Estimated Impact of Targeted Pre-Exposure Prophylaxis: Strategies for Men Who Have Sex with Men in the United States
Abstract
1. Introduction
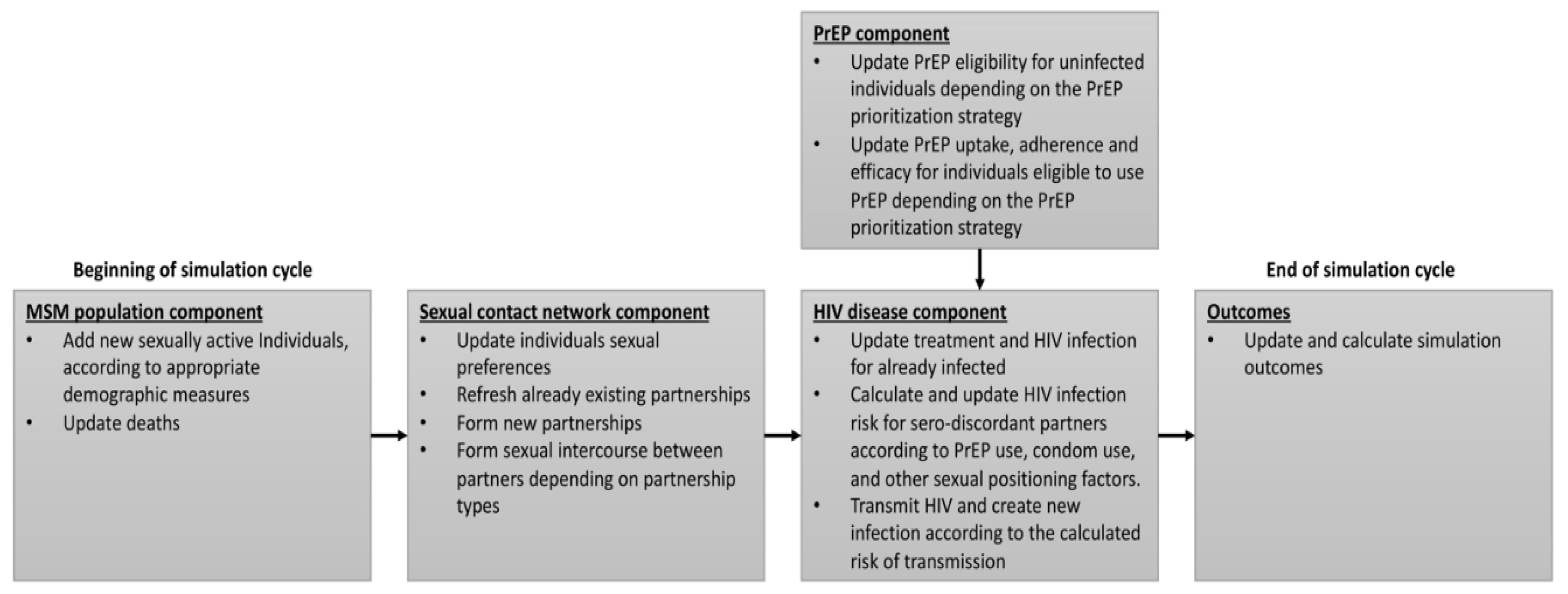
2. Materials and Methods
2.1. MSM Population Component
2.2. Sexual Contact Network
2.3. HIV Disease Model
2.4. PrEP Component
2.4.1. PrEP Prioritization Scenarios
2.4.2. PrEP Uptake, Adherence, and Effectiveness
2.5. Model Outcomes
2.6. Model Calibration
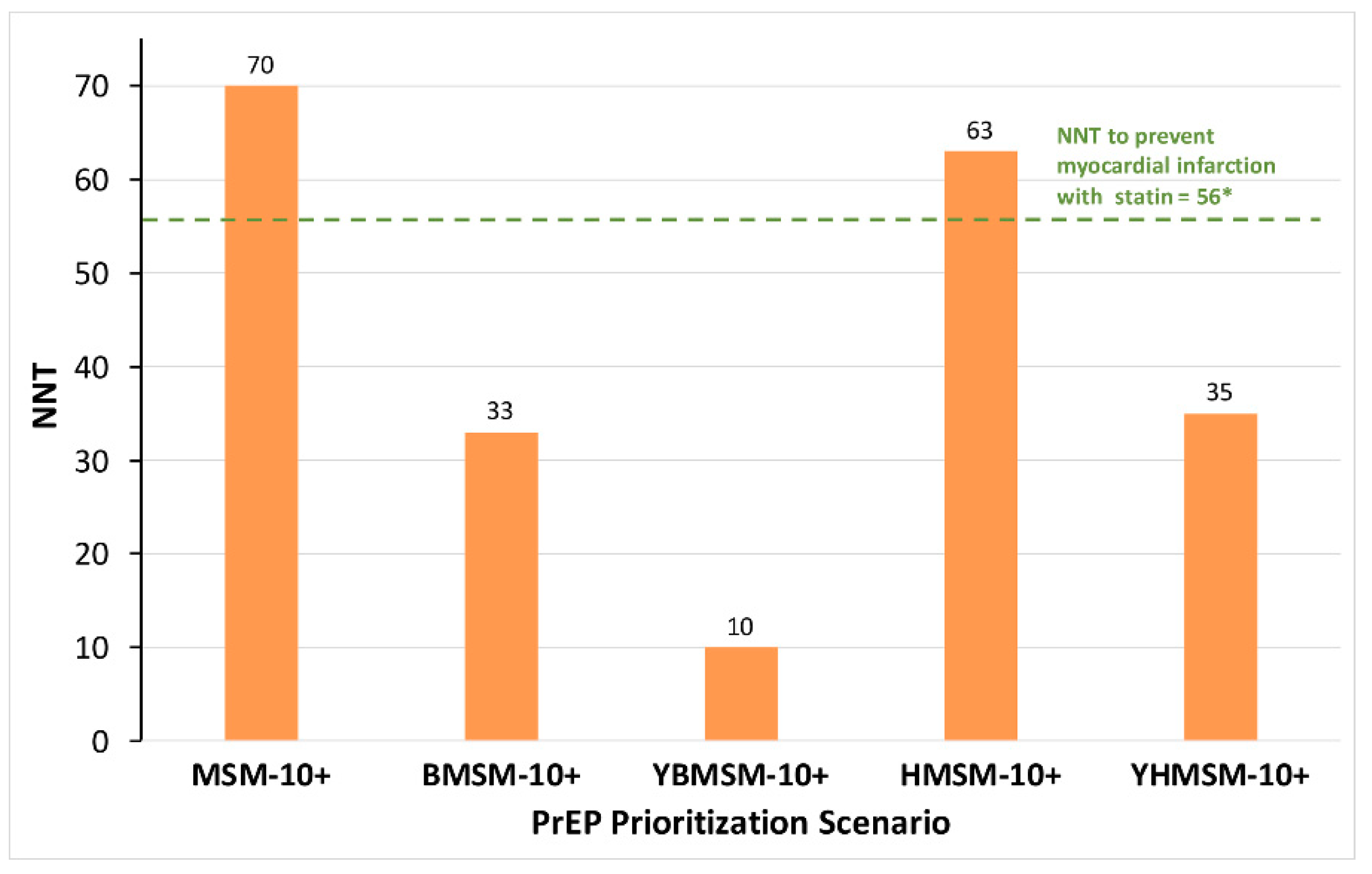
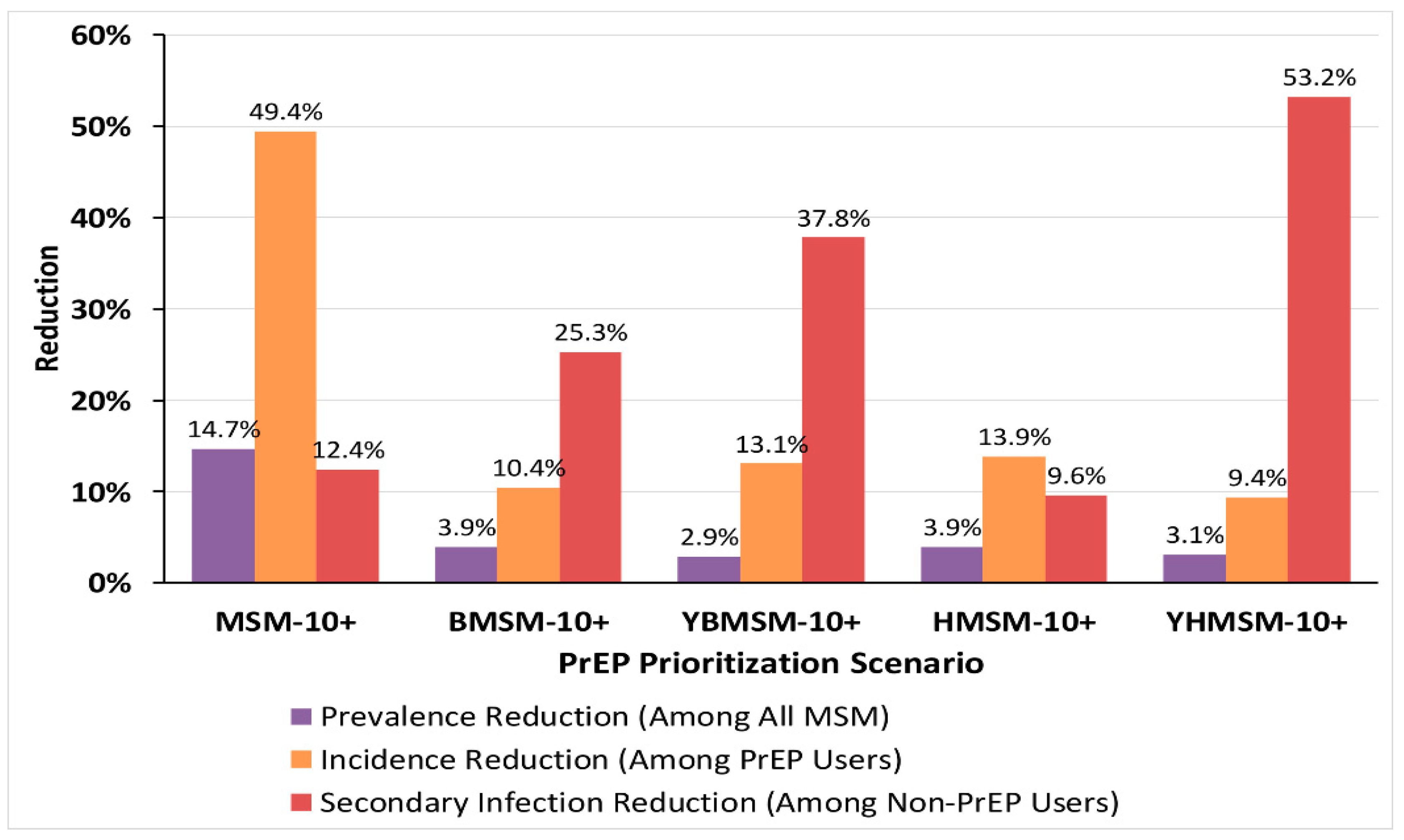
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- HIV/AIDS Basic Statistics. Centers for Disease Control and Prevention, 2016. Available online: https://www.cdc.gov/hiv/basics/statistics.html (accessed on 17 June 2018).
- HIV Prevention in the United States: New Opportunities, New Expectation. Centers for Disease Control and Prevention, National Center for HIV/AIDS, Viral Hepatitis, STD, and TB, 2015. Available online: https://www.cdc.gov/hiv/pdf/policies/cdc-hiv-prevention-bluebook.pdf. (accessed on 17 June 2018).
- TRUVADA® (Emtricitabine/Tenofovir Disoproxil Fumarate) Tablets Label. United States Food and Drug Administration, 2004. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/021752s047lbl.pdf (accessed on 17 June 2018).
- Mera Giler, R.; Trevor, H.; Bush, S.; Rawlings, K.; McCallister, S. Changes in Truvada (TVD) for HIV pre-exposure prophylaxis (PrEP) utilization in the United States: (2012–2016). In Proceedings of the 2017 International AIDS Society Conference on HIV Science, Paris, France, 23–26 July 2017; pp. 23–26. [Google Scholar]
- Hare, C.B.; Coll, J.; Ruane, P. The phase 3 DISCOVER Study: Daily F/TAF or F/TDF for HIV pre-exposure prophylaxis. In Proceedings of the 26th Conference on Retroviruses and Opportunistic Infections, Seattle, WA, USA, 4–7 March 2019. [Google Scholar]
- Smith, D.K.; Handel, M.V.; Gray, J.A. By Race/Ethnicity, Blacks Have Highest Number Needing PrEP in the United States, 2015. In Proceedings of the Conference on Retroviruses and Opportunistic Infections, Boston, MA, USA, 4–7 March 2018. [Google Scholar]
- HIV Infection Risk, Prevention, and Testing Behaviors Among Men Who Have Sex with Men—National HIV Behavioral Surveillance, 23 U.S. Cities, 2017. Centers for Disease Control and Prevention HIV Surveillance Special Report 22. , 2019. Available online: https://www.cdc.gov/hiv/library/reports/hiv-surveillance.html (accessed on 25 April 2019).
- Hall, H.I.; Lee, L.M.; Li, J.; Song, R.; Mckenna, M.T. Describing the HIV/AIDS epidemic: Using HIV case data in addition to AIDS case reporting. Ann. Epidemiol. 2005, 15, 5–12. [Google Scholar] [CrossRef]
- HIV among Gay and Bisexual Men. Centers for Disease Control and Prevention, 2018. Available online: https://www.cdc.gov/hiv/group/msm/index.html (accessed on 17 June 2018).
- Bush, S.; Magnuson, D.; Rawlings, M.K.; Hawkins, T.; McCallister, S.; Mera Giler, R. Racial characteristics of FTC/TDF for pre-exposure prophylaxis (PrEP) users in the US. In Proceedings of the ASM Microbe, Boston, MA, USA, 16–20 June 2016. [Google Scholar]
- Ya-lin, A.H.; Zhu, W.; Smith, D.K.; Harris, N.; Hoover, K.W. HIV preexposure prophylaxis, by race and ethnicity—United States, 2014–2016. Morb. Mortal. Wkly. Rep. 2018, 67, 1147. [Google Scholar]
- Hall, H.I.; Byers, R.H.; Ling, Q.; Espinoza, L. Racial/ethnic and age disparities in HIV prevalence and disease progression among men who have sex with men in the United States. Am. J. Public Health 2007, 97, 1060–1066. [Google Scholar] [CrossRef] [PubMed]
- Kessler, J.; Myers, J.E.; Nucifora, K.A.; Mensah, N.; Toohey, C.; Khademi, A.; Cutler, B.; Braithwaite, R.S. Evaluating the impact of prioritization of antiretroviral pre-exposure prophylaxis (PrEP) in New York City. AIDS 2011. [Google Scholar] [CrossRef]
- Chen, A.; Dowdy, D.W. Clinical effectiveness and cost-effectiveness of HIV pre-exposure prophylaxis in men who have sex with men: Risk calculators for real-world decision-making. PLoS ONE 2014, 9, e108742. [Google Scholar] [CrossRef]
- Juusola, J.L.; Brandeau, M.L.; Owens, D.K.; Bendavid, E. The cost-effectiveness of preexposure prophylaxis for HIV prevention in the United States in men who have sex with men. Ann. Intern. Med. 2012, 156, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Koppenhaver, R.T.; Sorensen, S.W.; Farnham, P.G.; Sansom, S.L. The cost-effectiveness of pre-exposure prophylaxis in men who have sex with men in the United States: An epidemic model. JAIDS J. Acquir. Immune Defic. Syndr. 2011, 58, e51–e52. [Google Scholar] [CrossRef]
- Paltiel, A.D.; Freedberg, K.A.; Scott, C.A.; Schackman, B.R.; Losina, E.; Wang, B.; Seage, G.R.; Sloan, C.E.; Sax, P.E.; Walensky, R.P. HIV preexposure prophylaxis in the United States: Impact on lifetime infection risk, clinical outcomes, and cost-effectiveness. Clin. Infect. Dis. 2009, 48, 806–815. [Google Scholar] [CrossRef]
- Desai, K.; Sansom, S.; Ackers, M.; Stewart, S.; Hall, H.; Hu, D. Modeling the impact of HIV chemoprophylaxis strategies among men who have sex with men in the United States: HIV infections prevented and cost-effectiveness. AIDS 2008, 1829–1839. [Google Scholar] [CrossRef]
- Bendavid, E.; Brandeau, M.L.; Wood, R.; Owens, D.K. Comparative effectiveness of HIV testing and treatment in highly endemic regions. Arch. Intern. Med. 2010, 170, 1347–1354. [Google Scholar] [CrossRef]
- Bershteyn, A.; Klein, D.J.; Eckhoff, P.A. Age-dependent partnering and the HIV transmission chain: A microsimulation analysis. J. R. Soc. Interface 2013, 10, 20130613. [Google Scholar] [CrossRef]
- Beck, E.C.; Birkett, M.; Armbruster, B.; Mustanski, B. A data-driven simulation of HIV spread among young men who have sex with men: The role of age and race mixing, and STIs. J. Acquir. Immune Defic. Syndr. (1999) 2015, 70, 186. [Google Scholar] [CrossRef] [PubMed]
- Goodreau, S.M.; Rosenberg, E.S.; Jenness, S.M.; Luisi, N.; Stansfield, S.E.; Millett, G.A.; Sullivan, P.S. Sources of racial disparities in HIV prevalence in men who have sex with men in Atlanta, GA, USA: A modelling study. Lancet HIV 2017, 4, e311–e320. [Google Scholar] [CrossRef]
- Kelley, C.F.; Rosenberg, E.S.; O’Hara, B.M.; Frew, P.M.; Sanchez, T.; Peterson, J.L.; del Rio, C.; Sullivan, P.S. Measuring population transmission risk for HIV: An alternative metric of exposure risk in men who have sex with men (MSM) in the US. PLoS ONE 2012, 7, e53284. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.K.; Pals, S.L.; Herbst, J.H.; Shinde, S.; Carey, J.W. Development of a clinical screening index predictive of incident HIV infection among men who have sex with men in the United States. J. Acquir. Immune Defic. Syndr. 2012, 60, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Bazghandi, A. Techniques, advantages and problems of agent based modeling for traffic simulation. Int. J. Comput. Sci. 2012, 9, 115–119. [Google Scholar]
- Halkitis, P.N.; Brockwell, S.; Siconolfi, D.E.; Moeller, R.W.; Sussman, R.D.; Mourgues, P.J.; Cutler, B.; Sweeney, M.M. Sexual behaviors of adolescent emerging and young adult men who have sex with men ages 13–29 in New York City. J. Acquir. Immune Defic. Syndr. 2011, 56, 285–291. [Google Scholar] [CrossRef]
- Health Statistics Lifetables. Centers for Disease Control and Prevention, 2011. Available online: ftp://ftp.cdc.gov/pub/Health_Statistics/NCHS/Publications/NVSR/64_11 (accessed on 15 July 2016).
- Zwahlen, M.; Harris, R.; May, M.; Hogg, R.; Costagliola, D.; de Wolf, F.; Gill, J.; Fatkenheuer, G.; Lewden, C.; Saag, M.; et al. Mortality of HIV-infected patients starting potent antiretroviral therapy: Comparison with the general population in nine industrialized countries. Int. J. Epidemiol. 2009, 38, 1624–1633. [Google Scholar] [CrossRef]
- Grey, J.A.; Bernstein, K.T.; Sullivan, P.S.; Purcell, D.W.; Chesson, H.W.; Gift, T.L.; Rosenberg, E.S. Estimating the population sizes of men who have sex with men in US states and counties using data from the American Community Survey. JMIR Public Health Surveill. 2016, 2, e14. [Google Scholar] [CrossRef]
- Purcell, D.; Johnson, C.; Lansky, A.; Prejean, J.; Stein, R.; Denning, P.; Crepaz, N. Calculating HIV and syphilis rates for risk groups: Estimating the national population size of men who have sex with men. In Proceedings of the National STD Prevention Conference, Atlanta, GA, USA, 8–11 March 2010; pp. 8–11. [Google Scholar]
- Lieb, S.; Fallon, S.J.; Friedman, S.R.; Thompson, D.R.; Gates, G.J.; Liberti, T.M.; Malow, R.M. Statewide estimation of racial/ethnic populations of men who have sex with men in the US. Public Health Rep. 2011, 126, 60–72. [Google Scholar] [CrossRef]
- Calabrese, S.K.; Rosenberger, J.G.; Schick, V.R.; Novak, D.S. Pleasure, affection, and love among Black men who have sex with men (MSM) versus MSM of other races: Countering dehumanizing stereotypes via cross-race comparisons of reported sexual experience at last sexual event. Arch. Sex. Behav. 2015, 44, 2001–2014. [Google Scholar] [CrossRef]
- NCHHSTP AtlasPlus. Centers for Disease Control and Prevention, 2017. Available online: https://www.cdc.gov/nchhstp/atlas/index.htm (accessed on 17 June 2018).
- Prestage, G.; Duncan, D.; Grierson, J.; Bradley, J.; Bavinton, B.; Kolstee, J.; Smith, A. Monopoly: A Study of Gay Men’s Relationships, Report 2014; The Kirby Institute, The University of New South Wales: Kensington, Australia, 2015. [Google Scholar]
- Mitchell, J.W. Aspects of gay male couples’ sexual agreements vary by their relationship length. Aids Care 2014, 26, 1164–1170. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Raymond, H.F. Preference for and maintenance of anal sex roles among men who have sex with men: Sociodemographic and behavioral correlates. Arch. Sex. Behav. 2011, 40, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Introcaso, C.E.; Xu, F.; Kilmarx, P.H.; Zaidi, A.; Markowitz, L.E. Prevalence of circumcision among men and boys aged 14 to 59 years in the United States, National Health and Nutrition Examination Surveys 2005–2010. Sex. Transm. Dis. 2013, 40, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Pines, H.A.; Gorbach, P.M.; Weiss, R.E.; Shoptaw, S.; Landovitz, R.J.; Javanbakht, M.; Ostrow, D.G.; Stall, R.D.; Plankey, M. Sexual risk trajectories among MSM in the United States: Implications for pre-exposure prophylaxis delivery. J. Acquir. Immune Defic. Syndr. 2014, 65, 579–586. [Google Scholar] [CrossRef] [PubMed]
- EXPLORE Study. A Randomized Clinical Trial of the Efficacy of a Behavioral Intervention to Prevent Acquisition of HIV Among Men who have Sex with Men. 1999–2003. Available online: https://www.hptn.org/sites/default/files/2016-05/hivnet_015.pdf (accessed on 20 March 2019).
- Paz-Bailey, G.; Hall, H.I.; Wolitski, R.J.; Prejean, J.; Van Handel, M.M.; Le, B.; LaFlam, M.; Koenig, L.J.; Mendoza, M.C.; Rose, C.; Valleroy, L.A. HIV testing and risk behaviors among gay, bisexual, and other men who have sex with men—United States. Morb. Mortal. Wkly. Rep. 2013, 62, 958. [Google Scholar]
- Centers for Disease Control and Prevention. HIV Surveillance Report; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2008. [Google Scholar]
- Millett, G.A.; Peterson, J.L.; Flores, S.A.; Hart, T.A.; Jeffries, W.L.t.; Wilson, P.A.; Rourke, S.B.; Heilig, C.M.; Elford, J.; Fenton, K.A.; et al. Comparisons of disparities and risks of HIV infection in black and other men who have sex with men in Canada, UK, and USA: A meta-analysis. Lancet 2012, 380, 341–348. [Google Scholar] [CrossRef]
- Patel, P.; Borkowf, C.B.; Brooks, J.T.; Lasry, A.; Lansky, A.; Mermin, J. Estimating per-act HIV transmission risk: A systematic review. AIDS 2014, 28, 1509–1519. [Google Scholar] [CrossRef]
- Hollingsworth, T.D.; Anderson, R.M.; Fraser, C. HIV-1 transmission, by stage of infection. J. Infect. Dis. 2008, 198, 687–693. [Google Scholar] [CrossRef]
- Grant, R.M.; Anderson, P.L.; McMahan, V.; Liu, A.; Amico, K.R.; Mehrotra, M.; Hosek, S.; Mosquera, C.; Casapia, M.; Montoya, O. Uptake of pre-exposure prophylaxis, sexual practices, and HIV incidence in men and transgender women who have sex with men: A cohort study. Lancet Infect. Dis. 2014, 14, 820–829. [Google Scholar] [CrossRef]
- Babiker, A.; Darby, S.; De Angelis, D.; Kwart, D.; Porter, K.; Beral, V.; Darbyshire, J.; Day, N.; Gill, N.; Coutinho, R.; et al. Time from HIV-1 seroconversion to AIDS and death before widespread use of highly-active antiretroviral therapy: A collaborative re-analysis. Collaborative Group on AIDS Incubation and HIV Survival including the CASCADE EU Concerted Action. Concerted Action on SeroConversion to AIDS and Death in Europe. Lancet 2000, 355, 1131–1137. [Google Scholar]
- Cohen, S.E.; Vittinghoff, E.; Bacon, O.; Doblecki-Lewis, S.; Postle, B.S.; Feaster, D.J.; Matheson, T.; Trainor, N.; Blue, R.W.; Estrada, Y.; et al. High interest in preexposure prophylaxis among men who have sex with men at risk for HIV infection: Baseline data from the US PrEP demonstration project. J. Acquir. Immune Defic. Syndr. 2015, 68, 439–448. [Google Scholar] [CrossRef]
- Liu, A.Y.; Cohen, S.E.; Vittinghoff, E.; Anderson, P.L.; Doblecki-Lewis, S.; Bacon, O.; Chege, W.; Postle, B.S.; Matheson, T.; Amico, K.R.; et al. Preexposure Prophylaxis for HIV Infection Integrated with Municipal- and Community-Based Sexual Health Services. JAMA Intern. Med. 2016, 176, 75–84. [Google Scholar] [CrossRef]
- Taylor, F.; Ward, K.; Moore, T.; Burke, M.; Davey Smith, G.; Casas, J.P.; Ebrahim, S. Statins for the primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2011, 1. [Google Scholar] [CrossRef]
- Gomez, G.B.; Borquez, A.; Case, K.K.; Wheelock, A.; Vassall, A.; Hankins, C. The cost and impact of scaling up pre-exposure prophylaxis for HIV prevention: A systematic review of cost-effectiveness modelling studies. PLoS Med. 2013, 10, e1001401. [Google Scholar] [CrossRef]
- National Research Council. HIV Screening and Access to Care: Exploring Barriers and Facilitators to Expanded HIV Testing; The National Academies Press: Washington, DC, USA, 2010. [Google Scholar]
- Washington, T.A.; D’Anna, L.; Meyer-Adams, N.; Malotte, C.K. From their voices: Barriers to HIV testing among black men who have sex with men remain. Healthcare 2015, 3, 933–947. [Google Scholar] [CrossRef]
- Robinson, R.; Moodie-Mills, A. HIV/AIDS Inequality: Structural Barriers to Prevention, Treatment, and Care in Communities of Color. Center for American Progress, 2012. Available online: https://www.americanprogress.org/issues/lgbt/reports/2012/07/27/11834/hivaids-inequality-structural-barriers-to-prevention-treatment-and-care-in-communities-of-color/ (accessed on 17 June 2018).
- Oster, A.M.; Wiegand, R.E.; Sionean, C.; Miles, I.J.; Thomas, P.E.; Melendez-Morales, L.; Le, B.C.; Millett, G.A. Understanding disparities in HIV infection between black and white MSM in the United States. AIDS 2011, 25, 1103–1112. [Google Scholar] [CrossRef]
- Sullivan, P.S.; Peterson, J.; Rosenberg, E.S.; Kelley, C.F.; Cooper, H.; Vaughan, A.; Salazar, L.F.; Frew, P.; Wingood, G.; DiClemente, R. Understanding racial HIV/STI disparities in black and white men who have sex with men: A multilevel approach. PLoS ONE 2014, 9, e90514. [Google Scholar] [CrossRef]
- Elopre, L.; Kudroff, K.; Westfall, A.O.; Overton, E.T.; Mugavero, M.J. The right people, right places, and right practices: Disparities in PrEP access among African American men, women and MSM in the Deep South. J. Acquir. Immune Defic. Syndr. (1999) 2017, 74, 56. [Google Scholar] [CrossRef]
- Brooks, R.A.; Etzel, M.A.; Hinojos, E.; Henry, C.L.; Perez, M. Preventing HIV among Latino and African American gay and bisexual men in a context of HIV-related stigma, discrimination, and homophobia: Perspectives of providers. AIDS Patient Care STDs 2005, 19, 737–744. [Google Scholar] [CrossRef]
| HIRI-MSM Risk Index [24] | |||
|---|---|---|---|
| 1. | How old are you today (years)? | <18 years | Score 0 |
| 18–28 years | Score 8 | ||
| 29–40 years | Score 5 | ||
| 41–48 years | Score 2 | ||
| ≥49 years | Score 0 | ||
| 2. | How many men have you had sex with in the last 6 months? | >10 male partners | Score 7 |
| 6–10 male partners | Score 4 | ||
| 0–5 male partners | Score 0 | ||
| 3. | In the last 6 months, how many times did you have receptive anal sex (you were the bottom) with a man? | 1 or more times | Score 10 |
| 0 times | Score 0 | ||
| 4. | How many of your male sex partners were HIV-positive? | >1 positive partner | Score 8 |
| 1 positive partner | Score 4 | ||
| <1 positive partner | Score 0 | ||
| 5. | In the last 6 months, how many times did you have insertive anal sex (you were the top) with a man who was HIV-positive? | 5 or more times | Score 6 |
| 0 times | Score 0 | ||
| 6. | In the last 6 months, have you used methamphetamines such as crystal or speed? | Yes | Score 5 |
| No | Score 0 | ||
| 7. | In the last 6 months, have you used poppers (amyl nitrate)? | Yes | Score 3 |
| No | Score 0 | ||
| The total of entries in right column is the calculated score. | Total Score | ||
| Model Parameter | Value | Source | |||
|---|---|---|---|---|---|
| MSM population (estimated during 2009 to 2013) | 4,503,080 (3.9% of the United States population) | [29,30] | |||
| Distribution of race/ethnicity among MSM (%) | [31] | ||||
| Black | 8.9 | ||||
| White | 71.4 | ||||
| Latino | 15.9 | ||||
| Other | 3.8 | ||||
| MSM age distribution by race/ethnicity (%) | Black | White | Latino | Other | [32] |
| 18–19 | 4.5 | 2.2 | 4.4 | 2.9 | |
| 20–29 | 34.2 | 24.8 | 39.8 | 44.2 | |
| 30–39 | 23.8 | 20.4 | 28.0 | 30.9 | |
| 40–49 | 25.9 | 28.5 | 21.1 | 16.1 | |
| 50–59 | 10.1 | 18.2 | 5.6 | 5.7 | |
| 60+ | 1.5 | 5.9 | 1.1 | 0.2 | |
| Prevalence of HIV among the MSM population (average during 2009 to 2013) | 448,026 (10% of the MSM population) | [33] | |||
| Distribution of race/ethnicity among HIV-infected MSM (average during 2009 to 2013) (%) | [33] | ||||
| Black | 29.9 | ||||
| White | 43.9 | ||||
| Latino | 20.7 | ||||
| Other | 5.5 | ||||
| Standardized mortality ratios for different HIV stages | [28] | ||||
| Acute stage, suppressed or unsuppressed viral load | 1 | ||||
| Chronic Stage, diagnosed, suppressed viral load | 1.05 | ||||
| Chronic Stage, diagnosed, unsuppressed viral load | 2.06 | ||||
| Chronic Stage, undiagnosed, unsuppressed viral load | 3.51 | ||||
| Early final stage (AIDS) | 23.4 | ||||
| End final stage (AIDS) | 24.9 | ||||
| Mean age of sexual onset (years) | [26] | ||||
| Black | 16.4 | ||||
| Latino | 16.3 | ||||
| White | 17.4 | ||||
| Other | 16.9 | ||||
| Distribution for the desired types of partnerships | [34] | ||||
| Casual | 14.6% | ||||
| Regular | 51.1% | ||||
| Both | 34.3% | ||||
| Distribution for the duration of regular partnership | [35] | ||||
| Short-term | 64% | ||||
| Long-term | 36% | ||||
| Rate of regular partnership dissolution | [35] | ||||
| Short-term | 0.38565 | ||||
| Long-term | 0.09977 | ||||
| Distribution for the positional preferences | |||||
| Insertive | 30.3% | [36] | |||
| Receptive | 14.2% | ||||
| Versatile | 55.4% | ||||
| Proportion of MSM with circumcision by race | [37] | ||||
| Black | 0.757 | ||||
| White | 0.908 | ||||
| Latino | 0.440 | ||||
| Other | 0.440 | ||||
| Rate of condom use by risk group | |||||
| Low-risk MSM | 100% | [38] | |||
| High-risk MSM—receptive intercourse with HIV-infected partner | 39% | [39] | |||
| High-risk MSM—insertive intercourse with HIV-infected partner | 45% | [39] | |||
| Testing frequency categories for HIV-uninfected MSM | [40] | ||||
| Never | 20.6% | ||||
| High frequency | 64.3% | ||||
| Low frequency | 15.1% | ||||
| Testing rate for the high-frequency category | 0.00509 | [40] | |||
| Testing rate for the low-frequency category | 0.00061 | ||||
| Proportion of MSM who remained in care upon HIV diagnosis | |||||
| Non-Black MSM | 0.40000 | [41] | |||
| Black MSM | 0.31596 | [41,42] | |||
| PrEP Prioritization Scenario | Eligibility Among All Noninfected MSM (%) | PrEP Uptake Among Eligible | Average Per-Person Time on PrEP (Years) | Reduction in Total New Infections 1 |
|---|---|---|---|---|
| MSM-10+ | 51% | 73% | 3.00 | 50% |
| BMSM-10+ | 5% | 67% | 2.38 | 14% |
| YBMSM-10+ | 2% | 66% | 2.50 | 8% |
| HMSM-10+ | 13% | 74% | 2.97 | 16% |
| YHMSM-10+ | 5% | 69% | 2.99 | 8% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elion, R.A.; Kabiri, M.; Mayer, K.H.; Wohl, D.A.; Cohen, J.; Beaubrun, A.C.; Altice, F.L. Estimated Impact of Targeted Pre-Exposure Prophylaxis: Strategies for Men Who Have Sex with Men in the United States. Int. J. Environ. Res. Public Health 2019, 16, 1592. https://doi.org/10.3390/ijerph16091592
Elion RA, Kabiri M, Mayer KH, Wohl DA, Cohen J, Beaubrun AC, Altice FL. Estimated Impact of Targeted Pre-Exposure Prophylaxis: Strategies for Men Who Have Sex with Men in the United States. International Journal of Environmental Research and Public Health. 2019; 16(9):1592. https://doi.org/10.3390/ijerph16091592
Chicago/Turabian StyleElion, Richard A., Mina Kabiri, Kenneth H. Mayer, David A. Wohl, Joshua Cohen, Anne C. Beaubrun, and Frederick L. Altice. 2019. "Estimated Impact of Targeted Pre-Exposure Prophylaxis: Strategies for Men Who Have Sex with Men in the United States" International Journal of Environmental Research and Public Health 16, no. 9: 1592. https://doi.org/10.3390/ijerph16091592
APA StyleElion, R. A., Kabiri, M., Mayer, K. H., Wohl, D. A., Cohen, J., Beaubrun, A. C., & Altice, F. L. (2019). Estimated Impact of Targeted Pre-Exposure Prophylaxis: Strategies for Men Who Have Sex with Men in the United States. International Journal of Environmental Research and Public Health, 16(9), 1592. https://doi.org/10.3390/ijerph16091592





