Zinc and Cadmium Mapping in the Apical Shoot and Hypocotyl Tissues of Radish by High-Resolution Secondary Ion Mass Spectrometry (NanoSIMS) after Short-Term Exposure to Metal Contamination
Abstract
1. Introduction
2. Experimental Set-Up and Methodology
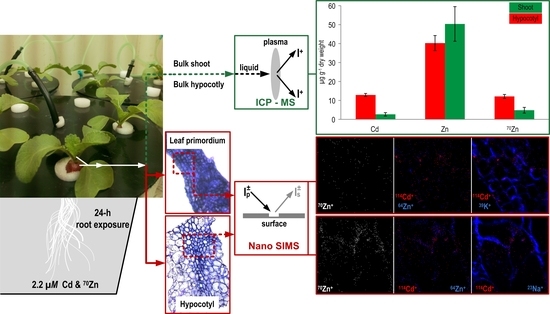
2.1. Plant Growth Conditions
2.2. Sampling and Sample Preparation for the NanoSIMS
2.3. NanoSIMS Detection
2.4. Sampling and Sample Preparation for ICP-MS Analysis
3. Result and Discussion
3.1. Total Concentration of Zn and Cd
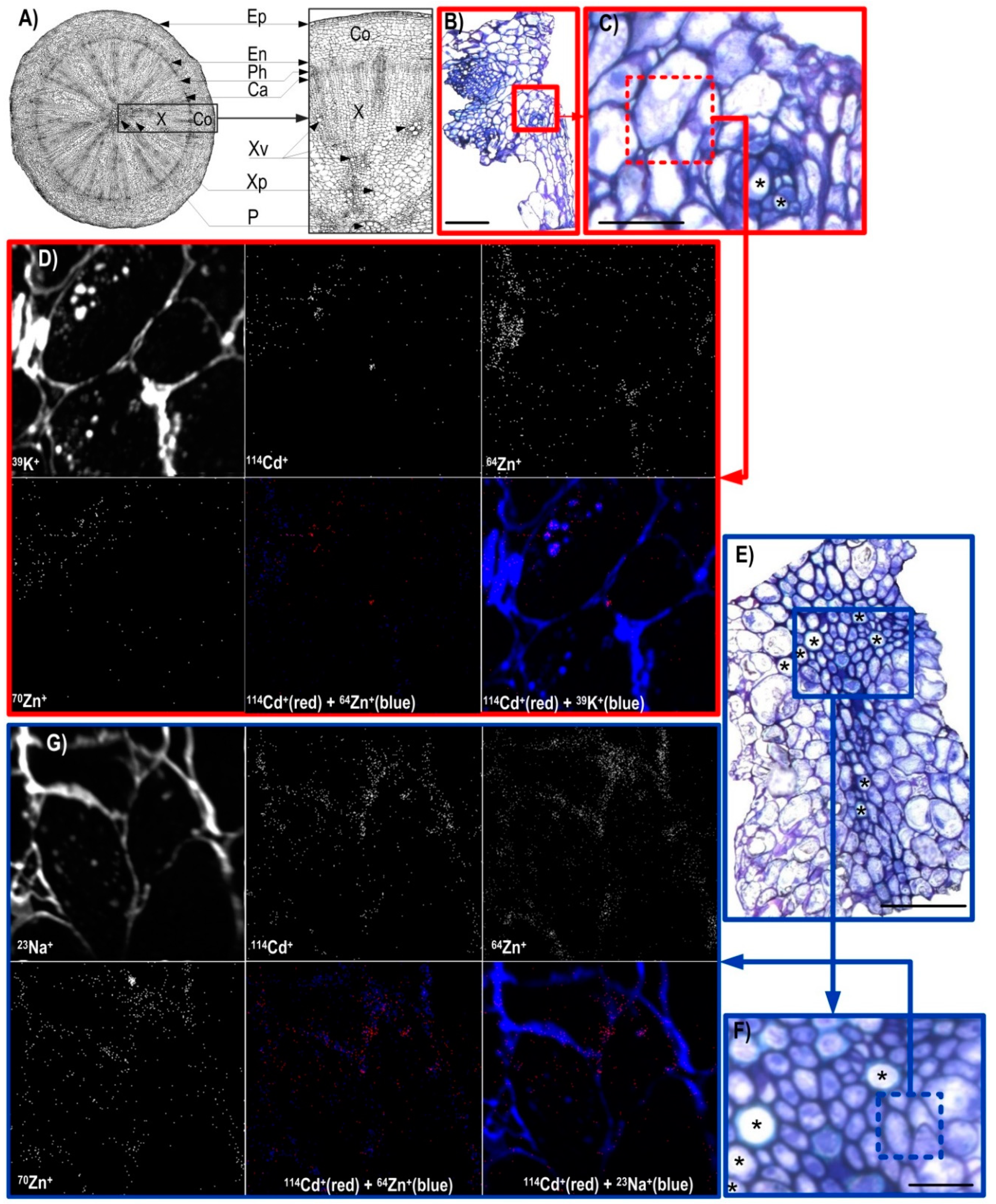
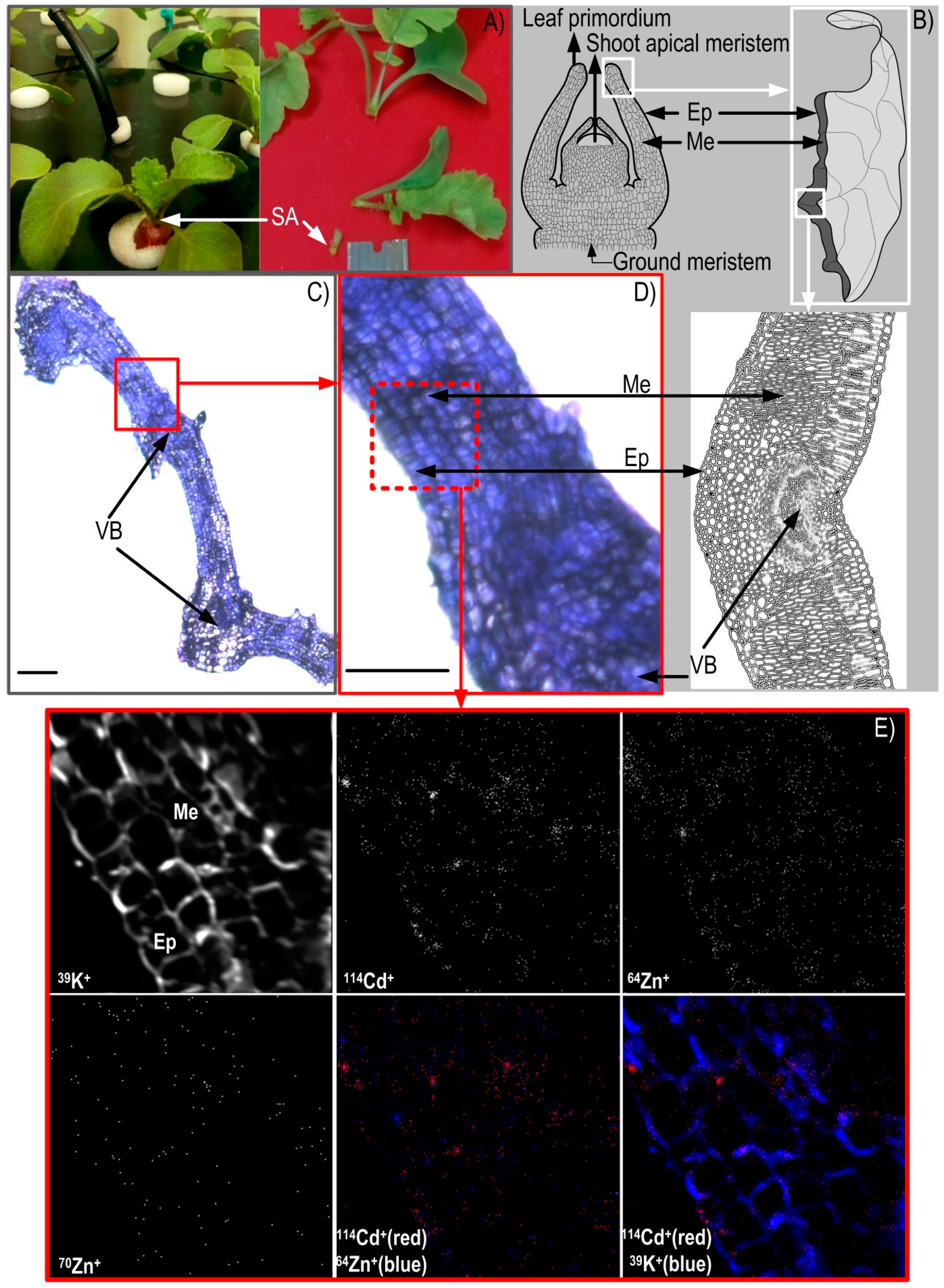
3.2. NanoSIMS Maps of Metals of Interest in the Hypocotyl and Shoot Apex
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Villafort Carvalho, M.T.; Pongrac, P.; Mumm, R.; van Arkel, J.; van Aelst, A.; Jeromel, L.; Vavpetič, P.; Pelicon, P.; Aarts, M.G.M. Gomphrena claussenii, a novel metal-hypertolerant bioindicator species, sequesters cadmium, but not zinc, in vacuolar oxalate crystals. New Phytol. 2015, 208, 763–775. [Google Scholar] [CrossRef]
- Rengel, Z. Availability of Mn, Zn and Fe in the rhizosphere. J. Soil Sci. Plant Nutr. 2015, 15, 397–409. [Google Scholar] [CrossRef]
- Ondrasek, G.; Rengel, Z. The Role of Soil Organic Matter in Trace Element Bioavailability and Toxicity; Springer: New York, NY, USA, 2012; ISBN 9781461406341. [Google Scholar]
- Ondrasek, G. The Responses of Salt-Affected Plants to Cadmium; Springer: New York, NY, USA, 2013; pp. 439–463. ISBN 978-1-4614-6107-4. [Google Scholar]
- Huang, Y.; He, C.; Shen, C.; Guo, J.; Mubeen, S.; Yuan, J.; Yang, Z. Toxicity of cadmium and its health risks from leafy vegetable consumption. Food Funct. 2017, 8, 1373–1401. [Google Scholar] [CrossRef] [PubMed]
- Lavado-García, J.M.; Puerto-Parejo, L.M.; Roncero-Martín, R.; Moran, J.M.; Pedrera-Zamorano, J.D.; Aliaga, I.J.; Leal-Hernández, O.; Canal-Macias, M.L. Dietary intake of cadmium, lead and mercury and its association with bone health in healthy premenopausal women. Int. J. Environ. Res. Public Health 2017, 14, 1437. [Google Scholar] [CrossRef] [PubMed]
- Piglowski, M. Heavy metals in notifications of rapid alert system for food and feed. Int. J. Environ. Res. Public Health 2018, 15, 365. [Google Scholar] [CrossRef]
- Lane, T.W.; Morel, F.M.M. A biological function for cadmium in marine diatoms. Proc. Natl. Acad. Sci. USA 2000, 97, 4627–4631. [Google Scholar] [CrossRef]
- Baeyens, W.; Gao, Y.; Davison, W.; Galceran, J.; Leermakers, M.; Puy, J.; Superville, P.-J.; Beguery, L. In situ measurements of micronutrient dynamics in open seawater show that complex dissociation rates may limit diatom growth. Sci. Rep. 2018, 8, 16125. [Google Scholar] [CrossRef]
- Xie, W.; Peng, C.; Wang, H.; Chen, W. Health risk assessment of trace metals in various environmental media, crops and human hair from a mining affected area. Int. J. Environ. Res. Public Health 2017, 14, 1595. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Qin, G.; Gu, H.; Qu, L.-J. Dof5.6/HCA2, a Dof Transcription Factor Gene, Regulates Interfascicular Cambium Formation and Vascular Tissue Development in Arabidopsis. Plant Cell 2009, 21, 3518–3534. [Google Scholar] [CrossRef] [PubMed]
- Myburg, A.A.; Lev-Yadun, S.; Sederoff, R.R. Xylem Structure and Function; John Wiley & Sons, Inc.: New York, NY, USA, 2013. [Google Scholar] [CrossRef]
- Liebsch, D.; Sunaryo, W.; Holmlund, M.; Norberg, M.; Zhang, J.; Hall, H.C.; Helizon, H.; Jin, X.; Helariutta, Y.; Nilsson, O.; et al. Class I KNOX transcription factors promote differentiation of cambial derivatives into xylem fibers in the Arabidopsis hypocotyl. Development 2014, 141, 4311–4319. [Google Scholar] [CrossRef] [PubMed]
- Rouhier, H.; Usuda, H. Spatial and temporal distribution of sucrose synthase in the radish hypocotyl in relation to thickening growth. Plant Cell Physiol. 2001, 42, 583–593. [Google Scholar] [CrossRef]
- Yu, R.; Wang, J.; Xu, L.; Wang, Y.; Wang, R.; Zhu, X.; Sun, X.; Luo, X.; Xie, Y.; Everlyne, M.; et al. Transcriptome Profiling of Taproot Reveals Complex Regulatory Networks during Taproot Thickening in Radish (Raphanus sativus L.). Front. Plant Sci. 2016, 7, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Xu, L.; Wang, Y.; Fan, L.; Chen, Y.; Tang, M.; Luo, X.; Liu, L. Comparative proteomic analysis provides insight into a complex regulatory network of taproot formation in radish (Raphanus sativus L.). Hortic. Res. 2018, 5, 51. [Google Scholar] [CrossRef] [PubMed]
- Ondrasek, G.; Rengel, Z.; Romic, D. Humic acids decrease uptake and distribution of trace metals, but not the growth of radish exposed to cadmium toxicity. Ecotoxicol. Environ. Saf. 2018, 151, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Suga, S.; Murai, M.; Kuwagata, T.; Maeshima, M. Differences in aquaporin levels among cell types of radish and measurement of osmotic water permeability of individual protoplasts. Plant Cell Physiol. 2003, 44, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Boxer, S.G.; Kraft, M.L.; Weber, P.K. Advances in Imaging Secondary Ion Mass Spectrometry for Biological Samples. Annu. Rev. Biophys. 2009, 38, 53–74. [Google Scholar] [CrossRef]
- Moore, K.L.; Lombi, E.; Zhao, F.J.; Grovenor, C.R.M. Elemental imaging at the nanoscale: NanoSIMS and complementary techniques for element localisation in plants. Anal. Bioanal. Chem. 2012, 402, 3263–3273. [Google Scholar] [CrossRef] [PubMed]
- Kilburn, M.R.; Jones, D.L.; Clode, P.L.; Cliff, J.B.; Stockdale, E.A.; Herrmann, A.M.; Murphy, D.V. Application of nanoscale secondary ion mass spectrometry to plant cell research. Plant Signal. Behav. 2010, 5, 760–762. [Google Scholar] [CrossRef]
- Smart, K.E.; Smith, J.A.C.; Kilburn, M.R.; Martin, B.G.H.; Hawes, C.; Grovenor, C.R.M. High-resolution elemental localization in vacuolate plant cells by nanoscale secondary ion mass spectrometry. Plant J. 2010, 63, 870–879. [Google Scholar] [CrossRef]
- Ondrasek, G.; Rengel, Z.; Clode, P.L.; Kilburn, M.R.; Guagliardo, P.; Romic, D. Zinc and cadmium mapping by NanoSIMS within the root apex after short-term exposure to metal contamination. Ecotoxicol. Environ. Saf. 2019, 171, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.L.; Chen, Y.; van de Meene, A.M.L.; Hughes, L.; Liu, W.; Geraki, T.; Mosselmans, F.; Mcgrath, S.P.; Grovenor, C.; Zhao, F.J. Combined NanoSIMS and synchrotron X-ray fluorescence reveal distinct cellular and subcellular distribution patterns of trace elements in rice tissues. New Phytol. 2014, 201, 104–115. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Liu, X.; Peijnenburg, W.J.G.M.; Zhao, J.; Chen, X.; Yu, J.; Wu, H. Pathways of cadmium fluxes in the root of the halophyte Suaeda salsa. Ecotoxicol. Environ. Saf. 2012, 75, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kilburn, M.R.; Clode, P.L. Elemental and Isotopic Imaging of Biological Samples Using NanoSIMS BT - Electron Microscopy: Methods and Protocols; Kuo, J., Ed.; Humana Press: Totowa, NJ, USA, 2014; pp. 733–755. ISBN 978-1-62703-776-1. [Google Scholar]
- Moore, K.L.; Schroder, M.; Wu, Z.; Martin, B.G.H.; Hawes, C.R.; McGrath, S.P.; Hawkesford, M.J.; Feng Ma, J.; Zhao, F.-J.; Grovenor, C.R.M. High-Resolution Secondary Ion Mass Spectrometry Reveals the Contrasting Subcellular Distribution of Arsenic and Silicon in Rice Roots. Plant Physiol. 2011, 156, 913–924. [Google Scholar] [CrossRef]
- Clode, P.L.; Kilburn, M.R.; Jones, D.L.; Stockdale, E.A.; Cliff, J.B.; Herrmann, A.M.; Murphy, D.V. In Situ Mapping of Nutrient Uptake in the Rhizosphere Using Nanoscale Secondary Ion Mass Spectrometry. Plant Physiol. 2009, 151, 1751–1757. [Google Scholar] [CrossRef] [PubMed]
- Gormanns, P.; Reckow, S.; Poczatek, J.C.; Turck, C.W.; Lechene, C. Segmentation of multi-isotope imaging mass spectrometry data for semi-automatic detection of regions of interest. PLoS ONE 2012, 7, e30576. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.L.; Schröder, M.; Lombi, E.; Zhao, F.J.; McGrath, S.P.; Hawkesford, M.J.; Shewry, P.R.; Grovenor, C.R.M. NanoSIMS analysis of arsenic and selenium in cereal grain. New Phytol. 2010, 185, 434–445. [Google Scholar] [CrossRef]
- Caruso, J.A.; Montes-Bayon, M. Elemental speciation studies—New directions for trace metal analysis. Ecotoxicol. Environ. Saf. 2003, 56, 148–163. [Google Scholar] [CrossRef]
- Khalid, S.; Shahid, M.; Natasha; Bibi, I.; Sarwar, T.; Shah, A.H.; Niazi, N.K. A review of environmental contamination and health risk assessment of wastewater use for crop irrigation with a focus on low and high-income countries. Int. J. Environ. Res. Public Health 2018, 15, 895. [Google Scholar] [CrossRef]
- Gupta, N.; Khan, D.K.; Santra, S.C. Heavy metal accumulation in vegetables grown in a long-term wastewater-irrigated agricultural land of tropical India. Environ. Monit. Assess. 2012, 184, 6673–6682. [Google Scholar] [CrossRef]
- Ondrasek, G.; Rengel, Z.; Romic, D.; Poljak, M.; Romic, M. Accumulation of non/essential elements in radish plants grown in salt-affected and cadmium-contaminated environment. Cereal Res. Commun. 2009, 37, 9–12. [Google Scholar]
- Cabrera, D.; Young, S.D.; Rowell, D.L. The toxicity of cadmium to barley plants as affected by complex formation with humic acid. Plant Soil 1988, 105, 195–204. [Google Scholar] [CrossRef]
- Curtis, I.S. The noble radish: past, present and future. Trends Plant Sci. 2003, 8, 305–307. [Google Scholar] [CrossRef]
- Filipović, L.; Romić, M.; Romić, D.; Filipović, V.; Ondrašek, G. Organic matter and salinity modify cadmium soil (phyto)availability. Ecotoxicol. Environ. Saf. 2018, 147, 824–831. [Google Scholar] [CrossRef] [PubMed]
- Marschner, P.; Crowley, D.; Rengel, Z. Rhizosphere interactions between microorganisms and plants govern iron and phosphorus acquisition along the root axis - model and research methods. Soil Biol. Biochem. 2011, 43, 883–894. [Google Scholar] [CrossRef]
- Smart, K.E.; Kilburn, M.R.; Salter, C.J.; Smith, J.A.C.; Grovenor, C.R.M. NanoSIMS and EPMA analysis of nickel localisation in leaves of the hyperaccumulator plant Alyssum lesbiacum. Int. J. Mass Spectrom. 2007, 260, 107–114. [Google Scholar] [CrossRef]
- Clemens, S. Molecular mechanisms of plant metal tolerance and homeostasis. Planta 2001, 212, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Clemens, S. Toxic metal accumulation, responses to exposure and mechanisms of tolerance in plants. Biochimie 2006, 88, 1707–1719. [Google Scholar] [CrossRef] [PubMed]
- Zaki, H.E.M.; Takahata, Y.; Yokoi, S. Analysis of the morphological and anatomical characteristics of roots in three radish (Raphanus sativus) cultivars that differ in root shape. J. Hortic. Sci. Biotechnol. 2012, 87, 172–178. [Google Scholar] [CrossRef]
- Jang, G.; Lee, J.-H.; Rastogi, K.; Park, S.; Oh, S.-H.; Lee, J.-Y. Cytokinin-dependent secondary growth determines root biomass in radish (Raphanus sativus L.). J. Exp. Bot. 2015, 66, 4607–4619. [Google Scholar] [CrossRef] [PubMed]
- Mitsui, Y.; Shimomura, M.; Komatsu, K.; Namiki, N.; Shibata-Hatta, M.; Imai, M.; Katayose, Y.; Mukai, Y.; Kanamori, H.; Kurita, K.; et al. The radish genome and comprehensive gene expression profile of tuberous root formation and development. Sci. Rep. 2015, 5, 10835. [Google Scholar] [CrossRef] [PubMed]
- Kitashiba, H.; Li, F.; Hirakawa, H.; Kawanabe, T.; Zou, Z.; Hasegawa, Y.; Tonosaki, K.; Shirasawa, S.; Fukushima, A.; Yokoi, S.; et al. Draft Sequences of the Radish (Raphanus sativus L.) Genome. DNA Res. 2014, 21, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Szilvia, V.; Gabrijel, O.; László, Z. Wheat Sensitivity to Nitrogen Supply under Different Climatic Conditions. Glob. Wheat Prod. 2018. [Google Scholar] [CrossRef]
- Yu, H.; Wang, J.; Fang, W.; Yuan, J.; Yang, Z. Cadmium accumulation in different rice cultivars and screening for pollution-safe cultivars of rice. Sci. Total Environ. 2006, 370, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Xin, J.; Huang, B.; Yang, Z.; Yuan, J.; Zhang, Y. Comparison of cadmium subcellular distribution in different organs of two water spinach (Ipomoea aquatica Forsk.) cultivars. Plant Soil 2013, 372, 431–444. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ondrasek, G.; Clode, P.L.; Kilburn, M.R.; Guagliardo, P.; Romić, D.; Rengel, Z. Zinc and Cadmium Mapping in the Apical Shoot and Hypocotyl Tissues of Radish by High-Resolution Secondary Ion Mass Spectrometry (NanoSIMS) after Short-Term Exposure to Metal Contamination. Int. J. Environ. Res. Public Health 2019, 16, 373. https://doi.org/10.3390/ijerph16030373
Ondrasek G, Clode PL, Kilburn MR, Guagliardo P, Romić D, Rengel Z. Zinc and Cadmium Mapping in the Apical Shoot and Hypocotyl Tissues of Radish by High-Resolution Secondary Ion Mass Spectrometry (NanoSIMS) after Short-Term Exposure to Metal Contamination. International Journal of Environmental Research and Public Health. 2019; 16(3):373. https://doi.org/10.3390/ijerph16030373
Chicago/Turabian StyleOndrasek, Gabrijel, Peta L. Clode, Matt R. Kilburn, Paul Guagliardo, Davor Romić, and Zed Rengel. 2019. "Zinc and Cadmium Mapping in the Apical Shoot and Hypocotyl Tissues of Radish by High-Resolution Secondary Ion Mass Spectrometry (NanoSIMS) after Short-Term Exposure to Metal Contamination" International Journal of Environmental Research and Public Health 16, no. 3: 373. https://doi.org/10.3390/ijerph16030373
APA StyleOndrasek, G., Clode, P. L., Kilburn, M. R., Guagliardo, P., Romić, D., & Rengel, Z. (2019). Zinc and Cadmium Mapping in the Apical Shoot and Hypocotyl Tissues of Radish by High-Resolution Secondary Ion Mass Spectrometry (NanoSIMS) after Short-Term Exposure to Metal Contamination. International Journal of Environmental Research and Public Health, 16(3), 373. https://doi.org/10.3390/ijerph16030373