Real Foliage Plants as Visual Stimuli to Improve Concentration and Attention in Elementary Students
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
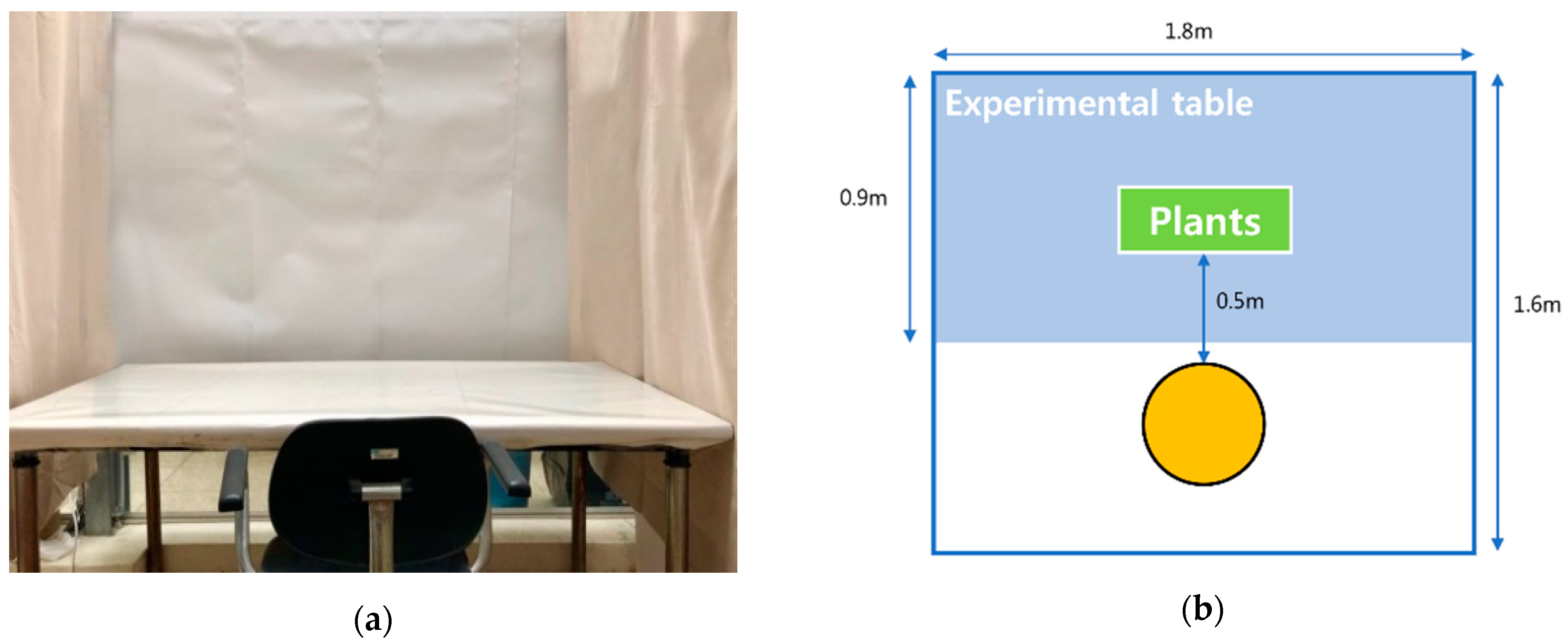
2.2. Experimental Environment
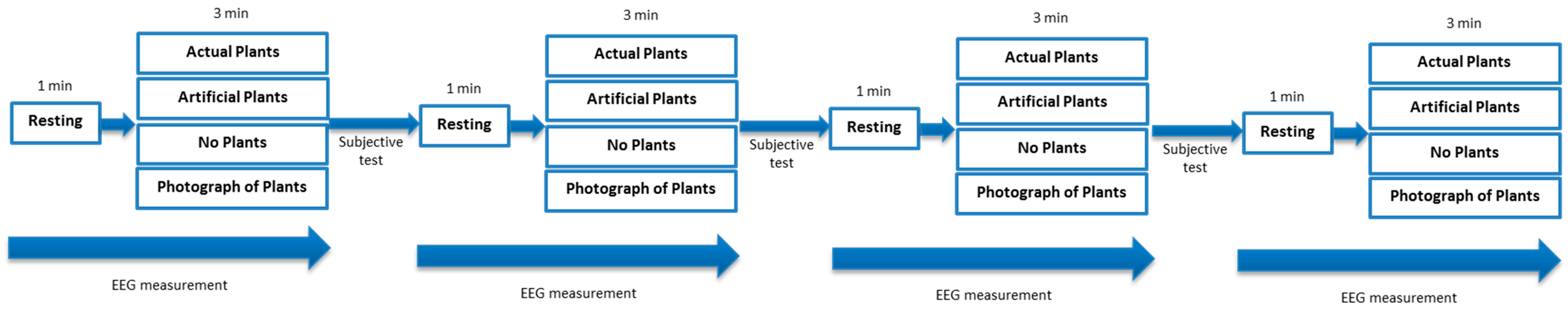
2.3. Experimental Protocol
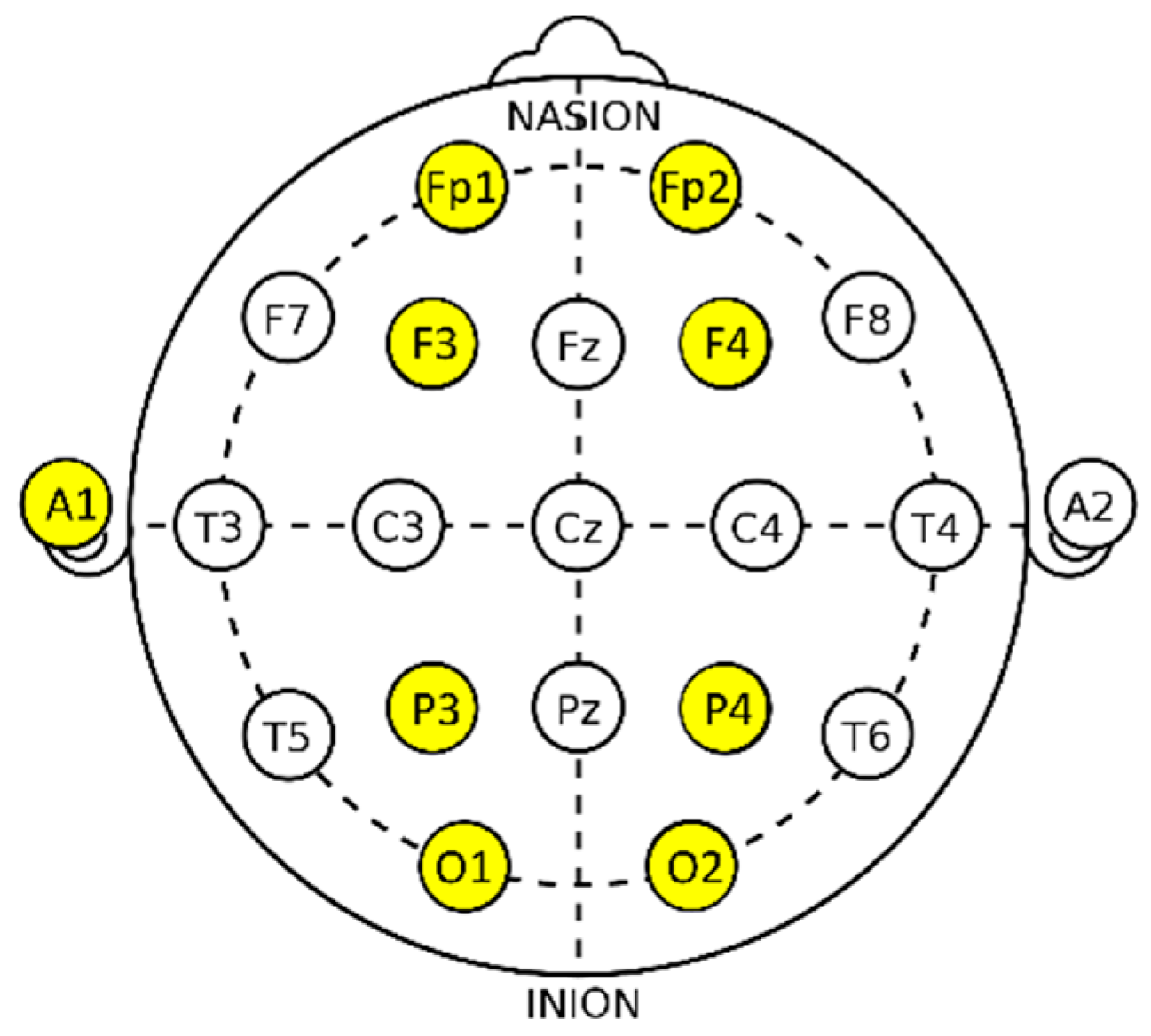
2.4. Measurement Items
2.5. Data Analysis
3. Results
3.1. Descriptive Characteristics
3.2. Electroencephalography (EEG)
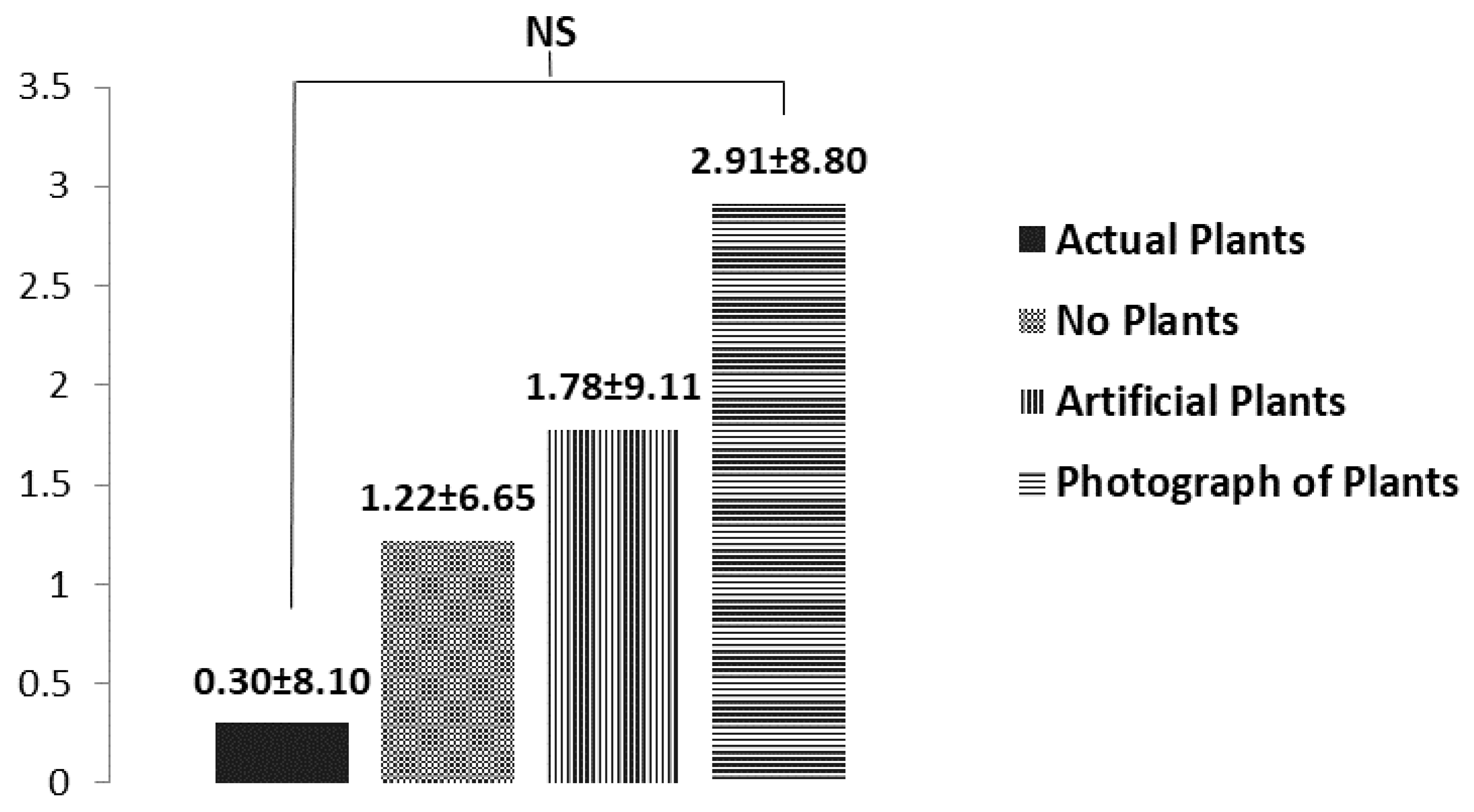
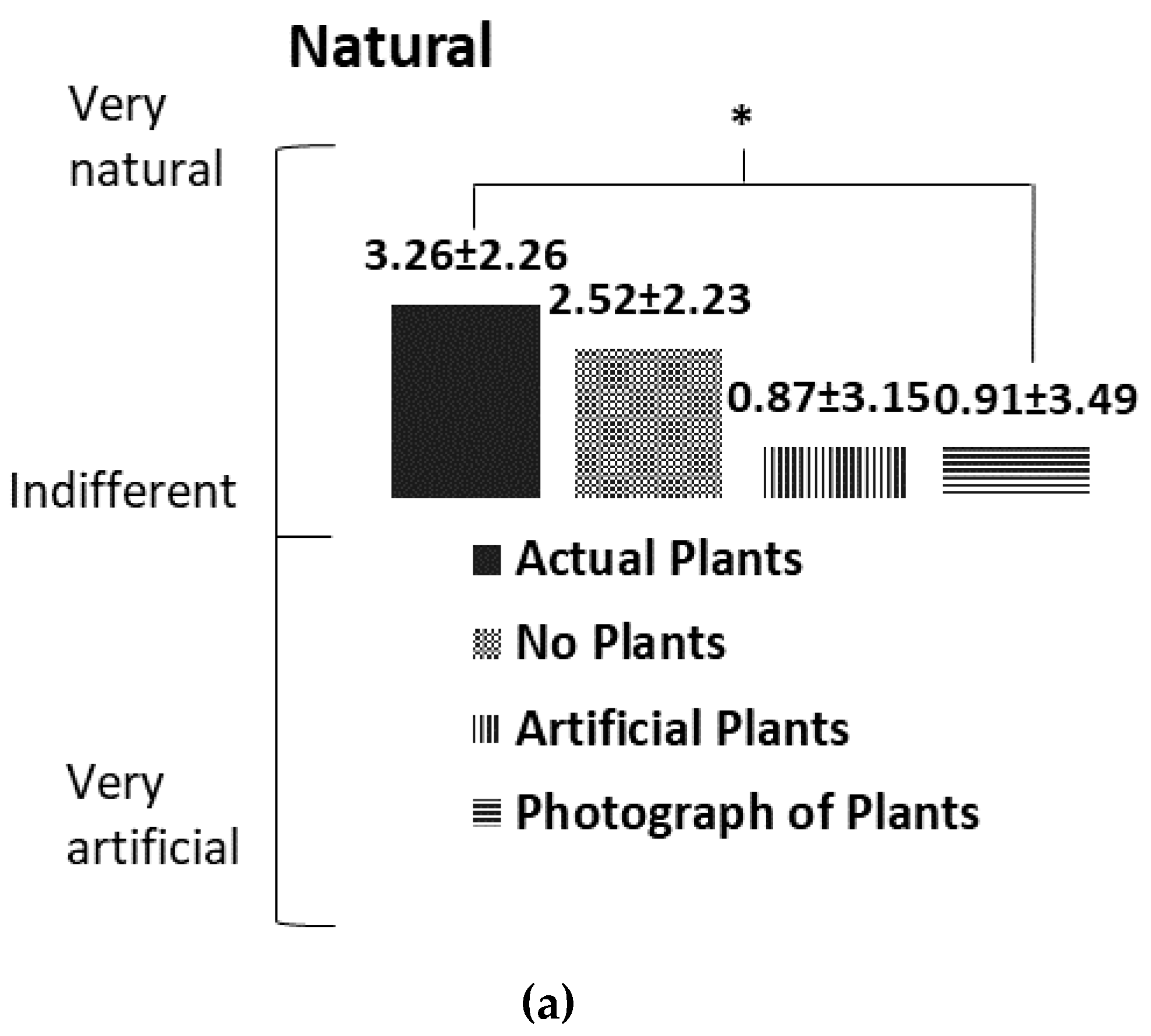
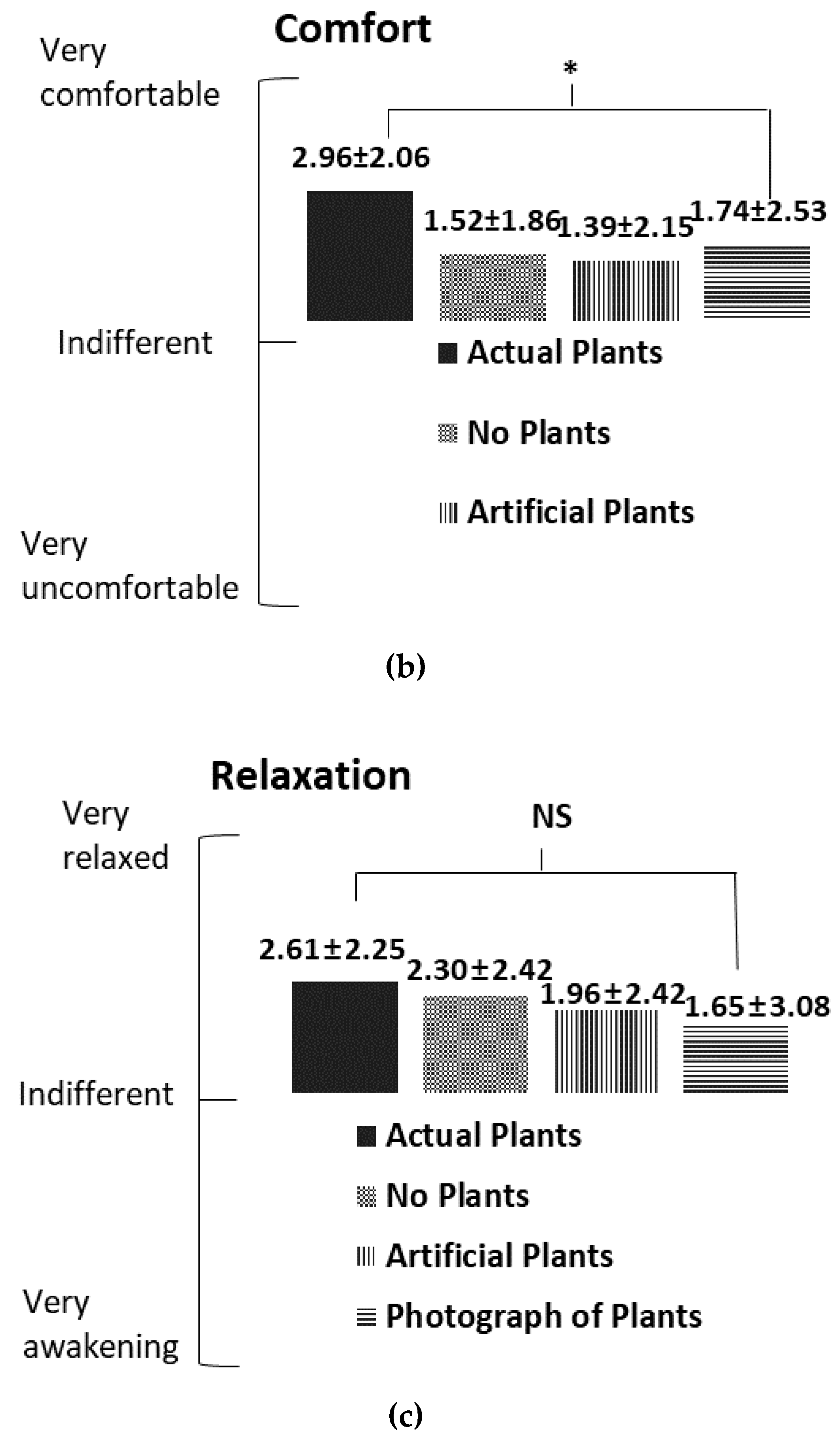
3.3. Subjective Mood Assessment
4. Discussion
4.1. Electroencephalography (EEG)
4.2. Subjective Mood Assessment
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kwon, T.E. Development and Status of Urban Agriculture in Seoul. Master’s Thesis, Korea University, Seoul, Korea, 2014. [Google Scholar]
- Orians, G.H. An Ecological and Evolutionary Approach to Landscape Aesthetics. In Landscape Meanings and Values; Allen and Unwin: London, UK, 1986; pp. 3–25. [Google Scholar]
- Wilson, E.O. Biophilia; Harvard Press: Cambridge, MA, USA, 1984. [Google Scholar]
- Bringslimark, T.; Hartig, T.; Patil, G.G. The Psychological Benefits of Indoor Plants: A Critical Review of the Experimental Literature. J. Environ. Psychol. 2009, 29, 422–433. [Google Scholar] [CrossRef]
- Lee, S.H. The Context Analysis of the Emotion-Improving Effects of the Index of Greenness: Home and Other Places. Korean J. Psychol. Health 2007, 12, 997–1017. [Google Scholar]
- Smith, A.; Tucker, M.; Pitt, M. Healthy, Productive Workplaces: Towards a Case for Interior Plantscaping. Facilities 2011, 29, 209–223. [Google Scholar] [CrossRef]
- Hartig, T.; Evans, G.W.; Jamner, L.D.; Davis, D.S.; Gärling, T. Tracking Restoration in Natural and Urban Field Settings. J. Environ. Psychol. 2003, 23, 109–123. [Google Scholar] [CrossRef]
- Kaplan, R.; Kaplan, S. The Experience of Nature: A Psychological Perspective; Cambridge University Press: New York, NY, USA, 1989. [Google Scholar]
- Ulrich, R.S. Aesthetic and affective response to natural environment. In Behavior and the Natural Environment; Springer: Boston, MA, USA, 1983; pp. 85–125. [Google Scholar]
- Hartig, T.; Mang, M.; Evans, G.W. Restorative Effects of Natural Environment Experiences. Environ. Behav. 1991, 23, 3–26. [Google Scholar] [CrossRef]
- Laumann, K.; Gärling, T.; Stormark, K.M. Selective Attention and Heart Rate Responses to Natural and Urban Environments. J. Environ. Psychol. 2003, 23, 125–134. [Google Scholar] [CrossRef]
- Park, B.; Tsunetsugu, Y.; Kasetani, T.; Hirano, H.; Kagawa, T.; Sato, M.; Miyazaki, Y. Physiological Effects of Shinrin-Yoku (Taking in the Atmosphere of the Forest)—Using Salivary Cortisol and Cerebral Activity as Indicators. J. Physiol. Anthropol. 2007, 26, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Tsunetsugu, Y.; Park, B.; Ishii, H.; Hirano, H.; Kagawa, T.; Miyazaki, Y. Physiological Effects of Shinrin-Yoku (Taking in the Atmosphere of the Forest) in an Old-Growth Broadleaf Forest in Yamagata Prefecture, Japan. J. Physiol. Anthropol. 2007, 26, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Environment. Korean Exposure Factors Handbook for Children; Ministry of Environment: Sejong, Korea, 2016.
- Park, S.; Lee, A.; Lee, G.; Kim, D.; Kim, W.; Shoemaker, C.A.; Son, K. Horticultural Activity Interventions and Outcomes. Hortic. Sci. Technol. 2016, 34, 513–527. [Google Scholar]
- Lee, B.J.; Kim, S.S.; Kim, N.H. An Exploratory Study on Korean Children’s Development—The Effects of Socioeconomic Factors and Social Capital. Korean Soc. Child Welf. 2010, 31, 107–141. [Google Scholar]
- Montessori, M. Pédagogie Scientifique, 5th ed.; Desclee De Brouwer, 1958 Et the Montessori Method; Schocken Books: New York, NY, USA, 1964. [Google Scholar]
- Wilson, R. Nature and Young Children. Young Child. 1995, 50, 4–11. [Google Scholar]
- Relf, D. Human Issues in Horticulture. HortTechnology 1992, 2, 159–171. [Google Scholar] [CrossRef]
- Bagot, K.L. Perceived Restorative Components: A Scale for Children. Child. Youth Environ. 2004, 14, 107–129. [Google Scholar]
- Corraliza, J.A.; Collado, S.; Bethelmy, L. Children’s Perceived Restoration: Adaptation of the PRCS for Children to a Spanish Sample. Psyecology 2012, 3, 195–204. [Google Scholar] [CrossRef]
- Bowler, D.E.; Buyung-Ali, L.M.; Knight, T.M.; Pullin, A.S. A Systematic Review of Evidence for the Added Benefits to Health of Exposure to Natural Environments. BMC Public Health 2010, 10, 456. [Google Scholar] [CrossRef] [PubMed]
- Ikei, H.; Komatsu, M.; Song, C.; Himoro, E.; Miyazaki, Y. The Physiological and Psychological Relaxing Effects of Viewing Rose Flowers in Office Workers. J. Physiol. Anthropol. 2014, 33, 6. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Song, C.; Choi, J.; Son, K.; Miyazaki, Y. Foliage Plants Cause Physiological and Psychological Relaxation as Evidenced by Measurements of Prefrontal Cortex Activity and Profile of Mood States. HortScience 2016, 51, 1308–1312. [Google Scholar] [CrossRef]
- Son, K.; Lee, J.; Song, J. Effect of Visual Recognition of Indoor Plants on Changes of Human Brain Electroencephalography. Hortic. Environ. Biotechnol. 1998, 39, 858–862. [Google Scholar]
- Park, S.; Song, C.; Oh, Y.; Miyazaki, Y.; Son, K. Comparison of Physiological and Psychological Relaxation using Measurements of Heart Rate Variability, Prefrontal Cortex Activity, and Subjective Indexes After Completing Tasks with and without Foliage Plants. Int. J. Environ. Res. Public Health 2017, 14, 1087. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Park, B.; Lee, J.; Park, K.; Ku, J.; Lee, J.; Oh, K.; Miyazaki, Y. Physiological Relaxation Induced by Horticultural Activity: Transplanting Work using Flowering Plants. J. Physiol. Anthropol. 2013, 32, 15. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Lee, J.; Park, B.; Miyazaki, Y. Interaction with Indoor Plants may Reduce Psychological and Physiological Stress by Suppressing Autonomic Nervous System Activity in Young Adults: A Randomized Crossover Study. J. Physiol. Anthropol. 2015, 34, 21. [Google Scholar] [CrossRef] [PubMed]
- Son, K.C.; Park, S.K.; Boo, H.O.; Bae, G.Y.; Paek, K.Y.; Lee, S.H. Horticultural Therapy; Seowon: Seoul, Korea, 1997. [Google Scholar]
- Banich, M.T.; Kim, M.S.; Kang, E.J.; Kang, Y.W.; Kim, H.T. Cognitive Neuroscience and Neuropsychology; Sigma Press: Seoul, Korea, 2008. [Google Scholar]
- Park, B.W. Introduction to Neurofeedback; Korea Mental Health Institute: Seoul, Korea, 2005. [Google Scholar]
- Kim, D.S.; Choi, J.W. Electroencephalogram; Koryo Medical: Seoul, Korea, 2001. [Google Scholar]
- Jasper, H.H. The Ten-Twenty Electrode System of the International Federation. Electroencephalogr. Clin. Neurophysiol. 1958, 10, 370–375. [Google Scholar]
- McNair, D.; Heuchert, J.; Shilony, E. Profile of Mood States Bibliography 1964–2002; Multi-Health Systems Inc.: New York, NY, USA, 2003. [Google Scholar]
- Yeun, E.J.; Shin-Park, K.K. Verification of the Profile of Mood States-brief: Cross-cultural Analysis. J. Clin. Psychol. 2006, 62, 1173–1180. [Google Scholar] [CrossRef] [PubMed]
- Osgood, C.E. The Nature and Measurement of Meaning. Psychol. Bull. 1952, 49, 197–237. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Mean Body Mass Index (BMI). Available online: https://www.who.int/gho/ncd/risk_factors/bmi_text/en/ (accessed on 1 January 2018).
- Hwang, M.C.; Son, J.H.; Kim, C.J. The Brain Wave of Emotional Characteristics by Sensory. J. Ergon. Soc. Korea 1996, 1, 66–71. [Google Scholar]
- Park, B.W. Optimization of Brain Function by Interrelationship of EEG Bands. Kor. Soc. Body-Mind-Spirit Sci. 2004, 21, 103–108. [Google Scholar]
- Son, K.C. Indoor Plants Revitalize People: Sick House Syndrome and Indoor Plant Wellbeing; Chungang Life Publish: Seoul, Korea, 2004. [Google Scholar]
- Kuo, F.E. Coping with Poverty: Impacts of Environment and Attention in the Inner City. Environ. Behav. 2001, 33, 5–34. [Google Scholar] [CrossRef]
- Kuo, F.E.; Taylor, A.F. A Potential Natural Treatment for Attention-Deficit/Hyperactivity Disorder: Evidence from a National Study. Am. J. Public Health 2004, 94, 1580–1586. [Google Scholar] [CrossRef] [PubMed]
- Wells, N.M. At Home with Nature: Effects of “greenness” on Children’s Cognitive Functioning. Environ. Behav. 2000, 32, 775–795. [Google Scholar] [CrossRef]
- Glass, A. Significance of EEG Alpha Asymmetries in Cerebral Dominance. Int. J. Psychophysiol. 1991, 11, 32–33. [Google Scholar] [CrossRef]
- Lee, J.; Son, K. Effects of Indoor Plant and Various Colors’ Stimuli on the Changes of Brain Activity and Emotional Responses. Hortic. Environ. Biotechnol. 1999, 40, 772–776. [Google Scholar]
- Jang, H.S. Influences of Visual Element of Ornamental Plants to Emotion or Electroencephalography of Human. Ph.D. Thesis, Korea University, Seoul, Korea, 2013. [Google Scholar]
- Igarashi, M.; Song, C.; Ikei, H.; Miyazaki, Y. Effect of Stimulation by Foliage Plant Display Images on Prefrontal Cortex Activity: A Comparison with Stimulation using Actual Foliage Plants. J. Neuroimaging 2015, 25, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Park, B.; Tsunetsugu, Y.; Ohira, T.; Kagawa, T.; Miyazaki, Y. Effect of Forest Bathing on Physiological and Psychological Responses in Young Japanese Male Subjects. Public Health 2011, 125, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Tsunetsugu, Y.; Lee, J.; Park, B.; Tyrväinen, L.; Kagawa, T.; Miyazaki, Y. Physiological and Psychological Effects of Viewing Urban Forest Landscapes Assessed by Multiple Measurements. Landsc. Urban Plan. 2013, 113, 90–93. [Google Scholar] [CrossRef]
- Park, B.; Tsunetsugu, Y.; Kasetani, T.; Morikawa, T.; Kagawa, T.; Miyazaki, Y. Physiological Effects of Forest Recreation in a Young Conifer Forest in Hinokage Town, Japan. Silva Fenn. 2009, 43, 291–301. [Google Scholar] [CrossRef]
- Wichrowski, M.; Whiteson, J.; Haas, F.; Mola, A.; Rey, M.J. Effects of Horticultural Therapy on Mood and Heart Rate in Patients Participating in an Inpatient Cardiopulmonary Rehabilitation Program. J. Cardiopulm. Rehabil. 2005, 25, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, A.E.; Koole, S.L.; Van der Wulp, N.Y. Environmental Preference and Restoration: (How) are they Related? J. Environ. Psychol. 2003, 23, 135–146. [Google Scholar] [CrossRef]

| Variables | |
|---|---|
| Gender | % (N) |
| Males | 39.1 (9) |
| Females | 60.9 (14) |
| Mean (SD) | |
| Age | 12.2 (1.0) |
| Height (cm) | 149.6 (11.3) |
| Weight (kg) | 44.3 (11.8) |
| Body mass index (kg·m−2) 1 | 19.4 (3.1) |
| EEG | Stimuli | F3 (µV) | F4 (µV) |
|---|---|---|---|
| Mean ± SD | |||
| Alpha | Actual Plants | 0.21 ± 0.79 | 0.21 ± 0.73 |
| No Plants | 0.22 ± 0.07 | 0.22 ± 0.07 | |
| Artificial Plants | 0.21 ± 0.05 | 0.20 ± 0.04 | |
| Photograph of Plants | 0.20 ± 0.06 | 0.19 ± 0.06 | |
| Significance | NS | NS | |
| Theta | Actual Plants | 0.37 ± 0.94 | 0.36 ± 0.82 |
| No Plants | 0.38 ± 0.10 | 0.37 ± 0.11 | |
| Artificial Plants | 0.44 ± 0.90 | 0.44 ± 0.10 | |
| Photograph of Plants | 0.43 ± 0.10 | 0.42 ± 0.11 | |
| Significance | 0.028 * | 0.048 * | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, Y.-A.; Kim, S.-O.; Park, S.-A. Real Foliage Plants as Visual Stimuli to Improve Concentration and Attention in Elementary Students. Int. J. Environ. Res. Public Health 2019, 16, 796. https://doi.org/10.3390/ijerph16050796
Oh Y-A, Kim S-O, Park S-A. Real Foliage Plants as Visual Stimuli to Improve Concentration and Attention in Elementary Students. International Journal of Environmental Research and Public Health. 2019; 16(5):796. https://doi.org/10.3390/ijerph16050796
Chicago/Turabian StyleOh, Yun-Ah, Seon-Ok Kim, and Sin-Ae Park. 2019. "Real Foliage Plants as Visual Stimuli to Improve Concentration and Attention in Elementary Students" International Journal of Environmental Research and Public Health 16, no. 5: 796. https://doi.org/10.3390/ijerph16050796
APA StyleOh, Y.-A., Kim, S.-O., & Park, S.-A. (2019). Real Foliage Plants as Visual Stimuli to Improve Concentration and Attention in Elementary Students. International Journal of Environmental Research and Public Health, 16(5), 796. https://doi.org/10.3390/ijerph16050796






