Risk of Kidney Injury among Construction Workers Exposed to Heat Stress: A Longitudinal Study from Saudi Arabia
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Questionnaire and Physical Exam
2.3. Urine Samples
2.4. Data Analysis
3. Results
3.1. Characteristics of Study Population
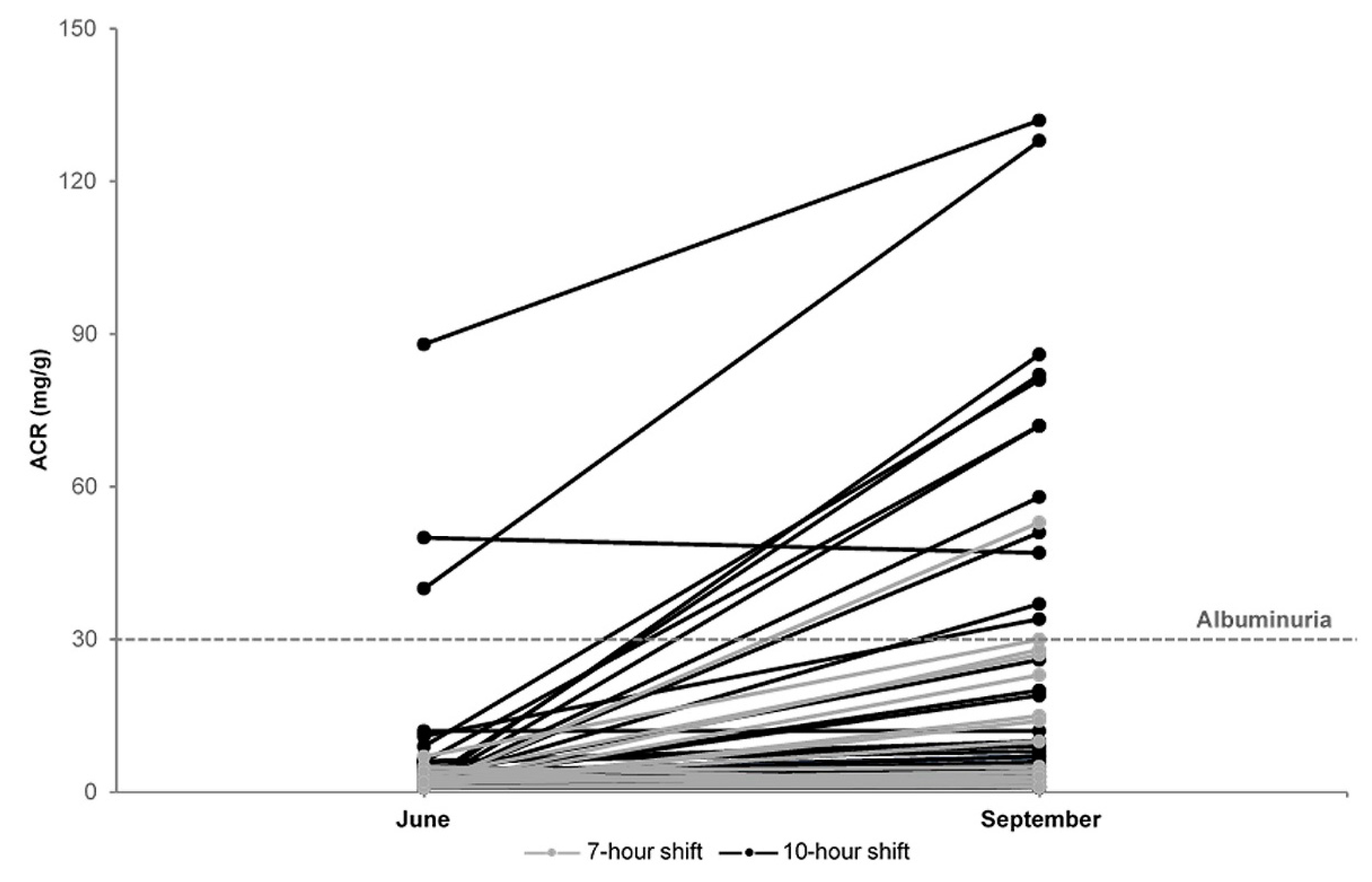
3.2. Cross Summer Changes
3.3. Incidence of Elevated ACR
3.4. Risk Factors
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Levy, B.S.; Roelofs, C. Impacts of climate change on workers’ health and safety. In Oxford Research Encyclopedia: Global Public Health; McQueen, D., Ed.; Oxford University Press, Inc.: New York, NY, USA, 2019. [Google Scholar]
- Moda, H.M.; Filho, W.L.; Minhas, A. Impacts of climate change on outdoor workers and their safety: Some research priorities. Int. J. Environ. Res. Public Health 2019, 16, 3458. [Google Scholar] [CrossRef]
- National Institute for Occupational Safety and Health (NIOSH). Criteria for a Recommended Standard: Occupational Exposure to Heat and Hot Environments; DHHS (NIOSH) Publication No. 2016-106; NIOSH: Cincinnati, OH, USA, 2016. Available online: https://www.cdc.gov/niosh/docs/2016-106/pdfs/2016-106.pdf?id=10.26616/NIOSHPUB2016106 (accessed on 8 April 2020).
- Larrañaga, M.D.; Bernard, T.E. Heat stress. In Patty’s Industrial Hygiene, 6th ed.; Rose, V.E., Cohrssen, B., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 1685–1752. [Google Scholar]
- Xiang, J.; Bi, P.; Pisaniello, D.; Hansen, A. Health impacts of workplace heat exposure: An epidemiological review. Ind. Health 2014, 52, 91–101. [Google Scholar] [CrossRef]
- International Labour Organization (ILO). Working on a Warmer Planet: The Impact of Heat Stress on Labour Productivity and Decent Work; ILO: Geneva, Switzerland, 2019; Available online: https://www.ilo.org/wcmsp5/groups/public/---dgreports/---dcomm/---publ/documents/publication/wcms_711919.pdf (accessed on 8 April 2020).
- Spector, J.T.; Masuda, Y.J.; Wolff, N.H.; Calkins, M.; Seixas, N. Heat exposure and occupational injuries: Review of the literature and implications. Curr. Environ. Health Rep. 2019, 6, 286–296. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.S.; West, G.H.; Holloway-Beth, A.; Wang, X.; Sokas, R.K. Heat-related deaths among construction workers in the United States. Am. J. Ind. Med. 2019, 62, 1047–1057. [Google Scholar] [CrossRef] [PubMed]
- Paula Santos, U.; Zanetta, D.M.; Terra-Filho, M.; Burdmann, E.A. Burnt sugarcane harvesting is associated with acute renal dysfunction. Kidney Int. 2015, 87, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Kamal, R.; Mudiam, M.K.; Gupta, M.K.; Satyanarayana, G.N.; Bihari, V.; Shukla, N.; Khan, A.H.; Kesavachandran, C.N. Heat and PAHs emissions in indoor kitchen air and its impact on kidney dysfunctions among kitchen workers in Lucknow, North India. PLoS ONE 2016, 11, e0148641. [Google Scholar] [CrossRef] [PubMed]
- Mix, J.; Elon, L.; Vi Thien Mac, V.; Flocks, J.; Economos, E.; Tovar-Aguilar, A.J.; Stover Hertzberg, V.; McCauley, L.A. Hydration status, kidney function, and kidney injury in Florida agricultural workers. J. Occup. Environ. Med. 2018, 60, e253–e260. [Google Scholar] [CrossRef]
- Nerbass, F.B.; Moist, L.; Clark, W.F.; Vieira, M.A.; Pecoits-Filho, R. Hydration status and kidney health of factory workers exposed to heat stress: A pilot feasibility study. Ann. Nutr. Metab. 2019, 74, 30–37. [Google Scholar] [CrossRef]
- Elinder, C.; Wernerson, A.; Wijkstrom, J. Mesoamerican nephropathy (MeN). A “new” chronic kidney disease related to occupational heat exposure with repeated deprivation of salts and water. Int. J. Nephrol. Kidney Fail. 2015, 1, 1–9. [Google Scholar]
- Glaser, J.; Lemery, J.; Rajagopalan, B.; Diaz, H.F.; Garcia-Trabanino, R.; Taduri, G.; Madero, M.; Amarasinghe, M.; Abraham, G.; Anutrakulchai, S.; et al. Climate change and the emergent epidemic of CKD from heat stress in rural communities: The case for heat stress nephropathy. Clin. J. Am. Soc. Nephrol. 2016, 11, 1472–1483. [Google Scholar] [CrossRef]
- Johnson, R.J.; Wesseling, C.; Newman, L.S. Chronic kidney disease of unknown cause in agricultural communities. N. Engl. J. Med. 2019, 380, 1843–1852. [Google Scholar] [CrossRef] [PubMed]
- Moyce, S.; Mitchell, D.; Armitage, T.; Tancredi, D.; Joseph, J.; Schenker, M. Heat strain, volume depletion and kidney function in California agricultural workers. Occup. Environ. Med. 2017, 74, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Schlader, Z.J.; Chapman, C.L.; Sarker, S.; Russo, L.; Rideout, T.C.; Parker, M.D.; Johnson, B.D.; Hostler, D. Firefighter work duration influences the extent of acute kidney injury. Med. Sci. Sports Exerc. 2017, 49, 1745–1753. [Google Scholar] [CrossRef] [PubMed]
- Chapman, C.L.; Johnson, B.D.; Vargas, N.T.; Hostler, D.; Parker, M.D.; Schlader, Z.J. Both hyperthermia and dehydration during physical work in the heat contribute to the risk of acute kidney injury. J. Appl. Physiol. 2020, 128, 715–728. [Google Scholar] [CrossRef]
- Nerbass, F.B.; Pecoits-Filho, R.; Clark, W.F.; Sontrop, J.M.; McIntyre, C.W.; Moist, L. Occupational heat stress and kidney health: From farms to factories. Kidney Int. Rep. 2017, 2, 998–1008. [Google Scholar] [CrossRef]
- Nerbass, F.B.; Pecoits-Filho, R. Can your work affect your kidney’s health? Rev. Environ. Health 2019, 34, 441–446. [Google Scholar] [CrossRef]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F., III; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Levey, A.S.; Coresh, J.; Bolton, K.; Culleton, B.; Harvey, K.S.; Ikizler, T.A.; Johnson, C.A.; Kausz, A.; Kimmel, P.L.; Kusek, J. K/DOQI clinical practice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Am. J. Kidney Dis. 2002, 39, S1–S266. [Google Scholar]
- Lopez-Giacoman, S.; Madero, M. Biomarkers in chronic kidney disease, from kidney function to kidney damage. World J. Nephrol. 2015, 4, 57–73. [Google Scholar] [CrossRef]
- Edelstein, C.L. Biomarkers of acute kidney injury. Adv. Chronic Kidney Dis. 2008, 15, 222–234. [Google Scholar] [CrossRef]
- Vaidya, V.S.; Ferguson, M.A.; Bonventre, J.V. Biomarkers of acute kidney injury. Annu. Rev. Pharmacol. Toxicol. 2008, 48, 463–493. [Google Scholar] [CrossRef] [PubMed]
- Bonventre, J.V.; Vaidya, V.S.; Schmouder, R.; Feig, P.; Dieterle, F. Next-generation biomarkers for detecting kidney toxicity. Nat. Biotechnol. 2010, 28, 436–440. [Google Scholar] [CrossRef] [PubMed]
- Kellum, J.A. How can we define recovery after acute kidney injury? Considerations from epidemiology and clinical trial design. Nephron Clin. Pract. 2014, 127, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Caplin, B.; Yang, C.W.; Anand, S.; Levin, A.; Madero, M.; Saran, R.; Jayasinghe, S.; De Broe, M.; Yeates, K.; Tonelli, M.; et al. The International Society of Nephrology’s International Consortium of Collaborators on Chronic Kidney Disease of Unknown Etiology: Report of the working group on approaches to population-level detection strategies and recommendations for a minimum dataset. Kidney Int. 2019, 95, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Gansevoort, R.T.; Matsushita, K.; van der Velde, M.; Astor, B.C.; Woodward, M.; Levey, A.S.; de Jong, P.E.; Coresh, J. Lower estimated GFR and higher albuminuria are associated with adverse kidney outcomes. A collaborative meta-analysis of general and high-risk population cohorts. Kidney Int. 2011, 80, 93–104. [Google Scholar] [CrossRef]
- Grams, M.E.; Sang, Y.; Ballew, S.H.; Gansevoort, R.T.; Kimm, H.; Kovesdy, C.P.; Naimark, D.; Oien, C.; Smith, D.H.; Coresh, J.; et al. A Meta-analysis of the association of estimated GFR, albuminuria, age, race, and sex with acute kidney injury. Am. J. Kidney Dis. 2015, 66, 591–601. [Google Scholar] [CrossRef]
- International Diabetes Federation (IDF). Global guideline for type 2 diabetes: Recommendations for standard, comprehensive, and minimal care. Diabet. Med. 2006, 23, 579–593. [Google Scholar] [CrossRef]
- Eknoyan, G.; Lameire, N.; Eckardt, K.; Kasiske, B.; Wheeler, D.; Levin, A.; Stevens, P.; Bilous, R.; Lamb, E.; Coresh, J.; et al. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. Suppl. 2013, 3, 1–150. [Google Scholar]
- Fleitmann, D.; Matter, A.; Pint, J.J.; Al-Shanti, M.A. The Speleothem Record of Climate Change in Saudi Arabia; Saudi Geological Survey: Jeddah, SA, USA, 2009. [Google Scholar]
- Krishna, L.V. Long term temperature trends in four different climatic zones of Saudi Arabia. Int. J. Appl. Sci. Technol. 2014, 4, 233–242. [Google Scholar]
- Al-Bouwarthan, M.; Quinn, M.M.; Kriebel, D.; Wegman, D.H. Assessment of heat stress exposure among construction workers in the hot desert climate of Saudi Arabia. Ann. Work Expo. Health 2019, 63, 505–520. [Google Scholar] [CrossRef] [PubMed]
- Al-Bouwarthan, M.; Quinn, M.M.; Kriebel, D.; Wegman, D.H. A field evaluation of construction workers’ activity, hydration status, and heat strain in the extreme summer heat of Saudi Arabia. Ann. Work Expo. Health 2020. [Google Scholar] [CrossRef] [PubMed]
- General Authority for Statistics of Saudi Arabia. Annual Economic Establishments Survey for 2018. Available online: https://www.stats.gov.sa/sites/default/files/nshr_lmsh_lqtsdy_llmwsst_2018_0.pdf (accessed on 8 April 2020).
- Jannadi, M.O.; Al-Sudairi, A. Safety management in the construction industry in Saudi Arabia: Survey shows that level of company practice influences safety performance. Build. Res. Inf. 1995, 23, 60–63. [Google Scholar] [CrossRef]
- Jannadi, M.O.; Assaf, S. Safety assessment in the built environment of Saudi Arabia. Saf. Sci. 1998, 29, 15–24. [Google Scholar] [CrossRef]
- Mosly, I. Safety performance in the construction industry of Saudi Arabia. Int. J. Civ. Eng. Manag. 2015, 4, 238–247. [Google Scholar] [CrossRef]
- Erogul, M.S.; Alyami, M.M. Construction site safety in small construction companies in Saudi Arabia. Int. J. Manag. Pract. 2017, 10, 406–421. [Google Scholar] [CrossRef]
- Bodin, T.; Garcia-Trabanino, R.; Weiss, I.; Jarquin, E.; Glaser, J.; Jakobsson, K.; Lucas, R.A.; Wesseling, C.; Hogstedt, C.; Wegman, D.H.; et al. Intervention to reduce heat stress and improve efficiency among sugarcane workers in El Salvador: Phase 1. Occup. Environ. Med. 2016, 73, 409–416. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Maresh, C.M.; Castellani, J.W.; Bergeron, M.F.; Kenefick, R.W.; LaGasse, K.E.; Riebe, D. Urinary indices of hydration status. Int. J. Sport Nutr. 1994, 4, 265–279. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Soto, J.A.; Hacker, F.T., Jr.; Casa, D.J.; Kavouras, S.A.; Maresh, C.M. Urinary indices during dehydration, exercise, and rehydration. Int. J. Sport Nutr. 1998, 8, 345–355. [Google Scholar] [CrossRef]
- McDonald, O.F.; Shanks, N.J.; Fragu, L. Heat stress: Improving safety in the Arabian Gulf oil and gas industry. Prof. Saf. 2008, 53, 31–36. [Google Scholar]
- Joubert, D.; Thomsen, J.; Harrison, O. Safety in the heat: A comprehensive program for prevention of heat illness among workers in Abu Dhabi, United Arab Emirates. Am. J. Public Health 2011, 101, 395–398. [Google Scholar] [CrossRef]
- El-Shafei, D.A.; Bolbol, S.A.; Awad Allah, M.B.; Abdelsalam, A.E. Exertional heat illness: Knowledge and behavior among construction workers. Environ. Sci. Pollut. Res. Int. 2018, 25, 32269–32276. [Google Scholar] [CrossRef] [PubMed]
- Mogensen, C.E.; Vestbo, E.; Poulsen, P.L.; Christiansen, C.; Damsgaard, E.M.; Eiskjaer, H.; Froland, A.; Hansen, K.W.; Nielsen, S.; Pedersen, M.M. Microalbuminuria and potential confounders. A review and some observations on variability of urinary albumin excretion. Diabetes Care 1995, 18, 572–581. [Google Scholar] [CrossRef]
- Witte, E.C.; Lambers Heerspink, H.J.; de Zeeuw, D.; Bakker, S.J.; de Jong, P.E.; Gansevoort, R. First morning voids are more reliable than spot urine samples to assess microalbuminuria. J. Am. Soc. Nephrol. 2009, 20, 436–443. [Google Scholar] [CrossRef] [PubMed]
- European Society of Cardiology (ESC). Definition of Hypertension and Pressure Goals during Treatment (ESC-ESH Guidelines 2018). Available online: https://www.escardio.org/Journals/E-Journal-of-Cardiology-Practice/Volume-17/definition-of-hypertension-and-pressure-goals-during-treatment-esc-esh-guidelin (accessed on 8 April 2020).
- World Health Organization (WHO). Body Mass Index (BMI) Classification. Available online: http://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi (accessed on 8 April 2020).
- Hirshkowitz, M.; Whiton, K.; Albert, S.M.; Alessi, C.; Bruni, O.; DonCarlos, L.; Hazen, N.; Herman, J.; Adams Hillard, P.J.; Katz, E.S.; et al. National Sleep Foundation’s updated sleep duration recommendations: Final report. Sleep Health 2015, 1, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Casa, D.J.; Armstrong, L.E.; Hillman, S.K.; Montain, S.J.; Reiff, R.V.; Rich, B.S.; Roberts, W.O.; Stone, J.A. National athletic trainers’ association position statement: Fluid replacement for athletes. J. Athl. Train. 2000, 35, 212–224. [Google Scholar] [PubMed]
- Won, J.C.; Lee, Y.J.; Kim, J.M.; Han, S.Y.; Noh, J.H.; Ko, K.S.; Rhee, B.D.; Kim, D.J. Prevalence of and factors associated with albuminuria in the Korean adult population: The 2011 Korea national health and nutrition examination survey. PLoS ONE 2013, 8, e83273. [Google Scholar] [CrossRef] [PubMed]
- Solbu, M.D.; Kronborg, J.; Eriksen, B.O.; Jenssen, T.G.; Toft, I. Cardiovascular risk-factors predict progression of urinary albumin-excretion in a general, non-diabetic population: A gender-specific follow-up study. Atherosclerosis 2008, 201, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Abo-Zenah, H.; El-Benayan, A.; El Nahas, A.M. Prevalence of increased albumin excretion rate in young saudi adults. Nephron. Clin. Pract. 2008, 108, c155–c162. [Google Scholar] [CrossRef]
- Jones, C.A.; Francis, M.E.; Eberhardt, M.S.; Chavers, B.; Coresh, J.; Engelgau, M.; Kusek, J.W.; Byrd-Holt, D.; Narayan, K.M.; Herman, W.H.; et al. Microalbuminuria in the US population: Third national health and nutrition examination survey. Am. J. Kidney Dis. 2002, 39, 445–459. [Google Scholar] [CrossRef]
- Flouris, A.D.; Dinas, P.C.; Ioannou, L.G.; Nybo, L.; Havenith, G.; Kenny, G.P.; Kjellstrom, T. Workers’ health and productivity under occupational heat strain: A systematic review and meta-analysis. Lancet Planet. Health 2018, 2, e521–e531. [Google Scholar] [CrossRef]
- Gallo-Ruiz, L.; Sennett, C.M.; Sanchez-Delgado, M.; Garcia-Urbina, A.; Gamez-Altamirano, T.; Basra, K.; Laws, R.L.; Amador, J.J.; Lopez-Pilarte, D.; Tripodis, Y.; et al. Prevalence and risk factors for CKD among brickmaking workers in La Paz Centro, Nicaragua. Am. J. Kidney Dis. 2019, 74, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Yu, I.T.; Li, Z.; Wang, X.; Sun, T.; Lin, H.; Wan, S.; Qiu, H.; Xie, S. Work-related injuries and musculoskeletal disorders among factory workers in a major city of China. Accid. Anal. Prev. 2012, 48, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Tomioka, K.; Morita, N.; Saeki, K.; Okamoto, N.; Kurumatani, N. Working hours, occupational stress and depression among physicians. Occup. Med. 2011, 61, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, M.; Ferrie, J.E.; Singh-Manoux, A.; Shipley, M.J.; Stansfeld, S.A.; Marmot, M.G.; Ahola, K.; Vahtera, J.; Kivimaki, M. Long working hours and symptoms of anxiety and depression: A 5-year follow-up of the Whitehall II study. Psychol. Med. 2011, 41, 2485–2494. [Google Scholar] [CrossRef]
- Kivimaki, M.; Jokela, M.; Nyberg, S.T.; Singh-Manoux, A.; Fransson, E.I.; Alfredsson, L.; Bjorner, J.B.; Borritz, M.; Burr, H.; Casini, A.; et al. Long working hours and risk of coronary heart disease and stroke: A systematic review and meta-analysis of published and unpublished data for 603,838 individuals. Lancet 2015, 386, 1739–1746. [Google Scholar] [CrossRef]
- Bongers, C.; Alsady, M.; Nijenhuis, T.; Tulp, A.D.M.; Eijsvogels, T.M.H.; Deen, P.M.T.; Hopman, M.T.E. Impact of acute versus prolonged exercise and dehydration on kidney function and injury. Physiol. Rep. 2018, 6, e13734. [Google Scholar] [CrossRef]
- Sato, Y.; Roncal-Jimenez, C.A.; Andres-Hernando, A.; Jensen, T.; Tolan, D.R.; Sanchez-Lozada, L.G.; Newman, L.S.; Butler-Dawson, J.; Sorensen, C.; Glaser, J.; et al. Increase of core temperature affected the progression of kidney injury by repeated heat stress exposure. Am. J. Physiol. Renal Physiol. 2019, 317, F1111–F1121. [Google Scholar] [CrossRef]
- Meier-Ewert, H.K.; Ridker, P.M.; Rifai, N.; Regan, M.M.; Price, N.J.; Dinges, D.F.; Mullington, J.M. Effect of sleep loss on C-reactive protein, an inflammatory marker of cardiovascular risk. J. Am. Coll. Cardiol. 2004, 43, 678–683. [Google Scholar] [CrossRef]
- Vgontzas, A.N.; Zoumakis, E.; Bixler, E.O.; Lin, H.M.; Follett, H.; Kales, A.; Chrousos, G.P. Adverse effects of modest sleep restriction on sleepiness, performance, and inflammatory cytokines. J. Clin. Endocrinol. Metab. 2004, 89, 2119–2126. [Google Scholar] [CrossRef]
- Patel, S.R.; Zhu, X.; Storfer-Isser, A.; Mehra, R.; Jenny, N.S.; Tracy, R.; Redline, S. Sleep duration and biomarkers of inflammation. Sleep 2009, 32, 200–204. [Google Scholar] [CrossRef]
- Upadhyay, A.; Larson, M.G.; Guo, C.Y.; Vasan, R.S.; Lipinska, I.; O’Donnell, C.J.; Kathiresan, S.; Meigs, J.B.; Keaney, J.F., Jr.; Rong, J.; et al. Inflammation, kidney function and albuminuria in the Framingham Offspring cohort. Nephrol. Dial. Transplant. 2011, 26, 920–926. [Google Scholar] [CrossRef] [PubMed]
- Hansson, E.; Glaser, J.; Weiss, I.; Ekstrom, U.; Apelqvist, J.; Hogstedt, C.; Peraza, S.; Lucas, R.; Jakobsson, K.; Wesseling, C.; et al. Workload and cross-harvest kidney injury in a Nicaraguan sugarcane worker cohort. Occup. Environ. Med. 2019, 76, 818–826. [Google Scholar] [CrossRef] [PubMed]
- Sallinen, M.; Kecklund, G. Shift work, sleep, and sleepiness—Differences between shift schedules and systems. Scand. J. Work Environ. Health 2010, 36, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Van der Hulst, M. Long workhours and health. Scand. J. Work Environ. Health 2003, 29, 171–188. [Google Scholar] [CrossRef] [PubMed]
- Plantinga, L.; Lee, K.; Inker, L.A.; Saran, R.; Yee, J.; Gillespie, B.; Rolka, D.; Saydah, S.; Powe, N.R.; Team, C.C.S. Association of sleep-related problems with CKD in the United States, 2005–2008. Am. J. Kidney Dis. 2011, 58, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, R.; Nagasawa, Y.; Iwatani, H.; Shinzawa, M.; Obi, Y.; Teranishi, J.; Ishigami, T.; Yamauchi-Takihara, K.; Nishida, M.; Rakugi, H.; et al. Self-reported sleep duration and prediction of proteinuria: A retrospective cohort study. Am. J. Kidney Dis. 2012, 59, 343–355. [Google Scholar] [CrossRef]
- McMullan, C.J.; Curhan, G.C.; Forman, J.P. Association of short sleep duration and rapid decline in renal function. Kidney Int. 2016, 89, 1324–1330. [Google Scholar] [CrossRef]
- Okamoto-Mizuno, K.; Mizuno, K. Effects of thermal environment on sleep and circadian rhythm. J. Physiol. Anthropol. 2012, 31, 14. [Google Scholar] [CrossRef]
- Park, J.B.; Nakata, A.; Swanson, N.G.; Chun, H. Organizational factors associated with work-related sleep problems in a nationally representative sample of Korean workers. Int. Arch. Occup. Environ. Health 2013, 86, 211–222. [Google Scholar] [CrossRef]
- Van Laethem, M.; Beckers, D.G.; Kompier, M.A.; Dijksterhuis, A.; Geurts, S.A. Psychosocial work characteristics and sleep quality: A systematic review of longitudinal and intervention research. Scand. J. Work Environ. Health 2013, 39, 535–549. [Google Scholar] [CrossRef]
- Dutta, M.J. Migration and health in the construction industry: Culturally centering voices of Bangladeshi workers in Singapore. Int. J. Environ. Res. Public Health 2017, 14, 132. [Google Scholar] [CrossRef] [PubMed]
- Magee, C.; Gopaldasani, V.; Bakand, S.; Coman, R. The physical work environment and sleep: A latent class analysis. J. Occup. Environ. Med. 2019, 61, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Hasle, P.; Limborg, H.J. A review of the literature on preventive occupational health and safety activities in small enterprises. Ind. Health 2006, 44, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Li, R.Y.M.; Poon, S.W. A literature review on the causes of construction accidents. In Construction Safety; Springer: Berlin, Germany, 2013; pp. 1–11. [Google Scholar]
- Marin, L.S.; Roelofs, C. Engaging small residential construction contractors in community-based participatory research to promote safety. Ann. Work Expo. Health 2018, 62, S72–S80. [Google Scholar] [CrossRef] [PubMed]
- Evanoff, B.; Rohlman, D.; Strickland, J.; Dale, A. Influence of work organization and work environment on missed work, productivity, and use of pain medications among construction apprentices. Am. J. Ind. Med. 2019, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.; Van Horn, L.; Jacobs, D.R., Jr.; Liu, K.; Muntner, P.; Newsome, B.; Shoham, D.A.; Durazo-Arvizu, R.; Bibbins-Domingo, K.; Reis, J.; et al. Lifestyle-related factors, obesity, and incident microalbuminuria: The CARDIA (Coronary Artery Risk Development in Young Adults) study. Am. J. Kidney Dis. 2013, 62, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Kramer, H.; Reboussin, D.; Bertoni, A.G.; Marcovina, S.; Lipkin, E.; Greenway, F.L., III; Brancati, F.L.; Look Ahead Research, G. Obesity and albuminuria among adults with type 2 diabetes: The Look AHEAD (Action for Health in Diabetes) study. Diabetes Care 2009, 32, 851–853. [Google Scholar] [CrossRef]
- Scheven, L.; Halbesma, N.; de Jong, P.E.; de Zeeuw, D.; Bakker, S.J.; Gansevoort, R.T. Predictors of progression in albuminuria in the general population: Results from the PREVEND cohort. PLoS ONE 2013, 8, e61119. [Google Scholar] [CrossRef]
- Garland, J.S. Elevated body mass index as a risk factor for chronic kidney disease: Current perspectives. Diabetes Metab. Syndr. Obes. 2014, 7, 347–355. [Google Scholar] [CrossRef]
- Donoghue, A.M.; Bates, G.P. The risk of heat exhaustion at a deep underground metalliferous mine in relation to body-mass index and predicted VO2max. Occup. Med. 2000, 50, 259–263. [Google Scholar] [CrossRef]
- Wallace, R.F.; Kriebel, D.; Punnett, L.; Wegman, D.H.; Wenger, C.B.; Gardner, J.W.; Kark, J.A. Risk factors for recruit exertional heat illness by gender and training period. Aviat. Space Environ. Med. 2006, 77, 415–421. [Google Scholar] [PubMed]
- Flouris, A.D.; McGinn, R.; Poirier, M.P.; Louie, J.C.; Ioannou, L.G.; Tsoutsoubi, L.; Sigal, R.J.; Boulay, P.; Hardcastle, S.G.; Kenny, G.P. Screening criteria for increased susceptibility to heat stress during work or leisure in hot environments in healthy individuals aged 31–70 years. Temperature 2018, 5, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Mykkanen, L.; Haffner, S.M.; Kuusisto, J.; Pyorala, K.; Laakso, M. Microalbuminuria precedes the development of NIDDM. Diabetes 1994, 43, 552–557. [Google Scholar] [CrossRef] [PubMed]
- Brantsma, A.H.; Bakker, S.J.; de Zeeuw, D.; de Jong, P.E.; Gansevoort, R.T. Urinary albumin excretion as a predictor of the development of hypertension in the general population. J. Am. Soc. Nephrol. 2006, 17, 331–335. [Google Scholar] [CrossRef]
- Kramer, B.K.; Kernz, M.; Ress, K.M.; Pfohl, M.; Muller, G.A.; Schmulling, R.M.; Risler, T. Influence of strenuous exercise on albumin excretion. Clin. Chem. 1988, 34, 2516–2518. [Google Scholar] [CrossRef]
- Robertshaw, M.; Cheung, C.K.; Fairly, I.; Swaminathan, R. Protein excretion after prolonged exercise. Ann. Clin. Biochem. 1993, 30, 34–37. [Google Scholar] [CrossRef]
- Wolyniec, W.; Kasprowicz, K.; Rita-Tkachenko, P.; Renke, M.; Ratkowski, W. Biochemical markers of renal hypoperfusion, hemoconcentration, and proteinuria after extreme physical exercise. Medicina 2019, 55, 154. [Google Scholar] [CrossRef]
- Fischer, R.S.B.; Mandayam, S.; Chavarria, D.; Vangala, C.; Nolan, M.S.; Garcia, L.L.; Palma, L.; Garcia, F.; Garcia-Trabanino, R.; Murray, K.O. Clinical evidence of acute Mesoamerican nephropathy. Am. J. Trop. Med. Hyg. 2017, 97, 1247–1256. [Google Scholar] [CrossRef]
| Worker Characteristics | All Workers |
|---|---|
| (n = 65) | |
| N (%) | |
| Age (years), Mean ± SD | 39.7 ± 9.4 |
| Sleeping hours a | |
| <8 h | 32 (49%) |
| ≥8 h | 33 (51%) |
| Current smokers | |
| Yes | 43 (66%) |
| No | 22 (34%) |
| BMI b | |
| ≥25 kg/m2 (overweight) | 14 (22%) |
| <25 kg/m2 (normal) | 51 (78%) |
| Diabetes c | |
| Yes | 6 (9%) |
| No | 59 (91%) |
| Hypertension d | |
| Yes | 10 (15%) |
| No | 55 (85%) |
| Work Characteristics | |
| Total work experience (years), Mean ± SD | 17.5 ± 8.6 |
| Work experience in SA (years), Mean ± SD | 7.0 ± 7.2 |
| Shift length | |
| 10 h | 44 (68%) |
| 7 h | 21 (32%) |
| Job title | |
| Carpenter | 8 (12%) |
| Block Layer | 4 (6%) |
| Laborer | 14 (22%) |
| Plasterer | 23 (35%) |
| Steel Fixer | 3 (5%) |
| Tiler | 13 (20%) |
| ACR < 30 mg/g (n = 51) | ACR ≥ 30 mg/g † (n = 14) | p-Value * | |
|---|---|---|---|
| Mean ± SD | Mean ± SD | ||
| Age (years) | 39.8 ± 9.6 | 39.1 ± 8.7 | 0.82 |
| Total work experience (years) | 17.6 ± 8.8 | 17.3 ± 8.3 | 0.91 |
| Work experience in SA (years) | 7.2 ± 6.9 | 5.7 ± 8.2 | 0.51 |
| ACR-June (mg/g) | 2.8 ± 2.0 | 15.7 ± 25.9 | <0.01 |
| Daily fluid intake-June | 5.3 ± 2.8 | 5.2 ± 3.2 | 0.91 |
| Daily fluid intake- September | 5.0 ± 0.7 | 5.0 ± 1.2 | 0.95 |
| N (%) | N (%) | ||
| Job title | |||
| Carpenter | 7 (14%) | 1 (7%) | 0.45 |
| Block Layer | 3 (6%) | 1 (7%) | 0.63 |
| Laborer | 11 (22%) | 3 (21%) | 0.65 |
| Plasterer | 20 (39%) | 3 (21%) | 0.18 |
| Steel Fixer | 3 (6%) | 0 | 0.48 |
| Tiler | 7 (14%) | 6 (43%) | 0.03 |
| Shift length | |||
| 10 h | 32 (63%) | 12 (86%) | 0.09 |
| 7 h | 19 (37%) | 2 (14%) | |
| Sleeping hours a | |||
| <8 h | 21 (41%) | 11 (79%) | 0.01 |
| ≥8 h | 30 (59%) | 3 (21%) | |
| Current smoker | |||
| Yes | 32 (63%) | 11 (79%) | 0.22 |
| No | 19 (37%) | 3 (21%) | |
| BMI b | |||
| ≥25 kg/m2 (overweight) | 9 (18%) | 5 (36%) | 0.14 |
| <25 kg/m2 (normal) | 42 (82%) | 9 (64%) | |
| Diabetes c | |||
| Yes | 5 (10%) | 1 (7%) | 0.62 |
| No | 46 (90%) | 13 (93%) | |
| Hypertension d | |||
| Yes | 6 (12%) | 4 (29%) | 0.13 |
| No | 45 (88%) | 10 (71%) | |
| Hydration status-June e | |||
| Significant dehydration | 4 (8%) | 3 (21%) | 0.16 |
| Minimal dehydration | 22 (43%) | 4 (29%) | 0.32 |
| Well hydrated | 25 (49%) | 7 (50%) | 0.95 |
| Hydration status-September e | |||
| Significant dehydration | 2 (4%) | 2 (14%) | 0.20 |
| Minimal dehydration | 26 (51%) | 4 (29%) | 0.14 |
| Well hydrated | 23 (45%) | 8 (57%) | 0.42 |
| Variables | ΔACR (mg/g) † (n = 65) | |
|---|---|---|
| β (95% CI) | p-Value * | |
| Age (years) | −0.18 (−0.83:0.47) | 0.59 |
| Total work experience (years) | −0.20 (−0.87:0.47) | 0.56 |
| Work experience in SA (years) | −0.26 (−1.25:0.72) | 0.60 |
| ACR-June (mg/g) | 0.41 (−0.09:0.90) | 0.11 |
| Daily fluid intake-June (L) | −0.42 (−3.06:2.22) | 0.76 |
| Daily fluid intake-September (L) | 1.64 (−6.16:9.44) | 0.68 |
| Hydration status-June a | ||
| Significant dehydration | 21.91 (1.20:42.62) | 0.04 |
| Minimal dehydration | −2.25 (−13.73:9.24) | 0.70 |
| Well hydrated | Ref | |
| Hydration status-September a | ||
| Significant dehydration | 16.29 (−11.00:43.58) | 0.24 |
| Minimal dehydration | −2.51 (−14.40:9.38) | 0.68 |
| Well hydrated | Ref | |
| Job title | ||
| Carpenter | −10.80 (−31.98:10.38) | 0.32 |
| Block Layer | −11.42 (−31.81:8.96) | 0.27 |
| Laborer | −6.00 (−27.07:15.08) | 0.58 |
| Plasterer | −11.97 (−31.81:7.88) | 0.24 |
| Steel Fixer | −17.59 (−36.20:1.02) | 0.06 |
| Tiler | Ref | |
| Shift length | ||
| 10 h | 8.63 (−1.40:18.65) | 0.09 |
| 7 h | Ref | |
| Sleeping hours b | ||
| <8 h | 12.70 (1.21:24.19) | 0.03 |
| ≥8 h | Ref | |
| Current smokers | ||
| Yes | 6.54 (−4.78:17.86) | 0.26 |
| No | Ref | |
| BMI c | ||
| ≥25 kg/m2 (overweight) | 15.32 (−1.62:32.25) | 0.08 |
| <25 kg/m2 (normal) | Ref | |
| Diabetes d | ||
| Yes | −12.55 (−22.29:−2.80) | 0.01 |
| No | Ref | |
| Hypertension e | ||
| Yes | 8.33 (−11.61:28.26) | 0.41 |
| No | Ref | |
| Variables † | Model 1 | p-Value * | Model 2 ‡ | p-Value * |
|---|---|---|---|---|
| β (95% CI) | β (95% CI) | |||
| Intercept | 31.67 (0.96:62.37) | 0.04 | 18.37 (−2.89:39.64) | 0.09 |
| Age (years) | −0.84 (−1.59:−0.10) | 0.03 | −0.57 (−1.19:0.05) | 0.07 |
| Hydration status-June a | ||||
| Significant dehydration | 28.41 (13.45:43.37) | <0.01 | 30.60 (15.88:45.32) | <0.01 |
| Minimal dehydration | −1.20 (−11.50:9.10) | 0.82 | 0.88 (−8.52:10.27) | 0.85 |
| Well hydrated | Ref | Ref | ||
| Sleeping hours b | ||||
| <8 h | 16.69 (6.59:26.79) | <0.01 | 17.38 (7.12:27.64) | <0.01 |
| ≥8 h | Ref | Ref | ||
| BMI c | ||||
| ≥25 kg/m2 (overweight) | 15.17 (0.49:29.84) | 0.04 | 16.13 (1.50:30.76) | 0.03 |
| <25 kg/m2 (normal) | Ref | Ref | ||
| Hypertension d | ||||
| Yes | 15.06 (0.17:29.95) | 0.05 | 12.55 (−3.17:28.28) | 0.12 |
| No | Ref | Ref | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Bouwarthan, M.; Quinn, M.M.; Kriebel, D.; Wegman, D.H. Risk of Kidney Injury among Construction Workers Exposed to Heat Stress: A Longitudinal Study from Saudi Arabia. Int. J. Environ. Res. Public Health 2020, 17, 3775. https://doi.org/10.3390/ijerph17113775
Al-Bouwarthan M, Quinn MM, Kriebel D, Wegman DH. Risk of Kidney Injury among Construction Workers Exposed to Heat Stress: A Longitudinal Study from Saudi Arabia. International Journal of Environmental Research and Public Health. 2020; 17(11):3775. https://doi.org/10.3390/ijerph17113775
Chicago/Turabian StyleAl-Bouwarthan, Mohammed, Margaret M. Quinn, David Kriebel, and David H. Wegman. 2020. "Risk of Kidney Injury among Construction Workers Exposed to Heat Stress: A Longitudinal Study from Saudi Arabia" International Journal of Environmental Research and Public Health 17, no. 11: 3775. https://doi.org/10.3390/ijerph17113775
APA StyleAl-Bouwarthan, M., Quinn, M. M., Kriebel, D., & Wegman, D. H. (2020). Risk of Kidney Injury among Construction Workers Exposed to Heat Stress: A Longitudinal Study from Saudi Arabia. International Journal of Environmental Research and Public Health, 17(11), 3775. https://doi.org/10.3390/ijerph17113775





