Rare Diseases with Periodontal Manifestations
Abstract
1. Introduction
2. Methods
2.1. System Behind the Search Criteria
2.2. Criteria for Inclusion and Exclusion
- -
- Gingiva, which surrounds the tooth and parts of the alveolar bone at the level of the neck of the tooth.
- -
- Alveolar bone, which is part of the jaw bone in which the teeth are set.
- -
- Root cementum, which covers the root surface of the teeth and parts of the apical root canal wall.
- -
- Periodontal fibre, which are bundles of collagen and (partly) oxytalan fibres that reach into the root element on one side, and into the alveolar inner bone tissue on the other side.
3. Results
4. Discussion
Limitations of the Study
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Regulation (EC) No 141/2000 of the European Parliament and of the Council of 16 December 1999 on Orphan Medical Products. 1999. Available online: http://ec.europa.eu/health/files/eudralex/vol-1/reg_2000_141_cons-2009-07/reg_2000_141_cons-2009-07_en.pdf (accessed on 21 July 2018).
- German Federal Ministry of Health. 2013. Available online: http://www.bmg.bund.de/praevention/gesundheitsgefahren/seltene-erkrankungen.html (accessed on 18 June 2017).
- Wetterauer, B.; Schuster, R. Rare diseases. Funding programs in Germany and Europe. Bundesgesund. Gesundheitsforsch. Gesundheitssch. 2008, 51, 519. [Google Scholar] [CrossRef] [PubMed]
- The Council of the European Union. Council Recommendation of 8 June 2009 on an Action in the Field of Rare Diseases (2009/C 151/02). Available online: http://eur-lex.europa.eu/LexUriServ.do?uri=OJ:C:2009:151:0007:0010:EN:PDF (accessed on 22 July 2018).
- National Plan of Action for People with Rare Diseases: Action Fields, Recommendations, Proposed Actions. German Federal Ministry of Health, German Federal Ministry of Education and Research, Alliance for Chronic Rare Diseases 2013. Available online: http://www.namse.de/english.html (accessed on 9 October 2017).
- Héon-Klin, V. European Reference networks for rare diseases: What is the conceptual framework? Orphanet. J. Rare Dis. 2017. [Google Scholar] [CrossRef] [PubMed]
- European Organisation for Rare Diseases (EURODIS). Understanding this Public Health Priority. 2005. Available online: http://www.eurordis.org/sites/default/files/publications/princeps_document-EN.pdf (accessed on 2 August 2018).
- Reimann, A.; Bend, J.; Dembski, B. Patient-centred care in rare diseases. A patient organisations’ perspective. Bundesgesund. Gesundheitsforsch. Gesundheitssch. 2007, 50, 1484. [Google Scholar] [CrossRef] [PubMed]
- Rare Disease Impact Report: Insights from patients and the medical community. Shire 2013. Available online: https://globalgenes.org/wp-content/uploads/2013/04/ShireReport-1.pdf (accessed on 25 October 2018).
- Kole, A.; Faurisson, F. Rare diseases social epidemiology: Analysis of inequalities. Adv. Exp. Med. Biol. 2010, 686, 223–250. [Google Scholar] [CrossRef] [PubMed]
- Jackowski, J.; Hanisch, M. Orofacial manifestations in 2006 rare diseases—A preliminary systematic review of the literature. J. German Soc. Dent. Oral Med. 2012, 67, D10. [Google Scholar]
- Hennekam, R.C.M.; Allanson, J.E.; Krantz, I.D.; Gorlin, R.J. Gorlin’s Syndromes of the Head and Neck, 5th ed.; Oxford University Press: Oxford, UK, 2010. [Google Scholar]
- Hanisch, M.; Fröhlich, L.F.; Kleinheinz, J. Gingival hyperplasia as first sign of recurrence of granulomatosis with polyangiitis (Wegener’s granulomatosis): Case report and review of the literature. BMC Oral Health 2016, 17, 33. [Google Scholar] [CrossRef] [PubMed]
- Eidt, D.; Frank, M.; Reimann, A.; Wagner, T.O.F.; Mittendorf, T.; Graf von der Schulenburg, J.M. Maßnahmen zur Verbesserung der Gesundheitlichen Situation von Menschen mit Seltenen Erkrankungen in Deutschland: Studie im Auftrag des Bundesministeriums für Gesundheit. Available online: https://www.bundesgesundheitsministerium.de/fileadmin/Dateien/5_Publikationen/Praevention/Berichte/110516_Forschungsbericht_Seltene_Krankheiten.pdf (accessed on 28 October 2017).
- Albandar, J.M.; Susin, C.; Hughes, F.J. Manifestations of systemic diseases and conditions that affect the periodontal attachment apparatus: Case definitions and diagnostic considerations. J. Periodontol. 2018, 89, S183–S203. [Google Scholar] [CrossRef]
- Berglundh, T.; Armitage, G.; Araujo, M.G.; Avila-Ortiz, G.; Blanco, J.; Camargo, P.M.; Chen, S.; Cochran, D.; Derks, J.; Figuero, E.; Hämmerle, C.H.F.; et al. Peri-implant diseases and conditions: Consensus report of workgroup 4 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Clin. Periodontol. 2018, 45, S286–S291. [Google Scholar] [CrossRef]
- Caton, J.; Armitage, G.; Berglundh, T.; Chapple, I.L.C.; Jepsen, S.; Kornman, K.L.; Mealey, B.; Papapanou, P.N.; Sanz, M.; Tonetti, M.S. A new classification scheme for periodontal and peri-implant diseases and conditions—Introduction and key changes from the 1999 classification. J. Clin. Periodontol. 2018, 45, S1–S8. [Google Scholar] [CrossRef]
- Chapple, I.L.C.; Mealey, B.L.; Van Dyke, T.E.; Bartold, P.M.; Dommisch, H.; Eickholz, P.; Geisinger, M.L.; Genco, R.J.; Glogauer, M.; Goldstein, M.; et al. Periodontal health and gingival diseases and conditions on an intact and a reduced periodontium: Consensus report of workgroup 1 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Clin. Periodontol. 2018, 45, S68–S77. [Google Scholar] [CrossRef]
- Cortellini, P.; Bissada, N.F. Mucogingival conditions in the natural dentition: Narrative review, case definitions, and diagnostic considerations. J. Clin. Periodontol. 2018, 45, S190–S198. [Google Scholar] [CrossRef] [PubMed]
- Herrera, D.; Retamal-Valdes, B.; Alonso, B.; Feres, M. Acute periodontal lesions (periodontal abscesses and necrotizing periodontal diseases) and endo-periodontal lesions. J. Clin. Periodontol. 2018, 45, S78–S94. [Google Scholar] [CrossRef] [PubMed]
- Holmstrup, P.; Plemons, J.; Meyle, J. Non-plaque-induced gingival diseases. J. Clin. Periodontol. 2018, 45, S28–S43. [Google Scholar] [CrossRef] [PubMed]
- Jepsen, S.; Caton, J.G.; Albandar, J.M.; Bissada, N.F.; Bouchard, P.; Cortellini, P.; Demirel, K.; de Sanctis, M.; Ercoli, C.; Fan, J.; et al. Periodontal manifestations of systemic diseases and developmental and acquired conditions: Consensus report of workgroup 3 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Clin. Periodontol. 2018, 45, S219–S229. [Google Scholar] [CrossRef] [PubMed]
- Papapanou, P.N.; Sanz, M.; Buduneli, N.; Dietrich, T.; Feres, M.; Fine, D.H.; Flemmig, T.F.; Garcia, R.; Giannobile, W.V.; Graziani, F.; Greenwell, H.; et al. Periodontitis: Consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Clin. Periodontol. 2018, 45, S162–S170. [Google Scholar] [CrossRef] [PubMed]
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J. Clin. Periodontol. 2018, 45, S149–S161. [Google Scholar] [CrossRef]
- ROMSE. Available online: http://romse.org (accessed on 2 January 2019).
- Hanisch, M.; Hanisch, L.; Benz, K.; Kleinheinz, J.; Jackowski, J. Development of a database to record orofacial manifestations in patients with rare diseases: A status report from the ROMSE (recording of orofacial manifestations in people with rare diseases) database. Br. J. Oral Maxillofac. Surg. 2017, 55, 500–503. [Google Scholar] [CrossRef]
- List of Rare Diseases and Synonyms in Alphabetical Order. May 2015 Version. Available online: http://www.orpha.net/consor4.01/www/cgi-bin/Education_Home.php?lng=DE#REPORT_RARE_DISEASES (accessed on 8 May 2015).
- PubMed. Available online: https://www.ncbi.nlm.nih.gov (accessed on 6 October 2018).
- Müller, A. Auflistung aller Parodontal-Relevanten Seltenen Erkrankungen mit den Anfangsbuchstaben A-K. Ph.D. Thesis, Dresden, Germany, 2017. [Google Scholar]
- Molina-García, A.; Castellanos-Cosano, L.; Machuca-Portillo, G.; Posada-de la Paz, M. Impact of rare diseases in oral health. Med. Oral Patol. Oral Cir. Bucal. 2016, 21, e587–e594. [Google Scholar]
- Corcuera-Flores, J.R.; Casttellanos-Cosano, L.; Torres-Lagares, D.; Serrera-Figallo, M.Á.; Rodríguez-Caballero, Á.; Machuca-Portillo, G. A systematic review of the oral and craniofacial manifestations of cri of the cat syndrome. Clin Anat. 2016, 29, 29–555. [Google Scholar] [CrossRef]
- Armitage, G.C. Development of classification system for periodontal diseases and conditions. Ann. Periodontol. 1999, 4, 1–6. [Google Scholar] [CrossRef]
- Khocht, A.; Albandar, J.M. Aggressive forms of periodontitis secondary to systemic disorders. Periodontology 2000, 65, 134–148. [Google Scholar] [CrossRef] [PubMed]
- Guan, G.; Firth, N. Oral manifestations as an early clinical sign of acute myeloid leukaemia: A case report. Aust. Dent. J. 2015, 60, 123–127. [Google Scholar] [CrossRef]
- Matsuo, F.S.; Barbosa de Paulo, L.F.; Servato, J.P.; de Faria, P.R.; Cardoso, S.V.; Loyola, A.M. Involvement of oral tissues by AL amyloidosis: A literature review and report of eight new cases. Clin. Oral Investig. 2016, 20, 1913–1920. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.; Michel, R.C.; Greghi, S.L.; Resende, M.L.; Sant’Ana, A.C.; Damante, C.A. Prevention and Periodontal Treatment in Down Syndrome Patients: A Systematic Review. PLoS ONE 2016, 11, e0158339. [Google Scholar] [CrossRef] [PubMed]
- Hanisch, M.; Jung, S.; Kleinheinz, J. Identification of rare diseases in the oral cavity. Internist 2018, 59, 972–980. [Google Scholar] [CrossRef] [PubMed]
- Berglund, B.; Björck, K. Women with Ehlers-Danlos Syndrome experience low oral health-related quality of life. J. Orofac. Pain 2012, 26, 307–314. [Google Scholar] [PubMed]
- Cicciù, M. Neurodegenerative Disorders and Periodontal Disease: Is There a Logical Connection? Neuroepidemiology 2016, 47, 94–95. [Google Scholar] [CrossRef] [PubMed]
- Dev, T.; Sethuraman, G. Diagnosis of acrodermatitis enteropathica in resource limited settings. BMJ Case Rep. 2017. [Google Scholar] [CrossRef]
- OMIM: Online Inheritance in Man. Available online: http://www.omim.org (accessed on 6 October 2018).
- Afifi, H.H.; Abdel-Hamid, M.S.; Eid, M.M.; Mostafa, I.S.; Abdel-Salam, G.M. De Novo Mutation in ABCC9 Causes Hypertrichosis Acromegaloid Facial Features Disorder. Pediatr. Dermatol. 2016, 2, e109–e113. [Google Scholar] [CrossRef]
- O’Donnell, M.R.; Tallman, M.S.; Abboud, C.N.; Altman, J.K.; Appelbaum, F.R.; Arber, D.A.; Bhatt, V.; Bixby, D.; Blum, W.; Coutre, S.E.; et al. Acute Myeloid Leukemia, Version 3.2017, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2017, 15, 926–957. [Google Scholar] [CrossRef]
- Sonoi, N.; Soga, Y.; Maeda, H.; Ichimura, K.; Yoshino, T.; Aoyama, K. Histological and immunohistochemical features of gingival enlargement in a patient with AML. Odontology 2012, 100, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Kini, U.; Clayton-Smith, J. Acromegaloid facial appearance syndrome: A further case report. Clin. Dysmorphol. 2004, 13, 251–253. [Google Scholar] [CrossRef] [PubMed]
- Li, L.L.; Liu, P.H.; Xie, X.H.; Ma, S.; Liu, C.; Chen, L. Loss of epithelial FAM20A in mice causes amelogenesis imperfecta, tooth eruption delay and gingival overgrowth. Int. J. Oral Sci. 2016, 8, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Venclikova, Z.; Benada, O.; Joska, L. Monitoring of selenium in oral cavity argyria—A clinical and microscopic study. Neuro Endocrinol. Lett. 2011, 32, 286–291. [Google Scholar] [PubMed]
- Donadieu, J.; Fenneteau, O.; Beaupain, B.; Mahlaoui, N.; Chantelot, C.B. Congenital neutropenia: Diagnosis, molecular bases and patient management. Orphanet. J. Rare Dis. 2011, 6, 26. [Google Scholar] [CrossRef] [PubMed]
- Hartman, M.J.; Caccamese, J.F., Jr.; Bergman, S.A. Perioperative management of a patient with Bernard-Soulier syndrome for third molar surgery. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod 2007, 103, 626–629. [Google Scholar] [CrossRef] [PubMed]
- Ward, D.M.; Shiflett, S.L.; Kaplan, J. Chediak-Higashi syndrome: A clinical and molecular view of a rare lysosomal storage disorder. Curr. Mol. Med. 2002, 2, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Buduneli, N.; Baylas, H.; Aksu, G.; Kutukculer, N. Prepubertal periodontitis associated with chronic granulomatous disease. J. Clin. Periodontol. 2001, 28, 589–593. [Google Scholar] [CrossRef]
- Girolami, A.; Santarossa, C.; Cosi, E.; Ferrari, S.; Lombardi, A.M.; Girolami, B. Bleeding manifestations in heterozygotes with prothrombin deficiency or abnormalities vs. unaffected family members as observed during a long follow-up study. Blood Coagul. Fibrinolysis 2017, 28, 623–626. [Google Scholar] [CrossRef]
- Naderi, M.; Tabibian, S.; Alizadeh, S.; Hosseini, S.; Zaker, F.; Bamedi, T. Congenital factor V deficiency: Comparison of the severity of clinical presentations among patients with rare bleeding disorders. Acta Haematol. 2015, 133, 148–154. [Google Scholar] [CrossRef]
- Weinstock, R.J.; Onyejiuwa, A.; Shnayder, G.; Clarkson, E.I. Use of recombinant factor VII for tooth extractions in a patient with severe congenital factor VII deficiency: A case report. J. Am. Dent. Assoc. 2015, 146, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Menegatti, M.; Peyvandi, F. Factor X deficiency. Semin. Thromb. Hemost. 2009, 35, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Bolton-Maggs, P.H. Factor XI deficiency and its management. Haemophilia 2000, 6, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Bender, L.; Weidmann, H.; Rose-John, S.; Renne, T.; Long, A.T. Factor XII-Driven Inflammatory Reactions with Implications for Anaphylaxis. Front Immunol. 2017, 8, 1115. [Google Scholar] [CrossRef] [PubMed]
- Karimi, M.; Cairo, A.; Safarpour, M.M.; Haghpanah, S.; Ekramzadeh, M.; Afrasiabi, A. Genotype and phenotype report on patients with combined deficiency of factor V and factor VIII in Iran. Blood Coagul. Fibrinolysis 2014, 25, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Scheper, H.J.; Brand, H.S. Oral aspects of Crohn’s disease. Int. Dent. J. 2002, 52, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Fang, L.; Yang, X. Cyclic neutropenia presenting as recurrent oral ulcers and periodontitis. J. Clin. Pediatr. Dent. 2013, 37, 307–308. [Google Scholar] [CrossRef] [PubMed]
- Migliaccio, A.R.; Migliaccio, G.; Dale, D.C.; Hammond, W.P. Hematopoietic progenitors in cyclic neutropenia: Effect of granulocyte colony-stimulating factor in vivo. Blood 1990, 75, 1951–1959. [Google Scholar]
- Wiebe, C.B.; Larjava, H.S. Do mutations in the basement membrane zone affect the human periodontium? Review with special reference to epidermolysis bullosa. J. West Soc. Periodontol. Periodontal. Abstr. 1998, 46, 5–18. [Google Scholar] [PubMed]
- Kapferer-Seebacher, I.; Lundberg, P.; Malfait, F.; Zschocke, J. Periodontal manifestations of Ehlers-Danlos syndromes: A systematic review. J. Clin. Periodontol. 2017, 44, 1088–1100. [Google Scholar] [CrossRef] [PubMed]
- Naz, A.; Biswas, A.; Khan, T.N.; Goodeve, A.; Ahmed, N.; Saqlain, N. Identification of novel mutations in congenital afibrinogenemia patients and molecular modeling of missense mutations in Pakistani population. Thromb. J. 2017, 15, 24. [Google Scholar] [CrossRef] [PubMed]
- Goswami, M.; Bhushan, U. Dental perspective of rare disease of Fanconi anemia: Case report with review. Clin. Med. Insights Case Rep. 2016, 17, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Gorlin, R.J. Focal palmoplantar and marginal gingival hyperkeratosis—A syndrome. Birth Defects Orig. Artic. Ser. 1976, 12, 239–242. [Google Scholar] [PubMed]
- Bethke, G.; Kolde, G.; Bethke, G.; Reichart, P.A. Focal palmoplantar and oral mucosa hyperkeratosis syndrome. Mund. Kiefer Gesichtschir. 2001, 5, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Li, N.; Dong, W.; Chen, Z.; Liu, Q.; Xu, Y.; He, G.; Shi, Y.; Li, X.; Hao, J.; et al. Copy-number mutations on chromosome 17q24.2-q24.3 in congenital generalized hypertrichosis terminalis with or without gingival hyperplasia. Am. J. Hum. Genet. 2009, 84, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Hartsfield, J.K., Jr.; Bixler, D.; Hazen, R.H. Gingival fibromatosis with sensorineural hearing loss: An autosomal dominant trait. Am. J. Med. Genet. 1985, 22, 623–627. [Google Scholar] [CrossRef]
- Goldblatt, J.; Singer, S.L. Autosomal recessive gingival fibromatosis with distinctive facies. Clin. Genet. 1992, 42, 306–308. [Google Scholar] [CrossRef]
- Toygar, H.U.; Guzeldemir, E. Excessive gingival bleeding in two patients with Glanzmann thrombasthenia. J. Periodontol. 2007, 78, 1154–1158. [Google Scholar] [CrossRef]
- Xiao, S.; Bu, L.; Zhu, L.; Zheng, G.; Yang, M.; Qian, M.; Hu, L.; Liu, J.; Zhao, G.; Kong, X. A new locus for hereditary gingival fibromatosis (GINGF2) maps to 5q13-q22. Genomics 2001, 74, 180–185. [Google Scholar] [CrossRef]
- Korkmaz, B.; Caughey, G.H.; Chapple, I.; Gauthier, F.; Hirschfeld, J.; Jenne, D.E.; Kettritz, R.; Lalmanach, G.; Lamort, A.S.; Lauritzen, C.; et al. Therapeutic targeting of cathepsin C: From pathophysiology to treatment. Pharmacol. Ther. 2018. [Google Scholar] [CrossRef]
- Hart, T.C.; Hart, P.S.; Michalec, M.D.; Zhang, Y.; Firatli, E.; Van Dyke, T.E.; Stabholz, A.; Zlotogorski, A.; Shapira, L.; Soskolne, W.A. Haim-Munk syndrome and Papillon-Lefèvre syndrome are allelic mutations in cathepsin C. J. Med. Genet. 2000, 37, 88–94. [Google Scholar] [CrossRef]
- Mannucci, P.M.; Tuddenham, E.G. The hemophilias—From royal genes to gene therapy. N. Engl. J. Med. 2001, 344, 1773–1779. [Google Scholar] [CrossRef] [PubMed]
- Scarcella, A.; De Lucia, A.; Pasquariello, M.B.; Gambardella, P. Early death in two sisters with Hennekam syndrome. Am. J. Med. Genet. 2000, 93, 181–183. [Google Scholar] [CrossRef]
- Davis, A.E. The pathophysiology of hereditary angioedema. Clin. Immunol. 2005, 114, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, C.A. Histoplasmosis: A clinical and laboratory update. Clin. Microbiol. Rev. 2007, 20, 115–132. [Google Scholar] [CrossRef] [PubMed]
- Xanthinaki, A.A.; Donta, C.; Gatou, V.; Tsichlakis, C. Hyalinosis cutis et mucosae: Diagnosis based on oral manifestations—Report of a case. J. Clin. Pediatr. Dent. 2008, 33, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.; Espanol-Boren, T.; Thomas, C.; Fischer, A.; Tovo, P.; Bordigoni, P.; Resnick, I.; Fasth, A.; Baer, M.; Gomez, L.; et al. Clinical spectrum of X-linked hyper-IgM syndrome. J. Pediatr. 1997, 131, 47–54. [Google Scholar] [CrossRef]
- Tadin-Strapps, M.; Salas-Alanis, J.C.; Moreno, L.; Warburton, D.; Martinez-Mir, A.; Christiano, A.M. Congenital universal hypertrichosis with deafness and dental anomalies inherited as an X-linked trait. Clin. Genet. 2003, 63, 418–422. [Google Scholar] [CrossRef]
- Fantauzzo, K.A.; Kurban, M.; Levy, B.; Christiano, A.M. Trps1 and its target gene Sox9 regulate epithelial proliferation in the developing hair follicle and are associated with hypertrichosis. PLoS Genet. 2012, 8, e1003002. [Google Scholar] [CrossRef]
- Foster, B.L.; Kuss, P.; Yadav, M.C.; Kolli, T.N.; Narisawa, S.; Lukashova, L.; Cory, E.; Sah, R.L.; Somerman, M.J.; Millán, J.L. Conditional Alpl Ablation Phenocopies Dental Defects of Hypophosphatasia. J. Dent. Res. 2017, 96, 81–91. [Google Scholar] [CrossRef]
- McKee, M.D.; Hoac, B.; Addison, W.N.; Barros, N.M.; Millan, J.L.; Chaussain, C. Extracellular matrix mineralization in periodontal tissues: Noncollagenous matrix proteins, enzymes, and relationship to hypophosphatasia and X-linked hypophosphatemia. Periodontology 2000, 63, 102–122. [Google Scholar] [CrossRef] [PubMed]
- Sulniute, R.; Lindh, T.; Wilczynska, M.; Li, J.; Ny, T. Plasmin is essential in preventing periodontitis in mice. Am. J. Pathol. 2011, 179, 819–828. [Google Scholar] [CrossRef] [PubMed]
- Ichimiya, Y.; Wada, Y.; Kunishima, S.; Tsukamoto, K.; Kosaki, R.; Sago, H.; Ishiguro, A.; Ito, Y. 11q23 deletion syndrome (Jacobsen syndrome) with severe bleeding: A case report. J. Med. Case Rep. 2018. [Google Scholar] [CrossRef] [PubMed]
- Denadai, R.; Raposo-Amaral, C.E.; Bertola, D.; Kim, C.; Alonso, N.; Hart, T.; Han, S.; Stelini, R.F.; Buzzo, C.L.; Raposo-Amaral, C.A.; et al. Identification of 2 novel ANTXR2 mutations in patients with hyaline fibromatosis syndrome and proposal of a modified grading system. Am. J. Med. Genet. 2012, 158, 732–742. [Google Scholar] [CrossRef] [PubMed]
- Miranda, L.A.; Fischer, R.G.; Sztajnbok, F.R.; Figueredo, C.M.; Gustafsson, A. Periodontal conditions in patients with juvenile idiopathic arthritis. J. Clin. Periodontol. 2003, 30, 969–974. [Google Scholar] [CrossRef] [PubMed]
- Boztug, K.; Ding, X.-Q.; Hartmann, H.; Ziesenitz, L.; Schaffer, A.A.; Diestelhorst, J.; Pfeifer, D.; Appaswamy, G.; Kehbel, S.; Simon, T.; et al. HAX1 mutations causing severe congenital neuropenia (sic) and neurological disease lead to cerebral microstructural abnormalities documented by quantitative MRI. Am. J. Med. Genet. 2010, 152, 3157–3163. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G.; Moutsopoulos, N.M.; Hajishengallis, E.; Chavakis, T. Immune and regulatory functions of neutrophils in inflammatory bone loss. Semin. Immunol. 2016, 28, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, P.J.; Chen, S.; Tayeh, M.K.; Ochoa, L.; Leal, S.M.; Pelet, A.; Munnich, A.; Lyonnet, S.; Majeed, H.A.; El-Shanti, H. Homozygous mutations in LPIN2 are responsible for the syndrome of chronic recurrent multifocal osteomyelitis and congenital dyserythropoietic anaemia (Majeed syndrome). J. Med. Genet. 2005, 42, 551–557. [Google Scholar] [CrossRef]
- Encarnacao, M.; Lacerda, L.; Costa, R.; Prata, M.J.; Coutinho, M.F.; Ribeiro, H.; Lopes, L.; Pineda, M.; Ignatius, J.; Galvez, H.; et al. Molecular analysis of the GNPTAB and GNPTG genes in 13 patients with mucolipidosis type II or type III—Identification of eight novel mutations. Clin. Genet. 2009, 76, 76–84. [Google Scholar] [CrossRef]
- Tartaglia, M.; Kalidas, K.; Shaw, A.; Song, X.; Musat, D.L.; van der Burgt, I.; Brunner, H.G.; Bertola, D.R.; Crosby, A.; Ion, A.; et al. PTPN11 mutations in Noonan syndrome: Molecular spectrum, genotype-phenotype correlation, and phenotypic heterogeneity. Am. J. Hum. Genet. 2002, 70, 1555–1563. [Google Scholar] [CrossRef]
- Moghaddasian, M.; Arab, H.; Dadkhah, E.; Boostani, H.; Babak, A.R.; Abbaszadegan, M.R. Protein modeling of cathepsin C mutations found in Papillon-Lefèvre syndrome. Gene 2014, 538, 182–187. [Google Scholar] [CrossRef] [PubMed]
- McNamara, K.M.; Hall, S.E.; Wilder, R.S.; Lawrence, H.P.; Offenbacher, S. Periodontitis and cytokine expression in CD14 deficient patients. J. Int. Acad. Periodontol. 1999, 1, 95–100. [Google Scholar] [PubMed]
- Gawron, K.; Łazarz-Bartyzel, K.; Potempa, J.; Chomyszyn-Gajewska, M. Gingival fibromatosis: Clinical, molecular and therapeutic issues. Orphanet. J. Rare Dis. 2016, 11, 9. [Google Scholar] [CrossRef] [PubMed]
- Bunn, K.J.; Daniel, P.; Rösken, H.S.; O’Neill, A.C.; Cameron-Christie, S.R.; Morgan, T.; Brunner, H.G.; Lai, A.; Kunst, H.P.; Markie, D.M.; et al. Mutations in DVL1 cause an osteosclerotic form of Robinow syndrome. Am. J. Hum. Genet. 2015, 96, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Murai, O.; Naruishi, K.; Ogihara, S.; Suwa, N.; Kanazawa, S.; Yaegashi, T.; Takeda, Y.; Kunimatsu, K. Cathepsin B, D, and L regulation in cyclosporin A-mediated gingival hyperplasia of a patient with sarcoidosis. Clin. Lab. 2011, 57, 535–541. [Google Scholar] [PubMed]
- Regen, A.; Nelson, L.P.; Woo, S.B. Dental manifestations associated with Seckel syndrome type II: A case report. Pediatr Dent. 2010, 32, 445–450. [Google Scholar] [PubMed]
- Agrawal, A.; Daniel, J.; Vijeev, V. Segmental odontomaxillary dysplasia. J. Indian Soc. Pedod Prev. Dent. 2014, 32, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Marques, C.P.; Maor, Y.; de Andrade, M.S.; Rodrigues, V.P.; Benatti, B.B. Possible evidence of systemic lupus erythematosus and periodontal disease association mediated by Toll-like receptors 2 and 4. Clin. Exp. Immunol. 2016, 183, 187–192. [Google Scholar] [CrossRef]
- Pischon, N.; Hoedke, D.; Kurth, S.; Lee, P.; Dommisch, H.; Steinbrecher, A.; Pischon, T.; Burmester, G.R.; Buttgereit, F.; Detert, J.; et al. Increased Periodontal Attachment Loss in Patients with Systemic Sclerosis. J. Periodontol. 2016, 87, 763–771. [Google Scholar] [CrossRef]
- Castaman, G.; Linari, S. Diagnosis and Treatment of von Willebrand Disease and Rare Bleeding Disorders. J. Clin. Med. 2017. [Google Scholar] [CrossRef]
- Prapanpoch, S.; Jorgenson, R.J.; Langlais, R.P.; Nummikoski, P.V. Winchester syndrome. A case report and literature review. Oral Surg. Oral Med. Oral Pathol. 1992, 74, 671–677. [Google Scholar] [CrossRef]
- Lemahieu, V.; Gastier, J.M.; Francke, U. Novel mutations in the Wiskott-Aldrich syndrome protein gene and their effects on transcriptional, translational, and clinical phenotypes. Hum. Mutat. 1999, 14, 54–66. [Google Scholar] [CrossRef]
- Balasubramanian, M.; Parker, M.J. Zimmermann-Laband syndrome in a child previously described with brachydactyly, extrahepatic biliary atresia, patent ductus arteriosus and seizures. Clin. Dysmorph. 2010, 19, 48–50. [Google Scholar] [CrossRef] [PubMed]
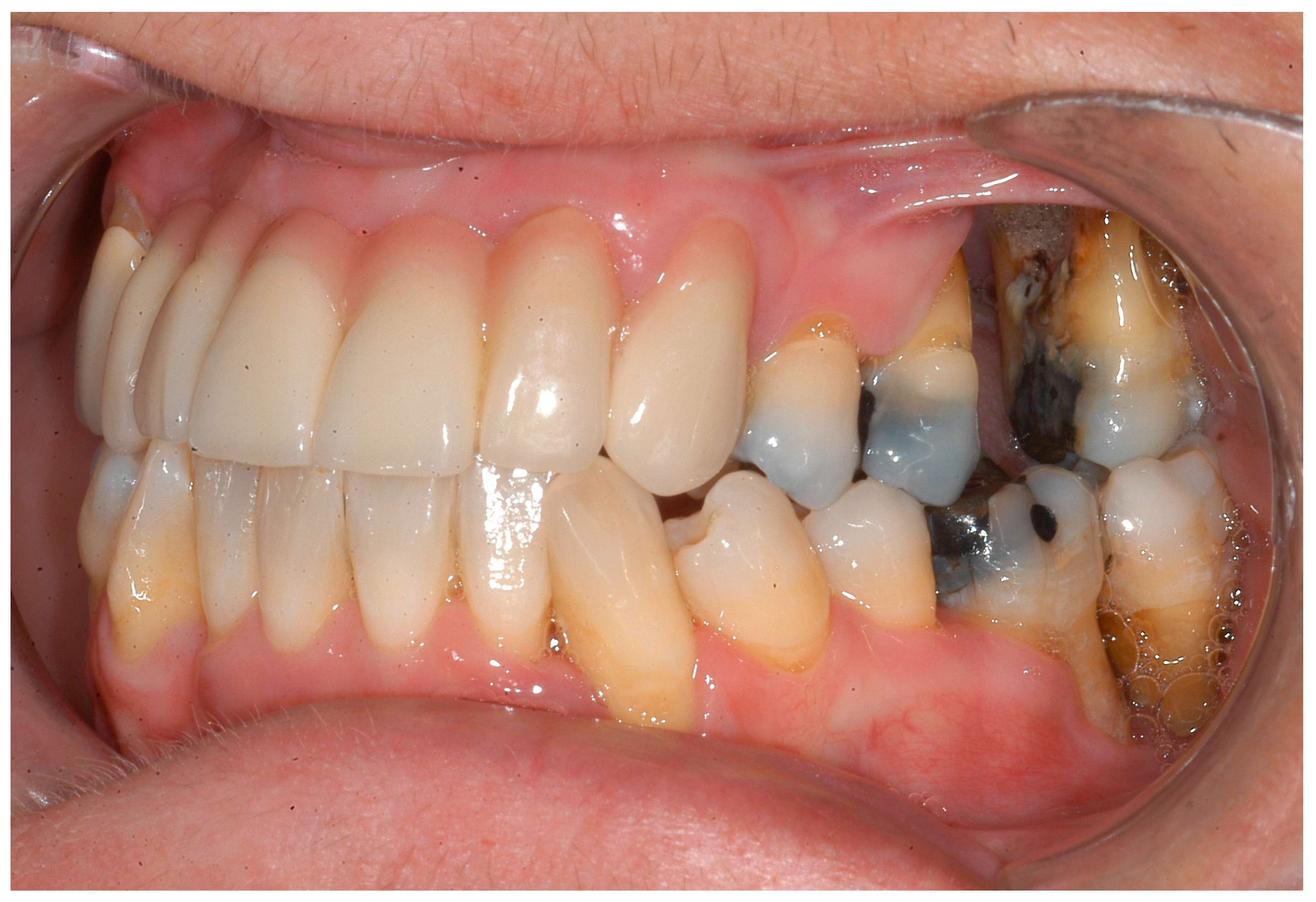
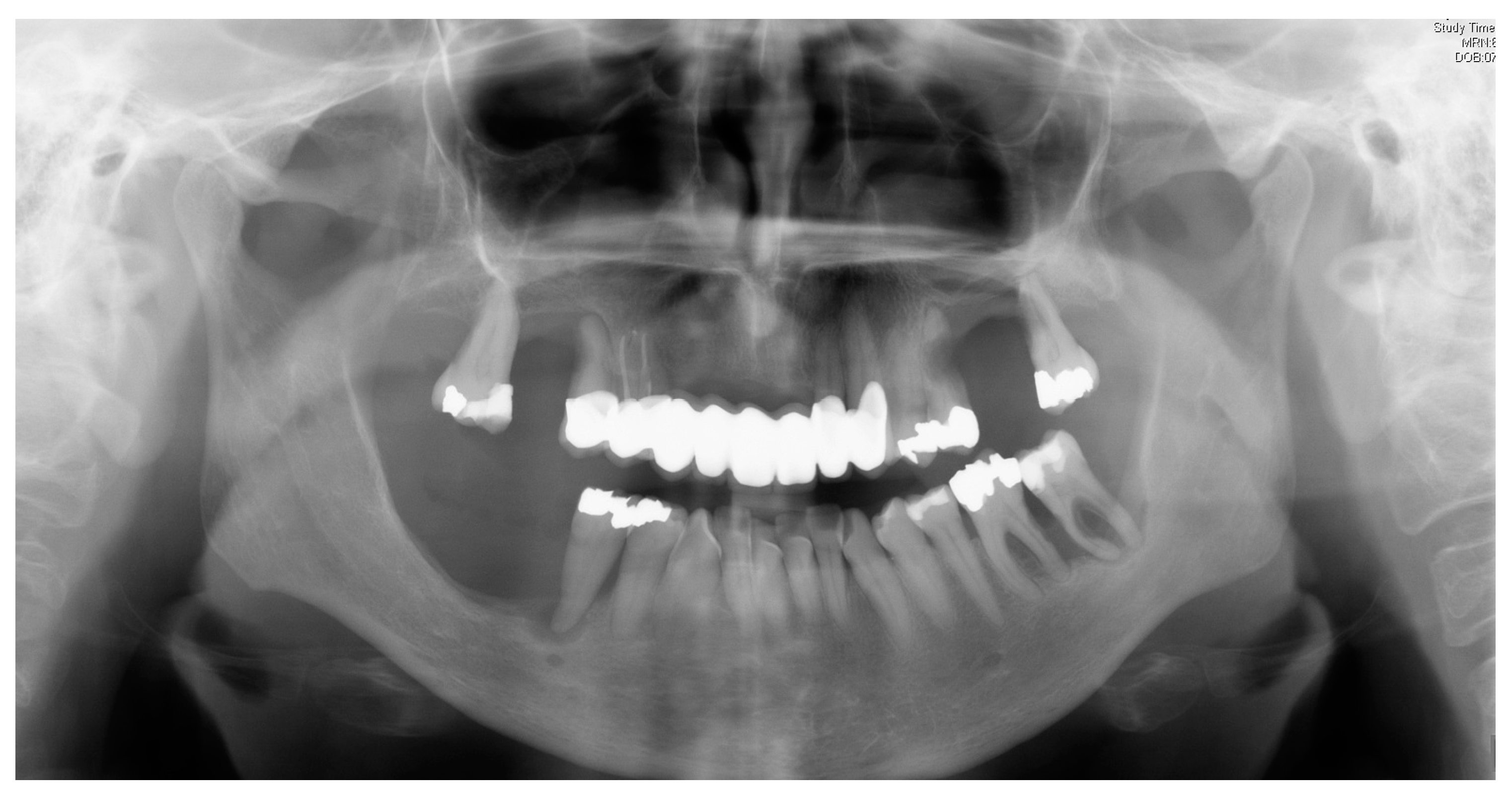
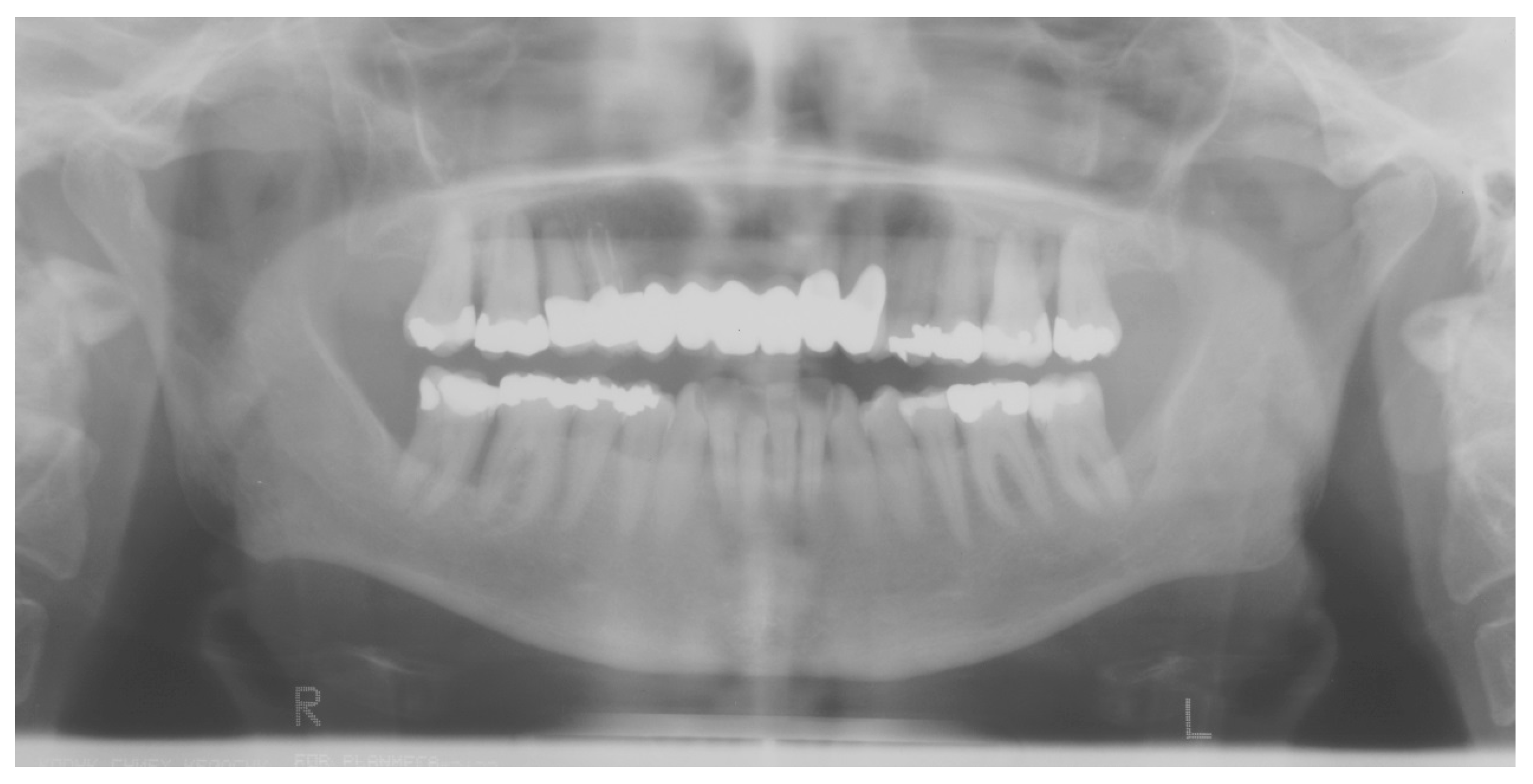
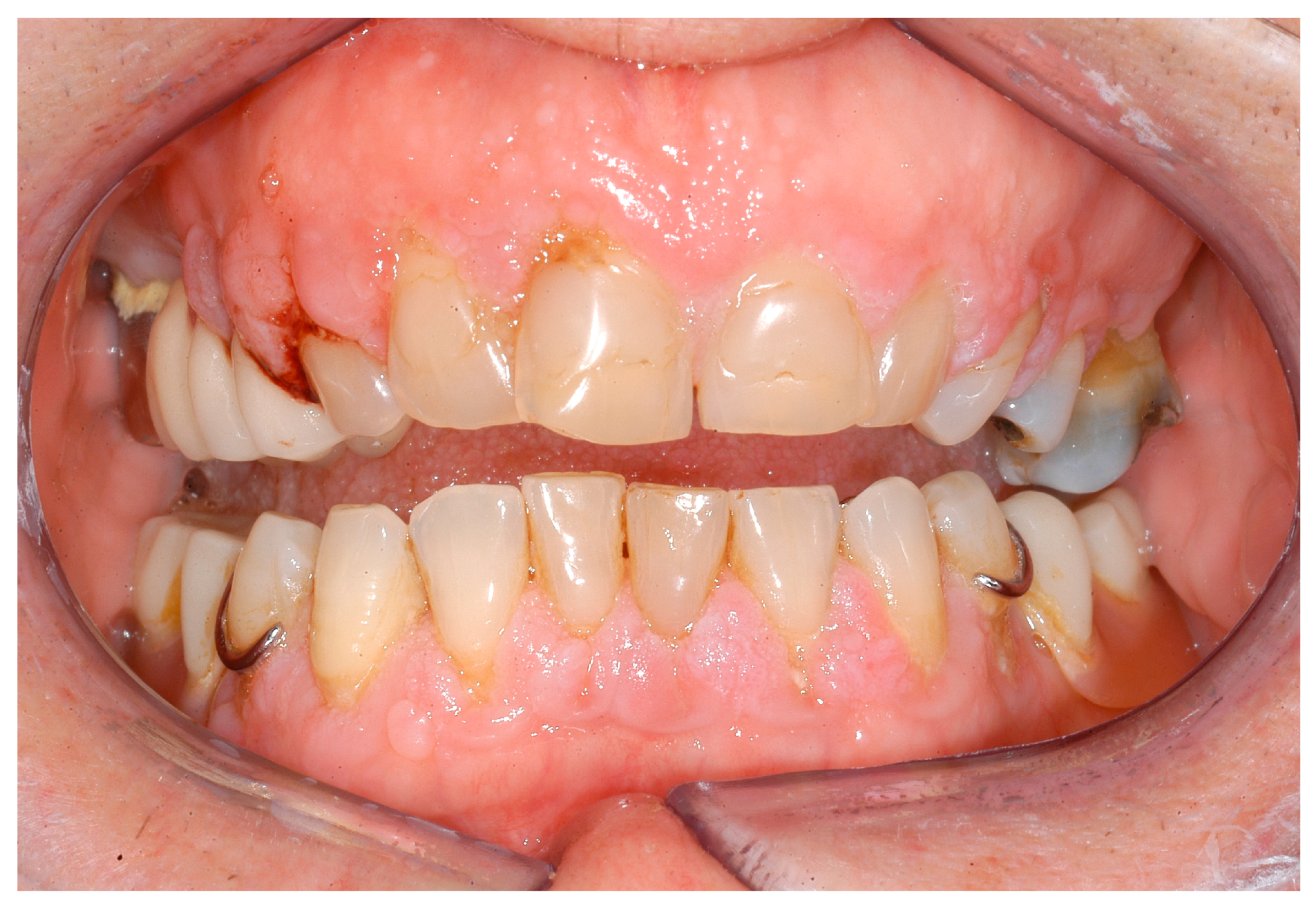
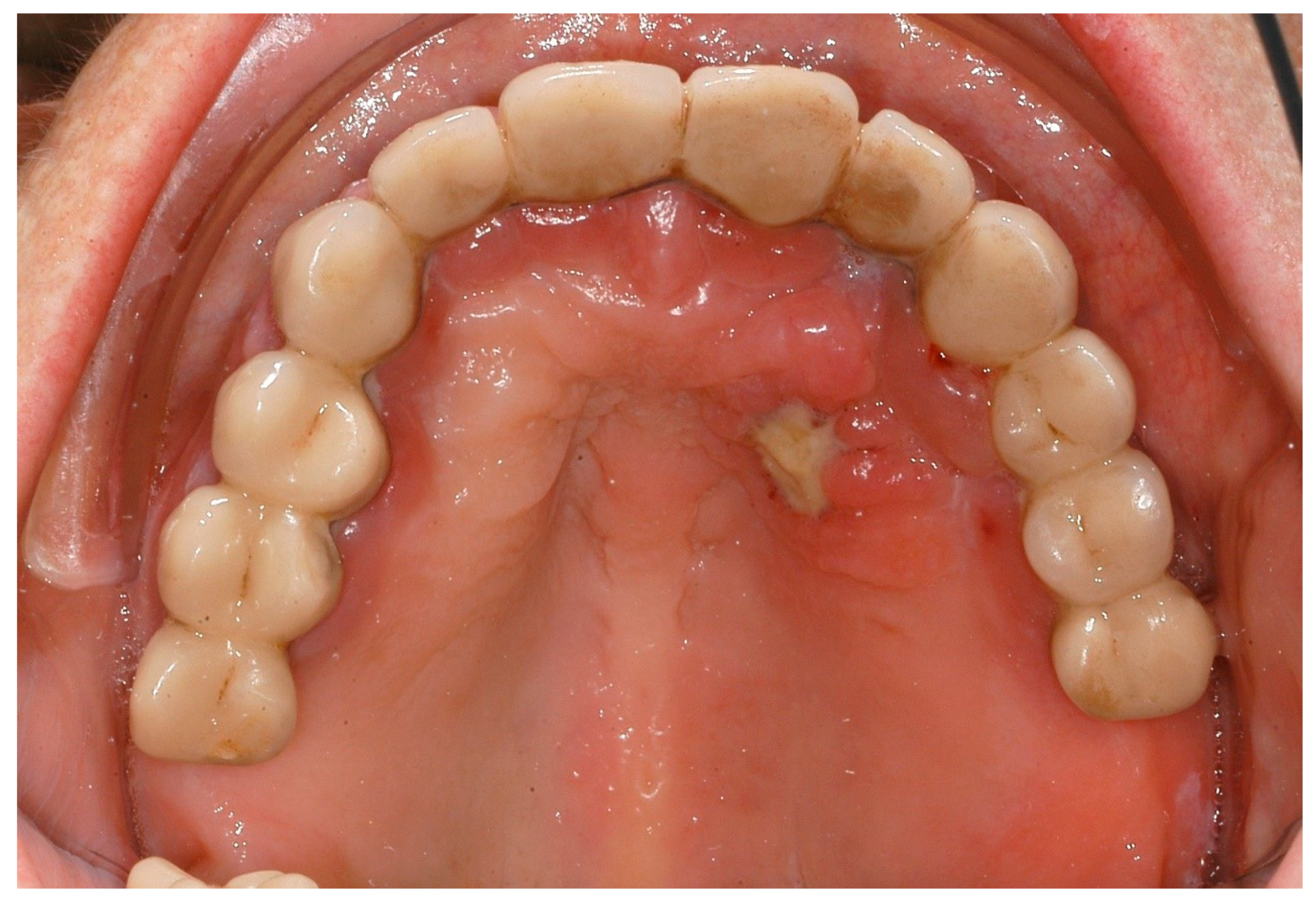
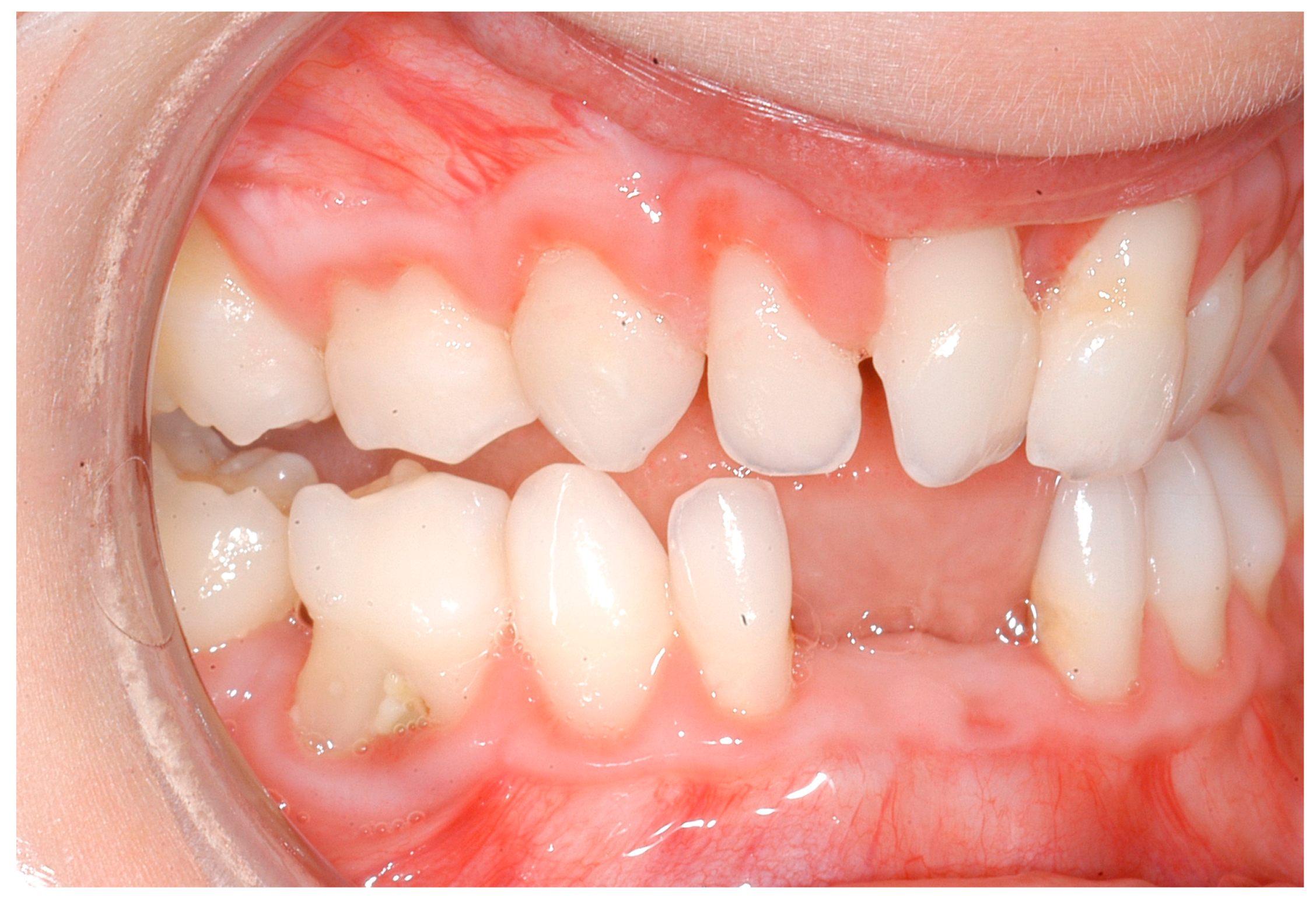
| Disease Name, ICD-10 | Symptoms | Possible Pathological Mechanisms Related with the Periodontal Manifestations | Gene, Location of the Disease | Classification of Periodontal and Peri-Implant Diseases and Conditions |
|---|---|---|---|---|
| Acrodermatitis enteropathica E83.2 | gingivitis | zinc deficiency [40] | SLC39A4, 8q24.3 [41] | non-plaque induced gingival diseases and conditions: endocrine, nutritional, and metabolic disease |
| Acromegaloid facial appearance syndrome Q87.0 | gingival enlargement | not reported [42] | ABCC9, exon 27 [41] | non-plaque induced gingival diseases and conditions: genetic/development disorder |
| Acroosteolysis, dominant type/Hajdu-Cheney Syndrome M89.5 | periodontitis [15] | not available | NOTCH2 1p12 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: metabolic and endocrine disorders |
| Acute megakaryo blastic leukemia C94.2 | gingival enlargement gum bleeding | heterogeneous hematologic malignancy characterized by the clonal expansion of myeloid blasts in the peripheral blood, bone marrow, and/or other tissues cause bleeding [43] | not genetically determined | non-plaque induced gingival diseases and conditions: neoplasms |
| Acute myeloid leukemia C92.0 | gingival enlargement gum bleeding | gingival infiltration of leukaemic cells, infiltration of myelomonocytic cells into the bone marrow, destruction of coagulation factors, hypofibrinogenemia [34] | not genetically determined | non-plaque induced gingival diseases and conditions: neoplasms |
| Acute myelomonocytic leukemia C92.5 | gingival enlargement gum bleeding | infiltration of myelomonocytic cells into the gingival tissue [44] | not genetically determined | non-plaque induced gingival diseases and conditions: neoplasms |
| AL amyloidosis E85.9 | gingival enlargement periodontitis | immunoglobulin light chain-derived deposition [35] | not genetically determined | non-plaque induced gingival diseases and conditions: neoplasms |
| Alopecia-epilepsy-pyorrhea-intellectual disability syndrome Q87.8 | gingivitis | unknown [45] | unknown [45] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Amelogenesis imperfecta-gingival hyperplasia syndrome K00.5 | gingival enlargement | exact pathomechanism unknown [46] | FAM20A,17 q24.2 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Argyria T56.8 | gray to gray-black staining of gingiva | deposition of silver and selenium [47] | not genetically determined | non-plaque induced gingival diseases and conditions: gingival pigmentation |
| Autosomal dominant severe congenital neutropenia D70 | periodontitis | neutropenia [48] | ELANE19p13.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Bernard Soulier syndrome/Giant platelet syndrome D69.1 | gum bleeding bleeding diathesis | low platelet count and macrothrombocytopenia [49] | GP1BA,17p 13.2 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Chediak–Higashi Syndrom E70.3 | gingivitis, periodontits | genetic disorder of granule morphology and function [50] | LYST1q42.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Chronic granulomatous disease D71 | periodontitis | defect in metabolic burst for the production of oxygen-free radicals [51] | depending on subtype [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Cohen syndrome Q87.8 | periodontitis | neutropenia [33] | VPS13B,8q 22.2 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Congenital factor II deficiency D68.2 | gum bleeding bleeding diathesis | low prothrombin antigen levels [52] | F2,11p11.2 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Congenital factor V deficiency D68.2 | gum bleeding bleeding diathesis | low blood coagulation factor V levels [53] | F5,1q24.2 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Congenital factor VII deficiency D68.2 | gum bleeding bleeding diathesis | low blood coagulation factor VII levels [54] | F7,13q34 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Congenital factor X deficiency D68.2 | gum bleeding bleeding diathesis | Factor X deficiency [55] | F10,13q34 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Congenital factor XI deficiency D68.1 | gum bleeding bleeding diathesis | Factor XI deficiency [56] | F11,4q35.2 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Congenital factor XII deficiency D68.2 | gum bleeding bleeding diathesis | Factor XII deficiency [57] | F12,5q35.3 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Congenital factor XIII deficiency D68.2 | gum bleeding bleeding diathesis | Factor XIII deficiency [58] | F13A1,6p25.1 F13B,1q31.3 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Crohn disease K50 | gingival enlargement | chronic inflammation [59] | not genetically determined | non-plaque induced gingival diseases and conditions: granulomatous inflammatory conditions (orofacial granulomatosis) |
| Cyclic neutropenia D70 | mucosal ulcers, periodontitis [60] | fluctuation in the number of blood neutrophils [61] | ELANE 19q13.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Down syndrome Q90 | periodontitis | poor oral hygiene and impairment of the immunological system [36] | 21q22.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Dystrophic epidermolysis bullosa Q81.2 | periodontitis | reduced resistance at the junctional epithelial complex [62] | COL7A1 3p21.31 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases affecting the oral mucosa and gingival tissue |
| Ehlers–Danlos syndrome Q79.6 | periodontitis | disturbance of fibrin collagens [63] | C1R and C1S [63] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases affecting connective tissues |
| Familial afibrinogenemi D68.2 | gum bleeding bleeding diathesis | fibrinogen deficiency [64] | FGA, FGB, FGG [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Fanconi anemia D61.0 | gingivitis | defective hemopoesis [65] | Depending on subtype [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Focal palmoplantar and gingival keratoderma Q82.8 [66] | gingival hyperkeratosis [66] | not available | Unknown [67] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Gaucher disease E75.2 [15] | periodontitis | not available | GBA1q22 (Type 1) [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: metabolic and endocrine disorders |
| Gingival fibromatosis/hypertrichosis syndrome L68.8 | gingival enlargement fibromatosis [68] | not available | 17q24.2-q24.3 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Gingival fibromatosis-progressive deafness syndrome H90.3 | gingival enlargement fibromatosis | unknown [69] | unknown [69] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Gingival fibromatosis with facial dysmorphism syndrome Q87.0 | gingival enlargement, fibromatosis | unknown [70] | unknown [70] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Glanzmann thrombasthenia D69.1 | gum bleeding, bleeding diathesis | platelet function disorder [71] | ITGA2B,17q21.31 ITGB3, 17q21.32 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Glycogen storage disease 1b E74.0 | periodontal breakdown | neutrophil dysfunction because of endoplasmic retic-ulum stress generated by disruption of endogenous glucose production [33] | SLC37A411q23.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: metabolic and endocrine disorders |
| Granulomatosis with polyangiitis M31.3 | gingival enlargement, periodontitis | production of inflammatory cytocines [13] | not genetically determined | non-plaque induced gingival diseases and conditions: granulomatous inflammatory conditions (orofacial granulomatosis) systemic diseases and conditions that affect the periodontal attachment apparatus: other disorders that may affect periodontal tissue |
| Hereditary gingival fibromatosis K06.1 | gingival enlargement fibromatosis | unknown [72] | SOS1, 2p22.1 (Type 1) 5q13-q22 (Type 2) 2p23.3-p22.3 (Type 3) 11p15 (Type 4) REST, 4q12 (Type 5) [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Haim–Munk syndrome/Keratosis palmoplantaris-periodontopathia-onychogry-posis syndrome Q82.8 | periodontitis | exact pathomechanism unknown [73] | CTSC, 11q14.2 [38,74] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Hemophilia A D66 | gum bleeding bleeding diathesis | Factor VIII deficiency [75] | F 8, Xq28 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| HemophiliaB D67 | gum bleeding bleeding diathesis | Factor IX deficiency [75] | F 9, Xq27.1 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Hennekam syndrome/Lymphedema-lymphangiectasia-intellectual disability syndrome Q87.8 | gingival enlargement | unknown [76] | CCBE1,18q21.32 (Type 1) FAT4,4q28. (Type 2) [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Hereditary angioedema (C-1-inhibitor deficiency) D84.1 | periodontitis | deficiency or functional alteration of the C1 inhibitor [77] | C1NH11q12.1 (Type 2) F12 5q35.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases affecting connective tissues |
| Histoplasmosis D39 | gingival candidosis | compromised immunological system [78] | not genetically determined | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Hyalinosis cutis et mucosae E78.8 | gingival enlargement | unknown [79] | ECM1, 1q21.2 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Hyper-IgE syndrome D82.4 | gingivitis, periodontitis [15] | not available | STAT3 17q21.2; DOCK8 9q24.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Hyper-IgM syndrome with susceptibility to opportunistic infections D80.5 | periodontitis | decreased levels of IgG and IgA; elevated concentrations ofIgM level [80] | TNFSF5,Xq26.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Hypertrichosis-acromegaloid facial appearance syndrome Q87.0 | gingival enlargement | exact pathomechanism unknown [68] | 17q24.2-q24.3 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Hypertrichosis lanuginosa congenita Q84.2 | gingival enlargement [81] | not available gingival hyperplasia, is caused by deletion or duplication at chromosome 17q24 or by mutation in the ABCA5 gene | deletion or duplication on 17q2417q24 or mutation in ABCA5ABCA5 [41,82] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Hypophosphatasia E83.38 | compromised periodontal attachment | exact pathomechanism unknown [83] | ALPL1 p36.12 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: metabolic and endocrine disorders |
| Hypophosphatemic rickets E83.30 | periodontitis | alteration of bone and tooth mineralization that may lead to malformed and feeble bone and teeth and premature tooth loss [84] | FGF23 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: metabolic and endocrine disorders |
| Hypoplasminogenemia L90.5 | Periodontitis, gingival enlargement | exact pathomechanism unknown [85] | PLG 6q26 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases affecting the oral mucosa and gingival tissue |
| Jacobsen syndrome Q93.5 | gum bleeding bleeding diathesis | abnormal platelet function and thrombocytopenia [86] | IC, 11q23 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Juvenile hyaline fibromatosis M72.8 | gingival enlargement fibromatosis | unknown [87] | ANTXR2, 4q21.21 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Juvenile idiopathic arthritis M08.0; M08.1; M08.2; M08.3; M08.4; M08.8; M08.9 | periodontal attachment loss [88] | dysregulation of the immune-inflammatory response [88] | not genetically determined | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Kindler syndrome Q81.8 | periodontitis | reduced resistance at the junctional epithelial complex [62] | KIND1 20p12.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases affecting the oral mucosa and gingival tissue |
| Kostmann syndrome/Severe congenital neutropenia type 3 D70 | periodontitis | neutropenia [89] | HAX1, 1q21.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Langerhans cell histiocytosis C96 | periodontitis | because proliferation of cells with characteristics similar to bone marrow-derived Langerhans cells [15] | not genetically determined | systemic diseases and conditions that affect the periodontal attachment apparatus: other disorders that may affect periodontal tissue |
| Leukocyte adhesion deficiency type II D84.8 | periodontitis | lack of circulating neutrophils due to defect of glycosylation [90] | SLC35C1, 11p11.2 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Majeed syndrome/Chronic recurrent multifocal osteomyelitis-congenital dyserythropoietic anemia-neutrophilic dermatosis syndrome ICD-10: not available | gum bleeding bleeding diathesis | Anemia with dyserythropoiesis [91] Homozygous mutations in LPIN2 Homozygous mutations in LPIN2 | LPIN2, 18p11.31 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Mucolipidosis type II E77.0 | gingival enlargement | unknown [92] | GNPTAB, 12q23.2 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Noonan syndrome Q87.1 | gum bleeding bleeding diathesis | bleeding diathesis [93] | Depending on subtype [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Papillon–Lefèvre syndrome/Keratosis palmoplantar-periodontopathy syndrome Q82.8 | periodontitis | exact pathomechanism unknown [72] | CTSC, 11q14.2 [41,94] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Paroxysmal nocturnal hemoglobinuria D59.5 | periodontitis | deficiency of monocytic CD14 [95] | PIGA, Xp22.2 (Type 1) PIGT, 20q13.12 (Type 2) [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Ramon syndrome/Cherubism-gingival fibromatosis-intellectual disability syndrome Q87.8 | gingival enlargement fibromatosis | unknown [96] | depending on subtype, mostly unknown [96] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Robinow syndrome/Acral dysostosis with facial and genital abnormalities Q87.1 | gingival enlargement | mutant transcript in fibroblasts [97] | ROR2, 9q22.31 (Type: autosomal-recessive) WNT5A, 3p14.3 (Type: dominant 1) DVL1, 1p36.33 (Type: dominant 2) DVL3, 3q27.1 (Type: dominant 3) [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Sarcoidosis D86 | gingival enlargement | Cathepsin dysregulation [98] | HLA-DRB1,6p21.32 (Type SS1) BTNL2, 6p21.32 (Type SS2) GRCh38, 10q22.3 (Type SS3) [41] | non-plaque induced gingival diseases and conditions: granulomatous inflammatory conditions (orofacial granulomatosis) |
| Seckel syndrome Q87.1 | gingival enlargement [99] | not available | depending on subtype [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Segmental odontomaxillary dysplasia K00.4 | gingival enlargement | unknown [100] | not genetically determined | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Severe congenital neutropenia D70 | periodontitis | neutropenia [48] | ELANE, 19p13.3 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases associated with immunologic disorders |
| Systemic lupus erythematosus M32 | gingivitis, periodontitis | increased accumulation of immune cells, antineutrophil cytoplasm antibodies and metalloproteinases [101] | PTPN221p13.2 FCGR2A1q23.3 FCGR2B1q23.3 CTLA42q33.2 TREX13p21.31 DNASE116p13 [41] | systemic diseases and conditions that affect the periodontal attachment apparatus: diseases affecting connective tissues |
| Systemic sclerosis M34 | periodontitis | unknown [102] | not genetically determined | systemic diseases and conditions that affect the periodontal attachment apparatus: other disorders that may affect periodontal tissue |
| Von Willebrand disease D68.0 | gum bleeding bleeding diathesis | deficiency of von Willebrand factor [103] | Depending on subtype [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Winchester syndrome M89.5 | gingival enlargement | mucopolysaccharides stored in tissue [104] | MMP14,14q11.2 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Wiskott–Aldrich syndrome/Eczema-thrombocytopenia-immunodeficiency syndrome D82.0 | gum bleeding bleeding diathesis | thrombozytopenia, neutropenia [105] | WAS, Xp11.4-p11.21 [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
| Zimmerman–Laband syndrome/Gingival fibromatosis-hepatosplenomegaly-other anomalies syndrome Q87.8 | gingival enlargement fibromatosis | unknown [106] | KCNH1, 1q32.2 (Type 1) ATP6V1B2, 8p21.3 (Type 2) [41] | non-plaque induced gingival diseases and conditions: genetic/developmental disorders |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hanisch, M.; Hoffmann, T.; Bohner, L.; Hanisch, L.; Benz, K.; Kleinheinz, J.; Jackowski, J. Rare Diseases with Periodontal Manifestations. Int. J. Environ. Res. Public Health 2019, 16, 867. https://doi.org/10.3390/ijerph16050867
Hanisch M, Hoffmann T, Bohner L, Hanisch L, Benz K, Kleinheinz J, Jackowski J. Rare Diseases with Periodontal Manifestations. International Journal of Environmental Research and Public Health. 2019; 16(5):867. https://doi.org/10.3390/ijerph16050867
Chicago/Turabian StyleHanisch, Marcel, Thomas Hoffmann, Lauren Bohner, Lale Hanisch, Korbinian Benz, Johannes Kleinheinz, and Jochen Jackowski. 2019. "Rare Diseases with Periodontal Manifestations" International Journal of Environmental Research and Public Health 16, no. 5: 867. https://doi.org/10.3390/ijerph16050867
APA StyleHanisch, M., Hoffmann, T., Bohner, L., Hanisch, L., Benz, K., Kleinheinz, J., & Jackowski, J. (2019). Rare Diseases with Periodontal Manifestations. International Journal of Environmental Research and Public Health, 16(5), 867. https://doi.org/10.3390/ijerph16050867






