Cancer Incidence Trends in the Oil Shale Industrial Region in Estonia
Abstract
1. Introduction
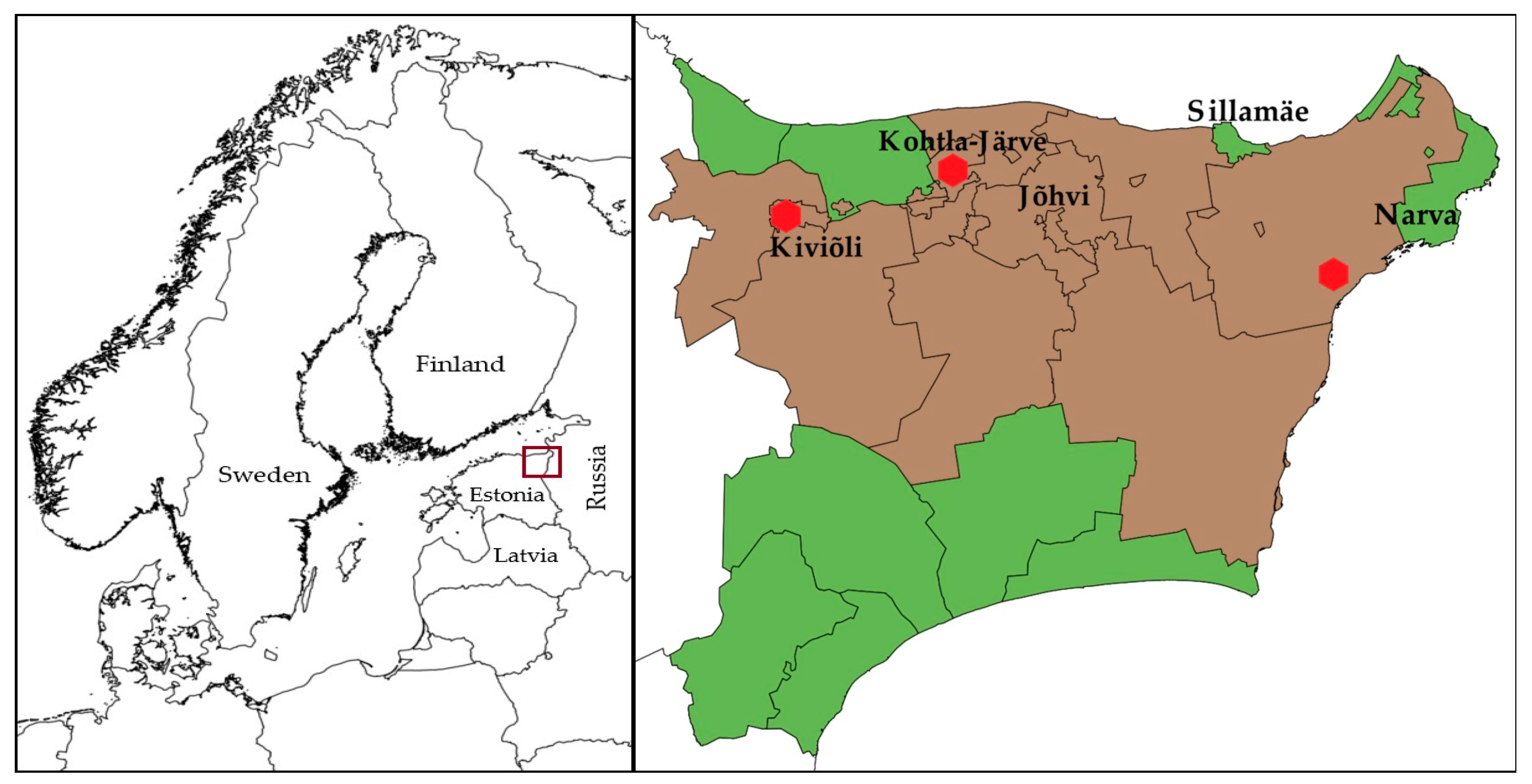
2. Materials and Methods
2.1. Exposure Assessment
2.2. Statistical Analyses
3. Results
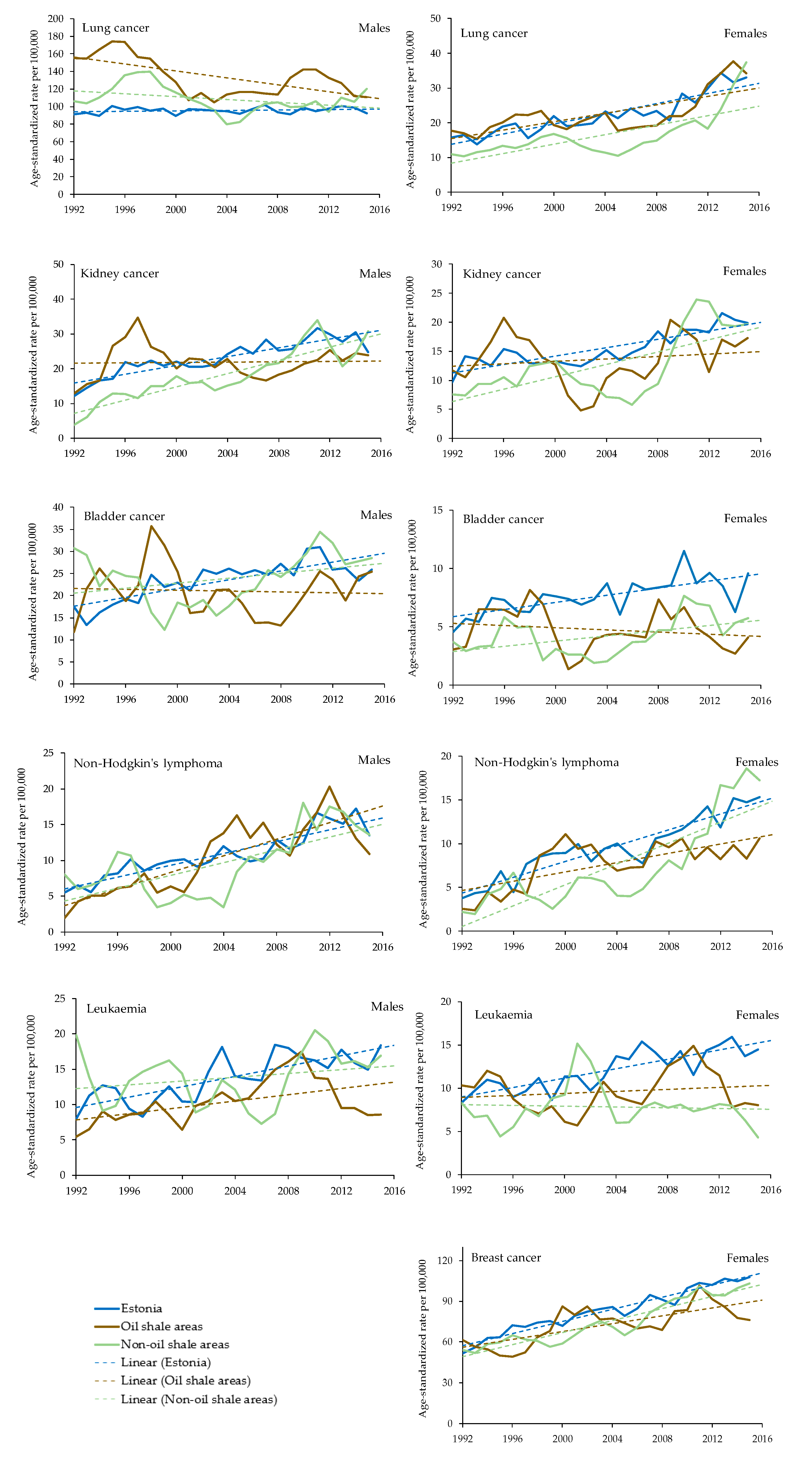
3.1. Lung Cancer
3.2. Kidney Cancer
3.3. Urinary Bladder Cancer
3.4. Non-Hodgkin’s Lymphoma
3.5. Leukaemia
3.6. Breast Cancer
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Appendix A
| Males | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lung cancer | Kidney cancer | Urinary bladder cancer | Leukaemia | Breast cancer | Non-Hodgkin’s lymphoma | Total of studied cancer sites | ||||||||||||||||||||||
| Ida-Viru County | Estonia | Ida-Viru County | Estonia | Ida-Viru County | Estonia | Ida-Viru County | Estonia | Ida-Viru County | Estonia | Ida-Viru County | Estonia | Ida-Viru County | Estonia | |||||||||||||||
| n | % | n | % | n | % | n | n | % | n | % | n | % | n | n | % | n | % | n | % | n | % | n | % | n | % | n | % | |
| Total | 2283 | 65.1 | 14740 | 57.3 | 393 | 11.2 | 3549 | 13.8 | 414 | 11.8 | 3580 | 13.9 | 231 | 6.6 | 2114 | 8.2 | 10 | 0.3 | 105 | 0.4 | 176 | 5.0 | 1646 | 6.4 | 3507 | 25734 | ||
| Age at diagnosis | ||||||||||||||||||||||||||||
| 0‒19 | 0 | 0.0 | 2 | 0.0 | 5 | 1.3 | 29 | 0.8 | 1 | 0.2 | 3 | 0.1 | 19 | 8.2 | 136 | 6.4 | 0 | 0.0 | 0 | 0.0 | 7 | 4.0 | 60 | 3.6 | 32 | 0.9 | 230 | 0.9 |
| 20‒29 | 2 | 0.1 | 9 | 0.1 | 0 | 0.0 | 9 | 0.3 | 0 | 0.0 | 4 | 0.1 | 7 | 3.0 | 52 | 2.5 | 0 | 0.0 | 0 | 0.0 | 16 | 9.1 | 76 | 4.6 | 25 | 0.7 | 150 | 0.6 |
| 30‒39 | 5 | 0.2 | 69 | 0.5 | 9 | 2.3 | 72 | 2.0 | 8 | 1.9 | 34 | 0.9 | 5 | 2.2 | 59 | 2.8 | 0 | 0.0 | 1 | 1.0 | 14 | 8.0 | 99 | 6.0 | 41 | 1.2 | 334 | 1.3 |
| 40‒49 | 102 | 4.5 | 605 | 4.1 | 37 | 9.4 | 302 | 8.5 | 11 | 2.7 | 121 | 3.4 | 14 | 6.1 | 133 | 6.3 | 1 | 10.0 | 7 | 6.7 | 22 | 12.5 | 142 | 8.6 | 187 | 5.3 | 1310 | 5.1 |
| 50‒59 | 447 | 19.6 | 2876 | 19.5 | 96 | 24.4 | 799 | 22.5 | 69 | 16.7 | 478 | 13.4 | 42 | 18.2 | 313 | 14.8 | 3 | 30.0 | 14 | 13.3 | 25 | 14.2 | 259 | 15.7 | 682 | 19.4 | 4739 | 18.4 |
| 60‒69 | 925 | 40.5 | 5621 | 38.1 | 144 | 36.6 | 1153 | 32.5 | 132 | 31.9 | 1104 | 30.8 | 64 | 27.7 | 560 | 26.5 | 3 | 30.0 | 36 | 34.3 | 41 | 23.3 | 413 | 25.1 | 1309 | 37.3 | 8887 | 34.5 |
| 70‒79 | 659 | 28.9 | 4389 | 29.8 | 76 | 19.3 | 911 | 25.7 | 147 | 35.5 | 1245 | 34.8 | 63 | 27.3 | 617 | 29.2 | 3 | 30.0 | 32 | 30.5 | 41 | 23.3 | 427 | 25.9 | 989 | 28.2 | 7621 | 29.6 |
| 80+ | 143 | 6.3 | 1169 | 7.9 | 26 | 6.6 | 274 | 7.7 | 46 | 11.1 | 591 | 16.5 | 17 | 7.4 | 244 | 11.5 | 0 | 0.0 | 15 | 14.3 | 10 | 5.7 | 170 | 10.3 | 242 | 6.9 | 2463 | 9.6 |
| Ethnicity | ||||||||||||||||||||||||||||
| Estonian | 544 | 23.8 | 9538 | 64.7 | 91 | 23.2 | 2422 | 68.2 | 105 | 25.4 | 2414 | 67.4 | 50 | 21.6 | 1496 | 70.8 | 4 | 40.0 | 68 | 64.8 | 41 | 23.3 | 1132 | 68.8 | 835 | 23.8 | 17070 | 66.3 |
| Russian | 1502 | 65.8 | 4169 | 28.3 | 254 | 64.6 | 897 | 25.3 | 268 | 64.7 | 951 | 26.6 | 148 | 64.1 | 471 | 22.3 | 5 | 50.0 | 27 | 25.7 | 121 | 68.8 | 383 | 23.3 | 2298 | 65.5 | 6898 | 26.8 |
| Ukrainian | 58 | 2.5 | 344 | 2.3 | 12 | 3.1 | 61 | 1.7 | 10 | 2.4 | 71 | 2.0 | 12 | 5.2 | 51 | 2.4 | 1 | 10.0 | 3 | 2.9 | 4 | 2.3 | 45 | 2.7 | 97 | 2.8 | 575 | 2.2 |
| Belorussian | 68 | 3.0 | 184 | 1.2 | 12 | 3.1 | 40 | 1.1 | 11 | 2.7 | 34 | 0.9 | 5 | 2.2 | 21 | 1.0 | 0 | 0.0 | 2 | 1.9 | 3 | 1.7 | 16 | 1.0 | 99 | 2.8 | 297 | 1.2 |
| Finnish | 24 | 1.1 | 106 | 0.7 | 8 | 2.0 | 23 | 0.6 | 1 | 0.2 | 13 | 0.4 | 1 | 0.4 | 12 | 0.6 | 0 | 0.0 | 0 | 0.0 | 2 | 1.1 | 8 | 0.5 | 36 | 1.0 | 162 | 0.6 |
| Other | 87 | 3.8 | 399 | 2.7 | 16 | 4.1 | 106 | 3.0 | 19 | 4.6 | 97 | 2.7 | 15 | 6.5 | 63 | 3.0 | 0 | 0.0 | 5 | 4.8 | 5 | 2.8 | 62 | 3.8 | 142 | 4.0 | 732 | 2.8 |
| Females | ||||||||||||||||||||||||||||
| Total | 505 | 15.6 | 3910 | 14.8 | 337 | 10.4 | 2716 | 10.3 | 124 | 3.8 | 1345 | 5.1 | 226 | 7.0 | 2132 | 8.1 | 1868 | 57.6 | 14169 | 56.7 | 185 | 5.7 | 1669 | 6.3 | 3245 | 26391 | ||
| Age at diagnosis | ||||||||||||||||||||||||||||
| 0‒19 | 0 | 0.0 | 1 | 0.0 | 1 | 0.3 | 29 | 1.1 | 0 | 0.0 | 1 | 0.1 | 16 | 7.1 | 142 | 6.7 | 1 | 0.1 | 1 | 0.0 | 5 | 2.7 | 26 | 1.6 | 23 | 0.7 | 200 | 0.8 |
| 20‒29 | 2 | 0.4 | 6 | 0.2 | 0 | 0.0 | 6 | 0.2 | 0 | 0.0 | 3 | 0.2 | 2 | 0.9 | 38 | 1.8 | 9 | 0.5 | 71 | 0.5 | 3 | 1.6 | 44 | 2.6 | 16 | 0.5 | 168 | 0.6 |
| 30‒39 | 8 | 1.6 | 37 | 0.9 | 7 | 2.1 | 36 | 1.3 | 1 | 0.8 | 8 | 0.6 | 4 | 1.8 | 58 | 2.7 | 93 | 5.0 | 636 | 4.5 | 11 | 5.9 | 70 | 4.2 | 124 | 3.8 | 845 | 3.2 |
| 40‒49 | 14 | 2.8 | 141 | 3.6 | 20 | 5.9 | 111 | 4.1 | 8 | 6.5 | 32 | 2.4 | 9 | 4.0 | 116 | 5.4 | 308 | 16.5 | 2321 | 16.4 | 16 | 8.6 | 95 | 5.7 | 375 | 11.6 | 2816 | 10.7 |
| 50‒59 | 74 | 14.7 | 539 | 13.8 | 52 | 15.4 | 448 | 16.5 | 13 | 10.5 | 128 | 9.5 | 33 | 14.6 | 257 | 12.1 | 493 | 26.4 | 3570 | 25.2 | 31 | 16.8 | 195 | 11.7 | 696 | 21.4 | 5137 | 19.5 |
| 60‒69 | 153 | 30.3 | 1140 | 29.2 | 117 | 34.7 | 805 | 29.6 | 30 | 24.2 | 292 | 21.7 | 48 | 21.2 | 487 | 22.8 | 434 | 23.2 | 3534 | 24.9 | 45 | 24.3 | 406 | 24.3 | 827 | 25.5 | 6664 | 25.3 |
| 70‒79 | 162 | 32.1 | 1354 | 34.6 | 102 | 30.3 | 885 | 32.6 | 45 | 36.3 | 490 | 36.4 | 80 | 35.4 | 637 | 29.9 | 391 | 20.9 | 3065 | 21.6 | 51 | 27.6 | 535 | 32.1 | 831 | 25.6 | 6966 | 26.4 |
| 80+ | 92 | 18.2 | 692 | 17.7 | 38 | 11.3 | 396 | 14.6 | 27 | 21.8 | 391 | 29.1 | 34 | 15.0 | 397 | 18.6 | 139 | 7.4 | 1421 | 10.0 | 23 | 12.4 | 298 | 17.9 | 353 | 10.9 | 3595 | 13.6 |
| Ethnicity | ||||||||||||||||||||||||||||
| Estonian | 112 | 22.2 | 2657 | 68.0 | 90 | 26.7 | 1862 | 68.6 | 25 | 20.2 | 961 | 71.4 | 45 | 19.9 | 1479 | 69.4 | 344 | 18.4 | 9472 | 66.9 | 46 | 24.9 | 1166 | 69.9 | 662 | 20.4 | 17597 | 66.7 |
| Russian | 335 | 66.3 | 1026 | 26.2 | 224 | 66.5 | 725 | 26.7 | 86 | 69.4 | 324 | 24.1 | 159 | 70.4 | 547 | 25.7 | 1361 | 72.9 | 4315 | 30.5 | 122 | 65.9 | 414 | 24.8 | 2287 | 70.5 | 7351 | 27.9 |
| Ukrainian | 13 | 2.6 | 55 | 1.4 | 4 | 1.2 | 37 | 1.4 | 4 | 3.2 | 14 | 1.0 | 3 | 1.3 | 21 | 1.0 | 46 | 2.5 | 237 | 1.7 | 6 | 3.2 | 31 | 1.9 | 76 | 2.3 | 395 | 1.5 |
| Belorussian | 17 | 3.4 | 38 | 1.0 | 4 | 1.2 | 17 | 0.6 | 3 | 2.4 | 8 | 0.6 | 6 | 2.7 | 18 | 0.8 | 52 | 2.8 | 189 | 1.3 | 7 | 3.8 | 22 | 1.3 | 89 | 2.7 | 292 | 1.1 |
| Finnish | 7 | 1.4 | 25 | 0.6 | 3 | 0.9 | 20 | 0.7 | 2 | 1.6 | 10 | 0.7 | 3 | 1.3 | 12 | 0.6 | 18 | 1.0 | 106 | 0.7 | 2 | 1.1 | 14 | 0.8 | 35 | 1.1 | 187 | 0.7 |
| Other | 21 | 4.2 | 109 | 2.8 | 12 | 3.6 | 55 | 2.0 | 4 | 3.2 | 28 | 2.1 | 10 | 4.4 | 55 | 2.6 | 47 | 2.5 | 300 | 2.1 | 2 | 1.1 | 22 | 1.3 | 96 | 3.0 | 569 | 2.2 |
References
- Global Burden of Disease 2017 Causes of Death Collaborators, A.R. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Lond. Engl. 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Dyba, T.; Randi, G.; Bettio, M.; Gavin, A.; Visser, O.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur. J. Cancer Oxf. Engl. 1990 2018, 103, 356–387. [Google Scholar] [CrossRef] [PubMed]
- Grasgruber, P.; Hrazdira, E.; Sebera, M.; Kalina, T. Cancer Incidence in Europe: An Ecological Analysis of Nutritional and Other Environmental Factors. Front. Oncol. 2018, 8, 151. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.-L.; Innos, K.; Härmaorg, P.; Mägi, M.; Baburin, A.; Aareleid, T. Vähihaigestumus 2009-2013 ja 20 aasta trendid Eestis. Eesti Arst 2017, 96, 11–20. [Google Scholar]
- You, W.; Henneberg, M. Cancer incidence increasing globally: The role of relaxed natural selection. Evol. Appl. 2017, 11, 140–152. [Google Scholar] [CrossRef]
- Loomis, D.; Grosse, Y.; Lauby-Secretan, B.; Ghissassi, F.E.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Baan, R.; Mattock, H.; Straif, K. The carcinogenicity of outdoor air pollution. Lancet Oncol. 2013, 14, 1262–1263. [Google Scholar] [CrossRef]
- WHO Regional Office for Europe. Contaminated Sites and Health; WHO Regional Office for Europe: Copenhagen, Denmark, 2012. [Google Scholar]
- Vaimann, T.; Risthein, E. Eesti Energeetika 100 Aastat; Post Factum: Tallinn, Estonia, 2018. [Google Scholar]
- Habicht, K. Oil-shale mines and their realizable production. Oil Shale 1994, 11, 259–264. [Google Scholar]
- Statistics Estonia. Statistical Yearbook of Estonia; Statistics Estonia: Tallinn, Estonia, 2016. [Google Scholar]
- OECD. OECD Environmental Performance Reviews; OECD: Tallinn, Estonia, 2017. [Google Scholar]
- Office of Technology Assessment. An Assessment of Oil Shale Technologies; Office of Technology Assessment: Washington, DC, USA, 1980. [Google Scholar]
- Raukas, A.; Punning, J.-M. Environmental problems in the Estonian oil shale industry. Energy Environ. Sci. 2009, 2, 723–728. [Google Scholar] [CrossRef]
- Idavain, J.; Julge, K.; Rebane, T.; Lang, A.; Orru, H. Respiratory symptoms, asthma, and levels of fractional exhaled nitric oxide in schoolchildren in the industrial areas of Estonia. Sci. Total Environ. 2019, 650, 65–72. [Google Scholar] [CrossRef]
- Orru, H.; Idavain, J.; Pindus, M.; Orru, K.; Kesanurm, K.; Lang, A.; Tomasova, J. Residents’ self-reported health effects and annoyance in relation to air pollution exposure in an industrial area in Eastern-Estonia. Int. J. Environ. Res. Public Health 2018, 15, 252. [Google Scholar] [CrossRef]
- Purde, M.; Rahu, M. Cancer patterns in the oil shale area of the Estonian S.S.R. Environ. Health Perspect. 1979, 30, 209–210. [Google Scholar] [CrossRef][Green Version]
- Этлин, С.Н. Гигиенические Оснoвы Охраны Атмoсфернoгo вoздуха в Райoне Размещения Предприятий Сланцевoй Химии и Энергетики; Академия Медицинских Наук СССР: Мoсква, Russia, 1989. [Google Scholar]
- Thomson, H.; Rahu, M.; Aareleid, T.; Gornoi, K. Cancer in Estonia 1968–1992. Incidence, Mortality, Prevalence, Survival.; Institute of Experimental and Clinical Medicine: Tallinn, Estonia, 1996. [Google Scholar]
- National Institute for Health Development. Overview of Health and Wellbeing in Ida-Viru County, 2000–2010; National Institute for Health Development: Tallinn, Estonia, 2012. [Google Scholar]
- International Agency for Research on Cancer. Polynuclear aromatic compounds, Part 4, Bitumens, coal-tars and derived products, shale-oils and soots. IARC Monogr. Eval. Carcinog. Risk Chem. Hum. 1985, 35, 1–247. [Google Scholar]
- Kivisto, H.; Pekari, K.; Peltonen, K.; Svinhufvud, J.; Veidebaum, T.; Sorsa, M.; Aitio, A. Biological monitoring of exposure to benzene in the production of benzene and in a cokery. Sci. Total Environ. 1997, 199, 49–63. [Google Scholar] [CrossRef]
- Kuljukka, T.; Vaaranrinta, R.; Veidebaum, T.; Sorsa, M.; Peltonen, K. Exposure to PAH compounds among cokery workers in the oil shale industry. Environ. Health Perspect. 1996, 104, 539–541. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. Polynuclear aromatic compounds, part 1, chemical, environmental, and experimental data. IARC Monogr. Eval. Carcinog. Risk Chem. Man IARC Sci. Publ. 1983, 32, 33–451. [Google Scholar]
- Loomis, D.; Guyton, K.Z.; Grosse, Y.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Vilahur, N.; Mattock, H.; Straif, K. Carcinogenicity of benzene. Lancet Oncol. 2017, 18, 1574–1575. [Google Scholar] [CrossRef]
- Bogovski, P.; Veidebaum, T.; Tamme, J.; Poldvere, E. Carcinogenicity and mutagenicity of the shale-oil produced in the Estonian Kiviter retort. IARC Sci. Publ. 1990, 354–362. [Google Scholar]
- Fernandez-Navarro, P.; Garcia-Perez, J.; Ramis, R.; Boldo, E.; Lopez-Abente, G. Industrial pollution and cancer in Spain: An important public health issue. Env. Res. 2017, 159, 555–563. [Google Scholar] [CrossRef]
- Lin, C.-K.; Hung, H.-Y.; Christiani, D.C.; Forastiere, F.; Lin, R.-T. Lung cancer mortality of residents living near petrochemical industrial complexes: A meta-analysis. Environ. Health 2017, 16, 101. [Google Scholar] [CrossRef]
- Pirastu, R.; Comba, P.; Iavarone, I.; Zona, A.; Conti, S.; Minelli, G.; Manno, V.; Mincuzzi, A.; Minerba, S.; Forastiere, F.; et al. Environment and Health in Contaminated Sites: The Case of Taranto, Italy. J. Environ. Public Health 2013, 2013, 20. [Google Scholar] [CrossRef]
- Yuan, T.-H.; Shen, Y.-C.; Shie, R.-H.; Hung, S.-H.; Chen, C.-F.; Chan, C.-C. Increased cancers among residents living in the neighborhood of a petrochemical complex: A 12-year retrospective cohort study. Int. J. Hyg. Environ. Health 2018, 221, 308–314. [Google Scholar] [CrossRef]
- Comba, P.; Ricci, P.; Iavarone, I.; Pirastu, R.; Buzzoni, C.; Fusco, M.; Ferretti, S.; Fazzo, L.; Pasetto, R.; Zona, A.; et al. Cancer incidence in Italian contaminated sites. Ann. Ist. Super. Sanita 2014, 50, 186–191. [Google Scholar] [CrossRef]
- Marinaccio, A.; Belli, S.; Binazzi, A.; Scarselli, A.; Massari, S.; Bruni, A.; Conversano, M.; Crosignani, P.; Minerba, A.; Zona, A.; et al. Residential proximity to industrial sites in the area of Taranto (Southern Italy). A case-control cancer incidence study. Ann. Ist. Super. Sanita 2011, 47, 192–199. [Google Scholar] [CrossRef]
- Cong, X. Air pollution from industrial waste gas emissions is associated with cancer incidences in Shanghai, China. Environ. Sci. Pollut. Res. 2018, 25, 13067–13078. [Google Scholar] [CrossRef]
- Gensburg, L.J.; Pantea, C.; Kielb, C.; Fitzgerald, E.; Stark, A.; Kim, N. Cancer incidence among former Love Canal residents. Environ. Health Perspect. 2009, 117, 1265–1271. [Google Scholar] [CrossRef]
- Garcia-Perez, J.; Perez-Abad, N.; Lope, V.; Castello, A.; Pollan, M.; Gonzalez-Sanchez, M.; Valencia, J.L.; Lopez-Abente, G.; Fernandez-Navarro, P. Breast and prostate cancer mortality and industrial pollution. Environ. Pollut. 2016, 214, 394–399. [Google Scholar] [CrossRef]
- Lewis-Michl, E.L.; Melius, J.M.; Kallenbach, L.R.; Ju, C.L.; Talbot, T.O.; Orr, M.F.; Lauridsen, P.E. Breast cancer risk and residence near industry or traffic in Nassau and Suffolk Counties, Long Island, New York. Arch. Environ. Health 1996, 51, 255–265. [Google Scholar] [CrossRef]
- Pan, S.Y.; Morrison, H.; Gibbons, L.; Zhou, J.; Wen, S.W.; DesMeules, M.; Mao, Y. Breast cancer risk associated with residential proximity to industrial plants in Canada. J. Occup. Environ. Med. 2011, 53, 522–529. [Google Scholar] [CrossRef]
- Boberg, E.; Lessner, L.; Carpenter, D.O. The role of residence near hazardous waste sites containing benzene in the development of hematologic cancers in upstate New York. Int. J. Occup. Med. Environ. Health 2011, 24, 327–338. [Google Scholar] [CrossRef]
- Bonzini, M.; Grillo, P.; Consonni, D.; Cacace, R.; Ancona, C.; Forastiere, F.; Cocco, P.L.; Satta, G.; Boldori, L.; Carugno, M.; et al. Cancer risk in oil refinery workers: A pooled mortality study in Italy. Med. Lav. 2019, 110, 3–10. [Google Scholar] [CrossRef]
- Garcia-Perez, J.; Lopez-Cima, M.F.; Boldo, E.; Fernandez-Navarro, P.; Aragones, N.; Pollan, M.; Perez-Gomez, B.; Lopez-Abente, G. Leukemia-related mortality in towns lying in the vicinity of metal production and processing installations. Environ. Int. 2010, 36, 746–753. [Google Scholar] [CrossRef]
- Ramis, R.; Fernandez-Navarro, P.; Garcia-Perez, J.; Boldo, E.; Gomez-Barroso, D.; Lopez-Abente, G. Risk of cancer mortality in spanish towns lying in the vicinity of pollutant industries. ISRN Oncol. 2012, 2012, 614198. [Google Scholar] [CrossRef]
- Zusman, M.; Dubnov, J.; Barchana, M.; Portnov, B.A. Residential proximity to petroleum storage tanks and associated cancer risks: Double Kernel Density approach vs. zonal estimates. Sci. Total Environ. 2012, 441, 265–276. [Google Scholar] [CrossRef]
- Ramis, R.; Diggle, P.; Cambra, K.; López-Abente, G. Prostate cancer and industrial pollution: Risk around putative focus in a multi-source scenario. Environ. Int. 2011, 37, 577–585. [Google Scholar] [CrossRef]
- López-Abente, G.; García-Pérez, J.; Fernández-Navarro, P.; Boldo, E.; Ramis, R. Colorectal cancer mortality and industrial pollution in Spain. BMC Public Health 2012, 12, 589. [Google Scholar] [CrossRef]
- Iavarone, I.; Buzzoni, C.; Stoppa, G.; Steliarova-Foucher, E. Cancer incidence in children and young adults living in industrially contaminated sites: From the Italian experience to the development of an international surveillance system. Epidemiol. Prev. 2018, 42, 76–85. [Google Scholar] [CrossRef]
- Ortega-Garcia, J.A.; Lopez-Hernandez, F.A.; Carceles-Alvarez, A.; Fuster-Soler, J.L.; Sotomayor, D.I.; Ramis, R. Childhood cancer in small geographical areas and proximity to air-polluting industries. Environ. Res. 2017, 156, 63–73. [Google Scholar] [CrossRef]
- Põder, J. Eesti Vähiregistri Andmete Täielikkuse Uuring, Tartu Ülikool; Tervishoiu Instituut: Tartu, Estonia, 2015. [Google Scholar]
- National Institute for Health Development. Estonian Cancer Registry. Available online: https://tai.ee/en/r-and-d/registers/estonian-cancer-registry (accessed on 14 May 2020).
- WHO. International Statistical Classification of Diseases and Related Health Problems; 10th revision; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Attfield, M.D.; Schleiff, P.L.; Lubin, J.H.; Blair, A.; Stewart, P.A.; Vermeulen, R.; Coble, J.B.; Silverman, D.T. The Diesel Exhaust in Miners study: A cohort mortality study with emphasis on lung cancer. J. Natl. Cancer Inst. 2012, 104, 869–883. [Google Scholar] [CrossRef]
- Belli, S.; Benedetti, M.; Comba, P.; Lagravinese, D.; Martucci, V.; Martuzzi, M.; Morleo, D.; Trinca, S.; Viviano, G. Case-Control Study on Cancer Risk Associated to Residence in the Neighborhood of a Petrochemical Plant. Eur. J. Epidemiol. 2004, 19, 49–54. [Google Scholar] [CrossRef]
- Edwards, R.; Pless-Mulloli, T.; Howel, D.; Chadwick, T.; Bhopal, R.; Harrison, R.; Gribbin, H. Does living near heavy industry cause lung cancer in women? A case-control study using life grid interviews. Thorax 2006, 61, 1076–1082. [Google Scholar] [CrossRef]
- Miller, B.G.; Cowie, H.A.; Middleton, W.G.; Seaton, A. Epidemiologic studies of Scottish oil shale workers: III. Causes of death. Am. J. Ind. Med. 1986, 9, 433–446. [Google Scholar] [CrossRef]
- Statistics Estonia. 2000 Population and Housing Census. Economically Active Population; Statistics Estonia: Tallinn, Estonia, 2001. [Google Scholar]
- Tiit, E.-M.; Servinski, M. Rahva ja Eluruumide Loendus 2011. Ülevaade Eesti Maakondade Rahvastikust; Statistics Estonia: Tallinn, Estonia, 2013; p. 102. [Google Scholar]
- EuroGeographics © EuroGeographics for the Administrative Boundaries. 2020. Available online: https://ec.europa.eu/eurostat/web/gisco/geodata/reference-data/administrative-units-statistical-units/countries (accessed on 20 March 2020).
- Estonian Land Board © Estonian Land Board for the administrative boundaries. 2019. Available online: https://geoportaal.maaamet.ee/eng/Spatial-Data/Administrative-and-Settlement-Division-p312.html (accessed on 1 October 2019).
- Orru, K.; Roose, A.; Ainsaar, M.; Gutman, R.; Gauk, M.; Orru, H. The Environment, Health and Well-being. In Escaping the Traps? Estonian Human Development Report 2014/2015; Estonian Cooperation Assembly: Tallinn, Estonia, 2015; pp. 171–179. [Google Scholar]
- Viru Keemia Grupp. VKG Aastaraamat 2009; Viru Keema Grupp: Kohtla-Järve, Estonia, 2010; Available online: https://www.vkg.ee/wp-content/uploads/2019/07/vkg-aastaraamat-est-2009.pdf (accessed on 26 May 2020).
- Richiardi, L.; Mirabelli, D.; Calisti, R.; Ottino, A.; Ferrando, A.; Boffetta, P.; Merletti, F. Occupational exposure to diesel exhausts and risk for lung cancer in a population-based case–control study in Italy. Ann. Oncol. 2006, 17, 1842–1847. [Google Scholar] [CrossRef]
- Gamble, J.F.; Nicolich, M.J.; Boffetta, P. Lung cancer and diesel exhaust: An updated critical review of the occupational epidemiology literature. Crit. Rev. Toxicol. 2012, 42, 549–598. [Google Scholar] [CrossRef]
- Gustavsson, P.; Jakobsson, R.; Nyberg, F.; Pershagen, G.; Järup, L.; Schéele, P. Occupational Exposure and Lung Cancer Risk: A Population-based Case-Referent Study in Sweden. Am. J. Epidemiol. 2000, 152, 32–40. [Google Scholar] [CrossRef]
- Silverman, D.T.; Samanic, C.M.; Lubin, J.H.; Blair, A.E.; Stewart, P.A.; Vermeulen, R.; Coble, J.B.; Rothman, N.; Schleiff, P.L.; Travis, W.D.; et al. The Diesel Exhaust in Miners study: A nested case-control study of lung cancer and diesel exhaust. J. Natl. Cancer Inst. 2012, 104, 855–868. [Google Scholar] [CrossRef]
- Kim, J.; Peters, C.E.; Arrandale, V.H.; Labreche, F.; Ge, C.B.; McLeod, C.B.; Song, C.; Lavoue, J.; Davies, H.W.; Nicol, A.-M.; et al. Burden of lung cancer attributable to occupational diesel engine exhaust exposure in Canada. Occup. Environ. Med. 2018, 75, 617–622. [Google Scholar] [CrossRef]
- Sorensen, M.; Poole, J.; Autrup, H.; Muzyka, V.; Jensen, A.; Loft, S.; Knudsen, L.E. Benzene exposure assessed by metabolite excretion in Estonian oil shale mineworkers: Influence of glutathione s-transferase polymorphisms. Cancer Epidemiol. Biomark. Prev. 2004, 13, 1729–1735. [Google Scholar]
- Muzyka, V.; Bogovski, S.; Viitak, A.; Veidebaum, T. Alterations of heme metabolism in lymphocytes and metal content in blood plasma as markers of diesel fuels effects on human organism. Sci. Total Environ. 2002, 286, 73–81. [Google Scholar] [CrossRef]
- Knudsen, L.E.; Gaskell, M.; Martin, E.A.; Poole, J.; Scheepers, P.T.J.; Jensen, A.; Autrup, H.; Farmer, P.B. Genotoxic damage in mine workers exposed to diesel exhaust, and the effects of glutathione transferase genotypes. Mutat. Res. Toxicol. Environ. Mutagen. 2005, 583, 120–132. [Google Scholar] [CrossRef]
- Tekkel, M.; Veideman, T. Eesti Täiskasvanud Rahvastiku Tervisekäitumise Uuring, 2016; National Institute for Health Development: Tallinn, Estonia, 2017; p. 177. [Google Scholar]
- Muzyka, V.; Scheepers, P.T.; Bogovski, S.; Lang, I.; Schmidt, N.; Ryazanov, V.; Veidebaum, T. Porphyrin metabolism in lymphocytes of miners exposed to diesel exhaust at oil shale mine. Sci. Total Environ. 2004, 322, 41–50. [Google Scholar] [CrossRef]
- Aareleid, T.; Zimmermann, M.-L.; Baburin, A.; Innos, K. Divergent trends in lung cancer incidence by gender, age and histological type in Estonia: A nationwide population-based study. BMC Cancer 2017, 17, 596. [Google Scholar] [CrossRef] [PubMed]
- Dobrzynski, L.; Fornalski, K.W.; Reszczynska, J. Meta-analysis of thirty-two case-control and two ecological radon studies of lung cancer. J. Radiat. Res. 2018, 59, 149–163. [Google Scholar] [CrossRef]
- Oh, S.-S.; Koh, S.; Kang, H.; Lee, J. Radon exposure and lung cancer: Risk in nonsmokers among cohort studies. Ann. Occup. Environ. Med. 2016, 28, 11. [Google Scholar] [CrossRef]
- Yoon, J.Y.; Lee, J.-D.; Joo, S.W.; Kang, D.R. Indoor radon exposure and lung cancer: A review of ecological studies. Ann. Occup. Environ. Med. 2016, 28, 15. [Google Scholar] [CrossRef]
- Petersell, V.; Täht-Kok, K.; Karimov, M.; Milvek, H.; Nirgi, S.; Raha, M.; Saarik, K. Radon in the soil air of Estonia. J. Environ. Radioact. 2017, 166, 235–241. [Google Scholar] [CrossRef]
- Ulp, S.; Kuusemäe, K.; Talk, M.; Raudsepp, T. 10 aastat rinnavähi sõeluuringut Eestis: Samm-sammult püsititatud eesmärkide poole. Eesti Arst 2010, 89, 493–501. [Google Scholar]
- Innos, K.; Magi, M.; Tekkel, M.; Aareleid, T. Place of residence predicts breast cancer stage at diagnosis in Estonia. Eur. J. Public Health 2011, 21, 376–380. [Google Scholar] [CrossRef]
- Adegoke, O.J.; Blair, A.; Shu, X.O.; Sanderson, M.; Jin, F.; Dosemeci, M.; Addy, C.L.; Zheng, W. Occupational history and exposure and the risk of adult leukemia in Shanghai. Ann. Epidemiol. 2003, 13, 485–494. [Google Scholar] [CrossRef]
- Stenehjem, J.S.; Kjærheim, K.; Rabanal, K.S.; Grimsrud, T.K. Cancer incidence among 41 000 offshore oil industry workers. Occup. Med. 2014, 64, 539–545. [Google Scholar] [CrossRef][Green Version]
- Lai, T.; Leinsalu, M. Trends and Inequalities in Mortality of Noncommunicable Diseases. Case Study for Estonia; WHO Regional Office for Europe: Copenhagen, Denmark, 2015. [Google Scholar]
- Metsoja, A.; Nelis, L.; Nurk, E. WHO Childhood Obesity Surveillance Initiative (COSI). Estonian Study Report for the Academic Year 2015/2016; National Institute for Health Development: Tallinn, Estonia, 2017. [Google Scholar]
- Leinsalu, M.; Stirbu, I.; Vågerö, D.; Kalėdienė, R.; Kovács, K.; Wojtyniak, B.; Wróblewska, W.; Mackenbach, J.P.; Kunst, A.E. Educational inequalities in mortality in four Eastern European countries: Divergence in trends during the post-communist transition from 1990 to 2000. Int. J. Epidemiol. 2008, 38, 512–525. [Google Scholar] [CrossRef] [PubMed]
- Statistics Estonia. 2011 Census. 2011. Available online: http://pub.stat.ee/px-web.2001/I_Databas/Population_census/PHC2011/04Study_migration/04Study_migration.asp (accessed on 19 April 2020).
- White, M.C.; Holman, D.M.; Boehm, J.E.; Peipins, L.A.; Grossman, M.; Jane Henley, S. Age and Cancer Risk: A Potentially Modifiable Relationship. Am. J. Prev. Med. 2014, 46, S7–S15. [Google Scholar] [CrossRef] [PubMed]
- Lang, K. Ethnic differences in cancer incidence in Estonia: Two cross-sectional unlinked census-based cancer incidence analyses. Popul. Health Metr. 2009, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Elliott, P.; Savitz, D.A. Design Issues in Small-Area Studies of Environment and Health. Environ. Health Perspect. 2008, 116, 1098–1104. [Google Scholar] [CrossRef]
- Pirastu, R.; Pasetto, R.; Zona, A.; Ancona, C.; Iavarone, I.; Martuzzi, M.; Comba, P. The Health Profile of Populations Living in Contaminated Sites: Sentieri Approach. J. Environ. Public Health 2013, 2013, 13. [Google Scholar] [CrossRef]
- Savitz, D.A. Commentary: A Niche for Ecologic Studies in Environmental Epidemiology. Epidemiology 2012, 23, 53–54. [Google Scholar] [CrossRef]
- Wakefield, J. Ecologic Studies Revisited. Annu. Rev. Public Health 2008, 29, 75–90. [Google Scholar] [CrossRef]
| Study Area | Average Population in Estonia | Average Population in Ida-Viru County | Average Population in oil Shale Areas | Average Population in non-oil Shale Areas |
|---|---|---|---|---|
| n (%) | n (%) | n (%) | n (%) | |
| Total | 1,376,819 | 175,765 | 67,230 | 107,452 |
| Gender | ||||
| Male | 640,146 (46.5) | 80,388 (45.7) | 31,002 (46.1) | 49,189 (45.8) |
| Female | 736,673 (53.5) | 95,377 (54.3) | 36,228 (53.4) | 58,263 (54.2) |
| Age groups | ||||
| 0‒19 | 330,460 (24.1) | 38,374 (21.8) | 14,201 (19.4) | 19,518 (19.3) |
| 20‒29 | 192,720 (14.1) | 21,654 (12.3) | 9220 (12.6) | 12,512 (12.4) |
| 30‒39 | 193,075 (14.1) | 24,160 (13.7) | 8991 (12.3) | 13,093 (13.0) |
| 40‒49 | 189,606 (13.8) | 26,418 (15.0) | 11,071 (15.1) | 15,482 (15.3) |
| 50‒59 | 174,188 (12.7) | 24,706 (14.1) | 11,168 (15.3) | 15,181 (15.0) |
| 60‒69 | 148,577 (10.8) | 21,373 (12.2) | 8490 (11.6) | 12,184 (12.1) |
| 70‒79 | 101,568 (7.4) | 13,987 (8.0) | 7161 (9.8) | 9732 (9.6) |
| 80+ | 46,616 (3.4) | 5093 (2.9) | 2885 (3.8) | 3366 (3.3) |
| Ethnicity | ||||
| Estonian | 910,842 (69.6) | 28,697 (19.3) | n/a | n/a |
| Russian | 332,566 (25.4) | 108,778 (73.3) | n/a | n/a |
| Ukrainian | 23,005 (1.8) | 3394 (2.3) | n/a | n/a |
| Belarusian | 12,514 (1.0) | 3325 (2.2) | n/a | n/a |
| Finnish | 7690 (0.6) | 1380 (0.9) | n/a | n/a |
| Other | 22,264 (1.7) | 2854 (1.9) | n/a | n/a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Idavain, J.; Lang, K.; Tomasova, J.; Lang, A.; Orru, H. Cancer Incidence Trends in the Oil Shale Industrial Region in Estonia. Int. J. Environ. Res. Public Health 2020, 17, 3833. https://doi.org/10.3390/ijerph17113833
Idavain J, Lang K, Tomasova J, Lang A, Orru H. Cancer Incidence Trends in the Oil Shale Industrial Region in Estonia. International Journal of Environmental Research and Public Health. 2020; 17(11):3833. https://doi.org/10.3390/ijerph17113833
Chicago/Turabian StyleIdavain, Jane, Katrin Lang, Jelena Tomasova, Aavo Lang, and Hans Orru. 2020. "Cancer Incidence Trends in the Oil Shale Industrial Region in Estonia" International Journal of Environmental Research and Public Health 17, no. 11: 3833. https://doi.org/10.3390/ijerph17113833
APA StyleIdavain, J., Lang, K., Tomasova, J., Lang, A., & Orru, H. (2020). Cancer Incidence Trends in the Oil Shale Industrial Region in Estonia. International Journal of Environmental Research and Public Health, 17(11), 3833. https://doi.org/10.3390/ijerph17113833





