The Association between Lead and Attention-Deficit/Hyperactivity Disorder: A Systematic Review
Abstract
1. Introduction
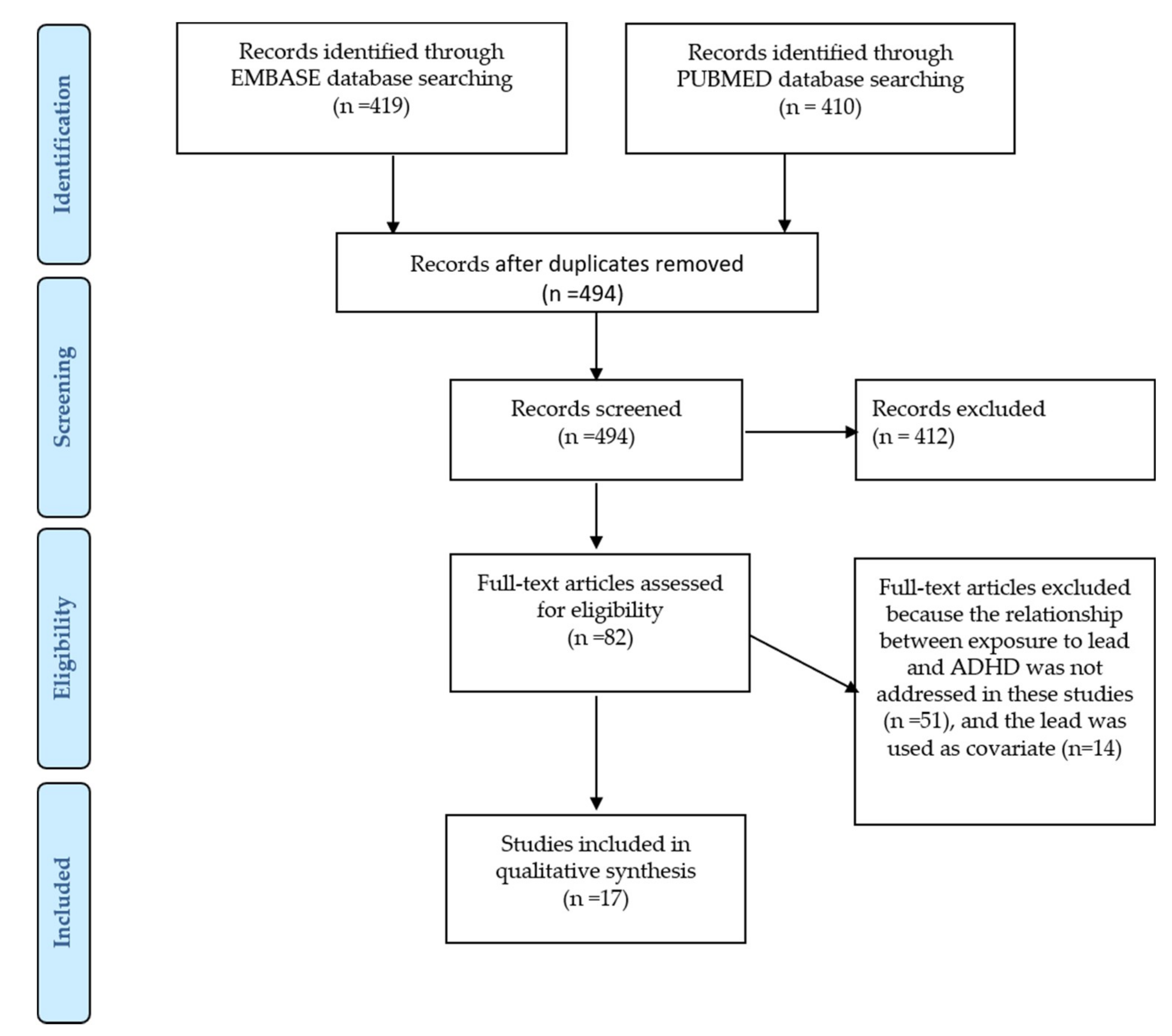
2. Methods
2.1. Study Identification and Eligibility Criteria
- ((“environmental” OR “pollution” OR “lead”) AND “hyperactivity disorder”)).
- the inclusion of humans as study subjects without restriction on the demographic characteristics of the population;
- the conduction of an exposure assessment to lead during pregnancy or early childhood; and
- the inclusion of measures of hyperactivity disorder symptoms or diagnosis.
2.2. Internal Validity
- a sufficiently large sample size;
- the specification of the inclusion and assessment criteria;
- an accurate diagnosis of ADHD and lead exposure assessment; and
- an adjustment for the confounding variables.
2.3. Data Extraction
3. Results
3.1. Characteristics of the Studies
3.2. Study Design and Population
3.3. Measurement of Lead Exposure
3.4. Cross-Sectional Studies
3.5. Cohort Studies
3.6. Case–Control Studies
4. Discussion
4.1. Summary of the Evidence
4.2. Strengths and Limitations of the Current Review
Conceptual Constraints
4.3. Strengths and Limitations of the Studies Included in the Review
4.3.1. ADHD Diagnosis
4.3.2. Observation and Exposure Periods
4.3.3. Lead Exposure Assessment
4.3.4. Measures of Association
4.3.5. Cofounders
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Polanczyk, G.; Willcutt, E.G.; Salum, A.G.; Kieling, C.; Rohde, L.A. ADHD prevalence estimates across three decades: An updated systematic review and meta-regression analysis. Int. J. Epidemiol. 2014, 43, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Parekh, R. American Psychiatric Association. 2017. Available online: https://www.psychiatry.org/patients-families/adhd/what-is-adhd (accessed on 26 August 2018).
- American Psychiatric Association. Attention Deficit/Hyperactivity Disorder. 2013. Available online: https://www.psychiatry.org/File%20Library/Psychiatrists/Practice/DSM/APA_DSM-5-ADHD.pdf (accessed on 9 November 2018).
- American Psychiatric Association (APA). Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV); APA: Washington, DC, USA, 2000. [Google Scholar]
- Liu, A.; Xu, Y.; Yan, Q.; Tong, L. The Prevalence of Attention Deficit/Hyperactivity Disorder among Chinese Children and Adolescents. Sci. Rep. 2018, 8, 11169. [Google Scholar] [CrossRef] [PubMed]
- Polanczyk, G.; de Lima, M.; Horta, B.; Biederman, J.; Rhode, L. The worldwide prevalence of ADHD: A systematic review and metaregression analysis. Am. J. Psychiatry 2007, 164, 942–948. [Google Scholar] [CrossRef] [PubMed]
- Willcutt, E. The Prevalence of DSM-IV Attention-Deficit/Hyperactivity Disorder: A Meta-Analytic Review. Neurotherapeutics 2012, 9, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Sciberras, E.; Mulraney, M.; Silva, D.; Coghill, D. Prenatal Risk Factors and the Etiology of ADHD—Review of Existing Evidence. Curr. Psychiatry Rep. 2017, 19. [Google Scholar] [CrossRef] [PubMed]
- Froehlich, T.E.; Anixt, J.S.; Loe, I.M.; Chirdkiatgumchai, V.; Kuan, L.; Gilman, R.C. Update on Environmental Risk Factors for Attention-Deficit/Hyperactivity Disorder. Curr. Psychiatry Rep. 2011, 13, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Walton, E.; Pingault, J.B.; Cecil, C.A.; Gaunt, T.R.; Relton, C.L.; Mill, J.; Barker, E.D. Epigenetic profiling of ADHD symptoms trajectories: A prospective, methylome-wide study. Mol. Psychiatry 2017, 22, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy Metals Toxicity and the Environment. Mol. Clin. Environ. Toxicol. 2012, 101, 133–164. [Google Scholar]
- Tong, S.; von Schirnding, Y.E.; Prapamontol, T. Environmental lead exposure: A public health problem of global dimensions. Bull. World Health Org. 2000, 78, 1068–1077. [Google Scholar]
- World Health Organization. International Lead Poisoning Prevention Awareness Campaign. 2017. Available online: http://www.who.int/ipcs/lead_campaign/QandA_lead_2017_en.pdf (accessed on 11 November 2018).
- Institute for Health Metrics and Evaluation. Lead Exposure. 2018. Available online: https://vizhub.healthdata.org/gbd-compare/ (accessed on 11 November 2018).
- Wildemann, T.M.; Weber, L.P.; Siciliano, S.D. Combined exposure to lead, inorganic mercury and methylmercury show deviation from additivity for cardiovascular toxicity in rats. J. Appl. Toxicol. 2015, 35, 918–926. [Google Scholar] [CrossRef] [PubMed]
- Beier, E.E.; Inzana, J.A.; Sheu, T.-J.; Shu, L.; Puzas, J.E.; Mooney, R.A. Effects of Combined Exposure to Lead and High-Fat Diet on Bone Quality in Juvenile Male Mice. Environ. Health Perspect. 2015, 123, 935–973. [Google Scholar] [CrossRef]
- World Health Organization. Childhood Lead Poisoning. 2010. Available online: http://www.who.int/ceh/publications/leadguidance.pdf (accessed on 11 December 2018).
- Centers for Disease Control and Prevention. Sources of Lead. 2015. Available online: https://www.cdc.gov/nceh/lead/tips/sources.htm (accessed on 11 December 2018).
- Centers for Disease Control and Prevention. Lead Toxicity. Where Is Lead Found? 2017. Available online: https://www.atsdr.cdc.gov/csem/csem.asp?csem=34&po=5 (accessed on 11 December 2018).
- Sanders, T.; Liu, Y.; Buchner, V.; Tchounwou, P.B. Neurotoxic Effects and Biomarkers of Lead Exposure: A Review. Rev. Environ. Health 2009, 24, 15–45. [Google Scholar] [CrossRef] [PubMed]
- Shukla, V.; Shukla, P.; Tiwari, A. Lead poisoning. Indian J. Med. Spec. 2018, 9, 146–149. [Google Scholar] [CrossRef]
- Amadi, C.N.; Igweze, Z.N.; Orisakwe, O.E. Heavy metals in miscarriages and stillbirths in developing nations. Middle East Fertil. Soc. J. 2017, 22, 91–100. [Google Scholar] [CrossRef]
- Hossain, N.; Nazli, E.W. Environmental factors implicated in the causation of adverse pregnancy outcome. Semin. Perinatol. 2007, 31, 240–242. [Google Scholar] [CrossRef]
- Vorvolakos, T.; Arseniou, S.; Samakouri, M. There is no safe threshold for lead exposure: A literature review. Psychiatriki 2016, 27, 204–214. [Google Scholar] [CrossRef]
- World Health Organization. Lead Poisoning and Health. 2018. Available online: http://www.who.int/news-room/fact-sheets/detail/lead-poisoning-and-health (accessed on 26 August 2018).
- Daneshparvar, M.; Mostafavi, S.A.; Jeddi, M.Z.; Yunesian, M.; Mesdaghinia, A.; Mahvi, A.H.; Akhondzadeh, S. The Role of Lead Exposure on Attention-Deficit/ Hyperactivity Disorder in Children: A Systematic Review. Iran J. Psychiatry 2016, 11, 1–14. [Google Scholar]
- He, J.; Ning, H.; Huang, R. Low blood lead levels and attention-deficit hyperactivity disorder in children: A systematic review and meta-analysis. Environ. Sci. Pollut. Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Friedenreich, C.M. Methods for pooled analyses of epidemiologic studies. Epidemiology 1993, 4, 295–302. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339. [Google Scholar] [CrossRef] [PubMed]
- SIGN. A Guideline Developer’s Handbook. 2008. Available online: https://www.sign.ac.uk/assets/sign50_2015.pdf (accessed on 11 September 2018).
- Baker, A.; Young, K.; Potter, J.; Madan, I. A review of grading systems for evidence-based guidelines produced by medical specialties. Clin. Med. 2010, 10, 358–363. [Google Scholar] [CrossRef]
- Sackett, D.L. Evidence-based medicine. Semin. Perinatol. 1997, 21, 3–5. [Google Scholar] [CrossRef]
- Huang, S.; Hu, H.; Sánchez, B.N.; Peterson, K.E.; Ettinger, A.S.; Lamadrid-Figueroa, H.; Schnaas, L.; Mercado-García, A.; Wright, R.O.; Basu, N.; et al. Childhood Blood Lead Levels and Symptoms of Attention Deficit Hyperactivity Disorder (ADHD): A Cross-Sectional Study of Mexican Children. Children’s Health 2016. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Huo, X.; Ho, G.; Chen, X.; Wang, H.; Wang, T.; Ma, L. Attention-deficit/hyperactivity symptoms in preschool children from an E-waste recycling town: Assessment by the parent report derived from DSM-IV. BMC Pediatrics 2015. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Hong, X.; Wang, G.; Chatterjee, N.; Riley, A.W.; Lee, L.-C.; Surkan, P.J.; Bartell, T.R.; Zuckerman, B.; Wang, X.; et al. A Prospective Birth Cohort Study on Early Childhood Lead Levels and Attention Deficit Hyperactivity Disorder: New Insight on Sex Differences. J. Pediatrics 2018. [Google Scholar] [CrossRef] [PubMed]
- Choi, W.J.; Kwon, H.J.; Lim, M.H.; Lim, J.A.; Ha, M. Blood lead, parental marital status and the risk of attention-deficit/hyperactivity disorder in elementary school children: A longitudinal study. Psychiatry Res. 2016, 236, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Neugebauer, J.; Wittsiepe, J.; Kasper-Sonnenberg, M.; Schöneck, N.; Schölmerich, A.; Wilhelm, M. The influence of low-level pre- and perinatal exposure to PCDD/Fs, PCBs, and lead on attention performance and attention-related related behavior among German school-aged children: Results from the Duisburg Birth Cohort Study. Int. J. Hyg. Environ. Health 2015. [Google Scholar] [CrossRef] [PubMed]
- Forns, J.; Fort, M.; Casas, M.; Cáceres, A.; Guxens, M.; Gascon, M.; Garcia-Esteban, R.; Julvez, J.; Grimalt, J.O.; Sunyer, J.; et al. Exposure to metals during pregnancy and neuropsychological development at the age of 4 years. NeuroToxicology 2014. [Google Scholar] [CrossRef] [PubMed]
- Sioen, I.; Den Hond, E.; Nelen, V.; Van de Mieroop, E.; Croes, K.; Van Larebeke, N.; Nawrot, T.S.; Schoeters, G. Prenatal exposure to environmental contaminants and behavioral problems at age 7–8 years. Environ. Int. 2013, 59, 225–231. [Google Scholar] [CrossRef]
- Yang, R.; Zhang, Y.; Gao, W.; Lin, N.; Li, R.; Zhao, Z. Blood Levels of Trace Elements in Children with Attention-Deficit Hyperactivity Disorder: Results from a Case-Control Study. Biol. Trace Element Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Chou, M.C.; Chou, W.J.; Huang, C.W.; Kuo, H.C.; Lee, S.Y.; Wang, L.J. Heavy Metals’ Effect on Susceptibility to Attention-Deficit/Hyperactivity Disorder: Implication of Lead, Cadmium, and Antimony. Int. J. Environ. Res. Public Health 2018. [Google Scholar] [CrossRef] [PubMed]
- Joo, H.; Lim, M.H.; Ha, M.; Kwon, H.J.; Yoo, S. J.; Choi, K.H.; Paik, K.C. Secondhand Smoke Exposure and Low Blood Lead Levels in Association With Attention-Deficit Hyperactivity Disorder and Its Symptom Domain in Children: A Community-Based Case–Control Study. Nicotine Tob. Res. 2017, 19, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Seo, J.H.; Hong, Y.S.; Kim, Y.M.; Kang, J.W.; Yoo, J.H.; Chueh, H.W.; Lee, J.H.; Kwak, M.J.; Kim, J.; et al. Blood lead concentrations and attention deficit hyperactivity disorder in Korean children: A hospital-based case control study. BMC Pediatrics 2016, 16, 156. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.J.; Du, J.C.; Chiou, H.C.; Feng, C.C.; Chung, M.Y.; Yang, W.; Chen, Y.S.; Chien, L.C.; Hwang, B.; Chen, M.L. Sugar-Sweetened Beverage Consumption Is Adversely Associated with Childhood Attention-Deficit/Hyperactivity Disorder. Int. J. Environ. Res. Public Health 2016. [Google Scholar] [CrossRef]
- Yu, C.J.; Du, J.C.; Chiou, H.C.; Yang, S.H.; Liao, K.W.; Yang, W.; Chung, M.Y.; Chien, L.C.; Hwang, B.; Chen, M.L. Attention Deficit/Hyperactivity Disorder and Urinary Nonylphenol Levels: A Case-Control Study in Taiwanese Children. PLoS ONE 2016. [Google Scholar] [CrossRef]
- Chan, T.J.; Gutierrez, C.; Ogunseitan, O.A. Metallic Burden of Deciduous Teeth and Childhood Behavioral Deficits. Int. J. Environ. Res. Public Health 2015. [Google Scholar] [CrossRef]
- Hong, S.B.; Im, M.H.; Kim, J.W.; Park, E.J.; Shin, M.S.; Kim, B.N.; Yoo, H.J.; Cho, I.H.; Bhang, S.Y.; Hong, Y.C.; et al. Environmental Lead Exposure and Attention Deficit/Hyperactivity Disorder Symptom Domains in a Community Sample of South Korean School-Age Children. Children’s Health 2015. [Google Scholar] [CrossRef]
- Kim, S.; Arora, M.; Fernandez, C.; Landero, J.; Caruso, J.; Chen, A. Lead, mercury, and cadmium exposure and attention deficit hyperactivity disorder in children. Environ. Res. 2013. [Google Scholar] [CrossRef]
- Dikme, G.; Arvas, A.; Gür, E. The relation between blood lead and mercury levels and chronic neurological diseases in children. Turk. Arch. Ped. 2013, 48, 221–225. [Google Scholar] [CrossRef]
- Faraone, S.V.; Asherson, P.; Banaschewski, T.; Biederman, J.; Buitelaar, J.K.; Ramos-Quiroga, J.A.; Rohde, L.A.; Sonuga-Barke, E.J.S.; Tannock, R.; Franke, B.; et al. Attention-deficit/hyperactivity disorder. Nat. Rev. Dis. Primers 2015. [Google Scholar] [CrossRef]
- Who, H.D.; Kim, D.W.; Hong, Y.S.; Kim, Y.M.; Seo, J.H.; Choe, B.M.; Park, J.H.; Kang, J.W.; Yoo, J.H.; Chueh, H.W.; et al. Dietary patterns in children with attention-deficit/hyperactivity disorder (ADHD). Nutrients 2014, 6, 1539–1553. [Google Scholar]
- Li, Y.; Zhang, H.; Kuang, H.; Fan, R.; Cha, C.; Li, G.; Luo, Z.; Pang, Q. Relationship between bisphenol A exposure and attention-deficit/hyperactivity disorder: A case-control study for primary school children in Guangzhou, China. Environ. Pollut. 2018, 235, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Perera, F.P.; Chang, H.; Tang, D.; Roen, E.L.; Herbstman, J.; Margolis, A.; Huang, T.J.; Miller, R.L.; Wang, S.; Rauh, V. Early-Life Exposure to Polycyclic Aromatic Hydrocarbons and ADHD Behavior Problems. PLoS ONE 2014. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.R.; Taylor, M.M.; Shalat, S.L.; Guillot, T.S.; Caudle, M.W.; Hossain, M.M.; Mathews, T.A.; Jones, S.R.; Cory-Slechta, D.A.; Miller, G.W. Developmental pesticide exposure reproduces features of attention deficit hyperactivity disorder. FASEB J. 2015, 29, 1960–1972. [Google Scholar] [CrossRef] [PubMed]
- Gulson, B.L.; Cameron, M.A.; Smith, A.J.; Mizon, K.J.; Korsch, M.J.; Vimpani, G.; McMichael, A.J.; Pisaniello, D.; Jameson, C.W.; Mahaffey, K.R. Blood Lead–Urine Lead Relationships in Adults and Children. Environ. Res. 1998, 78, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Dalton, J.E.; Bolen, S.D.; Mascha, E.J. Publication Bias: The Elephant in the Review. Anesth. Analg. 2016, 123, 812–813. [Google Scholar] [CrossRef] [PubMed]
- Bussing, R.; Fernandez, M.; Harwood, M.; Hou, W.; Garvan, C. W.; Eyberg, S.M.; Swanson, J.M. Parent and Teacher SNAP-IV Ratings of Attention Deficit/Hyperactivity Disorder Symptoms: Psychometric Properties and Normative Ratings from a School District Sample. Assessment 2008, 15, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Skelly, A.C.; Dettori, J.R.; Brodt, E.D. Assessing bias: The importance of considering confounding. Evid.-Based Spine-Care J. 2012, 3, 9–12. [Google Scholar] [CrossRef]
| Levels of Evidence | ||
|---|---|---|
| 1 | 1++ | High-quality meta-analyses, systematic reviews of RCTs, or RCTs with a very low risk of bias |
| 1+ | Well-conducted meta-analyses, systematic reviews of RCTs, or RCTswith a low risk of bias | |
| 1- | Meta-analyses, systematic reviews, or RCTs with a high risk of bias | |
| 2 | 2++ | High-quality systematic reviews of case–control or cohort studies High-quality case–control or cohort studies with a very low risk of confounding or bias and a high probability that the relationship is causal |
| 2+ | Well-conducted case–control or cohort studies with a low risk of confounding or bias and a moderate probability that the relationship is causal | |
| 2− | Case–control or cohort studies with a high risk of confounding or bias and a significant risk that the relationship is not causal | |
| 3 | Non-analytic studies, e.g., case reports, case series | |
| 4 | Expert opinion | |
| Grades of Recommendation | |
|---|---|
| A | At least one meta-analysis, systematic review, or RCT rated as 1++, and directly applicable to the target population A body of evidence consisting principally of studies rated as 1+, directly applicable to the target population, and demonstrating the overall consistency of results |
| B | A body of evidence including studies rated as 2++, directly applicable to the target population, and demonstrating the overall consistency of results Extrapolated evidence from studies rated as 1++ or 1+ |
| C | A body of evidence including studies rated as 2+, directly applicable to the target population, and demonstrating the overall consistency of results Extrapolated evidence from studies rated as 2++ |
| D | Evidence level 3 or 4 Extrapolated evidence from studies rated as 2+ |
| Citation | Location | Sample Size (Birth Years) | Study Design | ADHD Symptom Measured | Exposure Measurement | Results | LE | GR |
|---|---|---|---|---|---|---|---|---|
| Huang et al., 2016 [33] | Mexico | 4126–13 years | Cross-sectional | Inattention Hyperactivity/Impulsivity Combined ADHD | BLLs | BLLs among children with low exposure (≤5 μg/dL) was positively associated with hyperactive/impulsive behaviors but not with inattentiveness. | 2+ | C |
| Zhang et al., 2015 [34] | China | 2433–7 years | Cross-sectional | Inattention Hyperactivity/Impulsivity Combined ADHD | BLLs | The children with high BLLs (≥10 µg/dL) had a 2.4 times higher risk of ADHD than the children with low BLLs. | 2+ | C |
| Ji et al., 2018 [35] | Boston | 1479 mother–infant pairs (299/1176) | Birth cohort 1998–2013 | Combined ADHD | BLLs | Children with 5–10 μg/dL lead levels had 66% increased odds of having an ADHD diagnosis as compared with children with less than 5 μg/dL lead levels. | 2+ | C |
| Choi et al., 2016 [36] | South Korea | 2195 | Birth cohort 2005–2010 | Combined ADHD | BLLs | Relative risk for ADHD symptoms was 1.552 in children with blood lead levels > 2.17 μg/dL (highest quartile) compared with those with blood lead levels of ≤2.17 μg/dL. | 2+ | C |
| Neugebauer et al., 2014 [37] | Germany | 117 | Birth cohort 2000–2002 | Inattention Hyperactivity/Impulsivity Combined ADHD | BLLs | Lead exposure was positively associated with ADHD. Hyperactivity/Impulsivity and Combined ADHD significantly increased by 20% and 9% per each doubling of BLLs, respectively. | 2+ | C |
| Forns et al., 2014 [38] | Spain | 385 | Birth cohort 2004–2006 | Inattention Hyperactivity/Impulsivity | Urine sample | No statistically significant associations between lead and ADHD. | 2+ | C |
| Sioen et al., 2013 [39] | Belgium | 270 | Birth cohort 2002–2003 | Combined ADHD | BLLs | Doubling the prenatal lead exposure is associated with an odds ratio for hyperactivity of 3.43. | 2+ | C |
| Yang et al., 2018 [40] | China | 421/395 6–16 years | Case-control | Combined ADHD | BLLs | No statistically significant associations between lead and ADHD. | 2− | |
| Lee et al., 2018 [41] | Taiwan | 76/46 < 10 years | Case-control | Inattention Hyperactivity/Impulsivity | Urine sample | BLLs were positively correlated with inattention and hyperactivity/impulsivity symptoms (p < 0.05). | 2− | |
| Joo et al., 2017 [42] | South Korea | 214/2146–10 years | Case-control | Inattention Hyperactivity/Impulsivity Combined ADHD | BLLs | Exposure to low BLLs (geometric mean = 1.65 μg/dL) was associated with inattention symptoms but not with hyperactivity/impulsivity. | 2+ | C |
| Park et al., 2016 [43] | South Korea | 114/114 | Case-control | Inattention Hyperactivity/Impulsivity Combined ADHD | BLLs | Children with a blood lead concentration > 2.30 μg/dL had a 2.5 times higher risk of ADHD. | 2+ | C |
| Yu et al., 2016 (a) [44] | Taiwan | 173/159 | Case-control | Combined ADHD | BLLs | No statistically significant associations between lead and ADHD. | 2− | |
| Yu et al., 2016 (b) [45] | Taiwan | 97/1104–15 years | Case-control | Combined ADHD | BLLs | No statistically significant associations between lead and ADHD. | 2− | |
| Chan et al., 2015 [46] | USA | 266 11–13 years | Case-control | Inattention Hyperactivity/Impulsivity Combined ADHD | Analysis of teeth | BLLs were significantly associated with increased incidents of Hyperactivity/Impulsivity and Inattention. | 2+ | C |
| Hong et al., 2015 [47] | South Korea | 10018–11 years | Case-control | Inattention Hyperactivity/Impulsivity Combined ADHD | BLLs | BLLs were significantly associated with parent and teacher ratings for Hyperactivity/Impulsivity but not with Inattention. | 2+ | C |
| Kim et al., 2013 [48] | USA | 71/58 3–7 years | Case-control | Combined ADHD | BLLs | High BLLs were associated with a higher risk of ADHD. | 2+ | C |
| Dikme et al., 2013 [49] | Turkey | 59/591.6–16 years | Case-control | Combined ADHD | BLLs | No statistically significant associations between lead and ADHD. | 2− |
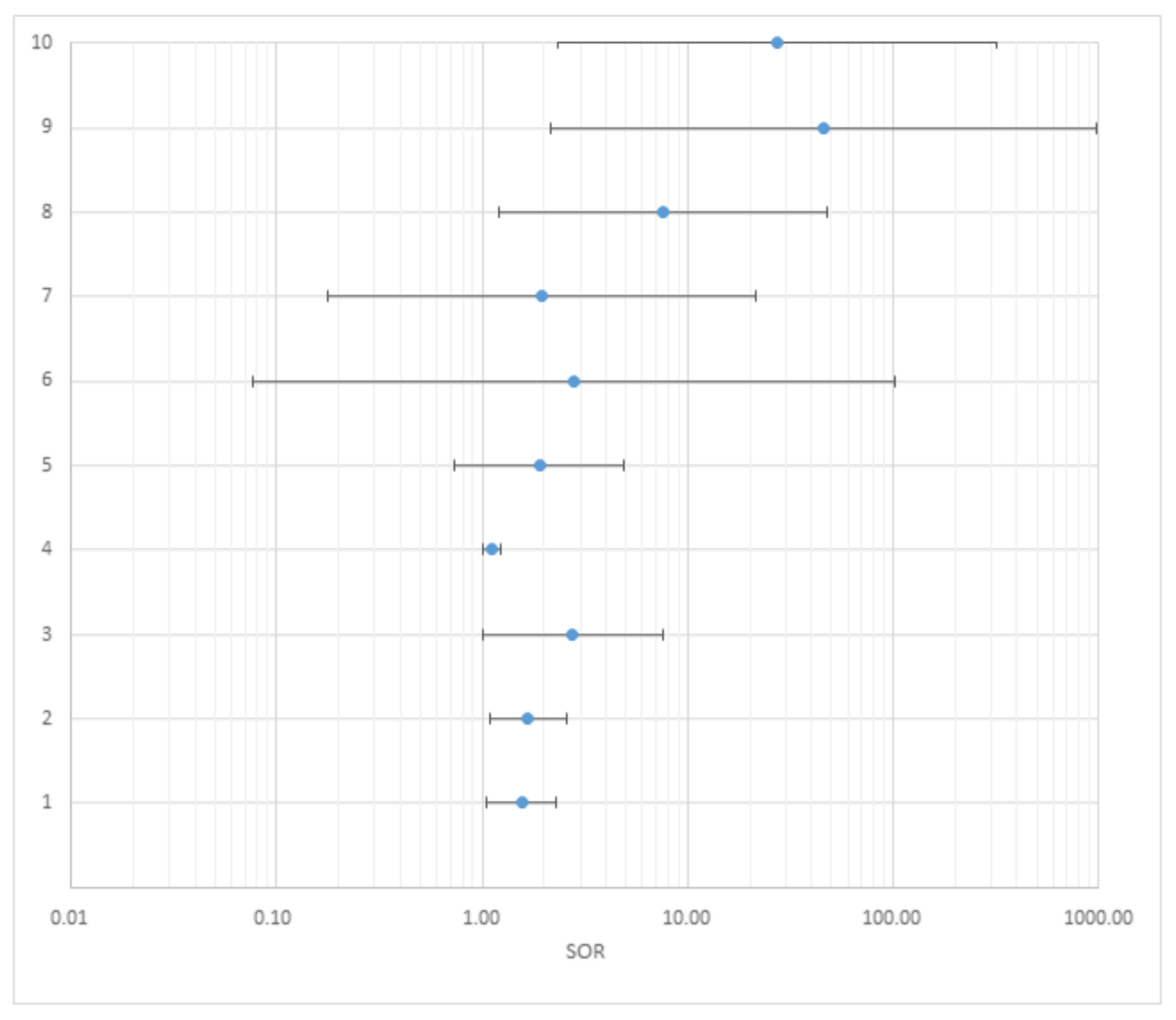
| Citation | N° | Adjusted ORs | Standardized ORs | ||||
|---|---|---|---|---|---|---|---|
| OR | Lower 95% CI | Upper 95% CI | OR | Lower 95% CI | Upper 95% CI | ||
| Zhang et al., 2015 [34] | |||||||
| —binary: cutoff 10 µg/dℓ | |||||||
| All ADHD | 1 | 2.4 | 1.1 | 5.2 | 1.55 | 1.05 | 2.28 |
| Ji et al., 2018 [35] | |||||||
| —binary: cutoff 5 µg/dL | |||||||
| All ADHD | 2 | 1.66 | 1.08 | 2.56 | 1.66 | 1.08 | 2.56 |
| Choi et al., 2016 [36] | |||||||
| —binary: cutoff 2.17 µg/dℓ | |||||||
| All ADHD | 3 | 1.552 | 1.002 | 2.403 | 2.753 | 1.005 | 7.539 |
| Neugebauer et al., 2014 [37] | |||||||
| —doubling of exposure concentrations | |||||||
| All ADHD | 4 | 1.09 | 1.01 | 1.17 | 1.12 | 1.01 | 1.22 |
| Joo et al., 2017 [42] | |||||||
| —binary: cutoff 1.90 µg/dℓ | |||||||
| All ADHD | 5 | 1.28 | 0.89 | 1.83 | 1.91 | 0.74 | 4.91 |
| Park et al., 2016 [43] | |||||||
| —categorical | |||||||
| All ADHD (1.13–1.71 μg/dℓ) | 6 | 1.26 | 0.56 | 2.84 | 2.78 | 0.08 | 101.35 |
| All ADHD (1.72–2.29 μg/dℓ) | 7 | 1.26 | 0.55 | 2.87 | 1.96 | 0.18 | 21.43 |
| All ADHD (2.30–5.35 μg/dℓ) | 8 | 2.54 | 1.09 | 5.94 | 7.59 | 1.21 | 48.10 |
| Kim et al., 2013 [48] | |||||||
| —categorical | |||||||
| All ADHD (>2 μg/dℓ) | 9 | 4.63 | 1.36 | 15.72 | 46.13 | 2.16 | 979.79 |
| All ADHD (>3 μg/dℓ) | 10 | 7.25 | 1.66 | 31.67 | 27.16 | 2.33 | 317.02 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donzelli, G.; Carducci, A.; Llopis-Gonzalez, A.; Verani, M.; Llopis-Morales, A.; Cioni, L.; Morales-Suárez-Varela, M. The Association between Lead and Attention-Deficit/Hyperactivity Disorder: A Systematic Review. Int. J. Environ. Res. Public Health 2019, 16, 382. https://doi.org/10.3390/ijerph16030382
Donzelli G, Carducci A, Llopis-Gonzalez A, Verani M, Llopis-Morales A, Cioni L, Morales-Suárez-Varela M. The Association between Lead and Attention-Deficit/Hyperactivity Disorder: A Systematic Review. International Journal of Environmental Research and Public Health. 2019; 16(3):382. https://doi.org/10.3390/ijerph16030382
Chicago/Turabian StyleDonzelli, Gabriele, Annalaura Carducci, Agustin Llopis-Gonzalez, Marco Verani, Agustin Llopis-Morales, Lorenzo Cioni, and María Morales-Suárez-Varela. 2019. "The Association between Lead and Attention-Deficit/Hyperactivity Disorder: A Systematic Review" International Journal of Environmental Research and Public Health 16, no. 3: 382. https://doi.org/10.3390/ijerph16030382
APA StyleDonzelli, G., Carducci, A., Llopis-Gonzalez, A., Verani, M., Llopis-Morales, A., Cioni, L., & Morales-Suárez-Varela, M. (2019). The Association between Lead and Attention-Deficit/Hyperactivity Disorder: A Systematic Review. International Journal of Environmental Research and Public Health, 16(3), 382. https://doi.org/10.3390/ijerph16030382







