Bioplastic Production from Microalgae: A Review
Abstract
1. Introduction
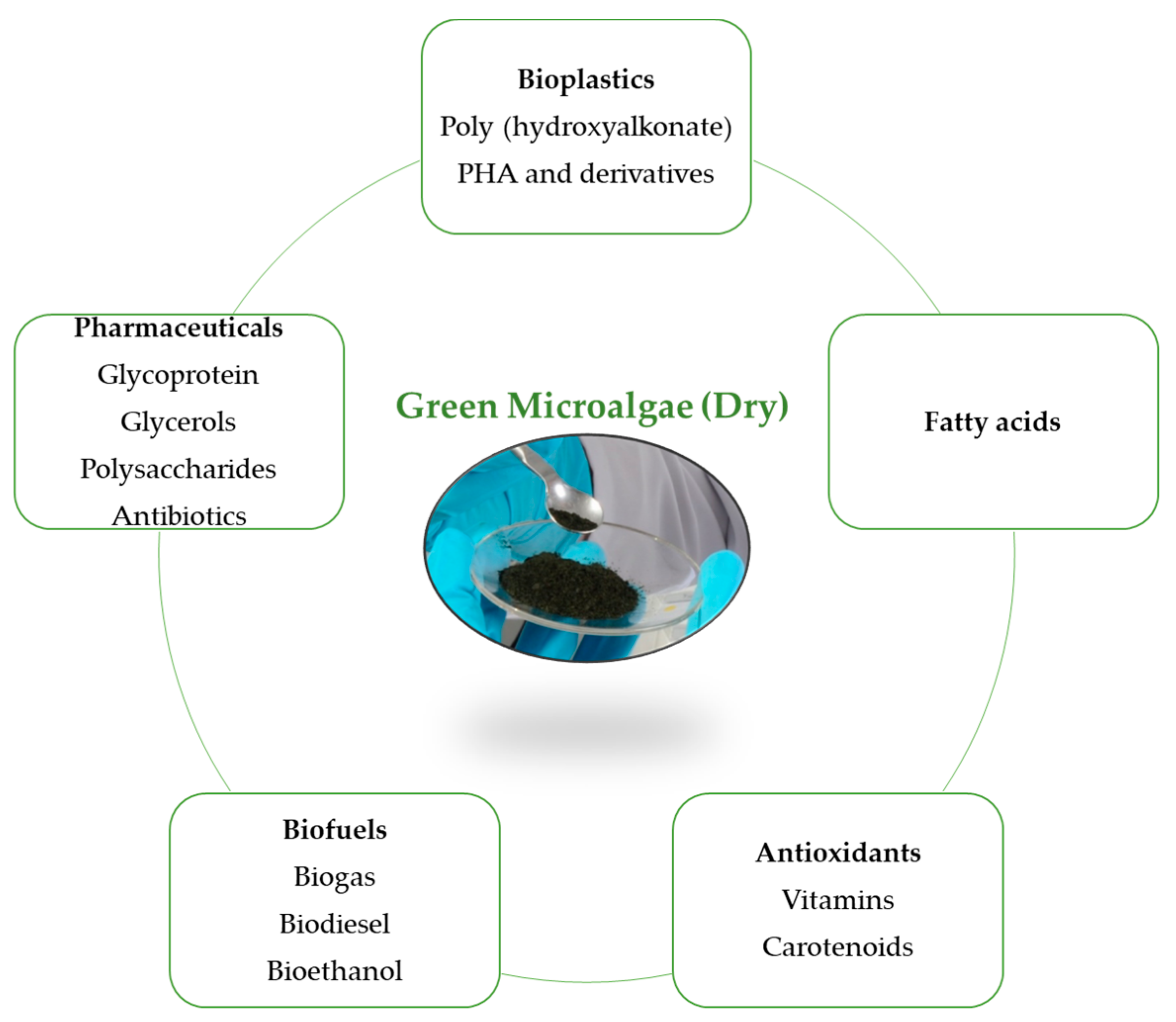
2. Microalgae and Additives
2.1. Chlorella
2.2. Spirulina
2.3. Other Microalgae Species Used for Bioplastic Production
2.4. Additives Used in the Production Process
2.4.1. Materials Blended with Microalgae Biomass
2.4.2. Plasticizers and Compatibilizers
2.4.3. Other Chemicals Used in the Process
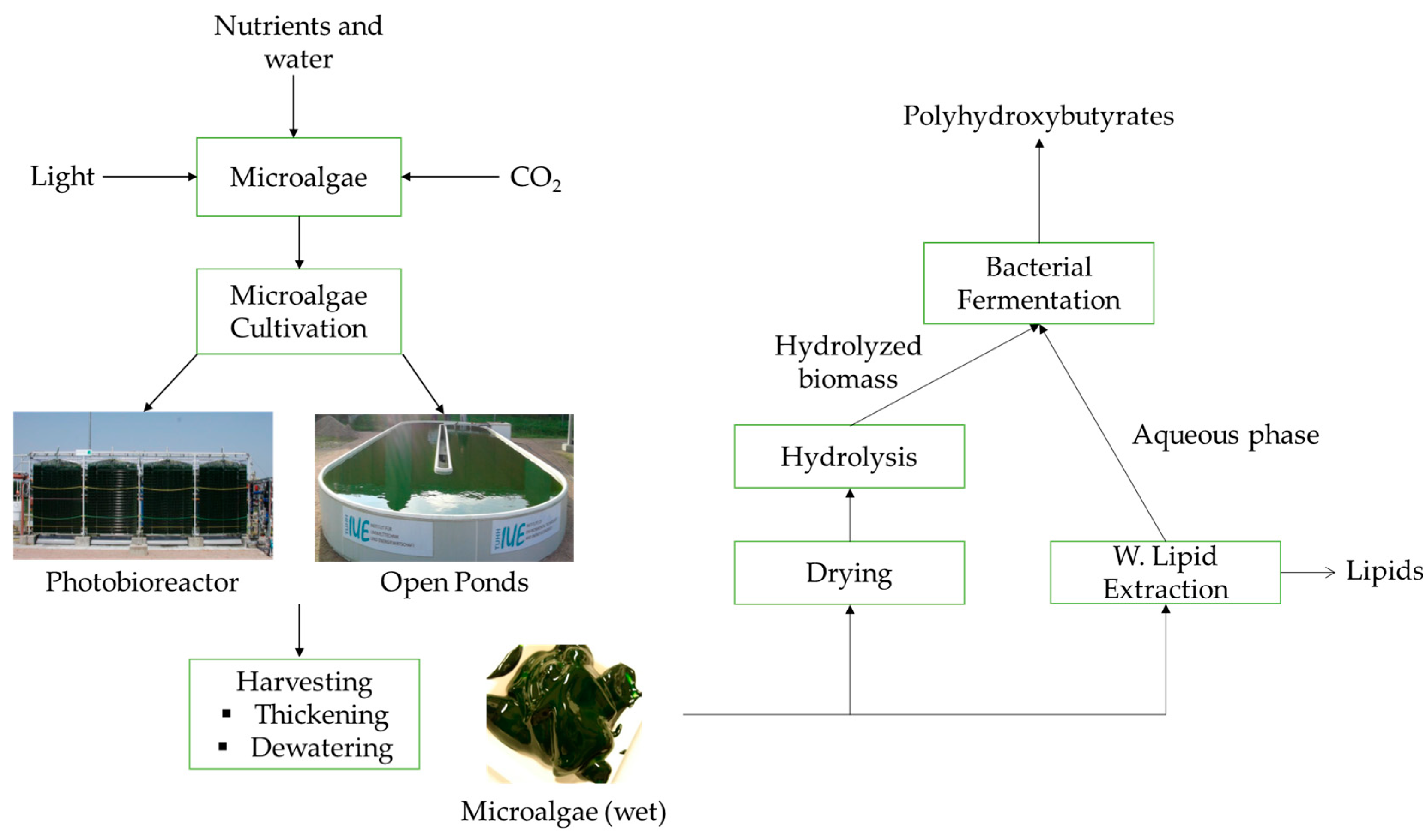
3. Cultivation of Microalgae
3.1. Production Systems
3.2. Harvesting and Processing Systems
4. Bioplastic Production Technologies
4.1. Production of Microalgae-Polymer Blends
Testing Methods Used for Blend Characterization and Performance Measurement
4.2. Production of Biopolymers Using Microalgae Cells
5. LCA Studies on Bioplastic Production from Microalgae
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rahman, A.; Miller, C.D. Microalgae as a Source of Bioplastics. In Algal Green Chemistry: Recent Progress in Biotechnology/Edited by Rajesh Prasad Rastogi, Datta Madamwar, Ashok Pandey; Rastogi, R.P., Madamwar, D., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 121–138. ISBN 9780444637840. [Google Scholar]
- Pandey, A.; Madamwar, D.; Rastogi, R.P. Algal Green Chemistry. Recent Progress in Biotechnology; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 9780444640413. [Google Scholar]
- MacArthur, E. Beyond plastic waste. Science 2017, 358, 843. [Google Scholar] [CrossRef] [PubMed]
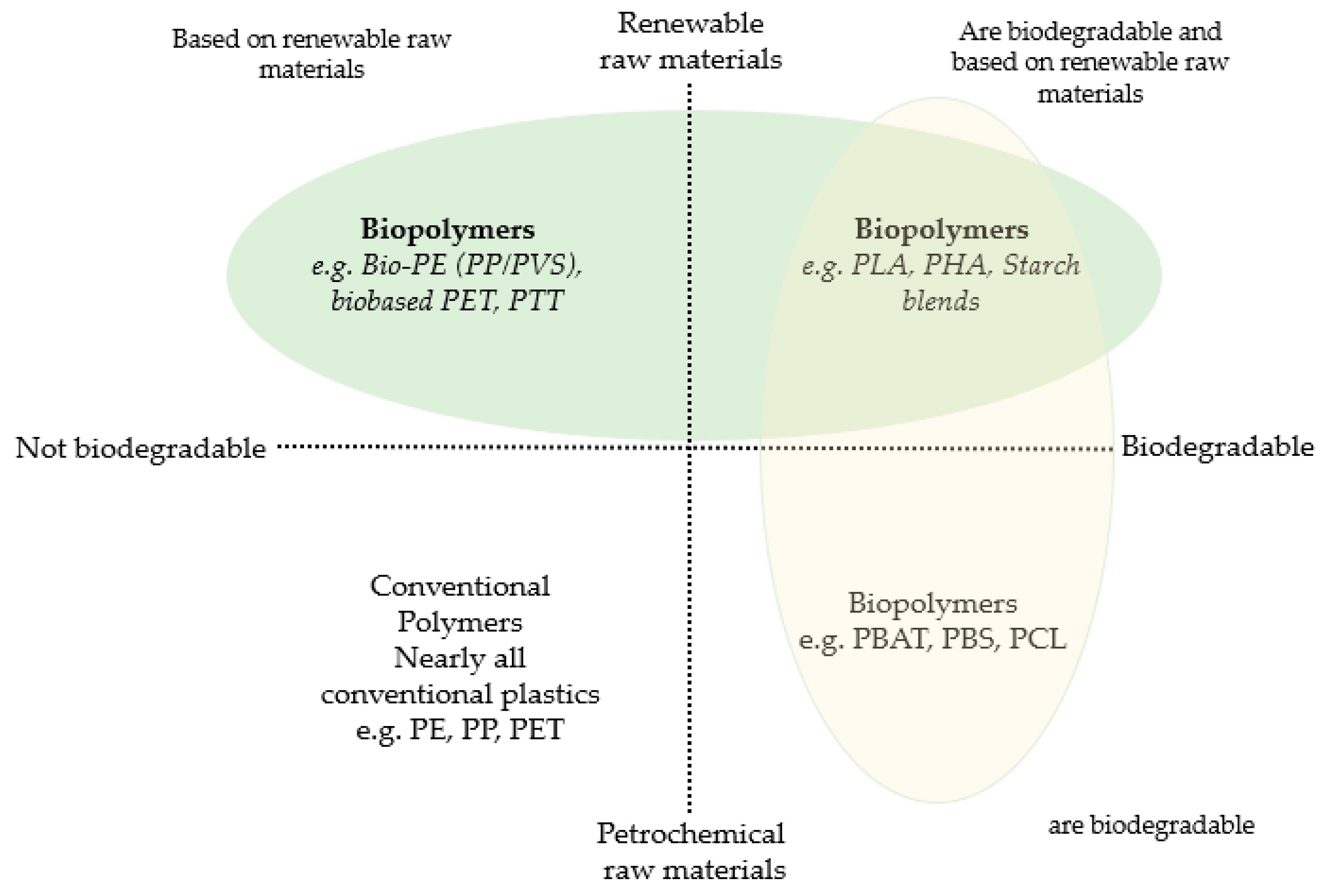
- European Bioplastics. Fact Sheet, European Bioplastics: What are the bioplastics? European Bioplastics e.V.: Berlin, Germany, 2018. [Google Scholar]
- Thielen, M. Bioplastics: Plants and Crops Raw Materials Products; Fachagentur Nachwachsende Rohstoffe eV (FNR) Agency for Renewable Resources: Gülzow, Germany, 2014. [Google Scholar]
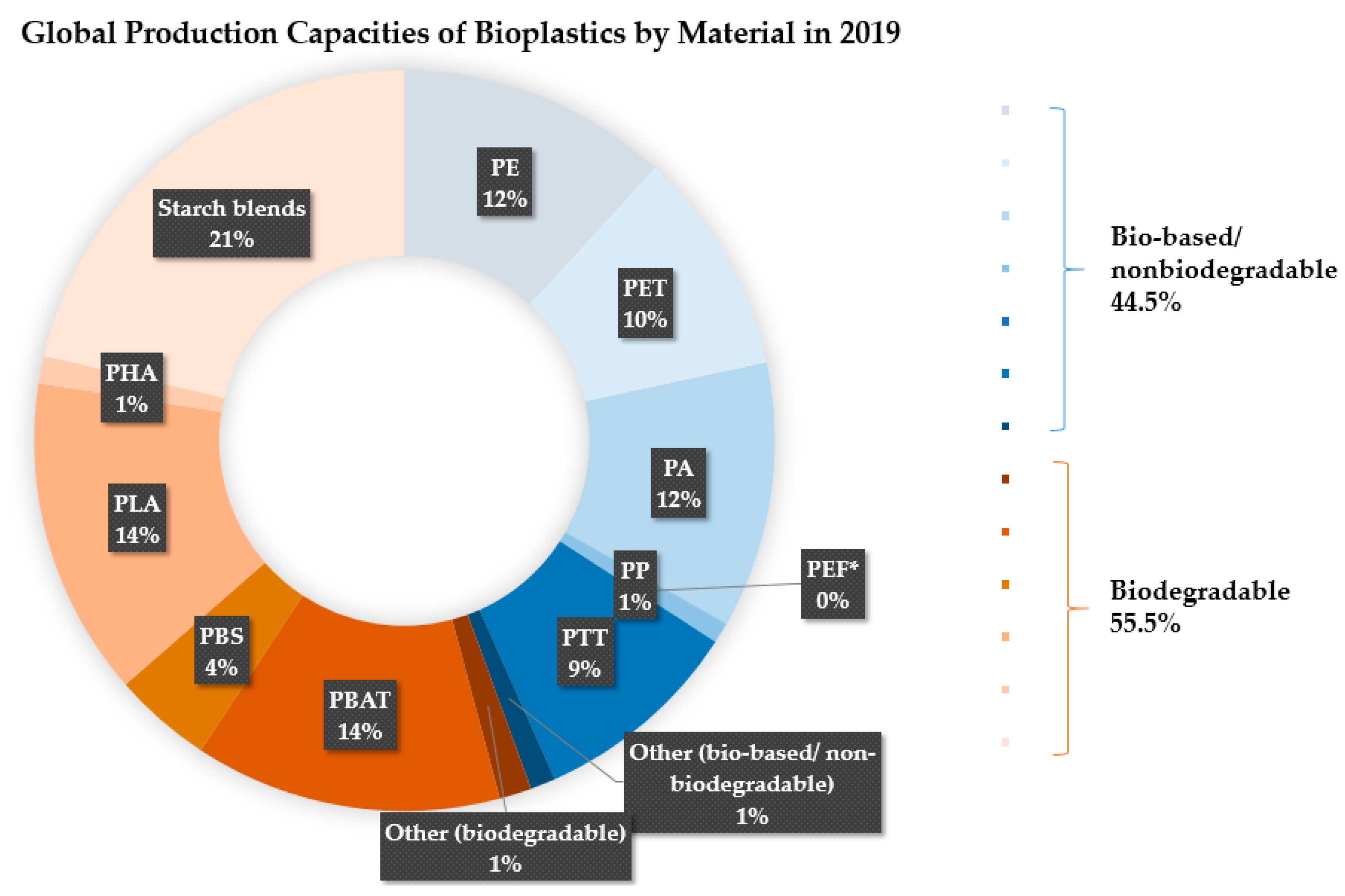
- European Bioplastics. Bioplastics Market Data. Available online: https://www.european-bioplastics.org/market/ (accessed on 1 April 2020).
- Alaerts, L.; Augustinus, M.; van Acker, K. Impact of bio-based plastics on current recycling of plastics. Sustainability 2018, 10, 1487. [Google Scholar] [CrossRef]
- Gilbert, M. Brydson’s Plastics Materials. Chapter 23—Bioplastics: New Routes, New Products; William Andrew: Amsterdam, The Netherlands, 2016; ISBN 0323370225. [Google Scholar]
- Zeller, M.A.; Hunt, R.; Jones, A.; Sharma, S. Bioplastics and their thermoplastic blends from Spirulina and Chlorella microalgae. J. Appl. Polym. Sci. 2013, 130, 3263–3275. [Google Scholar] [CrossRef]
- DiGregorio, B.E. Biobased performance bioplastic: Mirel. Chem. Biol. 2009, 16, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Hempel, F.; Bozarth, A.S.; Lindenkamp, N.; Klingl, A.; Zauner, S.; Linne, U.; Steinbüchel, A.; Maier, U.G. Microalgae as bioreactors for bioplastic production. Microb. Cell Factories 2011, 10, 81. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef]
- Mohan, S.V.; Hemalatha, M.; Chakraborty, D.; Chatterjee, S.; Ranadheer, P.; Kona, R. Algal biorefinery models with self-sustainable closed loop approach: Trends and prospective for blue-bioeconomy. Bioresour. Technol. 2019, 295, 122128. [Google Scholar] [CrossRef]
- Moreno-Garcia, L.; Adjallé, K.; Barnabé, S.; Raghavan, G.S.V. Microalgae biomass production for a biorefinery system: Recent advances and the way towards sustainability. Renew. Sustain. Energy Rev. 2017, 76, 493–506. [Google Scholar] [CrossRef]
- Scopus. 2020. Available online: https://www.scopus.com (accessed on 13 February 2020).
- Abu, Y. (Ed.) Microalgae Cultivation for Biofuels Production. Fundamentals of Microalgae Cultivation; Acedemic Press: Kumargaon, Bangladesh, 2020. [Google Scholar]
- Oh, Y.-K.; Hwang, K.-R.; Kim, C.; Kim, J.R.; Lee, J.-S. Recent developments and key barriers to advanced biofuels: A short review. Bioresour. Technol. 2018, 257, 320–333. [Google Scholar] [CrossRef]
- Pal, P.; Chew, K.W.; Yen, H.-W.; Lim, J.W.; Lam, M.K.; Show, P.L. Cultivation of Oily Microalgae for the Production of Third-Generation Biofuels. Sustainability 2019, 11, 5424. [Google Scholar] [CrossRef]
- Musa, M.; Ayoko, G.A.; Ward, A.; Rösch, C.; Brown, R.J.; Rainey, T.J. Factors Affecting Microalgae Production for Biofuels and the Potentials of Chemometric Methods in Assessing and Optimizing Productivity. Cells 2019, 8, 851. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.; Mohan, S.V. Enhanced bio-hydrogenesis by co-culturing photosynthetic bacteria with acidogenic process: Augmented dark-photo fermentative hybrid system to regulate volatile fatty acid inhibition. Int. J. Hydrogen Energy 2014, 39, 7604–7615. [Google Scholar] [CrossRef]
- Tibbetts, S.M.; Milley, J.E.; Lall, S.P. Chemical composition and nutritional properties of freshwater and marine microalgal biomass cultured in photobioreactors. J. Appl. Phycol. 2015, 27, 1109–1119. [Google Scholar] [CrossRef]
- Gozan, M.; Noviasari, C. The Effect of Glycerol Addition as Plasticizer in Spirulina Platensis Based Bioplastic; Dianursanti, M., Ed.; E3S Web of Conferences; EDP Sciences: Bali, Indonesia, 2018. [Google Scholar]
- Dianursanti, S.A.K.; Khalis, S.A. The Effect of Compatibilizer Addition on Chlorella Vulgaris Microalgae Utilization as a Mixture for Bioplastic; E3S Web of Conferences; EDP Sciences: Bali, Indonesia, 2018. [Google Scholar]
- Gifuni, I.; Olivieri, G.; Krauss, I.R.; D’Errico, G.; Pollio, A.; Marzocchella, A. Microalgae as new sources of starch: Isolation and characterization of microalgal starch granules. Chem. Eng. Trans. 2017, 57, 1423–1428. [Google Scholar]
- Zhang, F.; Endo, T.; Kitagawa, R.; Kabeya, H.; Hirotsu, T. Synthesis and characterization of a novel blend of polypropylene with Chlorella. J. Mater. Chem. 2000, 10, 2666–2672. [Google Scholar] [CrossRef]
- Sabathini, H.A.; Windiani, L.; Gozan, M. Mechanical Physicial Properties of Chlorella-PVA based Bioplastic with Ultrasonic Homogenizer. E3S Web Conf. 2018, 67, 3046. [Google Scholar] [CrossRef]
- Otsuki, T.; Zhang, F.; Kabeya, H.; Hirotsu, T. Synthesis and tensile properties of a novel composite ofChlorella and polyethylene. J. Appl. Polym. Sci. 2004, 92, 812–816. [Google Scholar] [CrossRef]
- Lupatini, A.L.; Colla, L.M.; Canan, C.; Colla, E. Potential application of microalga Spirulina platensis as a protein source. J. Sci. Food Agric. 2017, 97, 724–732. [Google Scholar] [CrossRef]
- Dianursanti, C.N.; Windiani, L.; Gozan, M. Effect of Compatibilizer Addition in Spirulina Platensis Based Bioplastic Production; American Institute of Physics Conference Series: Erbil, Iraq, 2019. [Google Scholar]
- Ciapponi, R.; Turri, S.; Levi, M. Mechanical Reinforcement by Microalgal Biofiller in Novel Thermoplastic Biocompounds from Plasticized Gluten. Materials 2019, 12, 1476. [Google Scholar] [CrossRef]
- Wang, K. Bio-Plastic Potential of Spirulina Microalgae. Master’s Thesis, The University of Georgia, Athens, GA, USA, 2014. [Google Scholar]
- Zhu, N.; Ye, M.; Shi, D.; Chen, M. Reactive compatibilization of biodegradable poly(butylene succinate)/Spirulina microalgae composites. Macromol. Res. 2017, 25, 165–171. [Google Scholar] [CrossRef]
- Monshupanee, T.; Nimdach, P.; Incharoensakdi, A. Two-stage (photoautotrophy and heterotrophy) cultivation enables efficient production of bioplastic poly-3-hydroxybutyrate in auto-sedimenting cyanobacterium. Sci. Rep. 2016, 6, 37121. [Google Scholar] [CrossRef] [PubMed]
- Torres, S.; Navia, R.; Campbell Murdy, R.; Cooke, P.; Misra, M.; Mohanty, A.K. Green Composites from Residual Microalgae Biomass and Poly(butylene adipate- co -terephthalate): Processing and Plasticization. ACS Sustain. Chem. Eng. 2015, 3, 614–624. [Google Scholar] [CrossRef]
- Johnsson, N.; Steuer, F. Bioplastic Material from Microalgae: Extraction of Starch and PHA from Microalgae to Create a Bioplastic Material; KTH Royal Institute of Technology: Stockholm, Sweden, 2018. [Google Scholar]
- Dixon, C.; Wilken, L.R. Green microalgae biomolecule separations and recovery. Bioresour. Bioprocess. 2018, 5, 1–24. [Google Scholar] [CrossRef]
- Eagan, J.M.; Xu, J.; Di Girolamo, R.; Thurber, C.M.; Macosko, C.W.; LaPointe, A.M.; Bates, F.S.; Coates, G.W. Combining polyethylene and polypropylene: Enhanced performance with PE/iPP multiblock polymers. Science 2017, 355, 814–816. [Google Scholar] [CrossRef]
- Valerie, C. Final report on the safety assessment of Polyethylene. Int. J. Toxicol. 2007, 26, 115–127. [Google Scholar]
- Park, S.-J.; Seo, M.-K. Interface Science and Composites; Academic Press: Burlington, NJ, USA, 2011; ISBN 0123750490. [Google Scholar]
- Yan, Y. Developments in fibers for technical nonwovens. In Advances in Technical Nonwovens; Elsevier: Sawston, UK, 2016; pp. 19–96. [Google Scholar]
- Proshad, R.; Kormoker, T.; Islam, M.S.; Haque, M.A.; Rahman, M.M.; Mithu, M.M.R. Toxic effects of plastic on human health and environment: A consequences of health risk assessment in Bangladesh. Int. J. Health 2018, 6, 1–5. [Google Scholar] [CrossRef]
- Wretfors, C.; Cho, S.-W.; Hedenqvist, M.S.; Marttila, S.; Nimmermark, S.; Johansson, E. Use of industrial hemp fibers to reinforce wheat gluten plastics. J. Polym. Environ. 2009, 17, 259. [Google Scholar] [CrossRef]
- Kalia, S.; Avérous, L. Biodegradable and Biobased Polymers for Environmental and Biomedical Applications; John Wiley & Sons: Beverly, MA, USA, 2016; ISBN 1119117348. [Google Scholar]
- Rudnik, E. Compostable Polymer Materials; Newnes: Amsterdam, The Netherlands, 2019; ISBN 0080994423. [Google Scholar]
- Xu, J.; Guo, B.-H. Poly (butylene succinate) and its copolymers: Research, development and industrialization. Biotechnol. J. 2010, 5, 1149–1163. [Google Scholar] [CrossRef]
- Morgott, D.A. Acetone. In Patty’s Toxicology, 5th ed.; Patty, F.A., Ed.; compl. rev. and updated.; Wiley: New York, NY, USA, 2001; p. 336. ISBN 9780471125471. [Google Scholar]
- Sereni, J.G.R. Reference module in materials science and materials engineering. Mater. Sci. Eng. A 2016. [Google Scholar] [CrossRef]
- Kutz, M. Applied Plastics Engineering Handbook: Processing and Materials; William Andrew: Amsterdam, The Netherlands, 2011; ISBN 1437735142. [Google Scholar]
- Vieira, M.G.A.; da Silva, M.A.; dos Santos, L.O.; Beppu, M.M. Natural-based plasticizers and biopolymer films: A review. Eur. Polym. J. 2011, 47, 254–263. [Google Scholar] [CrossRef]
- Ranade, V.V. The Future of Glycerol-New Usages for a Versatile Raw Material; LWW: Cambridge, MA, USA, 2009. [Google Scholar]
- Martin Chaplin. Carboxymethylcellulose (CMC). Available online: http://www1.lsbu.ac.uk/water/carboxymethylcellulose.html (accessed on 18 February 2020).
- Biron, M. Recycling: The First Source of Renewable Plastics. In Industrial Applications of Renewable Plastics; Elsevier: Amsterdam, The Netherlands, 2016; pp. 67–114. ISBN 9780323480666. [Google Scholar]
- Subramanian, V.D.V. (Ed.) Biopolymer Composites in Electronics. Thermoelectric Properties of Biopolymer Composites; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Chen, C.-Y.; Yeh, K.-L.; Aisyah, R.; Lee, D.-J.; Chang, J.-S. Cultivation, photobioreactor design and harvesting of microalgae for biodiesel production: A critical review. Bioresour. Technol. 2011, 102, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Scott, S.A.; Davey, M.P.; Dennis, J.S.; Horst, I.; Howe, C.J.; Lea-Smith, D.J.; Smith, A.G. Biodiesel from algae: Challenges and prospects. Curr. Opin. Biotechnol. 2010, 21, 277–286. [Google Scholar] [CrossRef]
- Sharma, K.K.; Schuhmann, H.; Schenk, P.M. High Lipid Induction in Microalgae for Biodiesel Production. Energies 2012, 5, 1532–1553. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae beats bioethanol. Trends Biotechnol. 2008, 26, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, J.; Aila, M.; Bangwal, D.P.; Kaul, S.; Garg, M.O. Algae based biorefinery—How to make sense? Renew. Sustain. Energy Rev. 2015, 47, 295–307. [Google Scholar] [CrossRef]
- Wijffels, R.H.; Barbosa, M.J.; Eppink, M.H.M. Microalgae for the production of bulk chemicals and biofuels. Biofuels Bioprod. Biorefin. 2010, 4, 287–295. [Google Scholar] [CrossRef]
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef]
- Singh, R.N.; Sharma, S. Development of suitable photobioreactor for algae production—A review. Renew. Sustain. Energy Rev. 2012, 16, 2347–2353. [Google Scholar] [CrossRef]
- Milledge, J.J.; Heaven, S. Disc Stack Centrifugation Separation and Cell Disruption of Microalgae: A Technical Note. Environ. Nat. Resour. Res. 2011, 1, 17–24. [Google Scholar] [CrossRef]
- Knuckey, R.M.; Brown, M.R.; Robert, R.; Frampton, D.M.F. Production of microalgal concentrates by flocculation and their assessment as aquaculture feeds. Aquac. Eng. 2006, 35, 300–313. [Google Scholar] [CrossRef]
- Hai, T.; Ahlers, H.; Gorenflo, V.; Steinbüchel, A. Axenic cultivation of anoxygenic phototrophic bacteria, cyanobacteria, and microalgae in a new closed tubular glass photobioreactor. Appl. Microbiol. Biotechnol. 2000, 53, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Tredici, M.R. Mass production of microalgae: Photobioreactors. Handb. Microalgal Cult. Biotechnol. Appl. Phycol. 2004, 1, 178–214. [Google Scholar]
- Harun, R.; Danquah, M.K.; Forde, G.M. Microalgal biomass as a fermentation feedstock for bioethanol production. J. Chem. Technol. Biotechnol. 2009, 99. [Google Scholar] [CrossRef]
- Ugwu, C.U.; Aoyagi, H.; Uchiyama, H. Photobioreactors for mass cultivation of algae. Bioresour. Technol. 2008, 99, 4021–4028. [Google Scholar] [CrossRef] [PubMed]
- Vonshak, A. Outdoor mass production of Spirulina: The basic concept. In Spirulina Platensis Arthrospira; CRC Press: Boca Raton, FL, USA, 1997; pp. 97–118. [Google Scholar]
- Pulz, O. Photobioreactors: Production systems for phototrophic microorganisms. Appl. Microbiol. Biotechnol. 2001, 57, 287–293. [Google Scholar] [CrossRef]
- Richmond, A.; Lichtenberg, E.; Stahl, B.; Vonshak, A. Quantitative assessment of the major limitations on productivity ofSpirulina platensis in open raceways. J. Appl. Phycol. 1990, 2, 195–206. [Google Scholar] [CrossRef]
- McBride, R.C.; Lopez, S.; Meenach, C.; Burnett, M.; Lee, P.A.; Nohilly, F.; Behnke, C. Contamination Management in Low Cost Open Algae Ponds for Biofuels Production. Ind. Biotechnol. 2014, 10, 221–227. [Google Scholar] [CrossRef]
- Doucha, J.; Straka, F.; Lívanský, K. Utilization of flue gas for cultivation of microalgae Chlorella sp.) in an outdoor open thin-layer photobioreactor. J. Appl. Phycol. 2005, 17, 403–412. [Google Scholar] [CrossRef]
- Carlozzi, P. Dilution of solar radiation through “culture” lamination in photobioreactor rows facing south-north: A way to improve the efficiency of light utilization by cyanobacteria (Arthrospira platensis). Biotechnol. Bioeng. 2003, 81, 305–315. [Google Scholar] [CrossRef]
- Olaizola, M. Commercial production of astaxanthin from Haematococcus pluvialis using 25,000-liter outdoor photobioreactors. J. Appl. Phycol. 2000, 12, 499–506. [Google Scholar] [CrossRef]
- Sato, T.; Usui, S.; Tsuchiya, Y.; Kondo, Y. Invention of outdoor closed type photobioreactor for microalgae. Energy Convers. Manag. 2006, 47, 791–799. [Google Scholar] [CrossRef]
- Lee, Y.-K. Microalgal mass culture systems and methods: Their limitation and potential. J. Appl. Phycol. 2001, 13, 307–315. [Google Scholar] [CrossRef]
- Shen, Y.; Yuan, W.; Pei, Z.J.; Wu, Q.; Mao, E. Microalgae Mass Production Methods. Trans. ASABE 2009, 52, 1275–1287. [Google Scholar] [CrossRef]
- Fu, W.; Gudmundsson, O.; Feist, A.M.; Herjolfsson, G.; Brynjolfsson, S.; Palsson, B.Ø. Maximizing biomass productivity and cell density of Chlorella vulgaris by using light-emitting diode-based photobioreactor. J. Biotechnol. 2012, 161, 242–249. [Google Scholar] [CrossRef]
- Salim, S.; Bosma, R.; Vermuë, M.H.; Wijffels, R.H. Harvesting of microalgae by bio-flocculation. J. Appl. Phycol. 2011, 23, 849–855. [Google Scholar] [CrossRef]
- Rawat, I.; Ranjith Kumar, R.; Mutanda, T.; Bux, F. Dual role of microalgae: Phycoremediation of domestic wastewater and biomass production for sustainable biofuels production. Appl. Energy 2011, 88, 3411–3424. [Google Scholar] [CrossRef]
- Uduman, N.; Qi, Y.; Danquah, M.K.; Forde, G.M.; Hoadley, A. Dewatering of microalgal cultures: A major bottleneck to algae-based fuels. J. Renew. Sustain. Energy 2010, 2, 12701. [Google Scholar] [CrossRef]
- Pragya, N.; Pandey, K.K.; Sahoo, P.K. A review on harvesting, oil extraction and biofuels production technologies from microalgae. Renew. Sustain. Energy Rev. 2013, 24, 159–171. [Google Scholar] [CrossRef]
- Barros, A.I.; Gonçalves, A.L.; Simões, M.; Pires, J.C.M. Harvesting techniques applied to microalgae: A review. Renew. Sustain. Energy Rev. 2015, 41, 1489–1500. Available online: http://www.sciencedirect.com/science/article/pii/S1364032114008107 (accessed on 11 April 2020). [CrossRef]
- Fabra, M.J.; Martínez-Sanz, M.; Gómez-Mascaraque, L.G.; Gavara, R.; López-Rubio, A. Structural and physicochemical characterization of thermoplastic corn starch films containing microalgae. Carbohydr. Polym. 2018, 186, 184–191. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, C.; Cao, G.; Wang, D.; Ho, S.-H. A sustainable solution to plastics pollution: An eco-friendly bioplastic film production from high-salt contained Spirulina sp. residues. J. Hazard. Mater. 2019, 121773. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Mandal, A.; Ayton, E.; Hunt, R.; Zeller, M.A.; Sharma, S. Chapter 6—Modification of Protein Rich Algal-Biomass to Form Bioplastics and Odor Removal. In Protein Byproducts; Academic Press: Cambridge, MA, USA, 2016; pp. 107–117. ISBN 978-0-12-802391-4. [Google Scholar]
- Charlie, M.; Pauline, P.; Benjamin, G.; Jean-François, S.; Florian, D.; Nicolas, L.M. Microalgae starch-based bioplastics: Screening of ten strains and plasticization of unfractionated microalgae by extrusion. Carbohydr. Polym. 2019, 208, 142–151. [Google Scholar] [CrossRef]
- Abdo, S.M.; Ali, G.H. Analysis of polyhydroxybutrate and bioplastic production from microalgae. Bull. Natl. Res. Cent. 2019, 43, 207. [Google Scholar] [CrossRef]
- Zhang, F.; Kabeya, H.; Kitagawa, R.; Hirotsu, T.; Yamashita, M.; Otsuki, T. An exploratory research of PVC-Chlorella composite material (PCCM) as effective utilization of Chlorella biologically fixing CO2. J. Mater. Sci. 2000, 35, 2603–2609. [Google Scholar] [CrossRef]
- Kato, N. Production of crude bioplastic-beads with microalgae: Proof-of-concept. Bioresour. Technol. Rep. 2019, 6, 81–84. [Google Scholar] [CrossRef]
- Osanai, T.; Numata, K.; Oikawa, A.; Kuwahara, A.; Iijima, H.; Doi, Y.; Tanaka, K.; Saito, K.; Hirai, M.Y. Increased bioplastic production with an RNA polymerase sigma factor SigE during nitrogen starvation in Synechocystis sp. PCC 6803. DNA Res. 2013, 20, 525–535. [Google Scholar] [CrossRef]
- Troschl, C. Bioplastic production with cyanobacteria. Ph.D. Thesis, Universität für Bodenkultur Wien, Wien, Austria, 2018. [Google Scholar]
- Das, S.K.; Sathish, A.; Stanley, J. Production of Biofuel and Bioplastic from Chlorella Pyrenoidosa. Mater. Today Proc. 2018, 5, 16774–16781. [Google Scholar] [CrossRef]
- Uggetti, E.; García, J.; Álvarez, J.A.; García-Galán, M.J. Start-up of a microalgae-based treatment system within the biorefinery concept: From wastewater to bioproducts. Water Sci. Technol. 2018, 78, 114–124. [Google Scholar] [CrossRef]
- Costa, S.S.; Miranda, A.L.; de Morais, M.G.; Costa, J.A.V.; Druzian, J.I. Microalgae as source of polyhydroxyalkanoates (PHAs)—A review. Int. J. Biol. Macromol. 2019, 131, 536–547. [Google Scholar] [CrossRef]
- Singh, A.K.; Sharma, L.; Mallick, N.; Mala, J. Progress and challenges in producing polyhydroxyalkanoate biopolymers from cyanobacteria. J. Appl. Phycol. 2017, 29, 1213–1232. [Google Scholar] [CrossRef]
- Kaparapu, J. Polyhydroxyalkanoate (PHA) Production by Genetically Engineered Microalgae: A Review. J. New Biol. Rep. 2018, 7, 68–73. [Google Scholar]
- Di Caprio, F.; Visca, A.; Altimari, P.; Toro, L.; Masciocchi, B.; Iaquaniello, G.; Pagnanelli, F. Two stage process of microalgae cultivation for starch and carotenoid production. Chem. Eng. Trans. 2016, 49, 415–420. [Google Scholar]
- Brányiková, I.; Maršálková, B.; Doucha, J.; Brányik, T.; Bišová, K.; Zachleder, V.; Vítová, M. Microalgae—Novel highly efficient starch producers. Biotechnol. Bioeng. 2011, 108, 766–776. [Google Scholar] [CrossRef]
- Khanra, S.; Mondal, M.; Halder, G.; Tiwari, O.N.; Gayen, K.; Bhowmick, T.K. Downstream processing of microalgae for pigments, protein and carbohydrate in industrial application: A review. Food Bioprod. Process. 2018, 110, 60–84. [Google Scholar] [CrossRef]
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Amaro, H.M.; Malcata, F.X.; Sousa-Pinto, I. Production and Supply Logistics of Microalgae as an Energy Feedstock. In Sustainable Bioenergy Production; CRC Press: Boca Raton, FL, USA, 2014; pp. 148–167. [Google Scholar]
- Rahman, A.; Putman, R.J.; Inan, K.; Sal, F.A.; Sathish, A.; Smith, T.; Nielsen, C.; Sims, R.C.; Miller, C.D. Polyhydroxybutyrate production using a wastewater microalgae based media. Algal Res. 2015, 8, 95–98. [Google Scholar] [CrossRef]
- Zhou, N.; Zhang, Y.; Wu, X.; Gong, X.; Wang, Q. Hydrolysis of Chlorella biomass for fermentable sugars in the presence of HCl and MgCl2. Bioresour. Technol. 2011, 102, 10158–10161. [Google Scholar] [CrossRef]
- Huo, Y.-X.; Cho, K.M.; Rivera, J.G.L.; Monte, E.; Shen, C.R.; Yan, Y.; Liao, J.C. Conversion of proteins into biofuels by engineering nitrogen flux. Nat. Biotechnol. 2011, 29, 346–351. [Google Scholar] [CrossRef]
- Bussa, M.; Eisen, A.; Zollfrank, C.; Röder, H. Life cycle assessment of microalgae products: State of the art and their potential for the production of polylactid acid. J. Clean. Prod. 2019, 213, 1299–1312. [Google Scholar] [CrossRef]
- Beckstrom, B.D. Bioplastic Production from Microalgae with Fuel Co-Products: A Techno-Economic and Life-Cycle Assessment. Master’s Thesis, Colorado State University, Fort Collins, CO, USA, 2019. [Google Scholar]
- Medeiros, D.L.; Sales, E.A.; Kiperstok, A. Energy production from microalgae biomass: Carbon footprint and energy balance. J. Clean. Prod. 2015, 96, 493–500. [Google Scholar] [CrossRef]
- Quinn, J.C.; Davis, R. The potentials and challenges of algae based biofuels: A review of the techno-economic, life cycle, and resource assessment modeling. Bioresour. Technol. 2015, 184, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Draaisma, R.B.; Wijffels, R.H.; Slegers, P.M.E.; Brentner, L.B.; Roy, A.; Barbosa, M.J. Food commodities from microalgae. Curr. Opin. Biotechnol. 2013, 24, 169–177. [Google Scholar] [CrossRef] [PubMed]


| Biomass Species | Type of Product | Ratio of Materials | Particle Size of Biomass | Characterization | Reference |
|---|---|---|---|---|---|
| C. vulgaris | 100% algae-based plastics and hybrid blends with PE and glycerol | Glycerol 0–30% (by weight) | 53–75 µm | 57% protein | [9] |
| C. vulgaris | Chlorella/PVA composites | Compatibilizer (MA) concentration 0–6% | - | 58.5% protein (on the dry weight basis) | [22,23] |
| C. sorokiniana | Starch granules | - | - | 38% starch (on the dry weight basis) | [24] |
| Chlorella | PP from Chlorella and MPP (maleic anhydride-modified polypropylene) | MPP/Chlorella: 0.5 | 50 µm | - | [25] |
| Chlorella | Chlorella/PVC composites | Stabilizer (PSE-227 and KM-55) concentration 0–2.5% | 62–66 µm | - | [25] |
| Chlorella | Chlorella/PVA blend film | Chlorella/distilled water: 2.8 g/8.4, 14, 28 mL | - | - | [26] |
| C. sp. | Chlorella/PE composites | Chlorella/MPE (modified PE):10–40% Chlorella/UPE (unmodified PE):10–40% | ~1 mm | - | [27] |
| Component | (wt %) |
|---|---|
| Protein | 60 |
| Lipid | 6 |
| Fatty acid | 265 mg·10 g−1 |
| Amino acid | 2410 mg·10 g−1 |
| Vitamin A | 2300 IU |
| Vitamin B1-B3 | 2.3 mg·10 g−1 |
| Vitamin B6 & B12 | 112 mcg |
| Vitamin E | 4 IU |
| Phycocyanin | 20% |
| Chlorophyll | 1.5% |
| B-Carotenoids | 0.15% |
| Pantothenic acid | 4 mg·100 g−1 |
| Folic acid | 100 mg·100 g−1 |
| Polysaccharide | 0.4 g·100 g−1 |
| Biomass Species | Type of Product | Ratio of Materials | Particle Size of Biomass | Characterization | Reference |
|---|---|---|---|---|---|
| S. platensis | 100% algae-based plastics Hybrid blends with PE and glycerol | Glycerol 0–30% (by weight) | 53–75 µm | 57% protein | [9] |
| S. platensis | Bioplastic biofilm | Compatibilizer concentartion: 0–6% | - | 60% protein | [29] |
| S. platensis | Bio filler | 10%, 20% and 30% microalgae | - | - | [30] |
| S. platensis | S. platensis-based plastic | Plasticizer concentrations: 15%, 20%, 25% and 30% | - | - | [22] |
| Spirulina | Plasticized spirulina | EG weight rations: 5%, 10%, 30% | - | - | [31] |
| Spirulina | Blend of Spirulina with UHMW-PE | PE weight ratios: 15%, 20% and 80% | - | - | [31] |
| Spirulina | Compatibilized bioplastic | weight percentage of 3% compatibilizer (PE-g-MA) | - | - | [31] |
| Spirulina | PBS/Spirulina composites | The formulated Spirulina (varying from 15% to 50% loading) and PBS with and without PBS-g-MAH | - | 60% protein (on dry weight basis) | [32] |
| Biomass Species | Type of Product | Ratio of Materials | Characterization | Reference |
|---|---|---|---|---|
| Chlorogloea fritschii | bioplastic poly-3-hydroxybutyrate | - | PHB levels at 14–17% (w/w DW) | [33] |
| Phaeodactylum tricornutum | bioplastic PHB | - | PHB levels of up to 10.6% of algal dry weight | [11] |
| Calothrix scytonemicola, Scenedesmus almeriensis and Neochloris oleoabundans, | bio-based plastic film | 1:2, Carboxymethyl Cellulose (CMC):biomass | - | [35] |
| Calothrix scytonemicola | PHA, plastic film | Product 1: 150 mg pure PH3B and 8 mL of chloroform | - | [35] |
| Product 2: 100 mg of PH3B and 50 mg CMC mixed with 8 mL of CMC. | ||||
| Product 3: 100 mg PH3B and 50 mg sucrose octa acetate in 8 mL of CMC | ||||
| Nannocloropsis gaditana | Bio composites: biomass and PBAT | Ratios of biomass: 10, 20, 30 | - | [34] |
| Blended Materials with Biomass | Chemical Formula | Purpose of Usage | Reference |
|---|---|---|---|
| PE | (C2H4)n | Blended with Chlorella and Spirulina | [9,27] |
| PP | (C3H6)n | Blended with Chlorella | [25] |
| PVA | (C2H4)n | Blended with Chlorella | [25] |
| Wheat gluten | Blended with Spirulina platensis | [30] | |
| PBS | (C8H12O4)n | Blended with Spirulina | [32] |
| UHMW-PE | C2H4 | Blended with Spirulina | [31] |
| PVA-g-MAH (maleic anhydride-grafted PVA) | Used in blending process | [25] | |
| Acetone | C3H6O | Used in blending process | [27,35] |
| Sodium sulfite | Na2SO3 | Used in blending process | [32] |
| BPO | C14H10O4 | Used in blending process | [22] |
| Plasticizers and Compatibilizers | Chemical Formula | Purpose of Usage | Reference |
|---|---|---|---|
| Glycerol | C3H8O3 | Plasticizer | [22,26,29,30,32,50] |
| Octanoic acid | C8H16O2 | Plasticizer | [30] |
| 1,4-butanediol | C4H10O2 | Plasticizer | [30] |
| EG | C2H6O2 | Plasticizer | [31] |
| CMC | Plasticizer | [35] | |
| MA | C4H2O3 | Compatibilizer and grafting PVA | [22,25] |
| PE-g-MA | Compatibilizer | [31] | |
| KPS | K2S2O8 | Compatibilizer initiator | [22] |
| DMSO | (CH3)2SO | Compatibilizer initiator | [22] |
| Other Chemicals Used in the Process | Chemical Formula | Purpose of Usage | Reference |
|---|---|---|---|
| Ethanol | C2H5OH | Suspension of biomass | [24] |
| IPP (isotactic polypropylene) | (C3H6)n | - | [25] |
| Citric acid | C6H8O7 | Bioplastic film preparation | [26] |
| DCP (Dicumyl peroxide) | C18H22O2 | In the synthesis of PBS-g-MAH | [32] |
| Methanol | CH3OH | To remove pigments in the PHB extraction process | [33] |
| CMC | CHCl3 | PHB extraction from Chlorella fritschii biomass | [33] |
| Phosphate buffered saline | Cl2H3K2Na3O8P2 | Cell washing | [11] |
| Sodium hypochlorite (aq) | NaClO | PHA extraction | [35] |
| Sucrose octa acetate | C28H38O19 | Casting of plastic film | [35] |
| Open Ponds | Closed PBR Systems | |
|---|---|---|
| Advantages |
|
|
| Disadvantages |
|
|
| Production Methods | Publications |
|---|---|
| Melt mixing | [22,23,27,32,84] |
| Compression molding | [9,27,30,31,32,84,86] |
| Hot molding | [22,23] |
| Injection molding | [34] |
| Twin screw extrusion | [34,87] |
| Solvent casting | [26,84,85,88] |
| Testing Methods | Publications |
|---|---|
| Mechanical testing | [9,22,23,26,27,30,31,32,34,84,85,86,88,90] |
| Thermal based analysis (TGA, DSC) | [9,26,30,31,32,34,85,86] |
| SEM | [9,23,26,30,31,34,84,85,88] |
| CLSM | [34] |
| FT-IR | [26,27,31,32,34,85] |
| WAXS, SAXS, XRD | [84] |
| Wetting and water permeability | [30,84] |
| Oxygen permeability | [84] |
| Transparency | [84] |
| Odor panel test | [86] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Onen Cinar, S.; Chong, Z.K.; Kucuker, M.A.; Wieczorek, N.; Cengiz, U.; Kuchta, K. Bioplastic Production from Microalgae: A Review. Int. J. Environ. Res. Public Health 2020, 17, 3842. https://doi.org/10.3390/ijerph17113842
Onen Cinar S, Chong ZK, Kucuker MA, Wieczorek N, Cengiz U, Kuchta K. Bioplastic Production from Microalgae: A Review. International Journal of Environmental Research and Public Health. 2020; 17(11):3842. https://doi.org/10.3390/ijerph17113842
Chicago/Turabian StyleOnen Cinar, Senem, Zhi Kai Chong, Mehmet Ali Kucuker, Nils Wieczorek, Ugur Cengiz, and Kerstin Kuchta. 2020. "Bioplastic Production from Microalgae: A Review" International Journal of Environmental Research and Public Health 17, no. 11: 3842. https://doi.org/10.3390/ijerph17113842
APA StyleOnen Cinar, S., Chong, Z. K., Kucuker, M. A., Wieczorek, N., Cengiz, U., & Kuchta, K. (2020). Bioplastic Production from Microalgae: A Review. International Journal of Environmental Research and Public Health, 17(11), 3842. https://doi.org/10.3390/ijerph17113842