Prepregnancy Endocrine, Autoimmune Disorders and the Risks of Gestational Hypertension-Preeclampsia in Primiparas: A Nationwide Population-Based Study in Taiwan
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source and Sample
2.2. Statement of Research Ethics
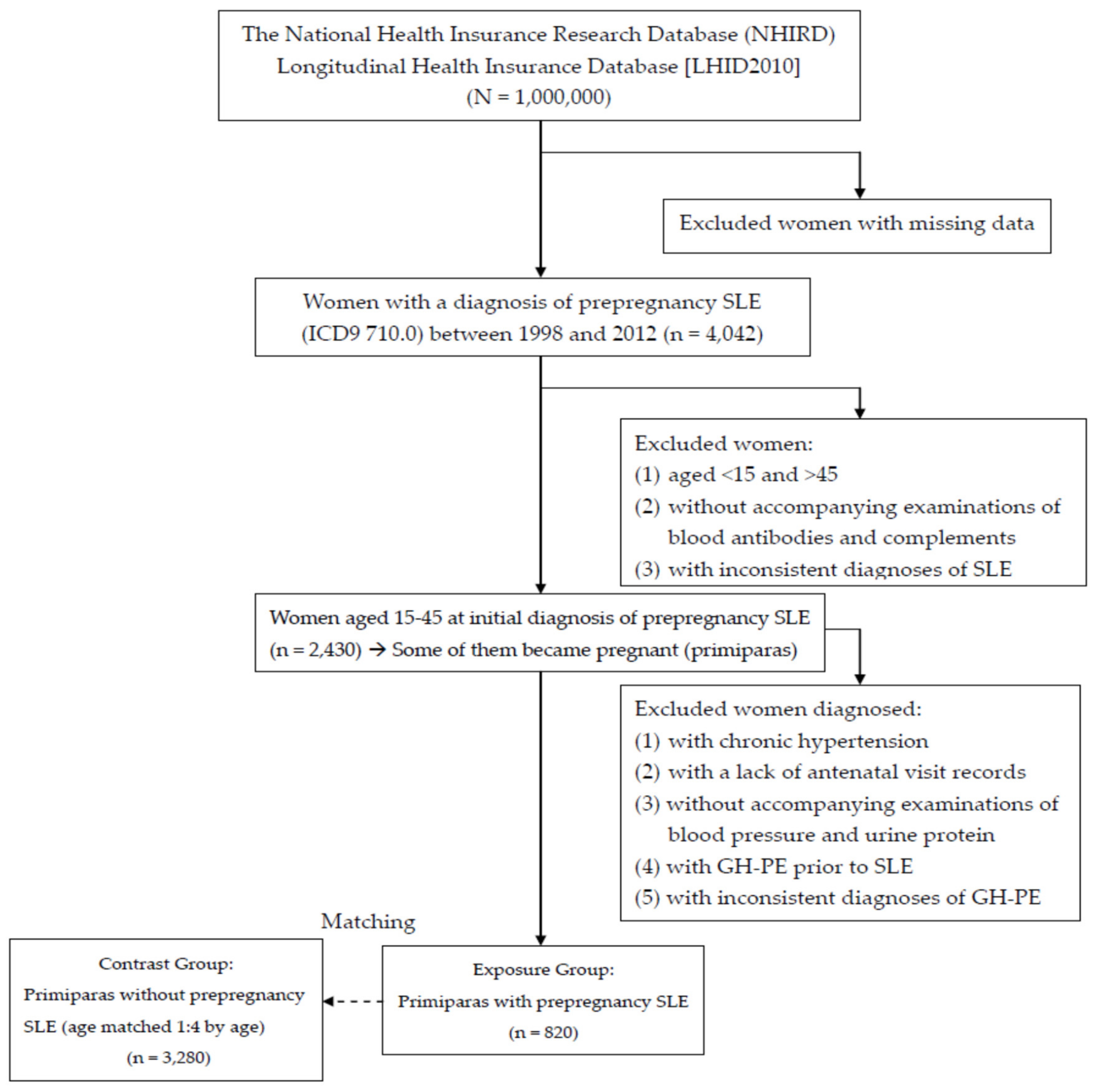
2.3. Inclusion and Exclusion Criteria
2.4. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lisnevskaia, L.; Murphy, G.; Isenberg, D. Systemic lupus erythematosus. Lancet 2014, 384, 1878–1888. [Google Scholar] [CrossRef]
- Shao, W.H.; Cohen, P.L. Disturbances of apoptotic cell clearance in systemic lupus erythematosus. Arthritis Res. Ther. 2011, 13, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum. Reprod. 2004, 19, 41–47. [Google Scholar] [CrossRef]
- Kousta, E.; Cela, E.; Lawrence, N.; Penny, A.; Millauer, B.; White, D. The prevalence of polycystic ovaries in women with a history of gestational diabetes. Clin. Endocrinol. 2000, 53, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Hart, R. Polycystic ovarian syndrome—Prognosis and treatment outcomes. Curr. Opin. Obstet. Gynecol. 2007, 19, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Dunaif, A. Insulin resistance and the polycystic ovary syndrome: Mechanism and implications for pathogenesis. Endocr. Rev. 1997, 18, 774–800. [Google Scholar] [PubMed]
- Eroglu, D.; Zeyneloglu, H.B. Metabolic disorders in patients with recent gestational diabetes mellitus. J. Obstet. Gynaecol. Res. 2006, 32, 408–415. [Google Scholar] [CrossRef]
- Ehrmann, D.A.; Barnes, R.B.; Rosenfield, R.L.; Cavaghan, M.K.; Imperial, J. Prevalence of impaired glucose tolerance and diabetes in women with polycystic ovary syndrome. Diabetes Care 1999, 22, 141–146. [Google Scholar] [CrossRef]
- Glintborg, D.; Henriksen, J.E.; Andersen, M.; Hagen, C.; Hangaard, J.; Rasmussen, P.E. Prevalence of endocrine diseases and abnormal glucose tolerance tests in 340 Caucasian premenopausal women with hirsutism as the referral diagnosis. Fertil. Steril. 2004, 82, 1570–1579. [Google Scholar] [CrossRef]
- Legro, R.S.; Dunaif, A. The role of insulin resistance in polycystic ovary syndrome. Endocrinologist 1996, 6, 307–321. [Google Scholar] [CrossRef]
- Petríková, J.; Lazúrová, I.; Yehuda, S. Polycystic ovary syndrome and autoimmunity. Eur. J. Intern. Med. 2010, 21, 369–371. [Google Scholar] [CrossRef] [PubMed]
- Fénichel, P.; Gobert, B.; Carré, Y.; Barbarino-Monnier, P.; Hiéronimus, S. Polycystic ovary syndrome in autoimmune disease. Lancet 1999, 353, 2210. [Google Scholar] [CrossRef]
- Haller, K.; Mathieu, C.; Rull, K.; Matt, K.; Béné, M.C.; Uibo, R. IgG, IgA and IgM antibodies against FSH: Serological markers of pathogenic autoimmunity or of normal immunoregulation? Am. J. Reprod. Immunol. 2005, 54, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Mohamed-Ali, A.F.; Fateen, B.; Ezzet, A.; Ramadan, A.; Badawy, H.; El-tobge, A. Polycystic ovary syndrome as an autoimmune disease: A new concept. Obstet. Gynecol. 2000, 95 (Suppl. 1), S48. [Google Scholar]
- Xiong, Y.L.; Liang, X.Y.; Yang, X.; Li, Y.; Wei, L.N. Low-grade chronic inflammation in the peripheral blood and ovaries of women with polycystic ovarian syndrome. Eur. J. Obstet. Gynecol. Reprod. Biol. 2011, 159, 148–150. [Google Scholar] [CrossRef]
- Mohamed-Hussein, Z.A.; Harun, S. Construction of a polycystic ovarian syndrome (PCOS) pathway based on the interactions of PCOS-related proteins retrieved from bibliomic data. Theor. Biol. Med. Model 2009, 6, 18. [Google Scholar] [CrossRef]
- Gleicher, N.; Weghofer, A.; Lee, I.H.; Barad, D.H. FMR1 genotype with autoimmunity-associated polycystic ovary-like phenotype and decreased pregnancy chance. PLoS ONE 2010, 5, e15303. [Google Scholar] [CrossRef]
- Menke, M.N.; Strauss, J.F., 3rd. Genetic approaches to polycystic ovarian syndrome. Curr. Opin. Obstet. Gynecol. 2007, 19, 355–359. [Google Scholar] [CrossRef]
- Hopkinson, Z.E.; Sattar, N.; Fleming, R.; Greer, I.A. Polycystic ovarian syndrome: The metabolic syndrome comes to gynecology. BMJ 1998, 317, 329–332. [Google Scholar] [CrossRef]
- Sibai, B.M. Diagnosis and management of gestational hypertension and preeclampsia. Obstet. Gynecol. 2003, 102, 181–192. [Google Scholar]
- Cunningham, F.G.; Leveno, K.J.; Bloom, S.L.; Hauth, J.C.; Rouse, D.J.; Spong, C.Y. Prenatal care, Pregnancy Hypertension, and Obesity. In Williams Obstetrics; Cunningham, F.G., Ed.; McGraw-Hill: New York, NY, USA, 2010; pp. 176–177. [Google Scholar]
- Mol, B.W.; Roberts, C.T.; Thangaratinam, S.; Magee, L.A.; de Groot, C.J.; Hofmeyr, G.J. Pre-eclampsia. Lancet 2016, 387, 999–1011. [Google Scholar] [CrossRef]
- Pauli, J.M.; Repke, J.T. Preeclampsia: Short-term and long-term implications. Obstet. Gynecol. Clin. North Am. 2015, 42, 299–313. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Make Every Mother and Child Count; World Health Report; WHO: Geneva, Switzerland, 2005. [Google Scholar]
- Visentin, S.; Londero, A.P.; Bellamio, B.; Giunta, G.; Cosma, C.; Faggian, D.; Plebani, M.; Cosmi, E. Fetal endothelial remodeling in late-onset gestational hypertension. Am. J. Hypertens. 2016, 29, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Visentin, S.; Grisan, E.; Zanardo, V.; Bertin, M.; Veronese, E.; Cavallin, F.; Ambrosini, G.; Trevisanuto, D.; Cosmi, E. Developmental programming of cardiovascular risk in intrauterine growth-restricted twin fetuses according to aortic intima thickness. J. Ultrasound Med. 2013, 32, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Veerbeek, J.H.; Hermes, W.; Breimer, A.Y.; van Rijn, B.B.; Koenen, S.V.; Mol, B.W.; Franx, A.; de Groot, C.J.; Koster, M.P. Cardiovascular disease risk factors after early-onset preeclampsia, late-onset preeclampsia, and pregnancy-induced hypertension. Hypertension 2015, 65, 600–606. [Google Scholar] [CrossRef] [PubMed]
- Farag, K.; Hassan, I.; Ledger, W.L. Prediction of preeclampsia: Can it be achieved? Obstet. Gynecol. Surv. 2004, 59, 464–482. [Google Scholar] [CrossRef]
- Barton, J.R.; Sibai, B.M. Prediction and prevention of recurrent preeclampsia. Obstet. Gynecol. 2008, 112, 359–372. [Google Scholar] [CrossRef]
- Telang, M.A.; Bhutkar, S.P.; Hirwani, R.R. Analysis of patents on preeclampsia detection and diagnosis: A perspective. Placenta 2013, 34, 2–8. [Google Scholar] [CrossRef]
- Jeyabalan, A. Epidemiology of preeclampsia: Impact of obesity. Nutr. Rev. 2013, 71 (Suppl. 1), S18–S25. [Google Scholar] [CrossRef]
- Aliyu, M.H.; Luke, S.; Kristensen, S.; Alio, A.P.; Salihu, H.M. Joint effect of obesity and teenage pregnancy on the risk of preeclampsia: A population-based study. J. Adolesc. Health 2010, 46, 77–82. [Google Scholar] [CrossRef]
- Frederick, I.O.; Rudra, C.B.; Miller, R.S.; Foster, J.C.; Williams, M.A. Adult weight change, weight cycling, and prepregnancy obesity in relation to risk of preeclampsia. Epidemiology 2006, 17, 428–434. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, T.E.; Ray, J.G.; Chan, W.S. Maternal body mass index and the risk of preeclampsia: A systematic overview. Epidemiology 2003, 14, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.H.; Seow, K.M.; Chen, L.R. Progression of gestational hypertension to pre-eclampsia: A cohort study of 20,103 pregnancies. Pregnancy Hypertens. 2017, 10, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.H.; Chen, L.R.; Lee, Y.H. Exploring the relationship between preterm placental calcification and adverse maternal and fetal outcome. Ultrasound Obstet. Gynecol. 2011, 37, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.H.; Chen, L.R.; Lee, Y.H. The role of preterm placental calcification in high-risk pregnancy as a predictor of poor uteroplacental blood flow and adverse pregnancy outcome. Ultrasound Med. Biol. 2012, 38, 1011–1018. [Google Scholar] [CrossRef]
- Chen, K.H.; Seow, K.M.; Chen, L.R. The role of preterm placental calcification on assessing the risks of stillbirths. Placenta 2015, 36, 1039–1044. [Google Scholar] [CrossRef]
- Arakaki, T.; Hasegawa, J.; Nakamura, M.; Hamada, S.; Muramoto, M.; Takita, H.; Ichizuka, K.; Sekizawa, A. Prediction of early- and late-onset pregnancy-induced hypertension using placental volume on three-dimensional ultrasound and uterine artery Doppler. Ultrasound Obstet. Gynecol. 2015, 45, 539–543. [Google Scholar] [CrossRef]
- Prefumo, F.; Sebire, N.J.; Thilaganathan, B. Decreased endovascular trophoblast invasion in first trimester pregnancies with high-resistance uterine artery Doppler indices. Hum. Reprod. 2004, 19, 206–209. [Google Scholar] [CrossRef]
- Roos, N.; Kieler, H.; Sahlin, L.; Ekman-Ordeberg, G.; Falconer, H.; Stephansson, O. Risk of adverse pregnancy outcomes in women with polycystic ovary syndrome: Population based cohort study. BMJ 2011, 343, d6309. [Google Scholar] [CrossRef]
- Kashyap, S.; Claman, P. Polycystic ovary disease and the risk of pregnancy-induced hypertension. J. Reprod. Med. 2000, 45, 991–994. [Google Scholar]
- Fridstrom, M.; Nisell, H.; Sjoblom, P.; Hillensjo, T. Are women with polycystic ovary syndrome at an increased risk of pregnancy-induced hypertension and/or preeclampsia? Hypertens. Pregnancy 1999, 18, 73–80. [Google Scholar] [CrossRef] [PubMed]
- De Vries, M.J.; Dekker, G.A.; Schoemaker, J. Higher risk of preeclampsia in the polycystic ovary syndrome. A case control study. Eur. J. Obstet. Gynecol. Reprod. Biol. 1998, 76, 91–95. [Google Scholar] [CrossRef]
- Nili, F.; McLeod, L.; O’Connell, C.; Sutton, E.; McMillan, D. Maternal and neonatal outcomes in pregnancies complicated by systemic lupus erythematosus: A population-based study. J. Obstet. Gynaecol. Can. 2013, 35, 323–328. [Google Scholar] [CrossRef]
- Chakravarty, E.F.; Colon, I.; Langen, E.S.; Nix, D.A.; El-Sayed, Y.Y.; Genovese, M.C.; Druzin, M.L. Factors that predict prematurity and preeclampsia in pregnancies that are complicated by systemic lupus erythematosus. Am. J. Obstet. Gynecol. 2005, 192, 1897–1904. [Google Scholar] [CrossRef] [PubMed]
- Wolfberg, A.J.; Lee-Parritz, A.; Peller, A.J.; Lieberman, E.S. Association of rheumatologic disease with preeclampsia. Obstet. Gynecol. 2004, 103, 1190–1193. [Google Scholar] [CrossRef]
- Naver, K.V.; Grinsted, J.; Larsen, S.O.; Hedley, P.L.; Jorgensen, F.S.; Christiansen, M.; Nilas, L. Increased risk of preterm delivery and pre-eclampsia in women with polycystic ovary syndrome and hyperandrogenaemia. BJOG 2014, 121, 575–581. [Google Scholar] [CrossRef]
- Mikola, M.; Hiilesmaa, V.; Halttunen, M.; Suhonen, L.; Tiitinen, A. Obstetric outcome in women with polycystic ovarian syndrome. Hum. Reprod. 2001, 16, 226–229. [Google Scholar] [CrossRef]
- Pan, M.L.; Chen, L.R.; Tsao, H.M.; Chen, K.H. Iron deficiency anemia as a risk factor for osteoporosis in Taiwan: A nationwide population-based study. Nutrients 2017, 9, 616. [Google Scholar] [CrossRef]
- Kim, M.Y.; Buyon, J.P.; Guerra, M.M.; Rana, S.; Zhang, D.; Laskin, C.A.; Petri, M.; Lockshin, M.D.; Sammaritano, L.R.; Branch, D.W.; et al. Angiogenic factor imbalance early in pregnancy predicts adverse outcomes in patients with lupus and antiphospholipid antibodies: Results of the PROMISSE study. Am. J. Obstet. Gynecol. 2016, 214, 108. [Google Scholar] [CrossRef]
- Ostensen, M.; Clowse, M. Pathogenesis of pregnancy complications in systemic lupus erythematosus. Curr. Opin. Rheumatol. 2013, 25, 591–596. [Google Scholar] [CrossRef]
- Moroni, G.; Ponticelli, C. Pregnancy in women with systemic lupus erythematosus (SLE). Eur. J. Intern. Med. 2016, 32, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Irastorza, G.; Khamashta, M.A. Lupus and pregnancy: Integrating clues from the bench and bedside. Eur. J. Clin. Investig. 2011, 41, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Mok, C.C.; Wong, R.W. Pregnancy in systemic lupus erythematosus. Postgrad. Med. J. 2001, 77, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.F.; Chen, H.S.; Rao, D.P.; Gong, J. Association between polycystic ovary syndrome and the risk of pregnancy complications: A PRISMA-compliant systematic review and meta-analysis. Medicine 2016, 95, e4863. [Google Scholar] [CrossRef] [PubMed]
- Boomsma, C.M.; Eijkemans, M.J.; Hughes, E.G.; Visser, G.H.; Fauser, B.C.; Macklon, N.S. A meta-analysis of pregnancy outcomes in women with polycystic ovary syndrome. Hum. Reprod. Update 2006, 12, 673–683. [Google Scholar] [CrossRef]
- Khan, G.H.; Galazis, N.; Docheva, N.; Layfield, R.; Atiomo, W. Overlap of proteomics biomarkers between women with pre-eclampsia and PCOS: A systematic review and biomarker database integration. Hum. Reprod. 2015, 30, 133–148. [Google Scholar] [CrossRef]
- Palomba, S.; de Wilde, M.A.; Falbo, A.; Koster, M.P.; La Sala, G.B.; Fauser, B.C. Pregnancy complications in women with polycystic ovary syndrome. Hum. Reprod. Update 2015, 21, 575–592. [Google Scholar] [CrossRef]
- Wallace, A.E.; Fraser, R.; Cartwright, J.E. Extravillous trophoblast and decidual natural killer cells: A remodelling partnership. Hum. Reprod. Update 2012, 18, 458–471. [Google Scholar] [CrossRef]
- Hutter, S.; Heublein, S.; Knabl, J.; Andergassen, U.; Vrekoussis, T.; Makrigiannakis, A.; Friese, K.; Mayr, D.; Jeschke, U. Macrophages: Are they involved in endometriosis, abortion and preeclampsia and how? J. Nippon Med. Sch. 2013, 80, 97–103. [Google Scholar] [CrossRef]

| Exposure Group | Contrast Group | Statistics | |||||
|---|---|---|---|---|---|---|---|
| Primiparas with prepregnancy PCOS | Primiparas without PCOS | ||||||
| (n = 1,953) | (n = 7,812) | ||||||
| N | % | N | % | OR | [95% CI] | p-value | |
| Age at diagnosis (y/o) | 27.49 ± 4.83 | 27.49 ± 4.83 | 1.0000 | ||||
| 15–25 | 672 | 34.41 | 2688 | 34.41 | |||
| 26–35 | 1186 | 60.73 | 4744 | 60.73 | |||
| 36–45 | 95 | 4.86 | 380 | 4.86 | |||
| Occupation | <0.0001*** | ||||||
| White collar | 1223 | 62.62 | 4192 | 53.66 | |||
| Blue collar | 321 | 16.44 | 1517 | 19.42 | |||
| Retired and other | 409 | 20.94 | 2103 | 26.92 | |||
| Urbanization | 0.0007** | ||||||
| Urban | 1319 | 67.54 | 4928 | 63.08 | |||
| Suburban | 510 | 26.11 | 2373 | 30.38 | |||
| Rural | 124 | 6.35 | 511 | 6.54 | |||
| Economic status (insurable wage) | <0.0001*** | ||||||
| ≧40,000 NTD | 443 | 22.68 | 1164 | 14.90 | |||
| 20,000–40,000 NTD | 833 | 42.65 | 3378 | 43.24 | |||
| <20,000 NTD | 405 | 20.74 | 1891 | 24.21 | |||
| Retired and others | 272 | 13.93 | 1379 | 17.65 | |||
| Comorbidities | |||||||
| Diabetes mellitus | 86 | 4.4 | 102 | 1.31 | 3.48 | [2.60–4.66] | <0.0001*** |
| Dyslipidemia | 87 | 4.45 | 218 | 2.79 | 1.62 | [1.26–2.09] | 0.0002** |
| Ischemic heart disease | 26 | 1.33 | 108 | 1.38 | 0.96 | [0.62–1.48] | 0.8619 |
| Cerebrovascular disease | 14 | 0.72 | 105 | 1.34 | 0.53 | [0.30–0.93] | 0.0238* |
| Chronic pulmonary disease | 304 | 15.57 | 1190 | 15.23 | 1.03 | [0.89–1.18] | 0.7148 |
| Exposure Group | Contrast Group | Statistics | |||||
|---|---|---|---|---|---|---|---|
| Primiparas with prepregnancy SLE | Primiparas without SLE | ||||||
| (n = 820) | (n = 3280) | ||||||
| N | % | N | % | OR | [95% CI] | p-value | |
| Age at diagnosis (y/o) | 29.51 ± 6.44 | 29.51 ± 6.44 | 1.0000 | ||||
| 15–25 | 232 | 28.29 | 928 | 28.29 | |||
| 26–35 | 430 | 52.44 | 1720 | 52.44 | |||
| 36–45 | 158 | 19.27 | 632 | 19.27 | |||
| Occupation | 0.4790 | ||||||
| White collar | 462 | 56.34 | 1772 | 54.02 | |||
| Blue collar | 166 | 20.24 | 689 | 21.01 | |||
| Retired and other | 192 | 23.41 | 819 | 24.97 | |||
| Urbanization | 0.3930 | ||||||
| Urban | 534 | 65.12 | 2101 | 64.05 | |||
| Suburban | 239 | 29.15 | 947 | 28.87 | |||
| Rural | 47 | 5.73 | 232 | 7.07 | |||
| Economic status (insurable wage) | 0.0146* | ||||||
| ≧40,000 NTD | 184 | 22.44 | 583 | 17.77 | |||
| 20,000–40,000 NTD | 344 | 41.95 | 1427 | 43.51 | |||
| <20,000 NTD | 164 | 20.00 | 752 | 22.93 | |||
| Retired and others | 128 | 15.61 | 518 | 15.79 | |||
| Comorbidities | |||||||
| Diabetes mellitus | 36 | 4.39 | 72 | 2.20 | 2.05 | [1.36–3.08] | 0.0004*** |
| Dyslipidemia | 52 | 6.34 | 121 | 3.69 | 1.77 | [1.27–2.47] | 0.0007*** |
| Ischemic heart disease | 26 | 3.17 | 49 | 1.49 | 2.16 | [1.33–3.50] | 0.0014** |
| Cerebrovascular disease | 29 | 3.54 | 53 | 1.62 | 2.23 | [1.41–3.53] | 0.0004*** |
| Chronic pulmonary disease | 166 | 20.24 | 487 | 14.85 | 1.46 | [1.20–1.77] | 0.0002*** |
| Group | No GH-PE | GH-PE | Statistics | |||
|---|---|---|---|---|---|---|
| N | % | N | % | Crude OR | Adjust OR | |
| [95% CI] | [95% CI] | |||||
| Contrast group (no PCOS; n = 7,812) | 7638 | 97.77 | 174 | 2.23 | Reference | Reference |
| Exposure group (PCOS: n = 1,953) | 1840 | 94.21 | 113 | 5.79 | 2.70*** | 2.36*** |
| [2.12–3.44] | [1.83–3.05] | |||||
| Subgroup in exposure group | ||||||
| No metformin use (n = 1,752) | 1654 | 94.41 | 98 | 5.59 | Reference | Reference |
| Metformin use (n = 201) | 186 | 92.54 | 15 | 7.46 | 1.36 | 1.44 |
| [0.78–2.39] | [0.81–2.56] | |||||
| Contrast group (no SLE; n = 3,280) | 3221 | 98.20 | 59 | 1.80 | Reference | Reference |
| Exposure group (SLE: n = 820) | 792 | 96.59 | 28 | 3.41 | 1.93** | 1.95** |
| [1.22–3.05] | [1.23–3.10] | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, M.-L.; Chen, L.-R.; Tsao, H.-M.; Chen, K.-H. Prepregnancy Endocrine, Autoimmune Disorders and the Risks of Gestational Hypertension-Preeclampsia in Primiparas: A Nationwide Population-Based Study in Taiwan. Int. J. Environ. Res. Public Health 2020, 17, 3657. https://doi.org/10.3390/ijerph17103657
Pan M-L, Chen L-R, Tsao H-M, Chen K-H. Prepregnancy Endocrine, Autoimmune Disorders and the Risks of Gestational Hypertension-Preeclampsia in Primiparas: A Nationwide Population-Based Study in Taiwan. International Journal of Environmental Research and Public Health. 2020; 17(10):3657. https://doi.org/10.3390/ijerph17103657
Chicago/Turabian StylePan, Mei-Lien, Li-Ru Chen, Hsiao-Mei Tsao, and Kuo-Hu Chen. 2020. "Prepregnancy Endocrine, Autoimmune Disorders and the Risks of Gestational Hypertension-Preeclampsia in Primiparas: A Nationwide Population-Based Study in Taiwan" International Journal of Environmental Research and Public Health 17, no. 10: 3657. https://doi.org/10.3390/ijerph17103657
APA StylePan, M.-L., Chen, L.-R., Tsao, H.-M., & Chen, K.-H. (2020). Prepregnancy Endocrine, Autoimmune Disorders and the Risks of Gestational Hypertension-Preeclampsia in Primiparas: A Nationwide Population-Based Study in Taiwan. International Journal of Environmental Research and Public Health, 17(10), 3657. https://doi.org/10.3390/ijerph17103657






