Effects of Surfactants on the Degradation of Diclofenac by Manganese Oxide
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Batch Experiments
2.3. Sample Preparation and Analysis
2.4. Identification of Oxidation Products
3. Results and Discussion
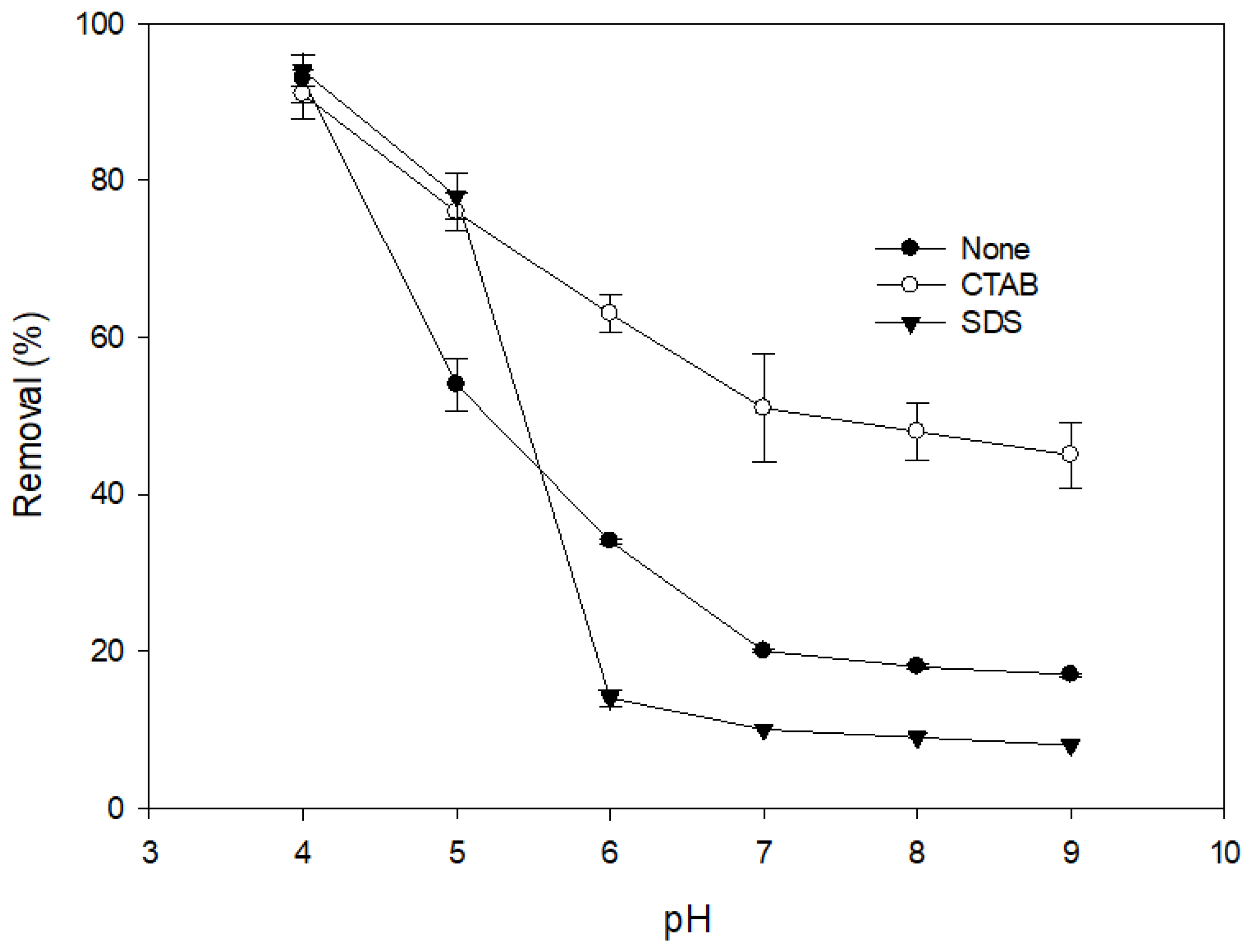
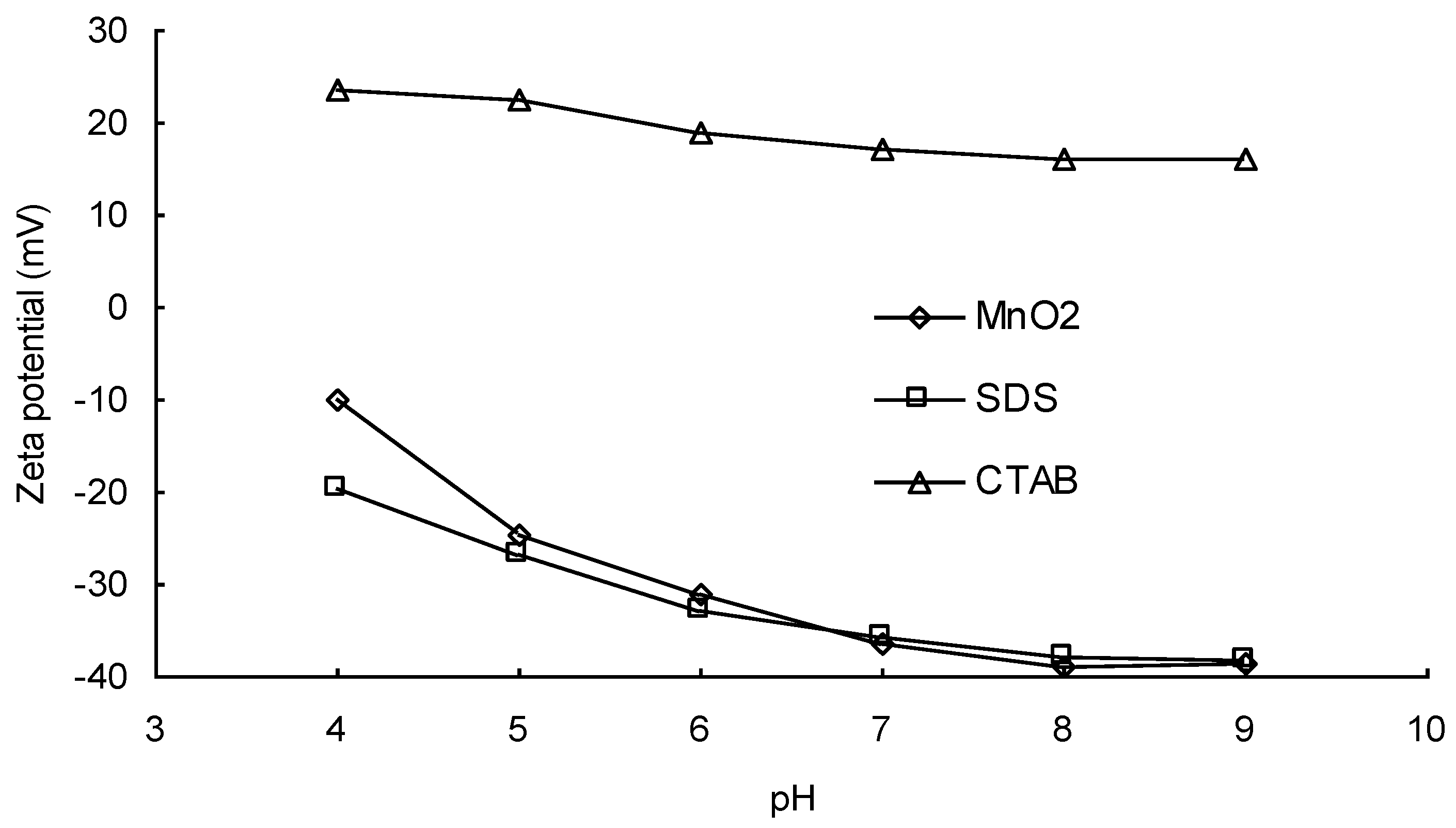
3.1. Effects of the Types of Surfactants on DIC Oxidation by MnO2
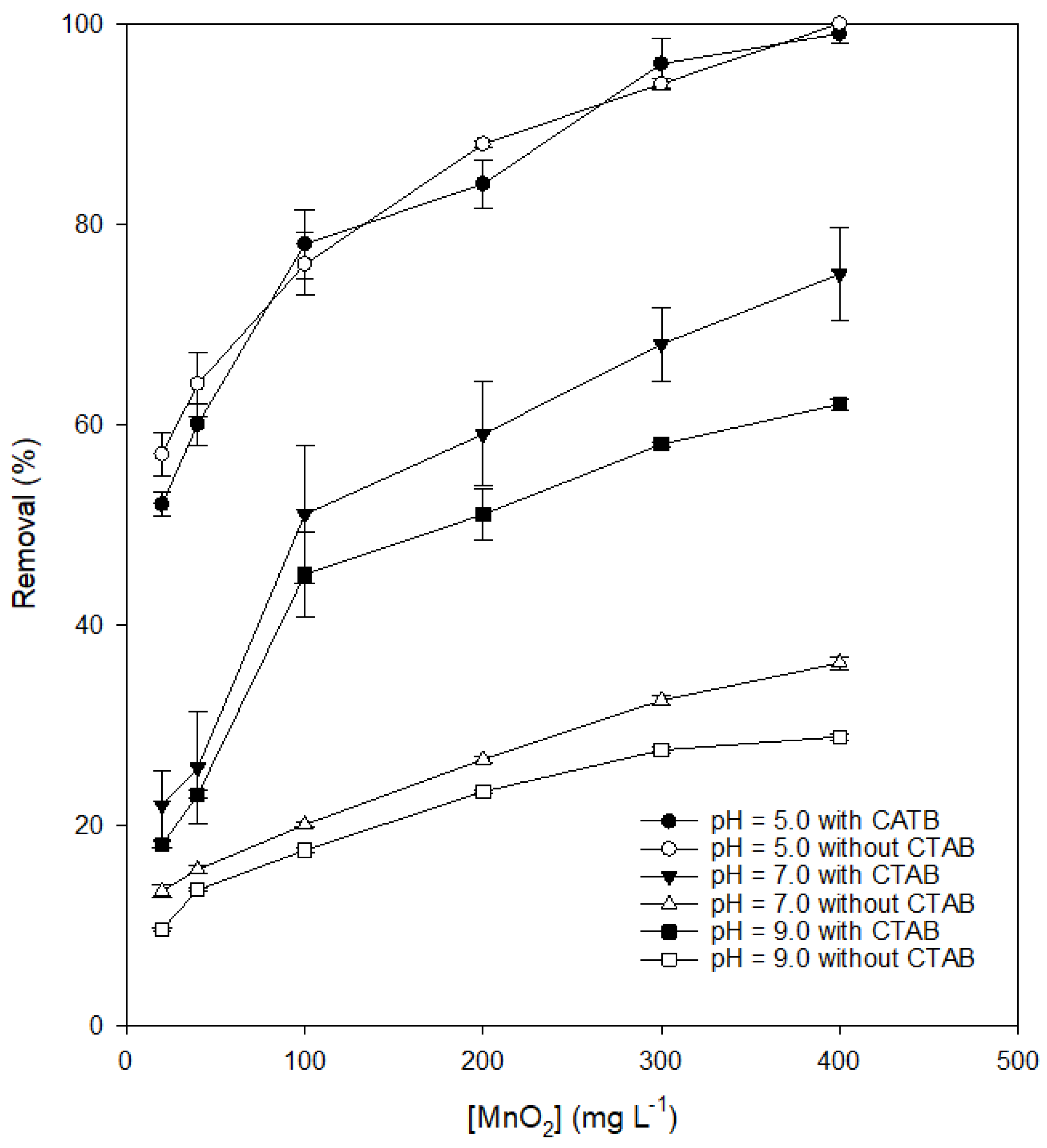
3.2. Effects of the Dosages of MnO2 and CTAB
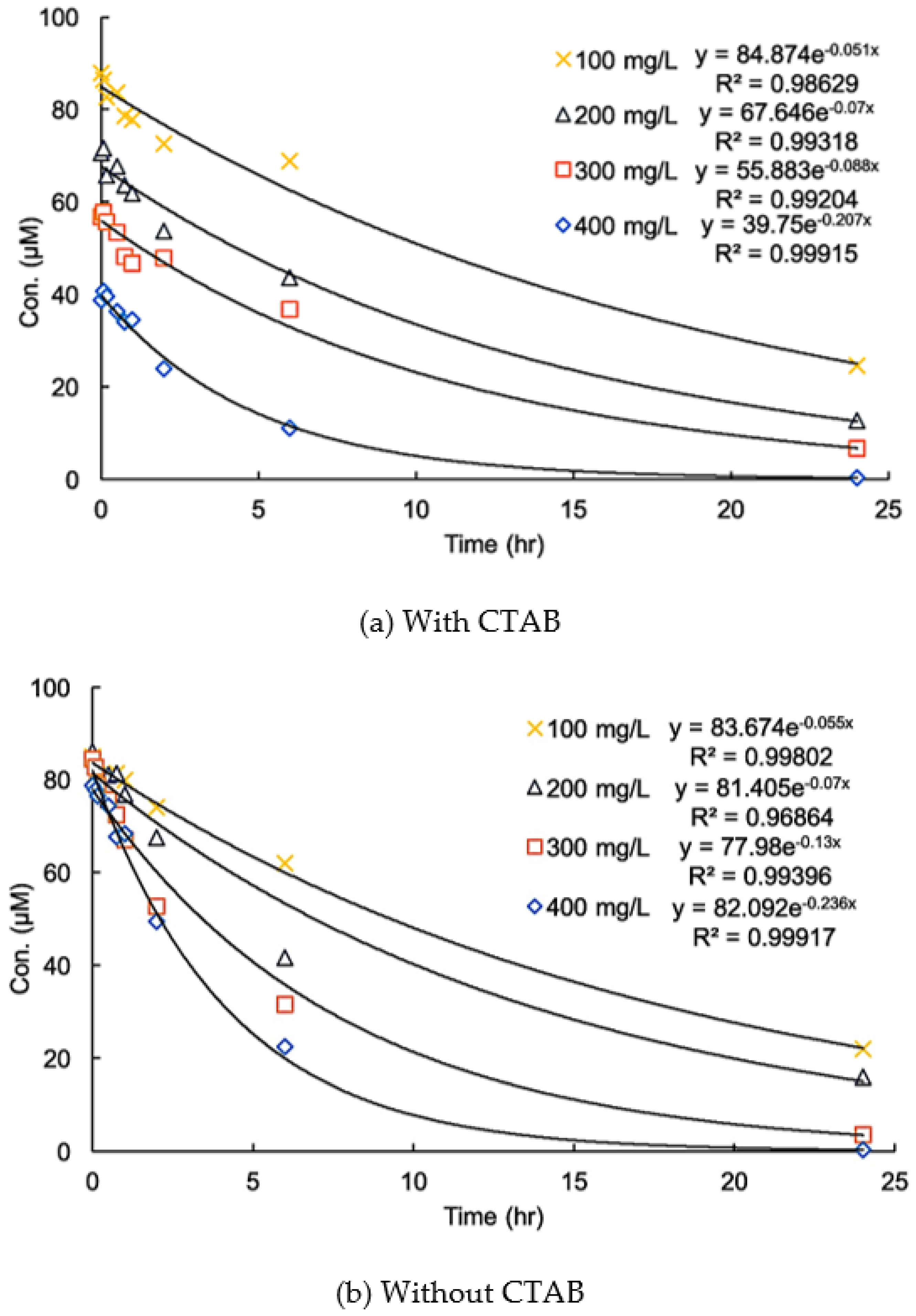
3.3. Effects of CTAB on the Kinetics of Degradation
3.4. Effects of CTAB on the Products
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ternes, T.A.; Herrmann, N.; Bonerz, M.; Knacker, T.; Siegrist, H.; Joss, A. A rapid method to measure the solid-water distribution coefficient (K-d) for pharmaceuticals and musk fragrances in sewage sludge. Water Res. 2004, 38, 4075–4084. [Google Scholar] [CrossRef]
- Joss, A.; Zabczynski, S.; Gobel, A.; Hoffmann, B.; Loffler, D.; McArdell, C.S.; Ternes, T.A.; Thomsen, A.; Siegrist, H. Biological degradation of pharmaceuticals in municipal wastewater treatment: Proposing a classification scheme. Water Res. 2006, 40, 1686–1696. [Google Scholar] [CrossRef] [PubMed]
- Camacho-Munoz, D.; Martin, J.; Santos, J.L.; Aparicio, I.; Alonso, E. Occurrence, temporal evolution and risk assessment of pharmaceutically active compounds in Donana Park (Spain). J. Hazard. Mater. 2010, 183, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Comeau, F.; Surette, C.; Brun, G.L.; Losier, R. The occurrence of acidic drugs and caffeine in sewage effluents and receiving waters from three coastal watersheds in Atlantic Canada. Sci. Total Environ. 2008, 396, 132–146. [Google Scholar] [CrossRef] [PubMed]
- Schwab, B.W.; Hayes, E.P.; Fiori, J.M.; Mastrocco, F.J.; Roden, N.M.; Cragin, D.; Meyerhoff, R.D.; D’Aco, V.J.; Anderson, P.D. Human pharmaceuticals in US surface waters: A human health risk assessment. Regul. Toxicol. Pharmacol. 2005, 42, 296–312. [Google Scholar] [CrossRef] [PubMed]
- Sidlova, P.; Podlipna, R.; Vanek, T. Cytotoxic Pharmaceuticals in the Environment. Chem. Listy 2011, 105, 8–14. [Google Scholar]
- Tauxe-Wuersch, A.; De Alencastro, L.F.; Grandjean, D.; Tarradellas, J. Occurrence of several acidic drugs in sewage treatment plants in Switzerland and risk assessment. Water Res. 2005, 39, 1761–1772. [Google Scholar] [CrossRef]
- Weber, S.; Khan, S.; Hollender, J. Human risk assessment of organic contaminants in reclaimed wastewater used for irrigation. Desalination 2006, 187, 53–64. [Google Scholar] [CrossRef]
- Triebskorn, R.; Casper, H.; Heyd, A.; Eikemper, R.; Kohler, H.R.; Schwaiger, J. Toxic effects of the non-steroidal anti-inflammatory drug diclofenac Part II. Cytological effects in liver, kidney, gills and intestine of rainbow trout (Oncorhynchus mykiss). Aquat. Toxicol. 2004, 68, 151–166. [Google Scholar] [CrossRef]
- Schwaiger, J.; Ferling, H.; Mallow, U.; Wintermayr, H.; Negele, R.D. Toxic effects of the non-steroidal anti-inflammatory drug diclofenac Part 1: Histopathological alterations and bioaccumulation in rainbow trout. Aquat. Toxicol. 2004, 68, 141–150. [Google Scholar] [CrossRef]
- Stone, A.T. Reductive Dissolution of Manganese(III/Iv) Oxides by Substituted Phenols. Environ. Sci. Technol. 1987, 21, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.C.; Huang, C.H. Oxidative transformation of triclosan and chlorophene by manganese oxides. Environ. Sci. Technol. 2003, 37, 2421–2430. [Google Scholar] [CrossRef]
- Park, J.W.; Dec, J.; Kim, J.E.; Bollag, J.M. Effect of humic constituents on the transformation of chlorinated phenols and anilines in the presence of oxidoreductive enzymes or birnessite. Environ. Sci. Technol. 1999, 33, 2028–2034. [Google Scholar] [CrossRef]
- Ulrich, H.J.; Stone, A.T. The oxidation of chlorophenols adsorbed to manganese oxide surfaces. Environ. Sci. Technol. 1989, 23, 421–428. [Google Scholar] [CrossRef]
- Pizzigallo, M.D.R.; Ruggiero, P.; Crecchio, C.; Mascolo, G. Oxidation of chloroanilines at metal oxide surfaces. J. Agric. Food Chem. 1998, 46, 2049–2054. [Google Scholar] [CrossRef]
- Li, H.; Lee, L.S.; Jafvert, C.T.; Graveel, J.G. Effect of substitution on irreversible binding and transformation of aromatic amines with soils in aqueous systems. Environ. Sci. Technol. 2000, 34, 3674–3680. [Google Scholar] [CrossRef]
- Lin, K.; Liu, W.; Gan, J. Oxidative removal of bisphenol A by manganese dioxide: Efficacy, products, and pathways. Environ. Sci. Technol. 2009, 43, 3860–3864. [Google Scholar] [CrossRef]
- Lu, Z.J.; Lin, K.D.; Gan, J. Oxidation of bisphenol F (BPF) by manganese dioxide. Environ. Pollut. 2011, 159, 2546–2551. [Google Scholar] [CrossRef]
- Sarmah, P.; Dutta, D.K. Manganese mediated aqueous reduction of aromatic nitro compounds to amines. J. Chem. Res. 2003, 2003, 236–237. [Google Scholar] [CrossRef]
- Rubert, K.F.; Pedersen, J.A. Kinetics of oxytetracycline reaction with a hydrous manganese oxide. Environ. Sci. Technol. 2006, 40, 7216–7221. [Google Scholar] [CrossRef]
- Sabirova, J.S.; Cloetens, L.F.F.; Vanhaecke, L.; Forrez, I.; Verstraete, W.; Boon, N. Manganese-oxidizing bacteria mediate the degradation of 17α-ethinylestradiol. Microb. Biotechnol. 2008, 1, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Xu, C.; Zhao, M.; Qiu, Y.; Sheng, G.D. Oxidative removal of aqueous steroid estrogens by manganese oxides. Water Res. 2008, 42, 5038–5044. [Google Scholar] [CrossRef] [PubMed]
- Forrez, I.; Carballa, M.; Verbeken, K.; Vanhaecke, L.; Schlusener, M.; Ternes, T.; Boon, N.; Verstraete, W. Diclofenac oxidation by biogenic manganese oxides. Environ. Sci. Technol. 2010, 44, 3449–3454. [Google Scholar] [CrossRef] [PubMed]
- Huguet, M.; Deborde, M.; Papot, S.; Gallard, H. Oxidative decarboxylation of diclofenac by manganese oxide bed filter. Water Res. 2013, 47, 5400–5408. [Google Scholar] [CrossRef]
- Ying, G.G. Fate, behavior and effects of surfactants and their degradation products in the environment. Environ. Int. 2006, 32, 417–431. [Google Scholar] [CrossRef]
- Huntsman, B.E.; Staples, C.A.; Naylor, C.G.; Williams, J.B. Treatability of nonylphenol ethoxylate surfactants in on-site wastewater disposal systems. Water Environ. Res. 2006, 78, 2397–2404. [Google Scholar] [CrossRef]
- Hari, A.C.; Paruchuri, R.A.; Sabatini, D.A.; Kibbey, T.C.G. Effects of pH and cationic and nonionic surfactants on the adsorption of pharmaceuticals to a natural aquifer material. Environ. Sci. Technol. 2005, 39, 2592–2598. [Google Scholar] [CrossRef]
- Gordon, A.K.; Muller, W.J.; Gysman, N.; Marshall, S.J.; Sparham, C.J.; O’Connor, S.M.; Whelan, M.J. Effect of laundry activities on in-stream concentrations of linear alkylbenzene sulfonate in a small rural South African river. Sci. Total Environ. 2009, 407, 4465–4471. [Google Scholar] [CrossRef]
- Jones-Hughes, T.; Turner, A. Sorption of ionic surfactants to estuarine sediment and their influence on the sequestration of phenanthrene. Environ. Sci. Technol. 2005, 39, 1688–1697. [Google Scholar] [CrossRef]
- Oleszczuk, P.; Xing, B.S. Influence of anionic, cationic and nonionic surfactants on adsorption and desorption of oxytetracycline by ultrasonically treated and non-treated multiwalled carbon nanotubes. Chemosphere 2011, 85, 1312–1317. [Google Scholar] [CrossRef]
- Pan, G.; Jia, C.X.; Zhao, D.Y.; You, C.; Chen, H.; Jiang, G.B. Effect of cationic and anionic surfactants on the sorption and desorption of perfluorooctane sulfonate (PFOS) on natural sediments. Environ. Pollut. 2009, 157, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Keller, A.A. Adsorption of hydrophobic organic compounds onto a hydrophobic carbonaceous geosorbent in the presence of surfactants. Environ. Toxicol. Chem. 2008, 27, 1237–1243. [Google Scholar] [CrossRef]
- Bui, T.X.; Choi, H. Influence of ionic strength, anions, cations, and natural organic matter on the adsorption of pharmaceuticals to silica. Chemosphere 2010, 80, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Scamehorn, J.F.; Schechter, R.S.; Wade, W.H. Adsorption of surfactants on mineral oxide surfaces from aqueous solutions: I: Isomerically pure anionic surfactants. J. Colloid Interface Sci. 1982, 85, 463–478. [Google Scholar] [CrossRef]
- Monteagudo, J.M.; El-taliawy, H.; Durán, A.; Caro, G.; Bester, K. Sono-activated persulfate oxidation of diclofenac: Degradation, kinetics, pathway and contribution of the different radicals involved. J. Hazard. Mater. 2018, 357, 457–465. [Google Scholar] [CrossRef]
- Yu, H.; Nie, E.; Xu, J.; Yan, S.; Cooper, W.J.; Song, W. Degradation of diclofenac by advanced oxidation and reduction processes: Kinetic studies, degradation pathways and toxicity assessments. Water Res. 2013, 47, 1909–1918. [Google Scholar] [CrossRef]
- Huguet, M.; Simon, V.; Gallard, H. Transformation of paracetamol into 1,4-benzoquinone by a manganese oxide bed filter. J. Hazard. Mater. 2014, 271, 245–251. [Google Scholar] [CrossRef]
- Li, H.; Lee, L.S.; Schulze, D.G.; Guest, C.A. Role of soil manganese in the oxidation of aromatic amines. Environ. Sci. Technol. 2003, 37, 2686–2693. [Google Scholar] [CrossRef]
- Xiao, H.; Song, H.; Xie, H.; Huang, W.; Tan, J.; Wu, J. Transformation of acetaminophen using manganese dioxide − mediated oxidative processes: Reaction rates and pathways. J. Hazard. Mater. 2013, 250–251, 138–146. [Google Scholar] [CrossRef]
- Liu, Q.; Luo, X.; Zheng, Z.; Zheng, B.; Zhang, J.; Zhao, Y.; Yang, X.; Wang, J.; Wang, L. Factors that have an effect on degradation of diclofenac in aqueous solution by gamma ray irradiation. Environ. Sci. Pollut. Res. 2011, 18, 1243–1252. [Google Scholar] [CrossRef]
- Martínez, C.; Canle L, M.; Fernández, M.I.; Santaballa, J.A.; Faria, J. Aqueous degradation of diclofenac by heterogeneous photocatalysis using nanostructured materials. Appl. Catal. B Environ. 2011, 107, 110–118. [Google Scholar] [CrossRef]
- Maximiano, F.A.; Chaimovich, H.; Cuccovia, I.M. Decarboxylation of 6-nitrobenzisoxazole-3-carboxylate in mixed micelles of zwitterionic and positively charged surfactants. Langmuir 2006, 22, 8050–8055. [Google Scholar] [CrossRef] [PubMed]

| Compound | RT (min) | Mode | m/z | Possible Structure | Reference |
|---|---|---|---|---|---|
| DIC | 3.18 | ESI+ | 296/298 (3/2) 278/280, 250/252, 215/217 | ||
| P1(A) | 2.09 | ESI+ | 346/348 (3/2) 328/330, 284/286, 244/246, 162/164 | Tri- or dihydroxyl DIC | (Yu et al., 2013) [36] |
| U1(A) | 2.48 | ESI+ ESI− | 298/300 (3/2) 267, 244 266/264 (3/2) 228/230 |  | |
| U2(A) | 2.94 | ESI+ | 597/599 (3/4) 575/577 (3/4), 551/553 (3/4), 507/509 (3/4), 308, 267, 255 | DCF+m/z 308 dimer or DCF+5OH-DCF dimer | |
| P2(A) | 3.05 | ESI+ ESI− | 308, 267 283 | 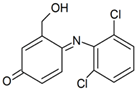 |
| Product | RT (min) | Mode | m/z | Possible Structure | Reference |
|---|---|---|---|---|---|
| DIC | 3.18 | ESI+ | 296/298 (3/2) 278/280, 250/252, 215/217 | ||
| P1(B) | 3.66 | ESI+ | 284 | Decarboxy-DIC | |
| P2(B) | 2.34 | ESI− | 266 228 | 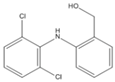 | [40,41] |
| P3(B) | 2.83 | ESI− | 250/252 (3/2) | 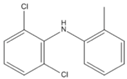 | (Martínez et al., 2011) [41] |
| or | |||||
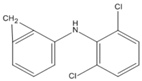 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuan, W.-H.; Liu, Y.-J.; Hu, C.-Y. Effects of Surfactants on the Degradation of Diclofenac by Manganese Oxide. Int. J. Environ. Res. Public Health 2020, 17, 4513. https://doi.org/10.3390/ijerph17124513
Kuan W-H, Liu Y-J, Hu C-Y. Effects of Surfactants on the Degradation of Diclofenac by Manganese Oxide. International Journal of Environmental Research and Public Health. 2020; 17(12):4513. https://doi.org/10.3390/ijerph17124513
Chicago/Turabian StyleKuan, Wen-Hui, Yu-Jung Liu, and Ching-Yao Hu. 2020. "Effects of Surfactants on the Degradation of Diclofenac by Manganese Oxide" International Journal of Environmental Research and Public Health 17, no. 12: 4513. https://doi.org/10.3390/ijerph17124513
APA StyleKuan, W.-H., Liu, Y.-J., & Hu, C.-Y. (2020). Effects of Surfactants on the Degradation of Diclofenac by Manganese Oxide. International Journal of Environmental Research and Public Health, 17(12), 4513. https://doi.org/10.3390/ijerph17124513







