Air Pollution Exposure Monitoring among Pregnant Women with and without Asthma
Abstract
1. Introduction
2. Methods
2.1. Study Sites
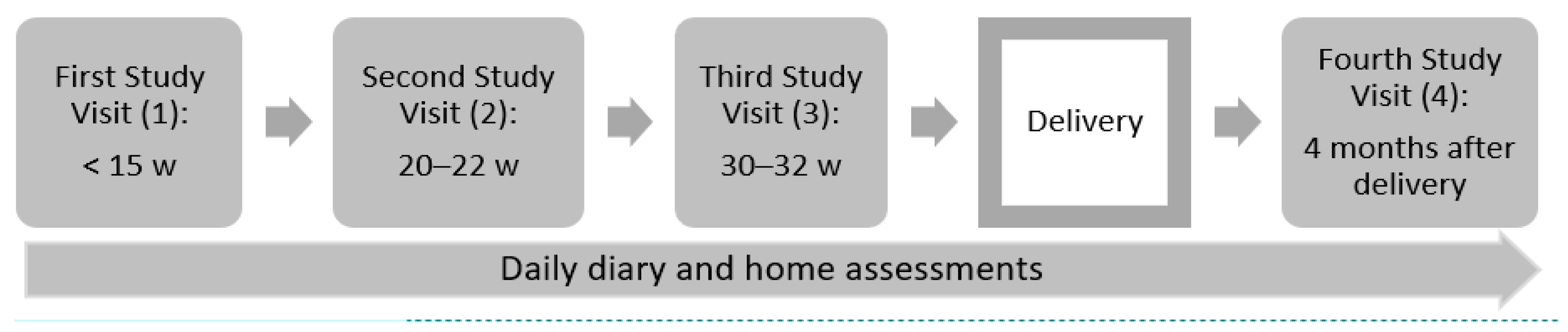
2.2. Data and Participants
2.3. Air Pollution Assessment
2.4. Daily Activities
2.5. Monitor Compliance Assessment
2.6. Predictors of Compliance
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kelly, W.; Massoumi, A.; Lazarus, A. Asthma in pregnancy: Physiology, diagnosis, and management. Postgrad Med. 2015, 127, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Gluck, J.C.; Gluck, P.A. The effect of pregnancy on the course of asthma. Immunol. Allergy Clin. North. Am. 2006, 26, 63–80. [Google Scholar] [CrossRef]
- Schatz, M.; Dombrowski, M.P.; Wise, R.; Thom, E.A.; Landon, M.; Mabie, W.; Newman, R.B.; Hauth, J.C.; Lindheimer, M.; Caritis, S.N.; et al. Asthma morbidity during pregnancy can be predicted by severity classification. J. Allergy Clin. Immunol. 2003, 112, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Guarnieri, M.; Balmes, J.R. Outdoor air pollution and asthma. Lancet 2014, 383, 1581–1592. [Google Scholar] [CrossRef]
- Orellano, P.; Quaranta, N.; Reynoso, J.; Balbi, B.; Vasquez, J. Effect of outdoor air pollution on asthma exacerbations in children and adults: Systematic review and multilevel meta-analysis. PLoS ONE 2017, 12, e0174050. [Google Scholar] [CrossRef]
- Mendola, P.; Wallace, M.; Liu, D.; Robledo, C.; Mnnist, T.; Grantz, K.L. Air pollution exposure and preeclampsia among US women with and without asthma. Environ. Res. 2016, 148, 248–255. [Google Scholar] [CrossRef]
- Mendola, P.; Wallace, M.; Hwang, B.S.; Liu, D.; Robledo, C.; Männistö, T.; Sundaram, R.; Sherman, S.; Ying, Q.; Grantz, K.L. Preterm birth and air pollution: Critical windows of exposure for women with asthma. J. Allergy Clin. Immunol. 2016, 138, 432–440. [Google Scholar] [CrossRef]
- Seeni, I.; Ha, S.; Nobles, C.; Liu, D.; Sherman, S.; Mendola, P. Air pollution exposure during pregnancy: Maternal asthma and neonatal respiratory outcomes. Ann. Epidemiol. 2018, 28, 612–618. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhang, C.; Liu, D.; Ha, S.; Kim, S.S.; Pollack, A.; Mendola, P. Ambient Air Pollution and Risk of Gestational Hypertension. Am. J. Epidemiol. 2017, 186, 334–343. [Google Scholar] [CrossRef]
- Klepac, P.; Locatelli, I.; Korosec, S.; Kunzli, N.; Kukec, A. Ambient air pollution and pregnancy outcomes: A comprehensive review and identification of environmental public health challenges. Environ. Res. 2018, 167, 144–159. [Google Scholar] [CrossRef]
- Mendola, P.; Ha, S.; Pollack, A.Z.; Zhu, Y.; Seeni, I.; Kim, S.S.; Sherman, S.; Liu, D. Chronic and Acute Ozone Exposure in the Week Prior to Delivery Is Associated with the Risk of Stillbirth. Int. J. Environ. Res. Public Health 2017, 14, 731. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Ha, S.; Roth, J.; Kearney, G.; Talbott, E.O.; Xu, X. Ambient Air Pollution and Hypertensive Disorders of Pregnancy: A Systematic Review and Meta-analysis. Atmos. Environ. (1994) 2014, 97, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, M.; Stayner, L.; Slama, R.; Sørensen, M.; Figueras, F.; Nieuwenhuijsen, M.J.; Raaschou-Nielsen, O.; Dadvand, P. Ambient air pollution and pregnancy-induced hypertensive disorders: A systematic review and meta-analysis. Hypertension 2014, 64, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.; Sundaram, R.; Buck Louis, G.M.; Nobles, C.; Seeni, I.; Sherman, S.; Mendola, P. Ambient air pollution and the risk of pregnancy loss: A prospective cohort study. Fertil. Steril. 2018, 109, 148–153. [Google Scholar] [CrossRef]
- Yuan, L.; Zhang, Y.; Gao, Y.; Tian, Y. Maternal fine particulate matter (PM2.5) exposure and adverse birth outcomes: An updated systematic review based on cohort studies. Environ. Sci. Pollut. Res. Int. 2019, 26, 13963–13983. [Google Scholar] [CrossRef]
- Dewulf, B.; Neutens, T.; Lefebvre, W.; Seynaeve, G.; Vanpoucke, C.; Beckx, C.; Van de Weghe, N. Dynamic assessment of exposure to air pollution using mobile phone data. Int. J. Health Geogr. 2016, 15, 14. [Google Scholar] [CrossRef]
- Klepeis, N.E.; Nelson, W.C.; Ott, W.R.; Robinson, J.P.; Tsang, A.M.; Switzer, P.; Behar, J.V.; Hern, S.C.; Engelmann, W.H. The National Human Activity Pattern Survey (NHAPS): A resource for assessing exposure to environmental pollutants. J. Expo. Anal. Environ. Epidemiol. 2001, 11, 231–252. [Google Scholar] [CrossRef] [PubMed]
- Bulletin, A.P. Asthma in Pregnancy. Clinical management guidelines for obstetrician-gynecologists. Obstet. Gynecol. 2008, 111, 457–464. [Google Scholar]
- RTI. Miniaturized, Wearable Device Gathers Real-Time Data to Support Environmental and Occupational Health Studies. 2020. Available online: https://www.rti.org/impact/micropem-sensor-measuring-exposure-air-pollution (accessed on 23 April 2020).
- Williams, R.; Kaufman, A.; Hanley, T.; Rice, J.; Garvey, S. Evaluation of Field-Deployed Low Cost PM Sensors; U.S. Environmental Protection Agency: Washington, DC, USA, 2014.
- Barzyk, T.; Huang, H.; Williams, R.; Kaufman, A.; Essoka, J. Advice and Frequently Asked Questions (FAQs) for Citizen-Science Environmental Health Assessments. Int. J. Environ. Res. Public Health 2018, 15, 960. [Google Scholar] [CrossRef]
- Wheeler, A.J.; Xu, X.; Kulka, R.; You, H.; Wallace, L.; Mallach, G.; Ryswyk, K.V.; MacNeill, M.; Kearney, J.; Rasmussen, P.E.; et al. Windsor, Ontario exposure assessment study: Design and methods validation of personal, indoor, and outdoor air pollution monitoring. J. Air Waste Manag. Assoc. 2011, 61, 142–156. [Google Scholar] [CrossRef]
- Cho, S.; Chartier, R.; Mortimer, K.; Dherani, M.; Tafatatha, T. A Personal Particulate Matter Exposure Monitor to Support Household Air Pollution Exposure and Health Studies. In Proceedings of the IEEE Global Humanitarian Technology Conference (GHTC), Seattle, WA, USA, 13–16 October 2016. [Google Scholar]
- EPA. NAAQS Table. 2016. Available online: https://www.epa.gov/criteria-air-pollutants/naaqs-table (accessed on 6 December 2019).
- Simons, E.; Dell, S.D.; Moineddin, R.; To, T. Neighborhood Material Deprivation Is Associated with Childhood Asthma Development: Analysis of Prospective Administrative Data. Can. Respir. J. 2019, 2019, 6808206. [Google Scholar] [CrossRef] [PubMed]
- Arnetz, B.B.; Arnetz, J.; Harkema, J.R.; Morishita, M.; Slonager, K.; Sudan, S.; Jamil, H. Neighborhood air pollution and household environmental health as it relates to respiratory health and healthcare utilization among elderly persons with asthma. J. Asthma 2019, 57, 1–12. [Google Scholar] [CrossRef]
- O’Lenick, C.R.; Winquist, A.; Mulholland, J.A.; Friberg, M.D.; Chang, H.H.; Kramer, M.R.; Darrow, L.A.; Sarnat, S.E. Assessment of neighbourhood-level socioeconomic status as a modifier of air pollution-asthma associations among children in Atlanta. J. Epidemiol. Community Health 2017, 71, 129–136. [Google Scholar] [CrossRef]
- Wilhelm, M.; Qian, L.; Ritz, B. Outdoor air pollution, family and neighborhood environment, and asthma in LA FANS children. Health Place 2009, 15, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Mohajerani, A.; Bakaric, J.; Jeffrey-Bailey, T. The urban heat island effect, its causes, and mitigation, with reference to the thermal properties of asphalt concrete. J. Environ. Manag. 2017, 197, 522–538. [Google Scholar] [CrossRef] [PubMed]
- Heaviside, C.; Macintyre, H.; Vardoulakis, S. The Urban Heat Island: Implications for Health in a Changing Environment. Curr. Environ. Health Rep. 2017, 4, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Murphy, V.E.; Jensen, M.E.; Gibson, P.G. Asthma during Pregnancy: Exacerbations, Management, and Health Outcomes for Mother and Infant. Semin. Respir. Crit. Care Med. 2017, 38, 160–173. [Google Scholar]
- Bozkurt, Z.; Dogan, G.; Arslanbas, D.; Pekey, B.; Pekey, H.; Dumanoğlu, Y.; Bayram, A.; Tuncel, G. Determination of the personal, indoor and outdoor exposure levels of inorganic gaseous pollutants in different microenvironments in an industrial city. Environ. Monit. Assess. 2015, 187, 590. [Google Scholar] [CrossRef]
- Weisel, C.P.; Zhang, J.; Turpin, B.J.; Morandi, M.T.; Colome, S.; Stock, T.H.; Spektor, D.M.; Korn, L.; Winer, A.M.; Kwon, J.; et al. Relationships of Indoor, Outdoor, and Personal Air (RIOPA). Part I. Collection methods and descriptive analyses. Res. Rep. Health Eff. Inst. 2005, 130 Pt 1, 1–107. [Google Scholar]
- Gold, D.R.; Allen, G.; Damokosh, A.; Serrano, P.; Hayes, C.; Castiilejos, M. Comparison of Outdoor and Classroom Ozone Exposures for School Children in Mexico City. J. Air Waste Manag. Assoc. 1996, 46, 335–342. [Google Scholar]
- Ouidir, M.; Giorgis-Allemand, L.; Lyon-Caen, S.; Morelli, X.; Cracowski, C.; Pontet, S.; Pin, I.; Lepeule, J.; Siroux, V.; Slama, R. Estimation of exposure to atmospheric pollutants during pregnancy integrating space-time activity and indoor air levels: Does it make a difference? Environ. Int. 2015, 84, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Hu, L.W.; Bai, Z. Human Exposure Assessment for Air Pollution. Adv. Exp. Med. Biol. 2017, 1017, 27–57. [Google Scholar] [PubMed]
- Lawless, P.; Thornburg, J.; Rodes, C.; Williams, R. Personal exposure monitoring wearing protocol compliance: An initial assessment of quantitative measurement. J. Expo. Sci. Environ. Epidemiol. 2012, 22, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Nethery, E.; Brauer, M.; Janssen, P. Time-activity patterns of pregnant women and changes during the course of pregnancy. J. Expo. Sci. Environ. Epidemiol. 2009, 19, 317–324. [Google Scholar] [CrossRef]
- Zielonka, T.; Zycinska, K. Are physicians specialists on the impact of air pollution on health? Eur. Respir. J. 2016, 48, PA4288. [Google Scholar]
- Mirabelli, M.C.; Damon, S.A.; Beavers, S.F.; Sircar, K.D. Patient-Provider Discussions About Strategies to Limit Air Pollution Exposures. Am. J. Prev. Med. 2018, 55, e49–e52. [Google Scholar] [CrossRef]
- Powell, P.; Brunekreef, B.; Grigg, J. How do you explain the risk of air pollution to your patients? Breathe (Sheff) 2016, 12, 201–203. [Google Scholar] [CrossRef][Green Version]
- Ramirez, A.S.; Ramondt, S.; Van Bogart, K.; Perez-Zuniga, R. Public Awareness of Air Pollution and Health Threats: Challenges and Opportunities for Communication Strategies to Improve Environmental Health Literacy. J. Health Commun. 2019, 24, 75–83. [Google Scholar] [CrossRef]
- Lovinsky-Desir, S.; Lawrence, J.; Jung, K.H.; Rundle, A.G.; Hoepner, L.A.; Yan, B.; Perera, F.; Perzanowski, M.S.; Miller, R.L.; Chillrud, S.N. Assessment of exposure to air pollution in children: Determining whether wearing a personal monitor affects physical activity. Environ. Res. 2018, 166, 340–343. [Google Scholar] [CrossRef]
| Characteristics | n (%) or Mean (SD) | ||||
|---|---|---|---|---|---|
| Overall (n = 40) | No Asthma (n = 9, 22.5%) | Well Controlled (n = 19, 47.5%) | Poorly Controlled (n = 12, 30.0%) | pa | |
| Study site (n, %) | |||||
| Site 1 | 36 (90.0) | 8 (88.9) | 18 (94.7) | 10 (83.3) | 0.67 |
| Site 2 | 4 (10.0) | 1 (11.1) | 1 (5.3) | 2 (16.7) | |
| Gestational age at enrollment (week, mean, SD) | 24.0 (4.6) | 27.1 (5.5) | 23.1 (4.5) | 23.1 (2.8) | 0.06 |
| Maternal age group (year, n, %) | |||||
| <23 | 1 (2.5) | 1 (11.1) | 0 (0) | 0 (0) | 0.42 |
| 23–29 | 10 (25.0) | 1 (11.1) | 5 (26.3) | 4 (33.3) | |
| ≥30 | 29 (72.5) | 7 (77.8) | 14 (73.7) | 8 (66.7) | |
| Maternal body mass index (kg/m2, n, %) | |||||
| Normal | 8 (20.0) | 1 (11.1) | 5 (26.3) | 2 (16.7) | 0.68 |
| Overweight/obese | 32 (80.0) | 8 (88.9) | 14 (73.7) | 10 (83.3) | |
| Monitor-wearing time (%, mean, range) | 38.4 (16.5–85.2) | 39.2 (16.6–69.2) | 40.4 (16.5–77.1) | 44.9 (24.3–85.2) | 0.73 |
| Waking-hour monitor-wearing time (%, mean, range) | 55.7(22.8–100.0) | 59.6(22.8–100) | 53.9(24–100) | 55.5(30.8–100) | 0.85 |
| GPS monitoring | |||||
| Yes | 24 (60.0) | 5 (55.6) | 13 (68.2) | 6 (50.0) | 0.64 |
| No | 16 (40.0) | 4 (44.4) | 6 (31.6) | 6 (50.0) | |
| Reported time spent outdoors per day (mins, mean, range) | 93.3 (0–480) | 94.2(0–360) | 98.6(0–480) | 83.1(0–300) | 0.67 |
| Average home distance from nearest EPA monitor (km, mean, range) | |||||
| PM2.5 | 6.6 (0.67–14.0) | 6.2(4.1–8.6) | 6.8(0.7–14) | 6.7(4.2–9.5) | 0.97 |
| Temperature | 23.5 (4.1–34.7) | 18.7(4.1–29.2) | 23.1(11.2–29.3) | 28.2(21.4–34.7) | 0.09 |
| Ozone | 8.5 (2.3–13.6) | 8.6(5.3–11.8) | 8.7(2.3–14.6) | 8.2(4.2–11.9) | 0.96 |
| NO2 | 6.3(0.75–14.0) | 6.6(0.8–13.4) | 7.1(1.0–14.0) | 4.3(1.8–8.3) | 0.38 |
| Average GPS distance from nearest EPA monitor (km, mean, range) | |||||
| PM2.5 | 6.9 (0.38–43.8) | 6.3(0.4–17.1) | 8.1(0.5–43.8) | 5.8(0.7–9.5) | <0.01 |
| Temperature | 25.6(0.38–63.5) | 19.1(0.4–63.5) | 24.7(2.1–30.9) | 30.8(12.6–42.3) | <0.01 |
| Ozone | 8.3 (0.38–25.2) | 8.1(0.4–25.2) | 9.3(0.8–25.1) | 7.2(1.4–13.9) | <0.01 |
| NO2 | 6.3(0.36–58.2) | 8.1(0.4–58.2) | 7(0.6–55.9) | 4.3(0.7–13.9) | <0.01 |
| Exhaled nitric oxide (ppb, mean, range) | 17.6 (5–52) | 21.5 (5–44) | 20 (7–48) | 15.1 (5–52) | 0.29 |
| Max peak flow morning (L/min, mean, range) | 377.3 (130–650) | 406.3 (150–650) | 418 (260–540) | 358 (160–550) | <0.01 |
| Max peak flow afternoon (L/min, mean, range) | 340.4 (100–540) | 384.8 (140–540) | 379.4 (210–500) | 323.2 (100–510) | <0.01 |
| Rate of events (per 100 person-day reported, mean, range) | |||||
| Wheeze | 7.3 (0–100) | 0 (0–0) | 3.1 (0–33.3) | 19.6 (0–100) | <0.01 |
| Cough | 27.2 (0–100) | 30.6 (0–100) | 27.9 (0–100) | 23.4 (0–75.0) | 0.10 |
| Shortness of breath | 31.5 (0–100) | 28.7 (0–100) | 26.9 (0–100) | 41.0 (0–100) | 0.20 |
| Chest tightness | 7.1 (0–100) | 4.0 (0–25.0) | 10.8 (0–100) | 3.7 (0–25) | 0.09 |
| Chest pain | 2 (0–25) | 5.0 (0–25.0) | 1.1 (0–20.0) | 1.2 (0–14.3) | 0.38 |
| Nausea | 17 (0–100) | 20.8 (0–75.0) | 12.8 (0–80.0) | 20.7 (0–100) | 0.65 |
| Runny nose | 47.5 (0–100) | 33.3 (0–100) | 56.1 (0–100) | 44.5 (0–100) | <0.01 |
| Missed work | 3.5 (0–60) | 2.8 (0–25.0) | 5.3 (0–60.0) | 1.2 (0–14.3) | 0.45 |
| Wake up at night | 14.8 (0–100) | 33.6 (0–100) | 4.2 (0–80.0) | 17.7 (0–100) | 0.07 |
| Exercise | 21.1 (0–100) | 12.9 (0–40.0) | 22.5 (0–100) | 25.1 (0–100) | 0.49 |
| Smoke | 2.5 (0–100) | 0 (0–0) | 0 (0–0) | 8.3 (0–100) | 0.31 |
| Around a smoker | 10 (0–100) | 16.1 (0–100) | 5.4 (0–66.7.0) | 12.8 (0–100) | 0.36 |
| Pollutant | Assessment Method a | Mean (Min–Max) | ||||
|---|---|---|---|---|---|---|
| All | No Asthma | Well Controlled | Poorly Controlled | p-Value b | ||
| PM2.5 (µg/m3) | Home + EPA monitor | 9.1 (1.7–28.5) | 9.7 (3.6–19) | 8.5 (1.7–16) | 9.2 (3.6–28.5) | 0.35 |
| GPS + EPA monitor | 9.3 (1.7–28.5) | 10.3 (3.6–28) | 8.4 (1.7–14.6) | 9.2 (3.6–28.5) | 0.11 | |
| Indoor | 14.2 (1–132.8) | 12.5 (2.3–76.5) | 15.8 (1–132.8) | 12.8 (2.5–85.8) | 0.69 | |
| Personal | 26.7 (0.9–665.6) | 15.1 (2.1–122.3) | 42.2 (0.9–665.6) | 12.1 (2.4–44.7) | 0.17 | |
| p-value exposure c | 0.001 | 0.42 | 0.01 | 0.22 | ||
| Ozone (ppb) | Home + EPA monitor | 28.9 (5.3–68.8) | 24.6 (5.3–65) | 28.4 (5.4–59.5) | 33.9 (6.3–68.8) | 0.01 |
| GPS + EPA monitor | 28.9 (5.3–68.8) | 24.6 (5.3–65) | 28.5 (5.4–59.5) | 33.9 (6.3–68.8) | 0.01 | |
| Indoor | 2.6 (1.1–23.9) | 2.2 (1.6–3) | 3.2 (1.1–23.9) | 2.1 (1.1–3.2) | 0.16 | |
| Personal | 3.1 (1.5–12.2) | 3.5 (1.9–7.6) | 2.7 (1.9–5.5) | 3.4 (1.5–12.2) | 0.13 | |
| p-value exposure c | <.0001 | <0.0001 | <0.0001 | <0.0001 | ||
| NO2 (ppb) | Home + EPA monitor | 16.3 (4–35.8) | 17.3 (6.9–31) | 15.7 (5.9–35.8) | 16.5 (4–35) | 0.42 |
| GPS + EPA monitor | 16.5 (4–35) | 18.3 (7.9–31.6) | 15.6 (5.5–34.7) | 16.4 (4–35) | 0.07 | |
| Indoor | 4.6 (0–39) | 3.5 (0–15.8) | 5.1 (0–27.9) | 4.7 (0–39) | 0.66 | |
| Personal | 5.1 (0–27.4) | 4.8 (0–17.4) | 5.6 (0–27.4) | 4.5 (0–23.1) | 0.67 | |
| p-value exposure c | <0.0001 | <0.0001 | <0.0001 | <0.001 | ||
| Temperature (°C) | Home + EPA monitor | 12.6 (−16.3–28.8) | 9.8 (−4.4–28.5) | 12.1 (−16.3–28.8) | 15.9 (−4.8–27) | 0.01 |
| GPS + EPA monitor | 12.6 (−16.3–28.8) | 9.7 (−4.4–27.8) | 12 (−16.3–28.8) | 15.9 (−4.8–27) | 0.01 | |
| Indoor | 24.3 (17.1–52.8) | 25 (19.5–31.7) | 24.4 (17.1–52.8) | 23.7 (18.1–31) | 0.38 | |
| Personal | 23.6 (13.1–38.7) | 24.5 (13.1–38.7) | 23.6 (16.8–33.7) | 23 (16–30.3) | 0.36 | |
| p-value exposure c | <0.0001 | <0.0001 | <0.001 | <0.001 | ||
| Activities (Yes vs. No) | PM2.5 (μg/m3) | NO2 (ppb) | Ozone (ppb) | |||
|---|---|---|---|---|---|---|
| Personal | Indoors | Personal | Indoors | Personal | Indoors | |
| Near heater | −43.0 (−88.7, 187.6) | - | 170.9 (−91.0, 8040.1) | - | 20.7 (−33.6, 119.2) | - |
| Near burning trash | 28.8 (−69.0, 435.1) | 68.3 (−54.7, 524.8) | 28.8 (−91.4, 1831.2) | 276.6 (−91.2, 16,070.4) | −8.2 (−45.7, 55.1) | 11.7 (−40.5, 109.8) |
| Used gas appliance | 46.4 (−21.3, 172.5) | 194.6 (55.0, 459.6) * | 33.2 (−53.5, 281.4) | 92.4 (−72.9, 1268.3) | −17.1 (−34, 4.0) | −3.1 (−35.5, 45.7) |
| Used clothes dryer | 11.6 (−48.1, 140.0) | 126.1 (−20.3, 541.4) | 58.2 (−70.6, 752.3) | 31.6 (−92.3, 2133.9) | 0.7 (−24.5, 34.3) | 10.9 (−34.8, 88.5) |
| Near water heater | −11.3 (−99.4, 12,893.3) | 68.4 (−61.5, 636.1) | 52.1 (−99.9, 209,422.1) | 549.8 (−60.6, 10,626.8) | 51.4 (−50.3, 361.0) | 40.8 (−25.5, 166.2) |
| Used printer | 4.0 (−46.3, 101.5) | - | −26.9 (−77.1, 133.7) | - | −19.4 (−36.5, 2.3) | - |
| Smoking | 88.2 (−83.5, 2048) | 398.7 (78.5, 1293.8) * | 66.9 (−95.8, 6499.9) | 1008.9 (−67.5, 37,779.1) | 47.4 (−5.5, 130.0) | 12.3 (−40.2, 110.8) |
| Near smoker | 29.9 (−87.8, 1283.9) | 3.1 (−67.7, 229.1) | −78.0 (−99.5, 844.9) | 18.5 (−99.3, 20,321.0) | 54.1 (−15.4, 180.7) | −0.2 (−33.7, 50.1) |
| Exercise | −1.6 (−47.1, 83.2) | 36.3 (−25.8, 150.3) | −0.6 (−56.2, 125.5) | −19.2 (−78.5, 203.1) | 1.8 (−13.8, 20.1) | 8.7 (−12.2, 34.6) |
| Characteristics | β (95% CI) | pa |
|---|---|---|
| Asthma status | ||
| No asthma | reference | |
| Well controlled | −4.23 (−23.1, 14.64) | 0.66 |
| Poorly controlled | −4.68 (−26.15, 16.78) | 0.67 |
| Site | ||
| Site 1 | reference | |
| Site 2 | 11.73 (−8.08, 31.54) | 0.25 |
| Maternal age group (years) | ||
| <23 | reference | |
| 23–29 | 7.60 (−7.93, 23.12) | 0.34 |
| ≥30 | 7.48 (−1.42, 16.38) | 0.10 |
| Maternal body mass index | ||
| Normal | reference | |
| Overweight/obese | −9.6 (−26.77, 7.57) | 0.27 |
| Exhaled nitric oxide (ppb) | 0.1 (−0.48, 0.68) | 0.74 |
| Max morning peak flow (L/min) | −0.01 (−0.06, 0.04) | 0.66 |
| Max afternoon peak flow (L/min) | −0.02 (−0.07, 0.03) | 0.45 |
| Symptoms (yes vs. no) | ||
| Wheeze | −5.66 (−14.94, 3.62) | 0.23 |
| Cough | 0.79 (−13.34, 14.93) | 0.91 |
| Shortness of breath | −2.99 (−14.16, 8.18) | 0.60 |
| Chest tightness | 9.88 (−10.92, 30.67) | 0.35 |
| Chest pain | 9.29 (−6.76, 25.34) | 0.26 |
| Nausea | 13.27 (−2, 28.54) | 0.09 |
| Runny nose | 2.06 (−9.57, 13.7) | 0.73 |
| Missed work | 19.81 (0.91, 38.72) | 0.04 |
| Wake up at night | 3.17 (−15.87, 22.2) | 0.74 |
| Exercise | −10.64 (−18.66, −2.61) | 0.01 |
| Smoke | −22.83 (−29.94, −15.71) | <0.001 |
| Around a smoker | −16.24 (−26.43, −6.04) | <0.001 |
| Used prescription drug | −13.39 (−26.42, −0.37) | 0.04 |
| Used over the counter drug | −10.37 (−26.39, 5.66) | 0.20 |
| Near heater | 42.05 (33.8, 50.31) | <0.001 |
| Near burning trash | −7.33 (−22.81, 8.16) | 0.35 |
| Used gas appliance | −8.74 (−22.99, 5.5) | 0.23 |
| Used clothes dryer | −6.29 (−20.58, 8) | 0.39 |
| Near water heater | 15.41 (−6.13, 36.95) | 0.16 |
| Used printer | −1.11 (−15.94, 13.72) | 0.88 |
| Times spent outdoors (minutes) | 0.03 (−0.04, 0.1) | 0.38 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ha, S.; Nobles, C.; Kanner, J.; Sherman, S.; Cho, S.-H.; Perkins, N.; Williams, A.; Grobman, W.; Biggio, J.; Subramaniam, A.; et al. Air Pollution Exposure Monitoring among Pregnant Women with and without Asthma. Int. J. Environ. Res. Public Health 2020, 17, 4888. https://doi.org/10.3390/ijerph17134888
Ha S, Nobles C, Kanner J, Sherman S, Cho S-H, Perkins N, Williams A, Grobman W, Biggio J, Subramaniam A, et al. Air Pollution Exposure Monitoring among Pregnant Women with and without Asthma. International Journal of Environmental Research and Public Health. 2020; 17(13):4888. https://doi.org/10.3390/ijerph17134888
Chicago/Turabian StyleHa, Sandie, Carrie Nobles, Jenna Kanner, Seth Sherman, Seung-Hyun Cho, Neil Perkins, Andrew Williams, William Grobman, Joseph Biggio, Akila Subramaniam, and et al. 2020. "Air Pollution Exposure Monitoring among Pregnant Women with and without Asthma" International Journal of Environmental Research and Public Health 17, no. 13: 4888. https://doi.org/10.3390/ijerph17134888
APA StyleHa, S., Nobles, C., Kanner, J., Sherman, S., Cho, S.-H., Perkins, N., Williams, A., Grobman, W., Biggio, J., Subramaniam, A., Ouidir, M., Chen, Z., & Mendola, P. (2020). Air Pollution Exposure Monitoring among Pregnant Women with and without Asthma. International Journal of Environmental Research and Public Health, 17(13), 4888. https://doi.org/10.3390/ijerph17134888







