Contribution of Chronic Fatigue to Psychosocial Status and Quality of Life in Spanish Women Diagnosed with Endometriosis
Abstract
1. Introduction
2. Material and Methods
2.1. Study Population
2.2. Assessment of Self-Reported Intensity of Chronic Fatigue and Pain
2.3. Psychosocial Assessment
2.4. Quality of Life
2.5. Statistical Analysis
3. Results
3.1. Intensity of Chronic Fatigue and Pain in Spanish Women Diagnosed with Endometriosis
3.2. Psychosocial Impairments and Quality of Life in Spanish Women Diagnosed with Endometriosis
3.3. Contribution of Fatigue Intensity to Psychosocial Impairment in Spanish Women
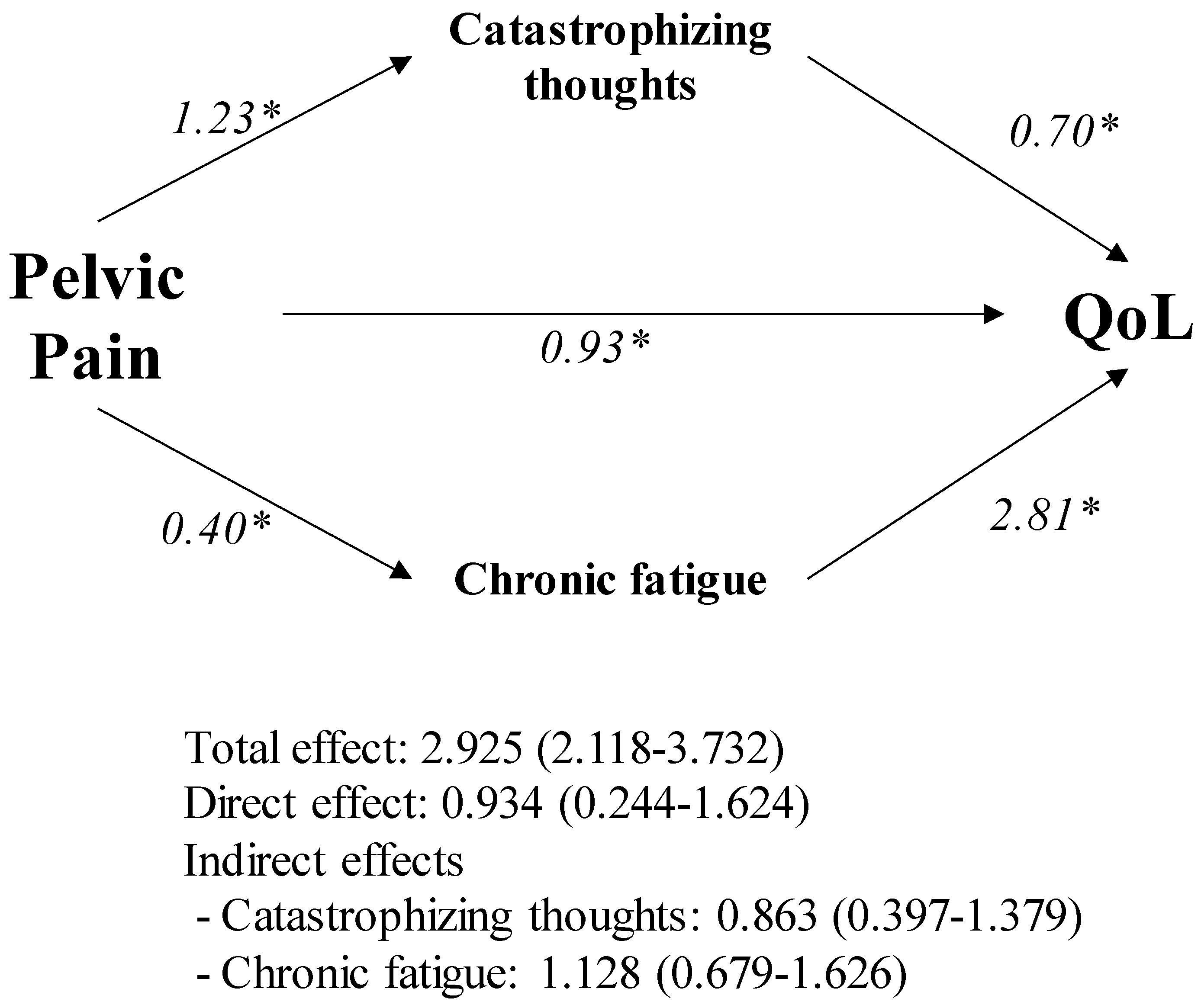
3.4. Contribution of Pain, Fatigue and Psychosocial Impairment to Quality of Life in Spanish Women
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vercellini, P.; Viganò, P.; Somigliana, E.; Fedele, L. Endometriosis: Pathogenesis and treatment. Nat. Rev. Endocrinol. 2014, 10, 261. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, V.H.; Weil, C.; Chodick, G.; Shalev, V. Epidemiology of endometriosis: A large population-based database study from a healthcare provider with 2 million members. BJOG 2018, 125, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Soliman, A.M.; Yang, H.; Du, E.X.; Kelley, C.; Winkel, C. The direct and indirect costs associated with endometriosis: A systematic literature review. Hum. Reprod. 2016, 31, 712–722. [Google Scholar] [CrossRef] [PubMed]
- Berkley, K.J.; Rapkin, A.J.; Papka, R.E. The pains of endometriosis. Science 2005, 308, 1587–1589. [Google Scholar] [CrossRef] [PubMed]
- Loeser, J.D.; Treede, R.-D. The Kyoto protocol of IASP Basic Pain Terminology. Pain 2008, 137, 473–477. [Google Scholar] [CrossRef] [PubMed]
- Soliman, A.M.; Coyne, K.S.; Zaiser, E.; Castelli-Haley, J.; Fuldeore, M.J. The burden of endometriosis symptoms on health-related quality of life in women in the United States: A cross-sectional study. J. Psychosom. Obstet. Gynaecol. 2017, 38, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Facchin, F.; Barbara, G.; Saita, E.; Mosconi, P.; Roberto, A.; Fedele, L.; Vercellini, P. Impact of endometriosis on quality of life and mental health: Pelvic pain makes the difference. J. Psychosom. Obstet. Gynaecol. 2015, 36, 135–141. [Google Scholar] [CrossRef]
- Nnoaham, K.E.; Hummelshoj, L.; Webster, P.; d’Hooghe, T.; de Cicco Nardone, F.; de Cicco Nardone, C.; Jenkinson, C.; Kennedy, S.H.; Zondervan, K.T.; World Endometriosis Research Foundation Global Study of Women’s Health consortium. Impact of endometriosis on quality of life and work productivity: A multicenter study across ten countries. Fertil. Steril. 2011, 96, 366–373.e8. [Google Scholar] [CrossRef]
- Eriksen, H.-L.F.; Gunnersen, K.F.; Sørensen, J.-A.; Munk, T.; Nielsen, T.; Knudsen, U.B. Psychological aspects of endometriosis: Differences between patients with or without pain on four psychological variables. Eur. J. Obstet. Gynecol. Reprod. Biol. 2008, 139, 100–105. [Google Scholar] [CrossRef]
- Nunes, F.R.; Ferreira, J.M.; Bahamondes, L. Pain threshold and sleep quality in women with endometriosis. Eur. J. Pain 2015, 19, 15–20. [Google Scholar] [CrossRef]
- Ramin-Wright, A.; Schwartz, A.S.K.; Geraedts, K.; Rauchfuss, M.; Wölfler, M.M.; Haeberlin, F.; von Orelli, S.; Eberhard, M.; Imthurn, B.; Imesch, P. Fatigue–a symptom in endometriosis. Hum. Reprod. 2018, 33, 1459–1465. [Google Scholar] [CrossRef] [PubMed]
- Gilmour, J.A.; Huntington, A.; Wilson, H.V. The impact of endometriosis on work and social participation. Int. J. Nurs. Pract. 2008, 14, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Jenkinson, C.; Kennedy, S. The impact of endometriosis upon quality of life: A qualitative analysis. J. Psychosom. Obstet. Gynaecol. 2004, 25, 123–133. [Google Scholar] [CrossRef] [PubMed]
- Pappalardo, A.; Reggio, E.; Patti, F.; Reggio, A. Management of fatigue in multiple sclerosis. Eur. J. Phys. Rehabil. Med. 2003, 39, 147. [Google Scholar]
- Ahlberg, K.; Ekman, T.; Gaston-Johansson, F.; Mock, V. Assessment and management of cancer-related fatigue in adults. Lancet 2003, 362, 640–650. [Google Scholar] [CrossRef]
- Omdal, R.; Koldingsnes, W.; Husby, G.; Mellgren, S.I. Fatigue in patients with systemic lupus erythematosus: The psychosocial aspects. J. Rheumatol. 2003, 30, 283–287. [Google Scholar]
- Trojan, D.; Arnold, D.; Collet, J.; Shapiro, S.; Bar-Or, A.; Robinson, A.; Le Cruguel, J.; Ducruet, T.; Narayanan, S.; Arcelin, K. Fatigue in multiple sclerosis: Association with disease-related, behavioural and psychosocial factors. Mult. Scler. 2007, 13, 985–995. [Google Scholar] [CrossRef]
- Piper, B.F.; Dibble, S.L.; Dodd, M.J.; Weiss, M.C.; Slaughter, R.E.; Paul, S.M. The revised Piper Fatigue Scale: Psychometric evaluation in women with breast cancer. Oncol. Nurs. Forum. 1998, 25, 677–684. [Google Scholar]
- Cantarero-Villanueva, I.; Fernández-Lao, C.; Díaz-Rodríguez, L.; Cuesta-Vargas, A.I.; Fernández-de-las-Peñas, C.; Piper, B.F.; Arroyo-Morales, M. The Piper Fatigue Scale-Revised: Translation and psychometric evaluation in Spanish-speaking breast cancer survivors. Qual. Life Res. 2014, 23, 271–276. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, F.; Zhang, H.; Yu, X.; Cong, Y. Effects of nurse-led home-based exercise & cognitive behavioral therapy on reducing cancer-related fatigue in patients with ovarian cancer during and after chemotherapy: A randomized controlled trial. Int. J. Nurs. Stud. 2018, 78, 52–60. [Google Scholar]
- Pozehl, B.; Duncan, K.; Hertzog, M. The effects of exercise training on fatigue and dyspnea in heart failure. Eur. J. Cardiovasc. Nurs. 2008, 7, 127–132. [Google Scholar] [CrossRef] [PubMed]
- O’Regan, P.; Hegarty, J. The importance of self-care for fatigue amongst patients undergoing chemotherapy for primary cancer. Eur. J. Oncol. Nurs. 2017, 28, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.P.; Turner, J.A.; Romano, J.M.; Fisher, L.D. Comparative reliability and validity of chronic pain intensity measures. Pain 1999, 83, 157–162. [Google Scholar] [CrossRef]
- Breivik, E.K.; Björnsson, G.A.; Skovlund, E. A comparison of pain rating scales by sampling from clinical trial data. Clin. J. Pain 2000, 16, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Boonstra, A.M.; Stewart, R.E.; Köke, A.J.; Oosterwijk, R.F.; Swaan, J.L.; Schreurs, K.M.; Schiphorst Preuper, H.R. Cut-off points for mild, moderate, and severe pain on the numeric rating scale for pain in patients with chronic musculoskeletal pain: Variability and influence of sex and catastrophizing. Front. Psychol. 2016, 7, 1466. [Google Scholar] [CrossRef] [PubMed]
- Forchheimer, M.B.; Richards, J.S.; Chiodo, A.E.; Bryce, T.N.; Dyson-Hudson, T.A. Cut point determination in the measurement of pain and its relationship to psychosocial and functional measures after traumatic spinal cord injury: A retrospective model spinal cord injury system analysis. Arch. Phys. Med. Rehabil. 2011, 92, 419–424. [Google Scholar] [CrossRef]
- Herrero, M.J.; Blanch, J.; Peri, J.M.; De Pablo, J.; Pintor, L.; Bulbena, A. A validation study of the hospital anxiety and depression scale (HADS) in a Spanish population. Gen. Hosp. Psychiatry 2003, 25, 277–283. [Google Scholar] [CrossRef]
- Zigmond, A.S.; Snaith, R.P. The hospital anxiety and depression scale. Acta Psychiatr Scand. 1983, 67, 361–370. [Google Scholar] [CrossRef]
- Quintana, J.; Padierna, A.; Esteban, C.; Arostegui, I.; Bilbao, A.; Ruiz, I. Evaluation of the psychometric characteristics of the Spanish version of the Hospital Anxiety and Depression Scale. Acta Psychiatr. Scand. 2003, 107, 216–221. [Google Scholar] [CrossRef]
- Bjelland, I.; Dahl, A.A.; Haug, T.T.; Neckelmann, D. The validity of the Hospital Anxiety and Depression Scale: An updated literature review. J. Psychosom. Res. 2002, 52, 69–77. [Google Scholar] [CrossRef]
- Olssøn, I.; Mykletun, A.; Dahl, A.A. The hospital anxiety and depression rating scale: A cross-sectional study of psychometrics and case finding abilities in general practice. BMC Psychiatry 2005, 5, 46. [Google Scholar] [CrossRef] [PubMed]
- Fernández, J.S. Un instrumento para evaluar la eficacia de los procedimientos de inducción de estado de ánimo: La “Escala de Valoración del Estado de Ánimo” (EVEA). Anal. Modif. Conduct. 2001, 27, 71–110. [Google Scholar]
- García, J.C.; Rodero, B.; Alda, M.; Sobradiel, N.; Montero, J.; Moreno, S. Validation of the Spanish version of the Pain Catastrophizing Scale in fibromyalgia. Med. Clin. (Barc.) 2008, 131, 487–492. [Google Scholar]
- Sullivan, M.J.; Bishop, S.R.; Pivik, J. The pain catastrophizing scale: Development and validation. Psychol. Assess. 1995, 7, 524. [Google Scholar] [CrossRef]
- Hita-Contreras, F.; Martínez-López, E.; Latorre-Román, P.A.; Garrido, F.; Santos, M.A.; Martínez-Amat, A. Reliability and validity of the Spanish version of the Pittsburgh Sleep Quality Index (PSQI) in patients with fibromyalgia. Rheumatol. Int. 2014, 34, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Buysse, D.J.; Reynolds, C.F., 3rd; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Blümel, J.E.; Binfa, L.; Cataldo, P.; Carrasco, A.; Izaguirre, H.; Sarrá, S. Índice de función sexual femenina: Un test para evaluar la sexualidad de la mujer. Rev. Chil. Obstet. Ginecol. 2004, 69, 118–125. [Google Scholar] [CrossRef]
- Rosen, C.B.; Heiman, J.; Leiblum, S.; Meston, C.; Shabsigh, R.; Ferguson, D.; D’Agostino, R., Jr. The Female Sexual Function Index (FSFI): A multidimensional self-report instrument for the assessment of female sexual function. J. Sex Marital Ther. 2000, 26, 191–208. [Google Scholar] [CrossRef]
- Wiegel, M.; Meston, C.; Rosen, R. The female sexual function index (FSFI): Cross-validation and development of clinical cutoff scores. J. Sex Marital Ther. 2005, 31, 1–20. [Google Scholar] [CrossRef]
- Quintana, J.; Cabriada, J.; de Tejada López, I.; Varona, M.; Oribe, V.; Barrios, B.; Perdigo, L.; Bilbao, A. Translation and validation of the gastrointestinal Quality of Life Index (GIQLI). Rev. Esp. Enferm. Dig. 2001, 93, 693–706. [Google Scholar]
- Nieveen Van Dijkum, E.J.; Terwee, C.B.; Oosterveld, P.; Van Der Meulen, J.H.; Gouma, D.J.; De Haes, J.C. Validation of the gastrointestinal quality of life index for patients with potentially operable periampullary carcinoma. Br. J. Surg. 2000, 87, 110–115. [Google Scholar] [CrossRef] [PubMed]
- De la Revilla, L.; Luna, J.; Bailón, E.; Medina, I. Validación del cuestionario MOS de apoyo social en Atención Primaria. Med. de Fam. 2005, 6, 10–18. [Google Scholar]
- Requena, G.C.; Salamero, M.; Gil, F. Validación del cuestionario MOS-SSS de apoyo social en pacientes con cáncer. Med. Clin. (Barc.) 2007, 128, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Kennedy, S.; Barnard, A.; Wong, J.; Jenkinson, C. Development of an endometriosis quality-of-life instrument: The Endometriosis Health Profile-30. Obstet. Gynecol. 2001, 98, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Hayes, A.F. Introduction to Mediation, Moderation, and Conditional Process Analysis: A Regression-Based Approach; Guilford Press: New York, NY, USA, 2013; Volume 17, p. 507. [Google Scholar]
- Grogan, S.; Turley, E.; Cole, J. ‘So many women suffer in silence’: A thematic analysis of women’s written accounts of coping with endometriosis. Psychol. Health 2018, 33, 1364–1378. [Google Scholar] [CrossRef] [PubMed]
- Surrey, E.S.; Soliman, A.M.; Agarwal, S.K.; Snabes, M.C.; Diamond, M.P. Impact of elagolix treatment on fatigue experienced by women with moderate to severe pain associated with endometriosis. Fertil. Steril. 2019, 112, 298–304.e3. [Google Scholar] [CrossRef] [PubMed]
- Symons, L.K.; Miller, J.E.; Kay, V.R.; Marks, R.M.; Liblik, K.; Koti, M.; Tayade, C. The immunopathophysiology of endometriosis. Trends Mol. Med. 2018, 24, 748–762. [Google Scholar] [CrossRef]
- Suryawanshi, S.; Huang, X.; Elishaev, E.; Budiu, R.A.; Zhang, L.; Kim, S.; Donnellan, N.; Mantia-Smaldone, G.; Ma, T.; Tseng, G. Complement pathway is frequently altered in endometriosis and endometriosis-associated ovarian cancer. Clin. Cancer Res. 2014, 20, 6163–6174. [Google Scholar] [CrossRef]
- Bower, J.E. Cancer-related fatigue: Links with inflammation in cancer patients and survivors. Brain Behav. Immun. 2007, 21, 863–871. [Google Scholar] [CrossRef]
- Louati, K.; Berenbaum, F. Fatigue in chronic inflammation-a link to pain pathways. Arthritis Res. Ther. 2015, 17, 254. [Google Scholar] [CrossRef]
- So, W.K.W.; Marsh, G.; Ling, W.M.; Leung, F.Y.; Lo, J.C.K.; Yeung, M.; Li, G.K.H. The symptom cluster of fatigue, pain, anxiety, and depression and the effect on the quality of life of women receiving treatment for breast cancer: A multicenter study. Oncol. Nurs. Forum. 2009, 36, E205–E214. [Google Scholar] [CrossRef] [PubMed]
- Matarese, G.; De Placido, G.; Nikas, Y.; Alviggi, C. Pathogenesis of endometriosis: Natural immunity dysfunction or autoimmune disease? Trends Mol. Med. 2003, 9, 223–228. [Google Scholar] [CrossRef]
- Marquardt, R.M.; Kim, T.H.; Shin, J.H.; Jeong, J.W. Progesterone and Estrogen Signaling in the Endometrium: What Goes Wrong in Endometriosis? Int. J. Mol. Sci. 2019, 20, 3822. [Google Scholar] [CrossRef] [PubMed]
- Matura, L.A.; Malone, S.; Jaime-Lara, R.; Riegel, B. A Systematic Review of Biological Mechanisms of Fatigue in Chronic Illness. Biol. Res. Nurs. 2018, 20, 410–421. [Google Scholar] [CrossRef] [PubMed]
- Sepulcri Rde, P.; do Amaral, V.F. Depressive symptoms, anxiety, and quality of life in women with pelvic endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 142, 53–56. [Google Scholar] [CrossRef]
- Pope, C.J.; Sharma, V.; Sharma, S.; Mazmanian, D. A Systematic Review of the Association Between Psychiatric Disturbances and Endometriosis. JOGC 2015, 37, 1006–1015. [Google Scholar] [CrossRef]
- Chen, L.C.; Hsu, J.W.; Huang, K.L.; Bai, Y.M.; Su, T.P.; Li, C.T.; Yang, A.C.; Chang, W.H.; Chen, T.J.; Tsai, S.J.; et al. Risk of developing major depression and anxiety disorders among women with endometriosis: A longitudinal follow-up study. J. Affect. Disord. 2016, 190, 282–285. [Google Scholar] [CrossRef]
- De Graaff, A.A.; Van Lankveld, J.; Smits, L.J.; Van Beek, J.J.; Dunselman, G.A. Dyspareunia and depressive symptoms are associated with impaired sexual functioning in women with endometriosis, whereas sexual functioning in their male partners is not affected. Hum. Reprod. (Oxford, England) 2016, 31, 2577–2586. [Google Scholar] [CrossRef]
- Santos, J.C.; Pyter, L.M. Neuroimmunology of behavioral comorbidities associated with cancer and cancer treatments. Front. Immunol. 2018, 9, 1195. [Google Scholar] [CrossRef]
- Webber, K.; Mok, K.; Bennett, B.; Lloyd, A.R.; Friedlander, M.; Juraskova, I.; Goldstein, D.; FolCan study group. If I am in the mood, I enjoy it: An exploration of cancer-related fatigue and sexual functioning in women with breast cancer. Oncologist 2011, 16, 1333–1344. [Google Scholar] [CrossRef]
- Ghasemi, V.; Simbar, M.; Ozgoli, G.; Nabavi, S.M.; Alavi Majd, H. Prevalence, dimensions, and predictor factors of sexual dysfunction in women of Iran Multiple Sclerosis Society: A cross-sectional study. Neurol. Sci. 2020. [Google Scholar] [CrossRef] [PubMed]
- Dote-Montero, M.; De-la-O, A.; Castillo, M.J.; Amaro-Gahete, F.J. Predictors of Sexual Desire and Sexual Function in Sedentary Middle-Aged Adults: The Role of Lean Mass Index and S-Klotho Plasma Levels. The FIT-AGEING Study. J. Sex Med. 2020, 17, 665–677. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Marsland, A.L.; Conley, Y.P.; Sereika, S.M.; Bender, C.M. Genes Involved in the HPA Axis and the Symptom Cluster of Fatigue, Depressive Symptoms, and Anxiety in Women With Breast Cancer During 18 Months of Adjuvant Therapy. Biol. Res. Nurs. 2020, 22, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Quinones, M.; Urrutia, R.; Torres-Reveron, A.; Vincent, K.; Flores, I. Anxiety, coping skills and hypothalamus-pituitary-adrenal (HPA) axis in patients with endometriosis. J. Reprod. Biol. Health 2015, 3, 2. [Google Scholar] [CrossRef] [PubMed]
- Aghaei, N.; Karbandi, S.; Gorji, M.A.; Golkhatmi, M.B.; Alizadeh, B. Social Support in Relation to Fatigue Symptoms Among Patients with Multiple Sclerosis. Indian J. Palliat. Care 2016, 22, 163–167. [Google Scholar] [PubMed]
- Young, K.; Fisher, J.; Kirkman, M. “Do mad people get endo or does endo make you mad?”: Clinicians’ discursive constructions of Medicine and women with endometriosis. Fem. Psychol. 2018, 29, 337–356. [Google Scholar] [CrossRef]
- Vercellini, P.; Giudice, L.C.; Evers, J.L.; Abrao, M.S. Reducing low-value care in endometriosis between limited evidence and unresolved issues: A proposal. Hum. Reprod. (Oxford, England) 2015, 30, 1996–2004. [Google Scholar] [CrossRef]
- Culley, L.; Law, C.; Hudson, N.; Denny, E.; Mitchell, H.; Baumgarten, M.; Raine-Fenning, N. The social and psychological impact of endometriosis on women’s lives: A critical narrative review. Hum. Reprod. Update 2013, 19, 625–639. [Google Scholar] [CrossRef]
- Wichniak, A.; Wierzbicka, A.; Walęcka, M.; Jernajczyk, W. Effects of Antidepressants on Sleep. Curr. Psychiatry Rep. 2017, 19, 63. [Google Scholar] [CrossRef]
- Angst, J.; Gamma, A.; Gastpar, M.; Lépine, J.P.; Mendlewicz, J.; Tylee, A. Gender differences in depression. Epidemiological findings from the European DEPRES I and II studies. Eur. Arch. Psychiatry Clin. Neurosci. 2002, 252, 201–209. [Google Scholar] [CrossRef]
- Márki, G.; Bokor, A.; Rigó, J.; Rigó, A. Physical pain and emotion regulation as the main predictive factors of health-related quality of life in women living with endometriosis. Hum. Reprod. 2017, 32, 1432–1438. [Google Scholar] [CrossRef] [PubMed]
- Petrelluzzi, K.; Garcia, M.; Petta, C.; Grassi-Kassisse, D.; Spadari-Bratfisch, R.C. Salivary cortisol concentrations, stress and quality of life in women with endometriosis and chronic pelvic pain. Stress 2008, 11, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Carey, E.T.; Martin, C.E.; Siedhoff, M.T.; Bair, E.D.; As-Sanie, S. Biopsychosocial correlates of persistent postsurgical pain in women with endometriosis. Int. J. Gynaecol. Obstet. 2014, 124, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, G.H.; Kangas, M.; David, D.; Hallquist, M.N.; Green, S.; Bovbjerg, D.H.; Schnur, J.B. Fatigue during breast cancer radiotherapy: An initial randomized study of cognitive-behavioral therapy plus hypnosis. Health Psychol. 2009, 28, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Anderson, F.J.; Winkler, A.E. An Integrated Model of Group Psychotherapy for Patients with Fibromyalgia. Int. J. Group Psychother. 2007, 57, 451–474. [Google Scholar] [CrossRef] [PubMed]
- Doerr, J.M.; Fischer, S.; Nater, U.M.; Strahler, J. Influence of stress systems and physical activity on different dimensions of fatigue in female fibromyalgia patients. J. Psychosom. Res. 2017, 93, 55–61. [Google Scholar] [CrossRef]
- Galiano-Castillo, N.; Cantarero-Villanueva, I.; Fernández-Lao, C.; Ariza-García, A.; Díaz-Rodríguez, L.; Del-Moral-Ávila, R.; Arroyo-Morales, M. Telehealth system: A randomized controlled trial evaluating the impact of an internet-based exercise intervention on quality of life, pain, muscle strength, and fatigue in breast cancer survivors. Cancer 2016, 122, 3166–3174. [Google Scholar] [CrossRef]
- De Graaff, A.A.; Dirksen, C.D.; Simoens, S.; De Bie, B.; Hummelshoj, L.; D’Hooghe, T.M.; Dunselman, G.A. Quality of life outcomes in women with endometriosis are highly influenced by recruitment strategies. Hum. Reprod. 2015, 30, 1331–1341. [Google Scholar] [CrossRef]
- Álvarez-Salvago, F.; Lara-Ramos, A.; Cantarero-Villanueva, I.; Mazheika, M.; Mundo-López, A.; Galiano-Castillo, N.; Fernández-Lao, C.; Arroyo-Morales, M.; Ocón-Hernández, O.; Artacho-Cordón, F. Chronic Fatigue, Physical Impairments and Quality of Life in Women with Endometriosis: A Case-Control Study. Int. J. Environ. Res. Public Health 2020, 17, 3610. [Google Scholar] [CrossRef]
- Skovlund, C.W.; Mørch, L.S.; Kessing, L.V.; Lidegaard, Ø. Association of Hormonal Contraception With Depression. JAMA Psychiatry 2016, 73, 1154–1162. [Google Scholar] [CrossRef]
- Dunselman, G.; Vermeulen, N.; Becker, C.; Calhaz-Jorge, C.; D’hooghe, T.; De Bie, B.; Heikinheimo, O.; Horne, A.; Kiesel, L.; Nap, A.; et al. ESHRE guideline: Management of women with endometriosis. Hum. Reprod. 2014, 29, 400–412. [Google Scholar] [CrossRef] [PubMed]

| N (%) | N (%) | ||
|---|---|---|---|
| Sociodemographic characteristics | Gynecological characteristics | ||
| Age (years)* | 36.4 ± 1.2 | N° children | |
| Schooling | None | 162 (70.4) | |
| Primary/secondary studies | 43 (18.7) | 1 | 40 (17.4) |
| Vocational training | 63 (27.4) | 2 | 23 (10.0) |
| University studies | 124 (53.9) | 3 | 5 (2.2) |
| Civil status | PMS severity | ||
| Single/Divorced | 112 (48.7) | None | 100 (43.5) |
| Married | 118 (51.3) | Mild | 14 (6.1) |
| Working outside home | Moderate | 77 (33.5) | |
| No | 26 (11.3) | Severe | 39 (17.0) |
| No, sick leave | 30 (13.0) | Endometriosis diagnosis | |
| No, loss due to endometriosis | 26 (11.3) | Laparoscopy | 155 (67.4) |
| Yes | 148 (64.3) | MRI and/or US | 62 (27.0) |
| Based on symptoms | 13 (5.7) | ||
| Time since endometriosis diagnosis (years) * | 2.4 ± 4.6 | ||
| N° surgeries | |||
| None | 75 (32.6) | ||
| 1 surgery | 87 (37.8) | ||
| 2 surgeries | 40 (17.4) | ||
| 3 or more surgeries | 28 (12.2) |
| N | |
|---|---|
| Chronic fatigue | 5.9 ± 1.7 * |
| Mild (<4) | 34 (14.8) |
| Moderate (4–7) | 82 (35.7) |
| Severe (>7) | 114 (49.6) |
| Last week pelvic pain intensity | 5.0 ± 1.9 * |
| Mild (<4) | 64 (26.7) |
| Moderate (4–7) | 111 (46.3) |
| Severe (>7) | 65 (27.1) |
| GM | GSD | Min. | Percentiles | Max. | |||
|---|---|---|---|---|---|---|---|
| 25 | 50 | 75 | |||||
| Quality of life | |||||||
| Total score | 55.0 | 1.7 | 1.7 | 47.2 | 62.5 | 73.5 | 96.6 |
| Gastrointestinal quality of life | |||||||
| Total score | 65.2 | 1.4 | 11.0 | 56.8 | 70.0 | 82.0 | 116.0 |
| Sexual function | |||||||
| Total score | 14.1 | 2.3 | 2.0 | 9.8 | 19.7 | 25.8 | 36.0 |
| Mental health | |||||||
| Anxiety | 9.4 | 1.7 | 1.0 | 7.0 | 10.0 | 13.3 | 20.0 |
| Depression | 6.6 | 1.9 | 0.0 | 4.0 | 7.0 | 11.0 | 21.0 |
| Pain catastrophizing scale | |||||||
| Total score | 23.4 | 2.0 | 0.0 | 17.0 | 28.0 | 39.0 | 52.0 |
| Sleep quality | |||||||
| Total score | 9.2 | 1.7 | 1.0 | 6.8 | 10.0 | 14.0 | 21.0 |
| Scale for Mood Assessment | |||||||
| Anger hostility | 12.2 | 2.6 | 0.0 | 4.0 | 13.0 | 26.0 | 40.0 |
| Happiness | 11.1 | 2.3 | 0.0 | 5.0 | 13.0 | 21.0 | 38.0 |
| Medical Outcomes Study-Social Support Survey | |||||||
| Total score | 70.8 | 1.4 | 23.0 | 59.8 | 76.5 | 90.0 | 95.0 |
| Bivariate Analysis | Adjusted Analysis * | Adjusted Analysis ** | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Exp (β) | 95%CI | p-Value | Exp (β) | 95%CI | p-Value | Exp (β) | 95%CI | p-Value | ||||
| Self-perceived pelvic pain severity (NRS) | ||||||||||||
| NRS = Moderate | 1.39 | 1.20 | 1.62 | <0.001 | 1.28 | 1.09 | 1.50 | 0.003 | - | - | - | - |
| NRS = Severe | 1.50 | 1.26 | 1.78 | <0.001 | 1.39 | 1.15 | 1.68 | 0.001 | - | - | - | - |
| Chronic fatigue (PFS) | ||||||||||||
| PFS = Moderate | 1.41 | 1.17 | 1.71 | <0.001 | 1.42 | 1.18 | 1.71 | <0.001 | 1.37 | 1.13 | 1.65 | 0.001 |
| PFS = Severe | 1.74 | 1.45 | 2.08 | <0.001 | 1.67 | 1.39 | 2.01 | <0.001 | 1.56 | 1.28 | 1.91 | <0.001 |
| Gastrointestinal quality of life (GIQLI) | ||||||||||||
| Total score | 1.96 | 1.66 | 2.32 | <0.001 | 1.83 | 1.51 | 2.23 | <0.001 | 1.83 | 1.51 | 2.23 | <0.001 |
| Sexual function (FSFI) | ||||||||||||
| Total score | 0.87 | 0.80 | 0.94 | <0.001 | 0.89 | 0.82 | 0.96 | 0.004 | 0.91 | 0.84 | 0.99 | 0.022 |
| Mental health (HADS) | ||||||||||||
| Anxiety | 1.43 | 1.27 | 1.61 | <0.001 | 1.33 | 1.18 | 1.50 | <0.001 | 1.29 | 1.15 | 1.46 | <0.001 |
| Depression | 1.29 | 1.18 | 1.41 | <0.001 | 1.25 | 1.13 | 1.37 | <0.001 | 1.21 | 1.09 | 1.34 | <0.001 |
| Pain catastrophizing scale (PCS) | ||||||||||||
| Total score | 1.52 | 1.41 | 1.63 | <0.001 | 1.49 | 1.38 | 1.61 | <0.001 | 1.47 | 1.35 | 1.59 | <0.001 |
| Sleep quality (PSQI) | ||||||||||||
| Total score | 1.30 | 1.16 | 1.46 | <0.001 | 1.21 | 1.06 | 1.38 | 0.005 | 1.14 | 0.99 | 1.31 | 0.059 |
| Scale for Mood Assessment (EVEA) | ||||||||||||
| Anger hostility | 1.13 | 1.06 | 1.22 | <0.001 | 1.09 | 1.02 | 1.17 | 0.012 | 1.08 | 1.01 | 1.15 | 0.031 |
| Happiness | 1.02 | 0.94 | 1.10 | 0.674 | 1.01 | 0.94 | 1.09 | 0.782 | 1.04 | 0.96 | 1.12 | 0.346 |
| Medical Outcomes Study-Social Support Survey (MOS-SSS) | ||||||||||||
| Total score | 0.73 | 0.59 | 0.91 | 0.005 | 0.79 | 0.63 | 1.00 | 0.048 | 0.83 | 0.66 | 1.03 | 0.096 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mundo-López, A.; Ocón-Hernández, O.; San-Sebastián, A.P.; Galiano-Castillo, N.; Rodríguez-Pérez, O.; Arroyo-Luque, M.S.; Arroyo-Morales, M.; Cantarero-Villanueva, I.; Fernández-Lao, C.; Artacho-Cordón, F. Contribution of Chronic Fatigue to Psychosocial Status and Quality of Life in Spanish Women Diagnosed with Endometriosis. Int. J. Environ. Res. Public Health 2020, 17, 3831. https://doi.org/10.3390/ijerph17113831
Mundo-López A, Ocón-Hernández O, San-Sebastián AP, Galiano-Castillo N, Rodríguez-Pérez O, Arroyo-Luque MS, Arroyo-Morales M, Cantarero-Villanueva I, Fernández-Lao C, Artacho-Cordón F. Contribution of Chronic Fatigue to Psychosocial Status and Quality of Life in Spanish Women Diagnosed with Endometriosis. International Journal of Environmental Research and Public Health. 2020; 17(11):3831. https://doi.org/10.3390/ijerph17113831
Chicago/Turabian StyleMundo-López, Antonio, Olga Ocón-Hernández, Ainhoa P. San-Sebastián, Noelia Galiano-Castillo, Olga Rodríguez-Pérez, María S. Arroyo-Luque, Manuel Arroyo-Morales, Irene Cantarero-Villanueva, Carolina Fernández-Lao, and Francisco Artacho-Cordón. 2020. "Contribution of Chronic Fatigue to Psychosocial Status and Quality of Life in Spanish Women Diagnosed with Endometriosis" International Journal of Environmental Research and Public Health 17, no. 11: 3831. https://doi.org/10.3390/ijerph17113831
APA StyleMundo-López, A., Ocón-Hernández, O., San-Sebastián, A. P., Galiano-Castillo, N., Rodríguez-Pérez, O., Arroyo-Luque, M. S., Arroyo-Morales, M., Cantarero-Villanueva, I., Fernández-Lao, C., & Artacho-Cordón, F. (2020). Contribution of Chronic Fatigue to Psychosocial Status and Quality of Life in Spanish Women Diagnosed with Endometriosis. International Journal of Environmental Research and Public Health, 17(11), 3831. https://doi.org/10.3390/ijerph17113831









