Dengue Vector Control through Community Empowerment: Lessons Learned from a Community-Based Study in Yogyakarta, Indonesia
Abstract
1. Introduction
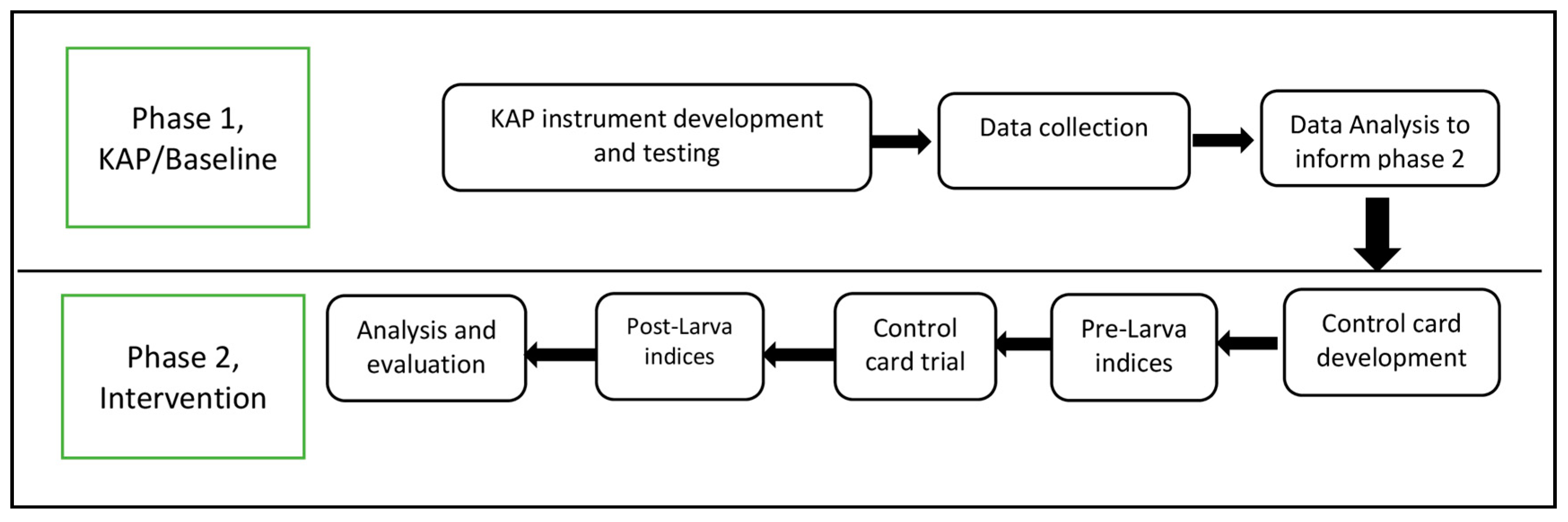
2. Materials and Methods
2.1. Study Design
2.2. Study Instruments
2.2.1. Baseline KAP Survey
2.2.2. Control Cards and Monitoring Forms
2.3. Sample and Procedure
2.3.1. Baseline Survey
2.3.2. Pre-Post Intervention
2.3.3. Statistical Analysis
2.4. Ethical Considerations
3. Results
3.1. Baseline Assessment through KAP Survey
3.1.1. Knowledge
3.1.2. Attitude
3.1.3. Practice
3.2. Control Card Feasibility Study
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wu, X.; Lu, Y.; Zhou, S.; Chen, L.; Xu, B. Impact of climate change on human infectious diseases: Empirical evidence and human adaptation. Environ. Int. 2016, 86, 14–23. [Google Scholar] [CrossRef]
- Liu-Helmersson, J.; Quam, M.; Wilder-Smith, A.; Stenlund, H.; Ebi, K.; Massad, E.; Rocklöv, J. Climate Change and Aedes Vectors: 21st Century Projections for Dengue Transmission in Europe. EBioMedicine 2016, 7, 267–277. [Google Scholar] [CrossRef]
- Ebi, K.L.; Nealon, J. Dengue in a changing climate. Environ. Res. 2016, 151, 115–123. [Google Scholar] [CrossRef]
- Saker, L.; Lee, K.; Cannito, B.; Gilmore, A.; Campbell-Lendrum, D. Globalization and Infectious Diseases: A Review of the Linkages; World Health Organization: Geneva, Switzerland, 2004; p. 63. [Google Scholar]
- Pratamawati, D.A. The role of “juru pantau jentik” in Dengue Haemorrhagic Fever early warning system in Indonesia. Kesmas 2010, 6, 243–248. [Google Scholar]
- Kusriastuti, R.; Sutomo, S. Evolution of Dengue Prevention and Control Programme in Indonesia. Dengue Bull. 2005, 29, 1–7. [Google Scholar]
- Indonesia Ministry of Health. Situasi DBD in Indonesia; Indonesia Ministry of Health: Jakarta, Indonesia, 2016; pp. 1–12.
- City Health Office oh Yogyakarta. Profil Kesehatan Tahun 2015 Kota Yogyakarta (Data Tahun 2014); City Health Office oh Yogyakarta: Yogyakarta, Indonesia, 2015. Available online: http://www.depkes.go.id/resources/download/profil/PROFIL_KAB_KOTA_2014/3471_DIY_Kota_Yogyakarta_2014.pdf (accessed on 1 February 2017).
- Indonesia Ministry of Health. Indonesia Health Profile Year 2015, 2016th ed.; Indonesia Ministry of Health: Jakarta, Indonesia, 2016; p. 403.
- Satria. 2017, Kasus DBD di Yogyakarta Mencapai 53 Kasus; Universitas Gadjah Mada: Yogyakarta, Indonesia, 2017; Available online: https://www.ugm.ac.id/id/berita/13206-2017.kasus.dbd.di.yogyakarta.mencapai.53.kasus (accessed on 11 September 2017).
- Indonesia Ministry of Health. Data and Information, Indonesia Health Profile Year 2016; Indonesia Ministry of Health: Jakarta, Indonesia, 2017; p. 100. Available online: http://www.depkes.go.id/resources/download/pusdatin/lain-lain/Data dan Informasi Kesehatan Profil Kesehatan Indonesia 2016-smaller size-web.pdf (accessed on 19 March 2019).
- Ika. DHF Cases in Yogyakarta City Increase (Bahasa). UGM News. Available online: https://ugm.ac.id/id/news/17658-kasus.dbd.di.kota.yogyakarta.meningkat (accessed on 8 February 2019).
- WHO. Dengue and Severe Dengue. 2018. Available online: http://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue (accessed on 17 October 2018).
- Achee, N.L.; Gould, F.; Perkins, T.A.; Reiner, R.C.; Morrison, A.C.; Ritchie S a Gubler, D.J.; Teyssou, R.; Scott, T.W. A critical assessment of vector control for Dengue prevention. PLoS Negl. Trop. Dis. 2015, 9, e0003655. [Google Scholar] [CrossRef]
- WHO. Global Strategy for Dengue Prevention and Control 2012–2020, 1st ed.; WHO: Geneva, Switzerland, 2012; pp. 1–43. Available online: http://apps.who.int/iris/bitstream/10665/75303/1/9789241504034_eng.pdf (accessed on 19 March 2019).
- Erlanger, T.E.; Keiser, J.; Utzinger, J. Effect of dengue vector control interventions on entomological parameters in developing countries: A systematic review and meta-analysis. Med. Vet. Entomol. 2008, 22, 203–221. [Google Scholar] [CrossRef]
- Tapia-Conyer, R.; Méndez-Galván, J.; Burciaga-Zúñiga, P. Community participation in the prevention and control of dengue: The patio limpio strategy in Mexico. Pediatr. Int. Child Health 2012, 32, 10–14. [Google Scholar] [CrossRef]
- Khun, S.; Manderson, L. Community participation and social engagement in the prevention and control of dengue fever in rural Cambodia. Dengue Bull. 2008, 32, 145–155. [Google Scholar]
- Ceptureanu, S.I.; Ceptureanu, E.G.; Luchian, C.E.; Luchian, I. Community based programs sustainability. A multidimensional analysis of sustainability factors. Sustainability 2018, 10, 870. [Google Scholar] [CrossRef]
- Indonesia Ministry of Health. General Information of Dengue Fever; Special; Indonesia Ministry of Health: Jakarta, Indonesia, 2011; pp. 1–5. Available online: http://www.pppl.depkes.go.id/_asset/_download/INFORMASI_UMUM_DBD_2011.pdf (accessed on 19 March 2019).
- Kusriastuti, R.; Suroso, T.; Nalim, S.; Kusumadi, W. “Together Picket”: Community Activities in Dengue Source Reduction in Purwokerto City, Central Java, Indonesia Country setting and background Planning innovation for dengue prevention and. Dengue Bull. 2004, 28, 35–38. [Google Scholar]
- Shay, M. Guidelines for Reliability, Confirmatory and Exploratory Factor Analysis. Statistic Data Analysis. 2015, pp. 1–15. Available online: http://www.wuss.org/proceedings09/09WUSSProceedings/papers/anl/ANL-SuhrShay.pdf (accessed on 19 March 2019).
- Andersson, N.; Nava-Aguilera, E.; Arosteguí, J.; Morales-Perez, A.; Suazo-Laguna, H.; Legorreta-Soberanis, J.; Hernandez-Alvarez, C.; Fernandez-Salas, I.; Paredes-Solís, S.; Balmaseda, A.; et al. Evidence based community mobilization for dengue prevention in Nicaragua and Mexico (Camino Verde, the Green Way): Cluster randomized controlled trial. BMJ 2015, 351, 1–9. [Google Scholar] [CrossRef]
- Heintze, C.; Garrido, M.V.; Kroeger, A. What do community-based dengue control programmes achieve? A systematic review of published evaluations. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 317–325. [Google Scholar] [CrossRef]
- Harapan, H.; Rajamoorthy, Y.; Anwar, S.; Bustamam, A.; Radiansyah, A.; Angraini, P.; Fasli, R.; Salwiyadi, S.; Bastian, R.A.; Oktiviyari, A.; et al. Knowledge, attitude, and practice regarding dengue virus infection among inhabitants of Aceh, Indonesia: A cross-sectional study. BMC Infect. Dis. 2018, 18, 96. [Google Scholar] [CrossRef]
- Wong, L.P.; Shakir, S.M.M.; Atefi, N.; AbuBakar, S. Factors affecting dengue prevention practices: Nationwide survey of the Malaysian public. PLoS ONE 2015, 10, e0122890. [Google Scholar] [CrossRef]
- Shuaib, F.; Todd, D.; Campbell-Stennett, D.; Ehiri, J.; Jolly, P.E. Knowledge, attitudes and practices regarding dengue infection in Westmoreland, Jamaica. West Indian Med. J. 2010, 59, 139–146. [Google Scholar]
- Kumaran, E.; Doum, D.; Keo, V.; Sokha, L.; Sam, B.L.; Chan, V.; Alexander, N.; Bradley, J.; Liverani, M.; Prasetyo, D.B.; et al. Dengue knowledge, attitudes and practices and their impact on community-based vector control in rural Cambodia. PLoS Negl. Trop. Dis. 2018, 12, e0006268. [Google Scholar] [CrossRef]
- Al-Zurfi, B.M.N.; Fuad, M.D.F.; Abdelqader, M.A.; Baobaid, M.F.; Elnajeh, M.; Ghazi, H.F.; Ibrahim, M.H.; Abdullah, M.R. Knowledge, attitude and practice of dengue fever and heath education programme among students of Alam Shah science school, Cheras, Malaysia. Malays. J. Public Health Med. 2015, 15, 69–74. [Google Scholar]
- University of Twente. Explaining Health Belief Model. 2017. Available online: https://www.utwente.nl/en/bms/communication-theories/sorted-by-cluster/Health Communication/Health_Belief_Model/ (accessed on 16 October 2018).
- Dhimal, M.; Aryal, K.K.; Dhimal, M.L.; Gautam, I.; Singh, S.P.; Bhusal, C.L.; Kuch, U. Knowledge, attitude and practice regarding dengue fever among the healthy population of highland and lowland communities in Central Nepal. PLoS ONE 2014, 9, e102028. [Google Scholar] [CrossRef]
- Spiegel, J.; Bennett, S.; Hattersley, L.; Hayden, M.H.; Kittayapong, P.; Nalim, S.; Wang, D.N.C.; Zielinski-Gutiérrez, E.; Gubler, D. Barriers and bridges to prevention and control of dengue: The need for a social-ecological approach. Ecohealth 2005, 2, 273–290. [Google Scholar] [CrossRef]
- Hinton, P.R.; Brownlow, C.; McMurray, I.; Cozens, B. SPSS Explained; Routledge Taylor and Francis Group: London, UK; New York, NY, USA, 2004. [Google Scholar]
- Higgins, E.; Tatham, L. Learning and Teaching in Action: Assessment. Learn. Teach. Action 2003, 2, 1–12. [Google Scholar]
- Tana, S.; Umniyati, S.; Petzold, M.; Kroeger, A.; Sommerfeld, J. Building and analyzing an innovative community-centered dengue-ecosystem management intervention in Yogyakarta, Indonesia. Pathog. Glob. Health 2012, 106, 469–478. [Google Scholar] [CrossRef]
| Socio-Demographic | N (%) | Mean Score | ||
|---|---|---|---|---|
| Knowledge ± SD | Attitude ± SD | Practice ± SD | ||
| All participants | 521 (100) | 3.7 ± 1.6 | 25.5 ± 4.0 | 9.2 ± 1.3 |
| Sub-district | ||||
| Mantrijeron | 257 (49.3) | 3.7 ± 1.6 | 25.8 ± 3.4 | 9.2 ± 1.2 |
| Demangan | 264 (50.7) | 3.7 ± 1.5 | 25.5 ± 3.7 | 9.2 ± 1.3 |
| Sex | ||||
| Male | 234 (44.9) | 3.6 ± 1.5 | 25.1 ± 4.1 | 8.9 ± 1.4 |
| Female | 287 (55.1) | 3.8 ± 1.7 | 26.1 ± 2.9 | 9.4 ± 1.1 |
| Age group (years) | ||||
| 15–29 | 71 (13.6) | 3.9 ± 1.4 | 26.5 ± 2.7 | 9.0 ± 1.3 |
| 30–44 | 142 (27.3) | 4.2 ± 1.6 | 26.7 ± 3.8 | 9.4 ± 1.2 |
| 45–59 | 180 (34.5) | 3.6 ± 1.6 | 25.2 ± 3.4 | 9.1 ± 1.4 |
| >60 | 128 (24.6) | 3.3 ± 1.5 | 24.7 ± 3.5 | 9.2 ± 1.1 |
| Education | ||||
| Primary | 61 (11.7) | 2.7 ± 1.5 | 23.8 ± 3.8 | 9.1 ± 1.6 |
| Secondary | 311 (59.7) | 3.8 ± 1.6 | 25.8 ± 3.5 | 9.2 ± 1.1 |
| University degree | 126 (24.2) | 4.3 ± 1.5 | 26.6 ± 3.2 | 9.2 ± 1.4 |
| Illiterate | 23 (4.4) | 2.7 ± 1.3 | 23.7 ± 2.0 | 8.9 ± 1.5 |
| Occupation | ||||
| Government sector | 27 (5.2) | 4.7 ± 1.2 | 27.6 ± 2.7 | 8.9 ± 1.7 |
| Private sector | 188 (36.1) | 3.7 ± 1.6 | 25.9 ± 3.4 | 9.1 ± 1.4 |
| Student | 25 (4.8) | 3.8 ± 1.5 | 25.9 ± 2.9 | 8.4 ± 1.2 |
| Seasonal worker | 16 (3.1) | 3.2 ± 1.9 | 24.2 ± 1.6 | 8.4 ± 1.9 |
| Retired | 46 (8.8) | 3.6 ± 1.5 | 25.5 ± 2.7 | 9.4 ± 0.9 |
| Housewife | 160 (30.7) | 3.8 ± 1.7 | 25.5 ± 3.2 | 9.4 ± 0.9 |
| Unemployed | 59 (11.3) | 3.2 ± 1.2 | 23.4 ± 5.2 | 9.2 ± 1.0 |
| Study Population | Mantrijeron N = 257 | Demangan N = 264 | Total N = 521 |
|---|---|---|---|
| Knowledge Item | Correct Answer N (%) | ||
| Dengue, agent, symptom, transmission, treatment | |||
| → DHF is an abbreviation for Dengue Haemorrhagic Fever | 93 (36.2) | 109 (41.3) | 202 (38.8) |
| → Using repellent in the morning till evening is one way to prevent dengue | 72 (28.0) | 71 (26.9) | 143 (27.4) |
| → Having several days of high fever is one dengue symptom | 71 (27.6) | 72 (27.3) | 143 (27.4) |
| → Paracetamol and sponging with tepid water are types of first aid when infected with dengue. | 72 (28.0) | 65 (24.6) | 137 (26.3) |
| → Discarded material and bathtubs are potential Aedes aegypti breeding sites inside the house. | 156 (60.7) | 145 (54.9) | 301 (57.8) |
| → Aedes aegypti biting time is in the morning till evening | 99 (38.5) | 96 (36.4) | 195 (37.4) |
| → Dengue cannot be transmitted by direct contact with a dengue patient | 210 (81.7) | 216 (81.8) | 426 (81.8) |
| → Ditches are not potential breeding sites for Aedes | 75 (29.2) | 72 (27.3) | 147 (28.2) |
| Study Population | Mantrijeron N = 257 | Demangan N = 264 | Total N = 521 |
|---|---|---|---|
| Attitude Statement | N (%) | ||
| I don’t bother with the larvae in the indoor water container | |||
| Strongly agree | 7 (2.7) | 6 (2.3) | 13 (2.5) |
| Agree | 4 (1.6) | 11 (4.2) | 15 (2.9) |
| Disagree | 125 (48.6) | 129 (48.9) | 254 (48.8) |
| Strongly disagree | 120 (46.7) | 116 (43.9) | 236 (45.3) |
| Don’t know | 1 (0.4) | 2 (0.8) | 3 (0.6) |
| I need to take my family to a hospital immediately if infected by DF | |||
| Strongly agree | 143 (55.6) | 131 (49.6) | 274 (52.6) |
| Agree | 110 (42.8) | 121 (45.8) | 231 (44.3) |
| Disagree | 1 (0.4) | 6 (2.3) | 7 (1.3) |
| Strongly disagree | 2 (0.8) | 4 (1.5) | 6 (1.2) |
| Don’t know | 1 (0.4) | 2 (0.8) | 3 (0.6) |
| It is not necessary to clean up the bathtub routinely if not dirty | |||
| Strongly agree | 4 (1.6) | 4 (1.5) | 8 (1.5) |
| Agree | 32 (12.5) | 43 (16.3) | 72 (14.4) |
| Disagree | 164 (63.8) | 166 (62.9) | 330 (63.3) |
| Strongly disagree | 56 (21.8) | 49 (18.6) | 105 (20.2) |
| Don’t know | 1 (0.4) | 2 (0.8) | 3 (0.6) |
| It is necessary to brush bathtubs to eliminate mosquito eggs | |||
| Strongly agree | 108 (42.0) | 112 (42.4) | 220 (42.2) |
| Agree | 133 (51.8) | 138 (52.3) | 271 (52.0) |
| Disagree | 6 (2.3) | 4 (1.5) | 10 (1.9) |
| Strongly disagree | 8 (3.1) | 8 (3.0) | 16 (3.1) |
| Don’t know | 2 (0.8) | 2 (0.8) | 4 (0.8) |
| I will leave the unused plastic mineral water cans outside my house | |||
| Strongly agree | 4 (1.6) | 5 (1.9) | 9 (1.7) |
| Agree | 9 (3.5) | 14 (5.3) | 23 (4.4) |
| Disagree | 149 (58.0) | 148 (56.1) | 297 (57.0) |
| Strongly disagree | 93 (36.2) | 95 (36.0) | 188 (36.1) |
| Don’t know | 2 (0.8) | 2 (0.8) | 4 (0.8) |
| I don’t need to monitor larvae in my environment | |||
| Strongly agree | 3 (1.2) | 3 (1.1) | 6 (1.2) |
| Agree | 22 (8.6) | 31 (11.7) | 53 (10.2) |
| Disagree | 160 (62.3) | 168 (63.6) | 328 (63.0) |
| Strongly disagree | 66 (25.7) | 59 (22.3) | 125 (24.0) |
| Don’t know | 6 (2.3) | 3 (1.1) | 9 (1.7) |
| If I have a fever for 3 consecutive days without any other symptoms (influenza, cough, diarrhea), I suspect that I have dengue fever | |||
| Strongly agree | 46 (17.9) | 49 (18.6) | 95 (18.2) |
| Agree | 182 (70.5) | 174 (65.9) | 356 (68.3) |
| Disagree | 23 (8.9) | 34 (12.9) | 57 (10.9) |
| Strongly disagree | 4 (1.6) | 3 (1.1) | 7 (1.3) |
| Don’t know | 2 (0.8) | 4 (1.5) | 6 (1.2) |
| In my opinion, everyone has the same risk to get infected by dengue fever | |||
| Strongly agree | 73 (28.4) | 80 (30.3) | 153 (29.4) |
| Agree | 161 (62.6) | 160 (60.6) | 321 (61.6) |
| Disagree | 19 (7.4) | 14 (5.3) | 33 (6.3) |
| Strongly disagree | 2 (0.8) | 6 (2.3) | 8 (1.5) |
| Don’t know | 2 (0.8) | 4 (1.5) | 6 (1.2) |
| Study Population | Mantrijeron N = 257 | Demangan N = 264 | Total N = 521 |
|---|---|---|---|
| Practice Items | Good Practice N (%) | ||
| → I pay attention to existing larvae in indoor water containers | 238 (92.2) | 248 (93.2) | 486 (92.7) |
| → I clean and brush water containers if any larvae inside | 236 (91.5) | 251 (94.4) | 487 (92.9) |
| → I clean my containers one to three times a week | 239 (92.6) | 242 (91.0) | 481 (91.8) |
| → I always keep water containers in my house closed | 174 (67.4) | 166 (62.4) | 340 (64.9) |
| → I cover or recycle discarded material outside the house | 255 (98.8) | 258 (97.0) | 513 (97.9) |
| → All my family members are responsible for cleaning water container | 254 (98.4) | 262 (98.5) | 516 (98.5) |
| → I clean water containers by draining and brushing | 246 (95.3) | 259 (97.4) | 505 (96.4) |
| → I use mosquito repellent | 161 (62.4) | 160 (60.2) | 321 (61.3) |
| → I use repellent or mosquito coil or mosquito spray in the morning and evening | 60 (23.3) | 53 (19.9) | 113 (21.6) |
| Variable | Effect of Control Card Intervention | |
|---|---|---|
| IRR (95% CI) | p-Value | |
| Number of containers positive with larvae | 1.71 (0.87–3.36) | 0.11 |
| Number of houses positive with larvae | 1.42 (0.69–2.92) | 0.33 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sulistyawati, S.; Dwi Astuti, F.; Rahmah Umniyati, S.; Tunggul Satoto, T.B.; Lazuardi, L.; Nilsson, M.; Rocklov, J.; Andersson, C.; Holmner, Å. Dengue Vector Control through Community Empowerment: Lessons Learned from a Community-Based Study in Yogyakarta, Indonesia. Int. J. Environ. Res. Public Health 2019, 16, 1013. https://doi.org/10.3390/ijerph16061013
Sulistyawati S, Dwi Astuti F, Rahmah Umniyati S, Tunggul Satoto TB, Lazuardi L, Nilsson M, Rocklov J, Andersson C, Holmner Å. Dengue Vector Control through Community Empowerment: Lessons Learned from a Community-Based Study in Yogyakarta, Indonesia. International Journal of Environmental Research and Public Health. 2019; 16(6):1013. https://doi.org/10.3390/ijerph16061013
Chicago/Turabian StyleSulistyawati, Sulistyawati, Fardhiasih Dwi Astuti, Sitti Rahmah Umniyati, Tri Baskoro Tunggul Satoto, Lutfan Lazuardi, Maria Nilsson, Joacim Rocklov, Camilla Andersson, and Åsa Holmner. 2019. "Dengue Vector Control through Community Empowerment: Lessons Learned from a Community-Based Study in Yogyakarta, Indonesia" International Journal of Environmental Research and Public Health 16, no. 6: 1013. https://doi.org/10.3390/ijerph16061013
APA StyleSulistyawati, S., Dwi Astuti, F., Rahmah Umniyati, S., Tunggul Satoto, T. B., Lazuardi, L., Nilsson, M., Rocklov, J., Andersson, C., & Holmner, Å. (2019). Dengue Vector Control through Community Empowerment: Lessons Learned from a Community-Based Study in Yogyakarta, Indonesia. International Journal of Environmental Research and Public Health, 16(6), 1013. https://doi.org/10.3390/ijerph16061013








