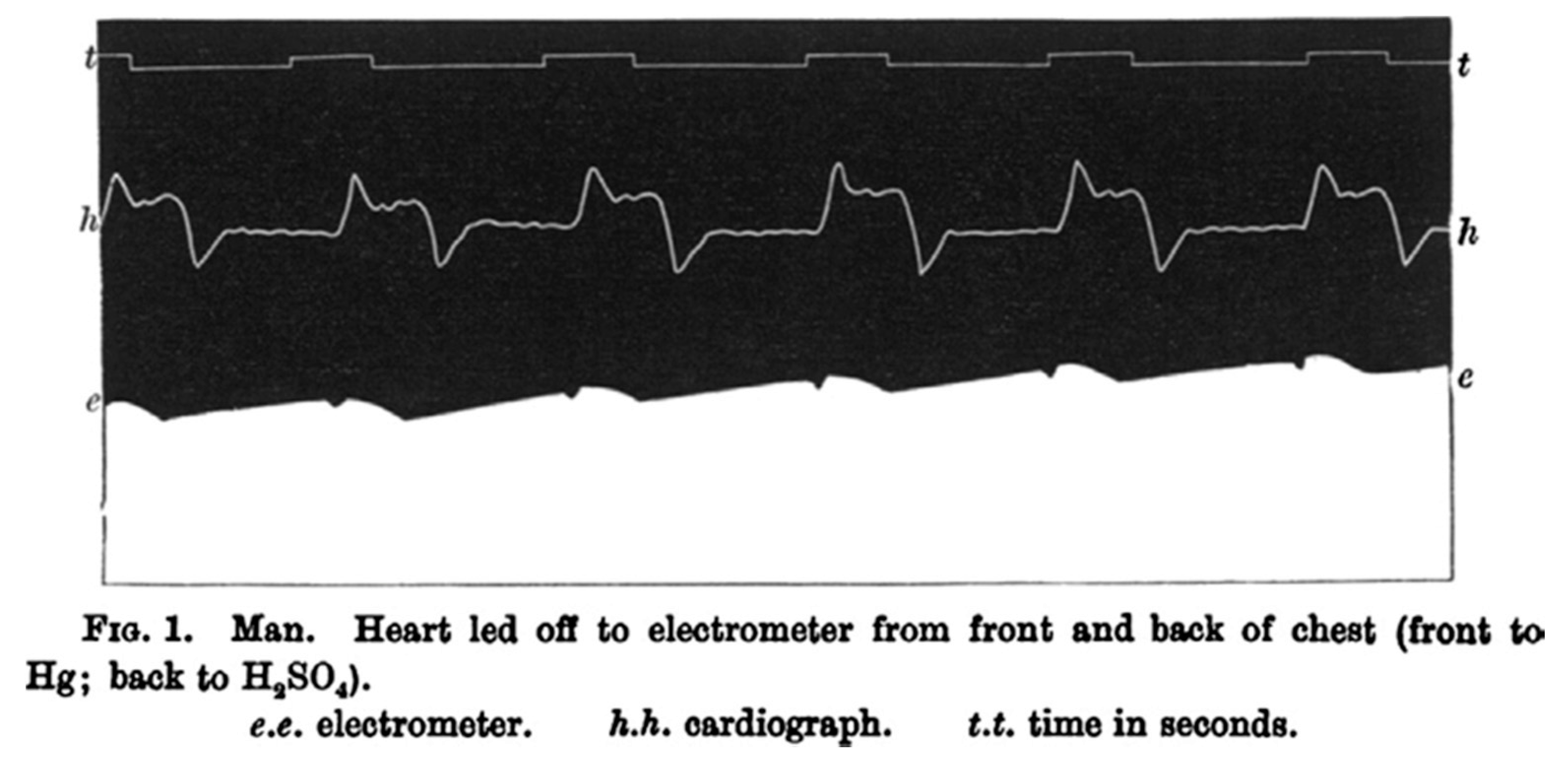
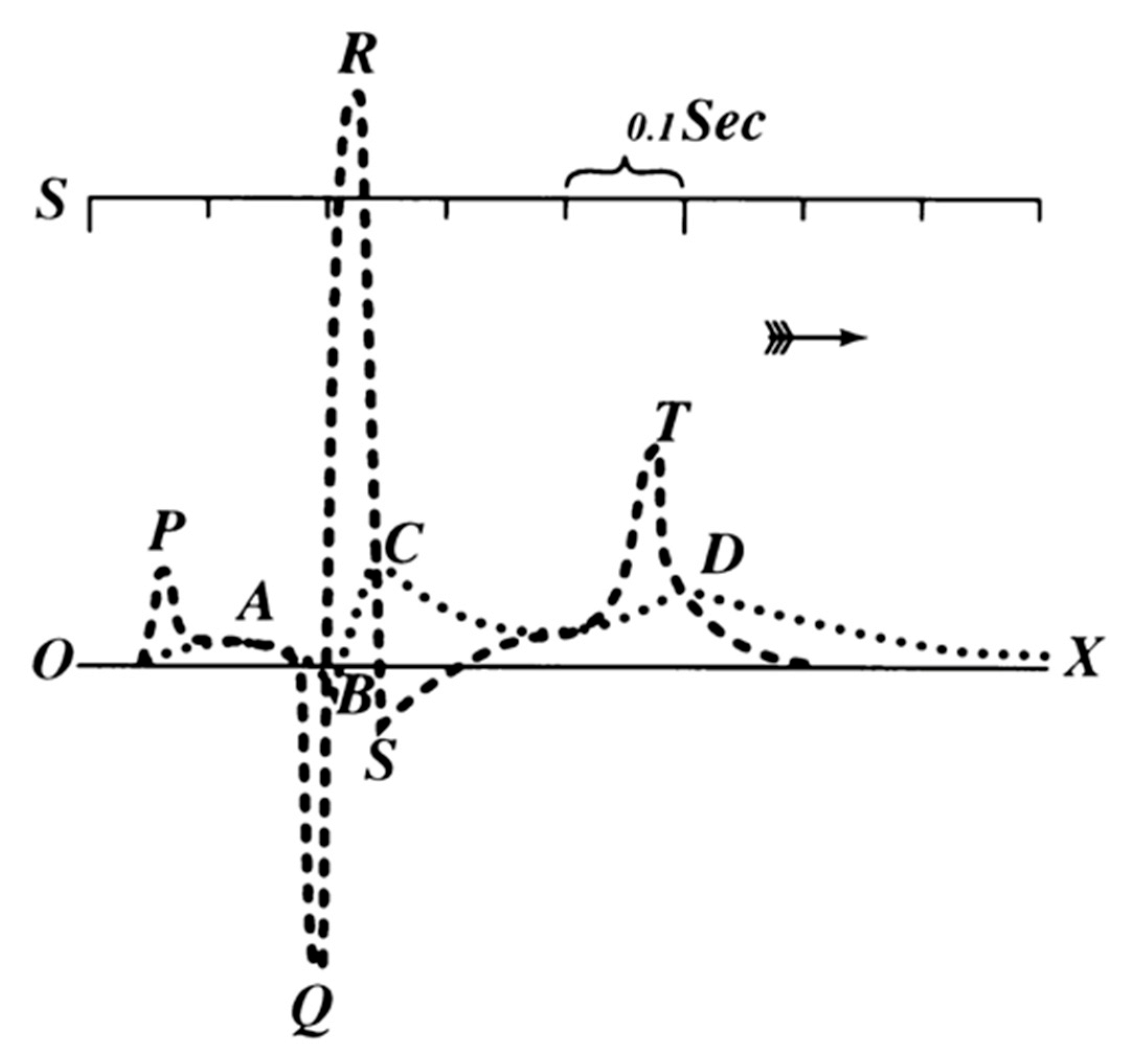
1. Dawn of Electrocardiography
2. Materials and Methods
- Population: workers;
- Interest: cardiovascular monitorization;
- Context: occupational medicine;
- Outcome: possible use in occupational safety and health (OSH)
- Setting: occupational settings;
- Perspective: worker;
- Intervention: cardiovascular monitorization with smart wearable devices;
- Comparison: cardiovascular monitorization with ambulatory instruments;
- Evaluation: possible use in occupational safety and health (OSH)
- Ad hoc string: (ECG OR electrocardiograph* OR electrocardiogram* OR EKG OR cardiogram*) AND ((wearable* OR mobile* OR portable*) AND (device* OR sensor* OR technology*))
- Article type: reviews and systematic reviews
- Publication date: last 5 years (since 2015)
- Language: English
- Ad hoc string: (‘worker’/exp OR ‘laborer’ OR ‘labourer’ OR ‘worker’) AND (‘cardiovascular monitoring device’/exp OR ‘wearable device’ OR ‘smart device’) AND ‘review’/it
3. Results
4. Evolution of Electrocardiography and Future Perspective
4.1. Wearable Smart Device Architecture
4.2. Monitoring of Cardiac Function in the Era of Wearable Devices
4.3. Wearable Devices: Potential and Criticality in the Working Context
| N | Title | Year | First Author | Country | Method | Key Message | PubMed | EMBASE |
|---|---|---|---|---|---|---|---|---|
| 1 | Lead-I ECG for detecting atrial fibrillation in patients with an irregular pulse using single time point testing: a systematic review and economic evaluation [17] | 2020 | Duarte R | UK | Systematic review | Single time point lead-I ECG devices appear to be a cost-effective compared with manual pulse palpation (MPP) followed by a 12-lead ECG. | ✓ | |
| 2 | Wearing Your Heart on Your Sleeve: the Future of Cardiac Rhythm Monitoring [41] | 2019 | Al-Alusi MA | USA | Review | Wearable ECG monitors are currently most useful to detect atrial fibrillation. Further study is needed to demonstrate whether wearable ECG monitors improve patient outcomes, and to expand their use into other indications. | ✓ | ✓ |
| 3 | How useful is the smartwatch ECG? [30] | 2019 | Isakadze N | USA | Review | The Apple Watch Series 4 ECG feature is FDA cleared for detection of presence of AF. AF represents only one area where the Apple Watch ECG shows promise to transform care. | ✓ | |
| 4 | Apple Watch, Wearables, and Heart Rhythm: where do we stand? [25] | 2019 | Raja JM | USA | Review | Health monitoring devices can be used as a non-invasive, ambulatory assessment of heart rate and rhythm. These devices have even shown to be cost-effective when used in a community screening program. | ✓ | ✓ |
| 5 | Electrode placement in electrocardiography smart garments: A review [13] | 2019 | Soroudi A | Sweden | Review | Since a successful ECG monitoring garment needs a multi factor design, a good strategy would be a cooperative design using techniques and experiences provided by medical experts, textile and garment designers. | ✓ | |
| 6 | Mobile Self-Monitoring ECG Devices to Diagnose Arrhythmia that Coincide with Palpitations: A Scoping Review [46] | 2019 | Marston HR | UK | Review | Mobile heart monitoring devices has several benefits: alleviation of patient anxiety, lowering the risk of morbidity and mortality, while progressively influencing national and international care pathway guidelines. | ✓ | |
| 7 | The use of photoplethysmography for assessing hypertension [33] | 2019 | Elgendi M | Canada | Narrative review | High Blood Pressure (BP) is a major source of mortality and morbidity around the world., Most PPG-based BP estimation is mainly divided into two research directions based on waveform morphology theory and waveform propagation theory. | ✓ | |
| 8 | Is There a Benefit to Patients Using Wearable Devices Such as Fitbit or Health Apps on Mobiles? A Systematic Review [40] | 2019 | Jo A | USA | Systematic review | This systematic review found a limited benefit to the use of a wearable device in chronic disease management. Findings showed inconsistent conclusions with respect to significant improvements in weight loss, blood pressure, and cholesterol level, except HbA1c. | ✓ | |
| 9 | Ambulatory ECG monitoring in the age of smartphones [24] | 2019 | Sanders D | USA | Narrative review | Consumer-oriented wearable devices are aimed at arrhythmia monitoring, which could lead to increased arrhythmia detection, but at the risk of more false-positive results and excessive use of healthcare resources. | ✓ | |
| 10 | Graphene-Based Sensors for Human Health Monitoring [12] | 2019 | Huang H | China | Narrative review | As a novel 2D material, graphene has aroused a boom in the field of sensor research around the world due to its advantages in mechanical, thermal, and electrical properties. Numerous graphene-based sensors used for human health monitoring have been reported. | ✓ | |
| 11 | Wearable devices for cardiac arrhythmia detection: a new contender? [22] | 2019 | Sajeev JK | Australia | Review | Potential patient-specific barriers may impede widespread screening using SDP. However, attitudes to SDP-based arrhythmia detection remain favourable compared with conventional Holter monitoring system for symptomatic arrhythmia. | ✓ | |
| 12 | Data management and wearables in older adults: A systematic review [36] | 2019 | Alharbi M | Australia | Systematic review | Wearable trackers are generally valid, reliable, and/or feasible when tracking step counts, MVPA, ECG and HR in aging populations. Thus, trackers may be ideal to help in diagnosing, measuring, monitoring, and/or motivating in this population cohort. There needs to be a framework and/or guideline and a standardized method for the collection and analysis of wearable tracker data. | ✓ | |
| 13 | The Current State of Mobile Phone Apps for Monitoring Heart Rate, Heart Rate Variability, and Atrial Fibrillation: Narrative Review [45] | 2019 | Li KHC | China | Narrative review | There is a role for mobile phone app in the diagnosis, monitoring, and screening of arrhythmias and HR. Within the context of HR monitoring and AF detection, given the impressive degree of sensitivity (>90%) and specificity (>90%) in most cases or apps, neither sensitivity nor specificity is more important than the other. | ✓ | |
| 14 | The Accuracy of Acquiring Heart Rate Variability from Portable Devices: A Systematic Review and Meta-Analysis [27] | 2019 | Dobbs WC | Switzerland | Systematic Review and Meta-Analysis | HRV measurements acquired using portable devices demonstrate a small amount of absolute error when compared to ECG. Portable devices were influenced by metric, position, and biological sex. Practitioners and researchers should consider the cost–benefit along with the simplicity of the measurement when attempting to increase compliance in acquiring HRV measurements. | ✓ | |
| 15 | The Clinical Value of Heart Rate Monitoring Using an Apple Watch [29] | 2019 | Karmen CL | USA | Review | Recent reports of the series 4 Apple Watch show that these devices can accurately discriminate atrial fibrillation from sinus rhythm with more than 93% sensitivity and 84% specificity compared with the ECG. | ✓ | |
| 16 | A systematic review of noninvasive electrocardiogram monitoring devices for the evaluation of suspected cardiovascular syncope [42] | 2019 | Solbiati M | Italy | Systematic review | This is an area of medicine where precision medicine, managing patients based on their own individual needs with the most appropriate device, is important. | ✓ | |
| 17 | Electronic textile electrocardiogram monitoring in cardiac patients: a scoping review protocol [14] | 2018 | Teferra MN | Australia | Systematic review | E-textiles are fabrics (or clothing) that contain electronic elements or circuits woven directly into the material and are an emerging interdisciplinary field of research. | ✓ | |
| 18 | Heart Rate Variability: An Old Metric with New Meaning in the Era of using mHealth Technologies for Health and Exercise Training Guidance. Part One: Physiology and Methods [31] | 2018 | Singh N | USA | Clinical review | Recently, the availability of commercially available heart rate (HR) monitoring systems has had important CV health implications and permits ambulatory CV monitoring on a scale not achievable with traditional cardiac diagnostics. | ✓ | |
| 19 | Heart Rate Variability: An Old Metric with New Meaning in the Era of Using mHealth technologies for Health and Exercise Training Guidance. Part Two: Prognosis and Training [32] | 2018 | Singh N | USA | Clinical review | Heart rate variability (HRV) is predictive of all-cause and cardiovascular mortality using clinical ECG recordings. the association between HRV and risk stratification is addressed by reviewing the current evidence from data acquired by resting ECG, exercise ECG and medical ambulatory devices. | ✓ | |
| 20 | Atrial fibrillation detection using single lead portable electrocardiographic monitoring: a systematic review and meta-analysis [26] | 2018 | Ramkumar S | Australia | Systematic review and meta-analysis | Single-lead portable ECG devices may offer an efficient screening option for AF compared with 24hours Holter monitoring. Total monitoring time is related to AF detection and a total of 19 min may achieve a similar detection rate to 24 h Holter monitoring. | ✓ | |
| 21 | The Emerging Role of Wearable Technologies in Detection of Arrhythmia [37] | 2018 | Cheung CC | Canada | Review | Future wearables will benefit from improved reliability and accuracy, collect additional health and fitness parameters, support management of chronic disease, and provide real-time connectivity and feedback that may supplant conventional medical monitoring. | ✓ | |
| 22 | Wearable Health Devices-Vital Sign Monitoring, Systems and Technologies [11] | 2018 | Dias D | Portugal | Narrative review | The technology revolution in the miniaturization of electronic devices is enabling to design more reliable and adaptable wearables, contributing for a world-wide change in the health monitoring approach. | ✓ | |
| 23 | Noncontact Wearable Wireless ECG Systems for Long-Term Monitoring [19] | 2018 | Majumder S | Canada | Review | A wireless ECG monitoring system is developed using flexible and dry capacitive electrodes for long-term monitoring of cardiovascular health. | ✓ | |
| 24 | Breathing Rate Estimation From the Electrocardiogram and Photoplethysmogram: A Review [34] | 2018 | Charlton PH | UK | Review | Breathing rate (BR) is a key physiological parameter used in a range of clinical settings. A plethora of algorithms have been proposed to estimate BR from the ECG and PPG signals. These BR algorithms provide opportunity for automated, electronic, and unobtrusive measurement of BR in both healthcare and fitness monitoring. | ✓ | |
| 25 | Can Wearable Devices Accurately Measure Heart Rate Variability? A systematic Review [21] | 2018 | Georgiou K | Greece | Systematic review | The correlation between classic ECG derived HRV and the wearable RV ranged from very good to excellent during rest, yet it declined progressively as exercise level increased. | ✓ | |
| 26 | Portable out-of-hospital electrocardiography: A review of current technologies [18] | 2018 | Bansal A | India | Narrative review | Number of first-generation devices using a single lead to record cardiac rhythm have been manufactured, tested and are approved by regulatory agencies. These devices are best suited for a short-term rhythm analysis. Second generation devices that can record multiple leads. | ✓ | |
| 27 | Smartphone Apps Using Photoplethysmography for Heart Rate Monitoring: Meta-Analysis [23] | 2018 | De Ridder B | Belgium | Systematic review and meta-analysis | Heart rate measured by smartphone apps performing PPG agrees with a validated method in an adult population in resting sinus rhythm, provided that during measurement the measuring point was kept still, and that appropriate pressure was maintained. In a pediatric population, the use of these apps can currently not be supported. | ✓ | |
| 28 | From Pacemaker to Wearable: Techniques for ECG Detection Systems [28] | 2018 | Kumar A | India | Review | Wearable ECG detectors can achieve sensitivity and specificities of around 99% without significant computational efforts, compared to implantable PMs. | ✓ | ✓ |
| 29 | Wearable Devices in Clinical Trials: Hype and Hypothesis [38] | 2018 | Izmailova ES | USA | Review | Wearable technologies are promising and have the potential to fundamentally change healthcare by changing the means of collecting, processing, and visualizing health data along with a reduction of healthcare costs. Remote data collection can bring new treatments and care management to all patients in need. | ✓ | |
| 30 | Smart health and innovation: facilitating health-related behaviour change [16] | 2017 | Redfern J | Australia | Narrative review | Smart health and innovation are evolving rapidly and may help with diagnosis, treatment, and management of chronic diseases including a focus on nutrition and its role in health. | ✓ | |
| 31 | ECG by mobile technologies [39] | 2016 | Guzik P | Poland | Narrative review | At present, the use of mobile ECG technology has not reached recognized guidelines and/or accepted clinical recommendations. The solid clinical evidence showing the real usefulness of new mobile ECG technologies should be available soon from large, randomized, multisite and prospective studies. | ✓ | |
| 32 | mHealth in Cardiovascular Health Care [15] | 2016 | Chow CK | Australia | Narrative review | mHealth has the potential to reduce socio economic disparity and alleviate the burden of CV disease. It includes simple strategies such the use of SMS for smoking-cessation, weight loss and diabetes management programmes. mHealth can also involve complex strategies such apps, GPS and Bluetooth technologies. | ✓ | |
| 33 | Use of smartphone technology in cardiology [35] | 2016 | Nguyen HH | USA | Review | The ever-broadening connectivity and increasing capabilities of smartphone-based technologies can better monitor, diagnose, and prevent cardiovascular diseases. Researchers can leverage the ubiquitous use of smartphone-based technologies and their constant stream of biometric data to establish large community based clinical research studies. | ✓ | |
| 34 | Electrocardiographic patch devices and contemporary wireless cardiac monitoring [20] | 2015 | Fung E | USA | Narrative review | Where continuous ECG monitoring in the short to medium term (days to weeks) is indicated, these cardiac devices and related digital mobile health technologies are reshaping the clinician-patient interface with important implications for future health care delivery. | ✓ | ✓ |
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fye, W.B. A History of the origin, evolution, and impact of electrocardiography. Am. J. Cardiol. 1994, 73, 937–949. [Google Scholar] [CrossRef]
- Henson, J.R. Descartes and the ECG lettering series. J. Hist. Med. Allied Sci. 1971, 26, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Pahlm, O.; Uvelius, B. The winner takes it all: Willem Einthoven, Thomas Lewis, and the Nobel prize 1924 for the discovery of the electrocardiogram. J. Electrocardiol. 2019, 57, 122–127. [Google Scholar] [CrossRef] [PubMed]
- AlGhatrif, M.; Lindsay, J. A brief review: History to understand fundamentals of electrocardiography. J. Community Hosp. Intern. Med. Perspect. 2012, 2, 529. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Ruiz, M.; Cajavilca, C.; Varon, J. Einthoven’s string galvanometer: The first electrocardiograph. Tex. Heart Inst. J. 2008, 35, 174–178. [Google Scholar] [PubMed]
- Yamagishi, T. A Short Biography of Takemi Taro, the President of the Japan Medical Association. J. Nanzan Acad. Soc. Soc. Sci. 2011, 1, 49–56. [Google Scholar]
- Yang, X.-L.; Liu, G.-Z.; Tong, Y.-H.; Yan, H.; Xu, Z.; Chen, Q.; Liu, X.; Zhang, H.-H.; Wang, H.-B.; Tan, S.-H. The history, hotspots, and trends of electrocardiogram. J. Geriatr. Cardiol. 2015, 12, 448–456. [Google Scholar] [CrossRef]
- Baule, G.M.; McFee, R. Detection of the magnetic field of the heart. Am. Heart J. 1963, 55, 95–96. [Google Scholar] [CrossRef]
- Miller, S.A.; Forrest, J.L. Enhancing your practice through evidence-based decision making: PICO, learning how to ask good questions. J. Evid. Based Dent. Pract. 2001, 1, 136–141. [Google Scholar] [CrossRef]
- Booth, A. Clear and present questions: Formulating questions for evidence-based practice. Libr. Hi. Tech. 2016, 24, 355–368. [Google Scholar] [CrossRef]
- Dias, D.; Paulo Silva Cunha, J. Wearable Health Devices - Vital Sign Monitoring, Systems and Technologies. Sensors 2018, 18, 2414. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Su, S.; Wu, N.; Wan, H.; Wan, S.; Bi, H.; Sun, L. Graphene-Based Sensors for Human Health Monitoring. Front. Chem. 2019, 7, 399. [Google Scholar] [CrossRef] [PubMed]
- Soroudi, A.; Hernández, N.; Berglin, L.; Nierstrasz, V. Electrode placement in electrocardiography smart garments: A review. J. Electrocardiol. 2019, 57, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Teferra, M.N.; Kourbelis, C.; Newman, P.; Ramos, J.S.; Hobbs, D.; Clark, R.A.; Reynolds, K.J. Electronic textile electrocardiogram monitoring in cardiac patients. JBI Database Syst. Rev. Implement. Rep. 2019, 17, 147–156. [Google Scholar] [CrossRef]
- Chow, C.K.; Ariyarathna, N.; Islam, S.M.; Thiagalingam, A.; Redfern, J. mHealth in Cardiovascular Health Care. Heart Lung Circ. 2016, 25, 802–807. [Google Scholar] [CrossRef]
- Redfern, J. Smart health and innovation: Facilitating health-related behavior change. Proc. Nutr. Soc. 2017, 76, 328–332. [Google Scholar] [CrossRef]
- Duarte, R.; Stainthorpe, A.; Mahon, J.; Greenhalgh, J.; Richardson, M.; Nevitt, S.; Kotas, E.; Boland, A.; Thom, H.; Marshall, T.; et al. Lead-I ECG for detecting atrial fibrillation in patients attending primary care with an irregular pulse using single-time point testing: A systematic review and economic evaluation. PLoS ONE 2019, 14, e0226671. [Google Scholar] [CrossRef]
- Bansal, A.; Joshi, R. Portable out-of-hospital electrocardiography: A review of current technologies. J. Arrhythm. 2018, 34, 129–138. [Google Scholar] [CrossRef]
- Majumder, S.; Chen, L.; Marinov, O.; Chen, C.H.; Mondal, T.; Deen, M.J. Noncontact Wearable Wireless ECG Systems for Long-Term Monitoring. IEEE Rev. Biomed. Eng. 2018, 11, 306–321. [Google Scholar] [CrossRef] [PubMed]
- Fung, E.; Jarvelin, M.; Doshi, R.N.; Shinbane, J.S.; Carlson, S.K.; Grazette, L.P.; Chang, P.M.; Sangha, R.S.; Huikuri, H.V.; Peters, N.S. Electrocardiographic patch devices and contemporary wireless cardiac monitoring. Front. Physiol. 2015, 6, 149. [Google Scholar] [CrossRef]
- Georgiou, K.; Larentzakis, A.V.; Khamis, N.N.; Alsuhaibani, G.I.; Alaska, Y.A.; Giallafos, E.J. Can Wearable Devices Accurately Measure Heart Rate Variability? A Systematic Review. Folia Med. (Plovdiv) 2018, 60, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Sajeev, J.K.; Koshy, A.N.; Teh, A.W. Wearable devices for cardiac arrhythmia detection: A new contender? Intern. Med. J. 2019, 49, 570–573. [Google Scholar] [CrossRef] [PubMed]
- De Ridder, B.; Van Rompaey, B.; Kampen, J.K.; Haine, S.; Dilles, T. Smartphone Apps Using Photoplethysmography for Heart Rate Monitoring: Meta-Analysis. JMIR Cardio. 2018, 2, e4. [Google Scholar] [CrossRef] [PubMed]
- Sanders, D.; Ungar, L.; Eskander, M.A.; Seto, A.H. Ambulatory ECG monitoring in the age of smartphones. Cleve Clin. J. Med. 2019, 86, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Raja, J.M.; Elsakr, C.; Roman, S.; Cave, B.; Pour-Ghaz, I.; Nanda, A.; Maturana, M.; Khouzam, R.N. Apple Watch, Wearables, and Heart Rhythm: Where do we stand? Ann. Transl. Med. 2019, 7, 417. [Google Scholar] [CrossRef]
- Ramkumar, S.; Nerlekar, N.; D’Souza, D.; Pol, D.J.; Kalman, J.M.; Marwick, T.H. Atrial fibrillation detection using single lead portable electrocardiographic monitoring: A systematic review and meta-analysis. BMJ Open 2018, 8, e024178. [Google Scholar] [CrossRef]
- Dobbs, W.C.; Fedewa, M.V.; Macdonald, H.V.; Holmes, C.J.; Cicone, Z.S.; Plews, D.J.; Esco, M.R. The Accuracy of Acquiring Heart Rate Variability from Portable Devices: A Systematic Review and Meta-Analysis. Sports Med. 2019, 49, 417–435. [Google Scholar] [CrossRef]
- Kumar, A.; Komaragiri, R.; Kumar, M. From Pacemaker to Wearable: Techniques for ECG Detection Systems. J. Med. Syst. 2018, 42, 34. [Google Scholar] [CrossRef]
- Karmen, C.L.; Reisfeld, M.A.; McIntyre, M.K.; Timmermans, R.; Frishman, W. The Clinical Value of Heart Rate Monitoring Using an Apple Watch. Cardiol. Rev. 2019, 27, 60–62. [Google Scholar] [CrossRef]
- Isakadze, N.; Martin, S.S. How useful is the smartwatch ECG? Trends Cardiovasc. Med. 2019. [Google Scholar] [CrossRef]
- Singh, N.; Moneghetti, K.J.; Christle, J.W.; Hadley, D.; Plews, D.; Froelicher, V. Heart Rate Variability: An Old Metric with New Meaning in the Era of using mHealth Technologies for Health and Exercise Training Guidance. Part One: Physiology and Methods. Arrhythm. Electrophysiol. Rev. 2018, 7, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Moneghetti, K.J.; Christle, J.W.; Hadley, D.; Froelicher, V.; Plews, D. Heart Rate Variability: An Old Metric with New Meaning in the Era of Using mHealth technologies for Health and Exercise Training Guidance. Part Two: Prognosis and Training. Arrhythm. Electrophysiol. Rev. 2018, 7, 247–255. [Google Scholar] [CrossRef]
- Elgendi, M.; Fletcher, R.; Liang, Y.; Howard, N.; Lovell, N.H.; Abbott, D.; Lim, K.; Ward, R. The use of photoplethysmography for assessing hypertension. NPJ Digit Med. 2019, 2, 60. [Google Scholar] [CrossRef]
- Charlton, P.H.; Birrenkott, D.A.; Bonnici, T.; Pimentel, M.A.; Johnson, A.E.W.; Alastruey, J.; Tarassenko, L.; Watkinson, P.; Beale, R.; Clifton, D. Breathing Rate Estimation From the Electrocardiogram and Photoplethysmogram: A Review. IEEE Rev. Biomed. Eng. 2018, 11, 2–20. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.H.; Silva, J.N. Use of smartphone technology in cardiology. Trends Cardiovasc. Med. 2016, 26, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Alharbi, M.; Straiton, N.; Smith, S.; Neubeck, L.; Gallagher, R. Data management and wearables in older adults: A systematic review. Maturitas 2019, 124, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Cheung, C.C.; Krahn, A.D.; Andrade, J.G. The Emerging Role of Wearable Technologies in Detection of Arrhythmia. Can. J. Cardiol. 2018, 34, 1083–1087. [Google Scholar] [CrossRef] [PubMed]
- Izmailova, E.S.; Wagner, J.A.; Perakslis, E.D. Wearable Devices in Clinical Trials: Hype and Hypothesis. Clin. Pharmacol. Ther. 2018, 104, 42–52. [Google Scholar] [CrossRef]
- Guzik, P.; Malik, M. ECG by mobile technologies. J. Electrocardiol. 2016, 49, 894–901. [Google Scholar] [CrossRef]
- Jo, A.; Coronel, B.D.; Coakes, C.E.; Mainous, A.G., 3rd. Is There a Benefit to Patients Using Wearable Devices Such as Fitbit or Health Apps on Mobiles? A Systematic Review. Am. J. Med. 2019, 132, 1394–1400.e1. [Google Scholar] [CrossRef]
- Al-Alusi, M.A.; Ding, E.; McManus, D.D.; Lubitz, S.A. Wearing Your Heart on Your Sleeve: The Future of Cardiac Rhythm Monitoring. Curr. Cardiol. Rep. 2019, 21, 158. [Google Scholar] [CrossRef] [PubMed]
- Solbiati, M.; Trombetta, L.; Sacco, R.; Erba, L.; Bozzano, V.; Costantino, G.; Raj, S.R.; Barbic, F.; Casazza, G.; DiPaola, F.; et al. A Systematic Review of Noninvasive Electrocardiogram Monitoring Devices for the Evaluation of Suspected Cardiovascular Syncope. J. Med. Devices. 2019, 13, 024001. [Google Scholar] [CrossRef]
- Jacobs, J.V.; Hettinger, L.J.; Huang, Y.H.; Jeffries, S.; Lesch, M.F.; Simmons, L.A.; Verma, S.K.; Willetts, J.L. Employee acceptance of wearable technology in the workplace. Appl. Ergon. 2019, 78, 148–156. [Google Scholar] [CrossRef]
- Maltseva, K. Wearables in the workplace: The brave new world of employee engagement. Bus. Horiz. 2020, 63, 493–505. [Google Scholar] [CrossRef]
- Li, C.K.; White, F.A.; Tipoe, T.; Liu, T.; Wong, M.C.; Jesuthasan, A.; Baranchuk, A.; Tse, G.; Yan, B.P.; Borges, L.; et al. The Current State of Mobile Phone Apps for Monitoring Heart Rate, Heart Rate Variability, and Atrial Fibrillation: Narrative Review. JMIR Mhealth Uhealth 2019, 7, e11606. [Google Scholar] [CrossRef]
- Marston, H.R.; Hadley, R.; Banks, D.; Duro, M.D.C.M. Mobile Self-Monitoring ECG Devices to Diagnose Arrhythmia that Coincide with Palpitations: A Scoping Review. Healthcare (Basel) 2019, 7, 96. [Google Scholar] [CrossRef]




© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baldassarre, A.; Mucci, N.; Padovan, M.; Pellitteri, A.; Viscera, S.; Lecca, L.I.; Galea, R.P.; Arcangeli, G. The Role of Electrocardiography in Occupational Medicine, from Einthoven’s Invention to the Digital Era of Wearable Devices. Int. J. Environ. Res. Public Health 2020, 17, 4975. https://doi.org/10.3390/ijerph17144975
Baldassarre A, Mucci N, Padovan M, Pellitteri A, Viscera S, Lecca LI, Galea RP, Arcangeli G. The Role of Electrocardiography in Occupational Medicine, from Einthoven’s Invention to the Digital Era of Wearable Devices. International Journal of Environmental Research and Public Health. 2020; 17(14):4975. https://doi.org/10.3390/ijerph17144975
Chicago/Turabian StyleBaldassarre, Antonio, Nicola Mucci, Martina Padovan, Alessia Pellitteri, Silvia Viscera, Luigi Isaia Lecca, Raymond P. Galea, and Giulio Arcangeli. 2020. "The Role of Electrocardiography in Occupational Medicine, from Einthoven’s Invention to the Digital Era of Wearable Devices" International Journal of Environmental Research and Public Health 17, no. 14: 4975. https://doi.org/10.3390/ijerph17144975
APA StyleBaldassarre, A., Mucci, N., Padovan, M., Pellitteri, A., Viscera, S., Lecca, L. I., Galea, R. P., & Arcangeli, G. (2020). The Role of Electrocardiography in Occupational Medicine, from Einthoven’s Invention to the Digital Era of Wearable Devices. International Journal of Environmental Research and Public Health, 17(14), 4975. https://doi.org/10.3390/ijerph17144975