PEN-13: A New Generic 13-Item Questionnaire for Measuring Patient Enablement (German Version)
Abstract
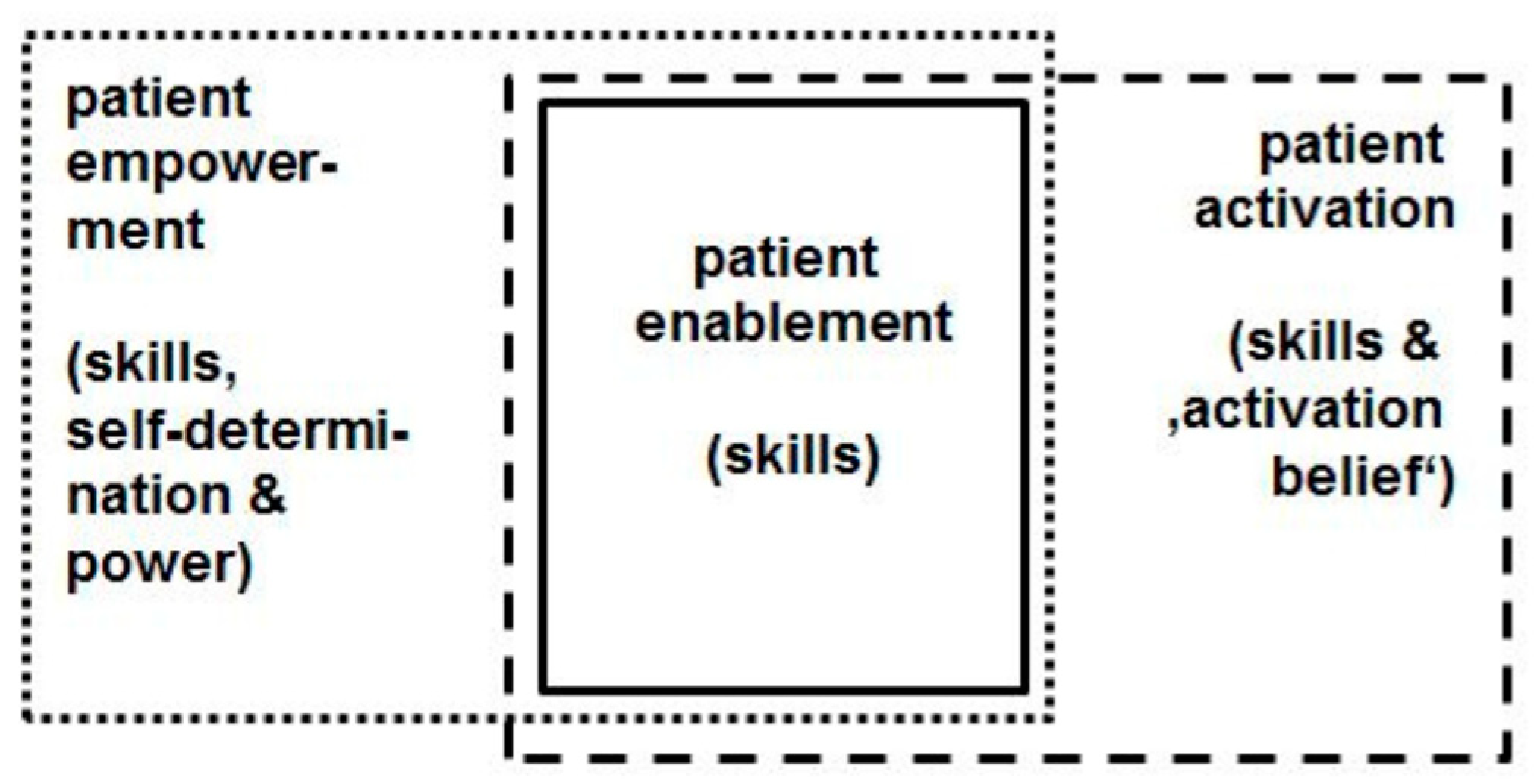
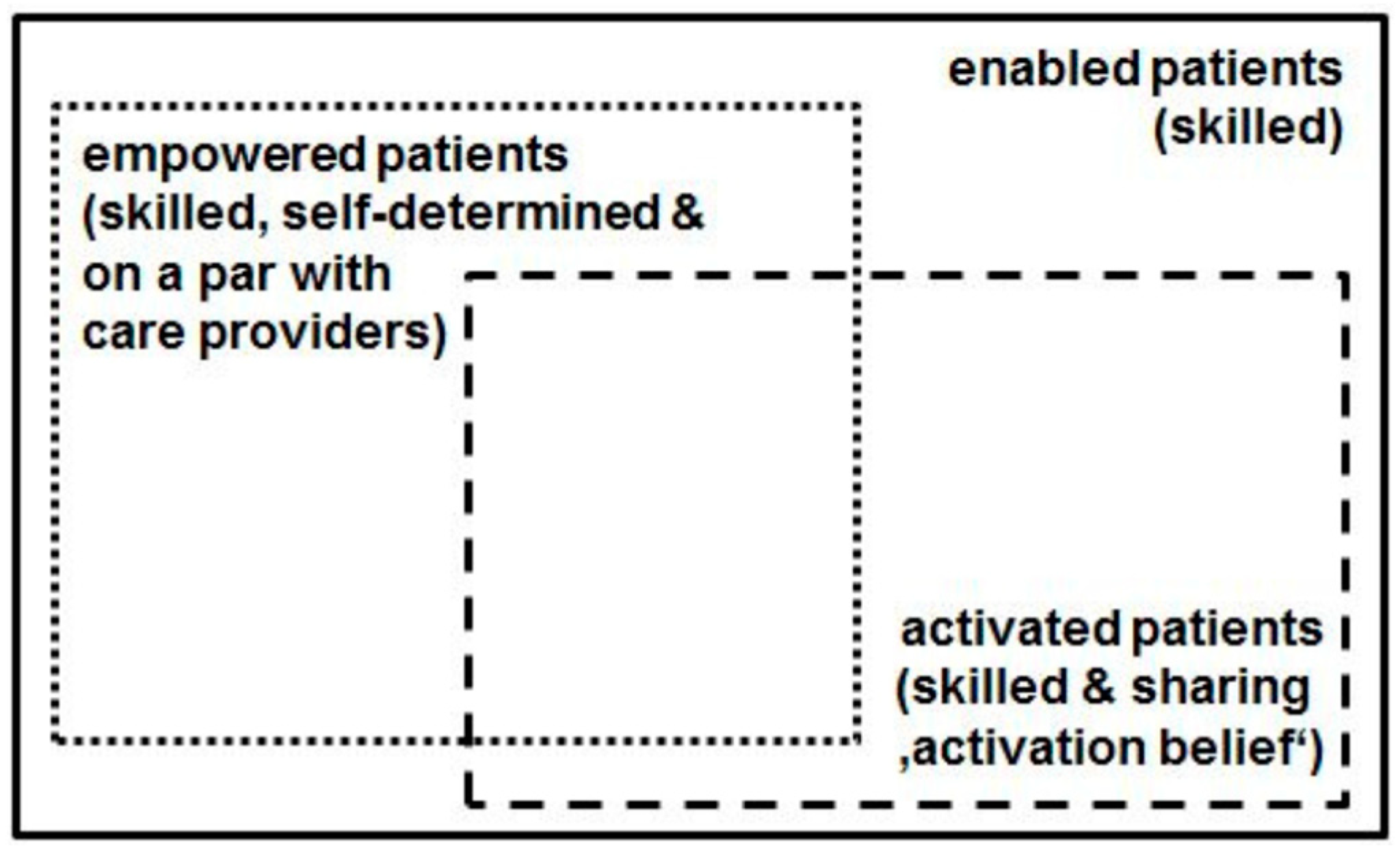
1. Introduction
2. Materials and Methods
2.1. Item Development
2.2. Data Collection and Study Population
2.3. Scales for Construct Validation by Hypotheses Testing
2.4. Statistical Analysis
2.5. Reliability
2.6. Construct Validity
2.6.1. Structural Validity
2.6.2. Hypotheses Testing
3. Results
3.1. Response and Socio-Demographic Characteristics
3.2. Exploratory Analysis
3.3. Internal Consistency
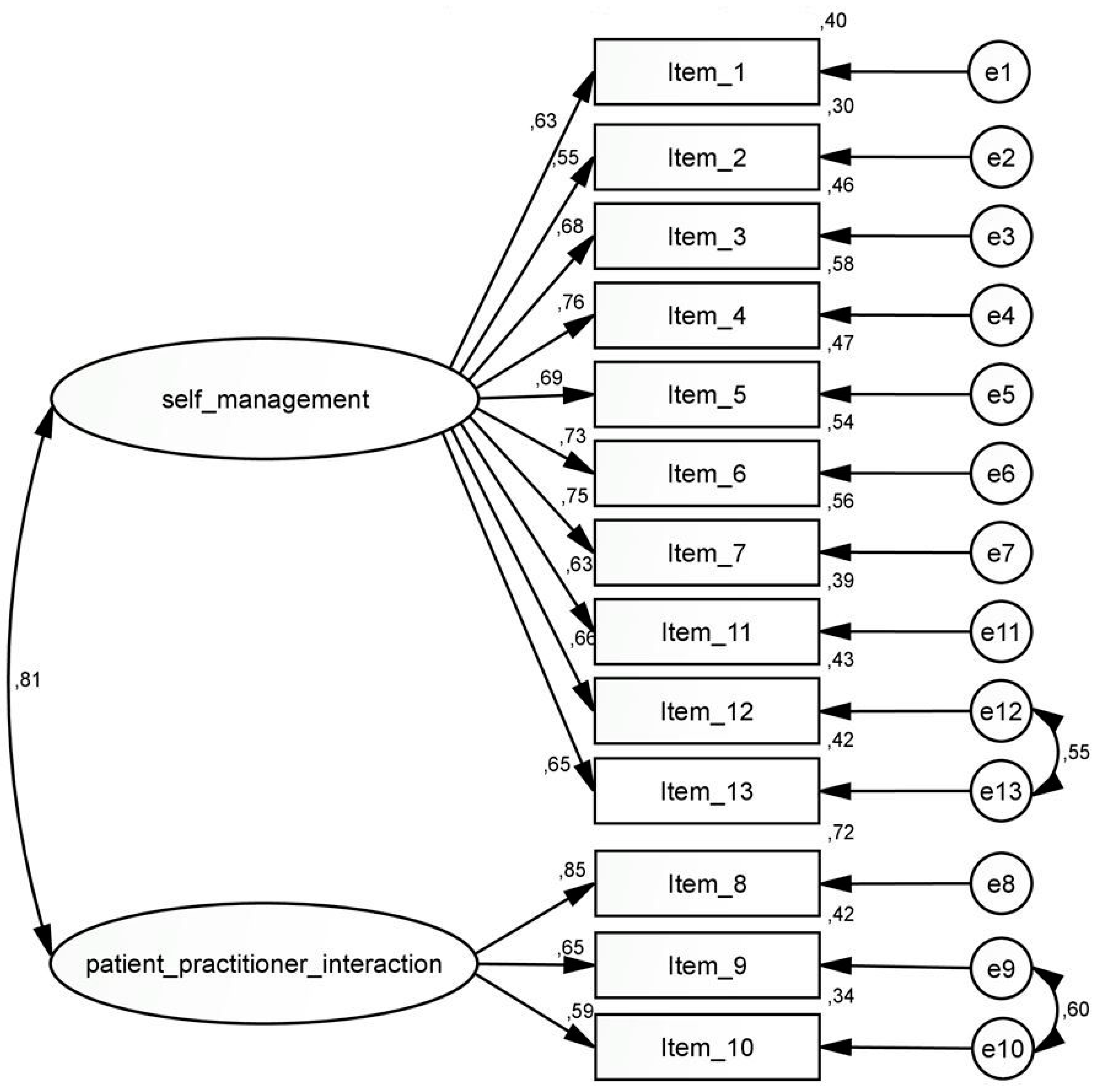
3.4. Confirmatory Analysis
3.5. Hypotheses Testing
4. Discussion
5. Conclusions
Practical Implications
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Availability of the German version of PEN-13
References
- Fumagalli, L.P.; Radaelli, G.; Lettieri, E.; Bertele’, P.; Masella, C. Patient Empowerment and its neighbours: Clarifying the boundaries and their mutual relationships. Health Policy 2015, 119, 384–394. [Google Scholar] [CrossRef] [PubMed]
- Howie, J.G.; Heaney, D.J.; Maxwell, M.; Walker, J.J. A comparison of a Patient Enablement Instrument (PEI) against two established satisfaction scales as an outcome measure of primary care consultations. Fam. Pract. 1998, 15, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Wallace, P.; Barber, J.; Clayton, W.; Currell, R.; Fleming, K.; Garner, P.; Haines, A.; Harrison, R.; Jacklin, P.; Jarrett, C.; et al. Virtual outreach: A randomised controlled trial and economic evaluation of joint teleconferenced medical consultations. Health Technol. Assess. 2004, 8, 1–106. [Google Scholar] [CrossRef] [PubMed]
- Wensing, M.; van Lieshout, J.; Jung, H.P.; Hermsen, J.; Rosemann, T. The Patients Assessment Chronic Illness Care (PACIC) questionnaire in The Netherlands: A validation study in rural general practice. BMC Health Serv. Res. 2008, 8, 182. [Google Scholar] [CrossRef]
- Pawlikowska, T.; Zhang, W.; Griffiths, F.; van Dalen, J.; van der Vleuten, C. Verbal and non-verbal behavior of doctors and patients in primary care consultations - how this relates to patient enablement. Patient Educ. Couns. 2012, 86, 70–76. [Google Scholar] [CrossRef]
- McKinstry, B.; Ashcroft, R.E.; Car, J.; Freeman, G.K.; Sheikh, A. Interventions for improving patients’ trust in doctors and groups of doctors. Cochrane Database Syst. Rev. 2006, CD004134. [Google Scholar] [CrossRef]
- Hudon, C.; St-Cyr Tribble, D.; Bravo, G.; Poitras, M.-E. Enablement in health care context: A concept analysis. J. Eval. Clin. Pract. 2011, 17, 143–149. [Google Scholar] [CrossRef]
- Frost, J.; Currie, M.J.; Cruickshank, M. An Integrative Review of Enablement in Primary Health Care. J. Prim. Care Community Health 2015, 6, 264–278. [Google Scholar] [CrossRef]
- Pawlikowska, T.R.B.; Walker, J.J.; Nowak, P.R.; Szumilo-Grzesik, W. Patient involvement in assessing consultation quality: A quantitative study of the Patient Enablement Instrument in Poland. Health Expect. 2010, 13, 13–23. [Google Scholar] [CrossRef]
- Barr, P.J.; Scholl, I.; Bravo, P.; Faber, M.J.; Elwyn, G.; McAllister, M. Assessment of patient empowerment—A systematic review of measures. PLoS ONE 2015, 10, e0126553. [Google Scholar] [CrossRef]
- Castro, E.M.; van Regenmortel, T.; Vanhaecht, K.; Sermeus, W.; van Hecke, A. Patient empowerment, patient participation and patient-centeredness in hospital care: A concept analysis based on a literature review. Patient Educ. Couns. 2016, 99, 1923–1939. [Google Scholar] [CrossRef]
- Hibbard, J.H.; Mahoney, E.R.; Stockard, J.; Tusler, M. Development and testing of a short form of the patient activation measure. Health Serv. Res. 2005, 40, 1918–1930. [Google Scholar] [CrossRef]
- Brenk-Franz, K.; Hibbard, J.H.; Herrmann, W.J.; Freund, T.; Szecsenyi, J.; Djalali, S.; Steurer-Stey, C.; Sönnichsen, A.; Tiesler, F.; Storch, M.; et al. Validation of the German version of the patient activation measure 13 (PAM13-D) in an international multicentre study of primary care patients. PLoS ONE 2013, 8, e74786. [Google Scholar] [CrossRef] [PubMed]
- Rademakers, J.; Maindal, H.T.; Steinsbekk, A.; Gensichen, J.; Brenk-Franz, K.; Hendriks, M. Patient activation in Europe: An international comparison of psychometric properties and patients’ scores on the short form Patient Activation Measure (PAM-13). BMC Health Serv. Res. 2016, 16, 570. [Google Scholar] [CrossRef]
- Hibbard, J.H.; Stockard, J.; Mahoney, E.R.; Tusler, M. Development of the Patient Activation Measure (PAM): Conceptualizing and measuring activation in patients and consumers. Health Serv. Res. 2004, 39, 1005–1026. [Google Scholar] [CrossRef]
- Siegel, A.; Niebling, W. Individueller Patientennutzen im „Gesunden Kinzigtal“ – Zwischenergebnisse einer Trendstudie. Z. Evidenz Fortbild. Qual. Gesundh. 2018, 130, 35–41. [Google Scholar] [CrossRef]
- Howie, J.G.; Heaney, D.J.; Maxwell, M. Measuring quality in general practice. Pilot study of a needs, process and outcome measure. Occas. Pap. R. Coll. Gen. Pract. 1997, 75, 1–32. [Google Scholar]
- Enthoven, P.; Peolsson, A.; Ludvigsson, M.L.; Wibault, J.; Peterson, G.; Öberg, B. Validity, Internal Consistency and Self-Rated Change of the Patient Enablement Instrument in Patients with Chronic Musculoskeletal Pain. J. Rehabil. Med. 2019, 51, 587–597. [Google Scholar] [CrossRef]
- Hudon, C.; Fortin, M.; Rossignol, F.; Bernier, S.; Poitras, M.-E. The Patient Enablement Instrument-French version in a family practice setting: A reliability study. BMC Fam. Pract. 2011, 12, 71. [Google Scholar] [CrossRef]
- Remelhe, M.; Teixeira, P.M.; Lopes, I.; Silva, L.; Correia de Sousa, J. The modified patient enablement instrument: A Portuguese cross-cultural adaptation, validity and reliability study. NPJ Prim. Care Respir. Med. 2017, 27, 16087. [Google Scholar] [CrossRef]
- Rööst, M.; Zielinski, A.; Petersson, C.; Strandberg, E.L. Reliability and applicability of the Patient Enablement Instrument (PEI) in a Swedish general practice setting. BMC Fam. Pract. 2015, 16, 31. [Google Scholar] [CrossRef][Green Version]
- Lam, C.L.K.; Yuen, N.Y.K.; Mercer, S.W.; Wong, W. A pilot study on the validity and reliability of the Patient Enablement Instrument (PEI) in a Chinese population. Fam. Pract. 2010, 27, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Pawlikowska, T.R.B.; Nowak, P.R.; Szumilo-Grzesik, W.; Walker, J.J. Primary care reform: A pilot study to test the evaluative potential of the Patient Enablement Instrument in Poland. Fam. Pract. 2002, 19, 197–201. [Google Scholar] [CrossRef][Green Version]
- Groene, O. Patient centredness and quality improvement efforts in hospitals: Rationale, measurement, implementation. Int. J. Qual. Health Care 2011, 23, 531–537. [Google Scholar] [CrossRef]
- Schwarzer, R.; Jerusalem, M. Skalen zur Erfassung von Lehrer-und Schülermerkmalen. Dokumentation der Psychometrischen Verfahren im Rahmen der Wissenschaftlichen Begleitung des Modellversuchs Selbstwirksame Schulen; R. Schwarzer: Berlin, Germany, 1999. [Google Scholar]
- Hildebrandt, H.; Hermann, C.; Knittel, R.; Richter-Reichhelm, M.; Siegel, A.; Witzenrath, W. Gesundes Kinzigtal Integrated Care: Improving population health by a shared health gain approach and a shared savings contract. Int. J. Integr. Care 2010, 10, e046. [Google Scholar] [CrossRef]
- Hildebrandt, H.; Schulte, T.; Stunder, B. Triple Aim in Kinzigtal: Improving Population Health, integrating health care and reducing costs of care - lessons for the UK? J. Integr. Care 2012, 20, 205–222. [Google Scholar] [CrossRef]
- The Euro Qol Group. Euro Qol a new facility for the measurement of health-related quality of life. Health Policy 1990, 16, 199–208. [Google Scholar] [CrossRef]
- Mokkink, L.B.; Terwee, C.B.; Patrick, D.L.; Alonso, J.; Stratford, P.W.; Knol, D.L.; Bouter, L.M.; de Vet, H.C.W. The COSMIN study reached international consensus on taxonomy, terminology, and definitions of measurement properties for health-related patient-reported outcomes. J. Clin. Epidemiol. 2010, 63, 737–745. [Google Scholar] [CrossRef]
- Mokkink, L.B.; Terwee, C.B.; Patrick, D.L.; Alonso, J.; Stratford, P.W.; Knol, D.L.; Bouter, L.M.; de Vet, H.C.W. The COSMIN checklist for assessing the methodological quality of studies on measurement properties of health status measurement instruments: An international Delphi study. Qual. Life Res. 2010, 19, 539–549. [Google Scholar] [CrossRef]
- Schwarzer, R.; Jerusalem, M. Self-Efficacy Measurement: Generalized Self-Efficacy Scale (GSES). In Measures in Health Psychology. A User’s Portfolio; Weinman, J., Wright, S., Johnston, M., Eds.; NFER-NELSON: Windsor, UK, 1995; pp. 35–37. [Google Scholar]
- Scholz, U.; Gutiérrez Doña, B.; Sud, S.; Schwarzer, R. Is General Self-Efficacy a Universal Construct? Psychometric Findings from 25 Countries. Eur. J. Psychol. Assess. 2002, 18, 242–251. [Google Scholar] [CrossRef]
- Röthlin, F.; Ganahl, K.; Nowak, P. Empfehlungen für eine international vergleichbare Erhebung von Gesundheitskompetenz (Hls-Neu) in Österreich. Diskussionspapier und Entscheidungshilfe; Fonds Gesundes Österreich: Vienna, Austria, 2017; Available online: https://jasmin.goeg.at/150/1/Empfehlungen_Erhebung%20Gesundheitskompetenz.pdf (accessed on 26 April 2018).
- Jordan, S.; Hoebel, J. Gesundheitskompetenz von Erwachsenen in Deutschland: Ergebnisse der Studie “Gesundheit in Deutschland aktuell” (GEDA). Bundesgesundheitsblatt Gesundh. Gesundh. 2015, 58, 942–950. [Google Scholar] [CrossRef] [PubMed]
- Mokkink, L.B.; Terwee, C.B.; Knol, D.L.; Stratford, P.W.; Alonso, J.; Patrick, D.L.; Bouter, L.M.; de Vet, H.C. The COSMIN checklist for evaluating the methodological quality of studies on measurement properties: A clarification of its content. BMC Med. Res. Methodol. 2010, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Rosenkoetter, U.; Tate, R.L. Assessing Features of Psychometric Assessment Instruments: A Comparison of the COSMIN Checklist with Other Critical Appraisal Tools. Brain Impair. 2018, 19, 103–118. [Google Scholar] [CrossRef]
- Tabachnick, B.G.; Fidell, L.S. Using Multivariate Statistics, 3rd ed.; Harper-Collins College Publishers: New York, NY, USA, 1996. [Google Scholar]
- Nunnally, J.C.; Bernstein, I.H. Psychometric Theory (McGraw-Hill Series in Psychology); McGraw-Hill: New York, NY, USA, 1994. [Google Scholar]
- Bentler, P.M. Comparative Fit Indexes in structural models. Psychol. Bull. 1990, 107, 238–246. [Google Scholar] [CrossRef]
- Byrne, B.M. Structural Equation Modeling with EQS Basic Concepts, Applications, and Programming, 2nd ed.; Taylor and Francis: New York, NY, USA, 2006. [Google Scholar]
- Bollen, K.A. Structural Equations with Latent Variables, 6th ed.; Wiley: New York, NY, USA, 1989. [Google Scholar]
- Moosbrugger, H.; Kelava, A. Testtheorie und Fragebogenkonstruktion, 2nd ed.; Springer: Berlin/Heidelberg, Germny, 2012. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Taylor and Francis: Hoboken, NJ, USA, 1988. [Google Scholar]
- Mead, N.; Bower, P.; Hann, M. The impact of general practitioners’ patient-centredness on patients’ post-consultation satisfaction and enablement. Soc. Sci. Med. 2002, 55, 283–299. [Google Scholar] [CrossRef]
- Smith, S.G.; Curtis, L.M.; Wardle, J.; von Wagner, C.; Wolf, M.S. Skill set or mind set? Associations between health literacy, patient activation and health. PLoS ONE 2013, 8, e74373. [Google Scholar] [CrossRef]
- Berkman, N.D.; Sheridan, S.L.; Donahue, K.E.; Halpern, D.J.; Crotty, K. Low health literacy and health outcomes: An updated systematic review. Ann. Intern. Med. 2011, 155, 97–107. [Google Scholar] [CrossRef]
- Magnezi, R.; Glasser, S. Psychometric properties of the hebrew translation of the patient activation measure (PAM-13). PLoS ONE 2014, 9, e113391. [Google Scholar] [CrossRef]
- Graffigna, G.; Barello, S.; Bonanomi, A.; Lozza, E.; Hibbard, J. Measuring patient activation in Italy: Translation, adaptation and validation of the Italian version of the patient activation measure 13 (PAM13-I). BMC Med. Inform. Decis. Mak. 2015, 15, 109. [Google Scholar] [CrossRef]
- Ahn, Y.-H.; Yi, C.-H.; Ham, O.-K.; Kim, B.-J. Psychometric properties of the Korean version of the “Patient Activation Measure 13”(PAM13-K) in patients with osteoarthritis. Eval. Health Prof. 2015, 38, 254–264. [Google Scholar] [CrossRef]
- Prey, J.E.; Qian, M.; Restaino, S.; Hibbard, J.; Bakken, S.; Schnall, R.; Rothenberg, G.; Vawdrey, D.K.; Creber, R.M. Reliability and validity of the patient activation measure in hospitalized patients. Patient Educ. Couns. 2016, 99, 2026–2033. [Google Scholar] [CrossRef] [PubMed]
- Yayla, K.; Caylan, A.; Oztora, S.; Çeçen, C.E.; Yılmaz, A.C.; Dağdeviren, H.N. Reliability Analysis Of The Turkish Version Of The Patient Activation Measure. Eurasian J. Fam. Med. 2016, 5, 20–24. [Google Scholar]
- Ngooi, B.X.; Packer, T.L.; Kephart, G.; Warner, G.; Koh, K.W.L.; Wong, R.C.C.; Lim, S.P. Validation of the Patient Activation Measure (PAM-13) among adults with cardiac conditions in Singapore. Qual. Life Res. 2017, 26, 1071–1080. [Google Scholar] [CrossRef] [PubMed]
- Kosar, C.; Besen, D.B. Adaptation of a patient activatıon measure (PAM) into Turkish: Reliability and validity test. Afr. Health Sci. 2019, 19, 1811–1820. [Google Scholar] [CrossRef]
- Sørensen, K.; van den Broucke, S.; Pelikan, J.M.; Fullam, J.; Doyle, G.; Slonska, Z.; Kondilis, B.; Stoffels, V.; Osborne, R.H.; Brand, H. Measuring health literacy in populations: Illuminating the design and development process of the European Health Literacy Survey Questionnaire (HLS-EU-Q). BMC Public Health 2013, 13, 948. [Google Scholar] [CrossRef] [PubMed]
- Groene, O.; Bolíbar, I.; Brotons, C. Impact, barriers and facilitators of the ‘Ask Me 3’ Patient Communication Intervention in a primary care center in Barcelona, Spain: A mixed-methods analysis. Int. J. Pers. Cent. Med. 2012, 2, 853–861. [Google Scholar]
- Ožvačić Adžić, Z.; Katić, M.; Kern, J.; Lazić, Đ.; Nekić, V.C.; Soldo, D. Patient, Physician, and Practice Characteristics Related to Patient Enablement in General Practice in Croatia: Cross-sectional Survey Study. Croat. Med. J. 2008, 49, 813–823. [Google Scholar] [CrossRef]
- Mercer, S.W.; Jani, B.D.; Maxwell, M.; Wong, S.Y.S.; Watt, G.C.M. Patient enablement requires physician empathy: A cross-sectional study of general practice consultations in areas of high and low socioeconomic deprivation in Scotland. BMC Fam. Pract. 2012, 13, 6. [Google Scholar] [CrossRef]
- Weenink, J.-W.; Braspenning, J.; Wensing, M. Patient reported outcome measures (PROMs) in primary care: An observational pilot study of seven generic instruments. BMC Fam. Pract. 2014, 15, 88. [Google Scholar] [CrossRef]
- Mead, N.; Bower, P.; Roland, M. Factors associated with enablement in general practice: Cross-sectional study using routinely-collected data. Br. J. Gen. Pract. 2008, 58, 346–352. [Google Scholar] [CrossRef]
- Ernstmann, N.; Wirtz, M.; Nitzsche, A.; Gross, S.E.; Ansmann, L.; Gloede, T.D.; Jung, J.; Pfaff, H.; Baumann, W.; Schmitz, S.; et al. Patients’ Trust in Physician, Patient Enablement, and Health-Related Quality of Life During Colon Cancer Treatment. J. Cancer Educ. 2017, 32, 571–579. [Google Scholar] [CrossRef] [PubMed]
| Total | Calibration Sample | Validation Sample | ||||
|---|---|---|---|---|---|---|
| N = 1168 | n = 584 | n = 584 | ||||
| n | % | n | % | n | % | |
| Gender | ||||||
| Male | 506 | 43.3 | 255 | 43.7 | 251 | 43.0 |
| Female | 662 | 56.7 | 329 | 56.3 | 333 | 57.0 |
| Age (Years) | ||||||
| Mean (Standard Deviation) | 62.3 (15.9) | 61.65 (15.8) | 63.00 (16.0) | |||
| Range | 19–95 | 19–92 | 19–95 | |||
| Chronic Disease | ||||||
| Yes | 650 | 55.7 | 327 | 56.0 | 323 | 55.3 |
| No | 412 | 35.3 | 212 | 36.3 | 200 | 34.2 |
| Don’t know | 70 | 6.0 | 25 | 4.3 | 45 | 7.7 |
| Missing | 36 | 3.1 | 20 | 3.4 | 16 | 2.7 |
| Education level | ||||||
| No school leaving certificate | 19 | 1.6 | 11 | 1.9 | 8 | 1.4 |
| Secondary school certificate | 735 | 62.9 | 363 | 62.2 | 372 | 63.7 |
| Intermediate maturity | 255 | 21.8 | 130 | 22.3 | 125 | 21.4 |
| Polytechnic secondary school | 11 | 0.9 | 5 | 0.9 | 6 | 1.0 |
| Advanced technical college certificate | 58 | 5.0 | 30 | 5.1 | 28 | 4.8 |
| Abitur (a-level) | 45 | 3.9 | 19 | 3.3 | 26 | 4.5 |
| Missing | 45 | 3.9 | 26 | 4.5 | 19 | 3.3 |
| Employment status | ||||||
| Currently employed | 497 | 42.6 | 256 | 43.8 | 241 | 41.3 |
| Currently not employed | 587 | 50.3 | 292 | 50.0 | 295 | 50.5 |
| Missing | 84 | 7.1 | 36 | 6.2 | 48 | 8.2 |
| Total | Calibration Sample | Validation Sample | ||||
|---|---|---|---|---|---|---|
| n | Mean (SD) | n | Mean (SD) | n | Mean (SD) | |
| EQ-5D Index | 1090 | 0.84 (0.20) | 544 | 0.84 (0.20) | 546 | 0.83 (0.20) |
| EQ-VAS | 1052 | 68.69 (20.30) | 527 | 69.11 (19.92) | 525 | 68.27 (20.69) |
| HLS-EU-Q16 | - | - | 126 | 12.19 (4.18) | ||
| GSE scale | - | - | 162 | 29.01 (5.58) | ||
| Item 1 | Factor Loading Factor 1 | Factor Loading Factor 2 | Mean (SD) 2 | Missing Values n (%) 3 | |
|---|---|---|---|---|---|
| 1 | I know how I can promote my health. | 0.70 | 0.16 | 4.12 (0.86) | 51 (4.5) |
| 2 | It is easy for me to practice health-promoting behavior in everyday life (e.g., nutrition, exercise). | 0.71 | 0.01 | 3.72 (0.94) | 49 (4.2) |
| 3 | I am well informed regarding my health condition. | 0.59 | 0.36 | 4.20 (0.87) | 73 (6.3) |
| 4 | I am able to cope with my health problems. | 0.74 | 0.30 | 4.01 (0.88) | 58 (5.0) |
| 5 | I know various treatment possibilities for my diseases. | 0.66 | 0.24 | 3.71 (1.0) | 113 (9.7) |
| 6 | I am able to prevent a deterioration of my health condition as much as this is possible. | 0.75 | 0.22 | 3.83 (0.94) | 79 (6.8) |
| 7 | I know when to seek medical or therapeutic help, or when I can deal with my complaints on my own. | 0.63 | 0.45 | 3.99 (0.91) | 56 (4.8) |
| 8 | I am able to get medical or therapeutic help when I need it. | 0.50 | 0.63 | 4.26 (0.88) | 50 (4.3) |
| 9 | I have no difficulty in telling my doctor about my concerns and fears, even if he or she does not address them directly. | 0.18 | 0.90 | 4.12 (0.98) | 37 (3.2) |
| 10 | It is easy for me to ask my questions or express my wishes during a medical consultation. | 0.19 | 0.87 | 4.19 (0.95) | 35 (3.0) |
| 11 | I am convinced that I can practice a healthy lifestyle even in strenuous times. | 0.64 | 0.32 | 3.63 (0.96) | 39 (3.3) |
| 12 | In general, I am coping well with life. | 0.63 | 0.37 | 4.19 (0.86) | 26 (2.2) |
| 13 | On the whole, I am able to look after myself. | 0.65 | 0.34 | 4.04 (0.94) | 30 (2.6) |
| Item_1 | Item_2 | Item_3 | Item_4 | Item_5 | Item_6 | Item_7 | Item_8 | Item_9 | Item_10 | Item_11 | Item_12 | Item_13 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Item_1 | 1 | 0.506 | 0.462 | 0.479 | 0.521 | 0.482 | 0.501 | 0.429 | 0.316 | 0.358 | 0.426 | 0.412 | 0.443 |
| Item_2 | 0.506 | 1 | 0.361 | 0.395 | 0.377 | 0.449 | 0.397 | 0.327 | 0.242 | 0.274 | 0.524 | 0.378 | 0.372 |
| Item_3 | 0.462 | 0.361 | 1 | 0.622 | 0.539 | 0.512 | 0.513 | 0.517 | 0.438 | 0.447 | 0.412 | 0.368 | 0.357 |
| Item_4 | 0.479 | 0.395 | 0.622 | 1 | 0.552 | 0.619 | 0.583 | 0.54 | 0.406 | 0.411 | 0.494 | 0.553 | 0.564 |
| Item_5 | 0.521 | 0.377 | 0.539 | 0.552 | 1 | 0.556 | 0.523 | 0.445 | 0.388 | 0.367 | 0.428 | 0.378 | 0.423 |
| Item_6 | 0.482 | 0.449 | 0.512 | 0.619 | 0.556 | 1 | 0.636 | 0.52 | 0.375 | 0.345 | 0.517 | 0.492 | 0.528 |
| Item_7 | 0.501 | 0.397 | 0.513 | 0.583 | 0.523 | 0.636 | 1 | 0.66 | 0.475 | 0.444 | 0.458 | 0.484 | 0.539 |
| Item_8 | 0.429 | 0.327 | 0.517 | 0.54 | 0.445 | 0.52 | 0.66 | 1 | 0.586 | 0.55 | 0.437 | 0.536 | 0.562 |
| Item_9 | 0.316 | 0.242 | 0.438 | 0.406 | 0.388 | 0.375 | 0.475 | 0.586 | 1 | 0.775 | 0.423 | 0.423 | 0.382 |
| Item_10 | 0.358 | 0.274 | 0.447 | 0.411 | 0.367 | 0.345 | 0.444 | 0.55 | 0.775 | 1 | 0.457 | 0.41 | 0.391 |
| Item_11 | 0.426 | 0.524 | 0.412 | 0.494 | 0.428 | 0.517 | 0.458 | 0.437 | 0.423 | 0.457 | 1 | 0.558 | 0.508 |
| Item_12 | 0.412 | 0.378 | 0.368 | 0.553 | 0.378 | 0.492 | 0.484 | 0.536 | 0.423 | 0.41 | 0.558 | 1 | 0.732 |
| Item_13 | 0.443 | 0.372 | 0.357 | 0.564 | 0.423 | 0.528 | 0.539 | 0.562 | 0.382 | 0.391 | 0.508 | 0.732 | 1 |
| Item_1 | Item_2 | Item_3 | Item_4 | Item_5 | Item_6 | Item_7 | Item_8 | Item_9 | Item_10 | Item_11 | Item_12 | Item_13 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.619 | 0.527 | 0.645 | 0.728 | 0.637 | 0.704 | 0.728 | 0.715 | 0.606 | 0.607 | 0.657 | 0.666 | 0.673 |
| Criterion | Two-Factor Model Without Adjustment | Two-Factor Model with Two Additional Correlations 1 | One-Factor Model | One-Factor Model with Two Additional Correlations 1 |
|---|---|---|---|---|
| Chi2 | 768.357 | 443.723 | 992.045 | 495.927 |
| p-value of Chi2 | <0.001 | <0.001 | <0.001 | <0.001 |
| Df | 64 | 62 | 65 | 63 |
| Chi2/df | 12.006 | 7.157 | 15.262 | 7.872 |
| Comparative Fit Index (CFI) | 0.821 | 0.903 | 0.765 | 0.890 |
| Tucker Lewis Index (TLI) | 0.782 | 0.878 | 0.718 | 0.864 |
| Root Mean Square Error of Approximation (RMSEA) (90% CI) | 0.137 (0.129–0.146) | 0.103 (0.094–0.112) | 0.156 (0.148–0.165) | 0.109 (0.100–0.118) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siegel, A.; Ehmann, A.T.; Meyer, I.; Gröne, O.; Niebling, W.; Martus, P.; Rieger, M.A. PEN-13: A New Generic 13-Item Questionnaire for Measuring Patient Enablement (German Version). Int. J. Environ. Res. Public Health 2019, 16, 4867. https://doi.org/10.3390/ijerph16234867
Siegel A, Ehmann AT, Meyer I, Gröne O, Niebling W, Martus P, Rieger MA. PEN-13: A New Generic 13-Item Questionnaire for Measuring Patient Enablement (German Version). International Journal of Environmental Research and Public Health. 2019; 16(23):4867. https://doi.org/10.3390/ijerph16234867
Chicago/Turabian StyleSiegel, Achim, Anna T. Ehmann, Ingo Meyer, Oliver Gröne, Wilhelm Niebling, Peter Martus, and Monika A. Rieger. 2019. "PEN-13: A New Generic 13-Item Questionnaire for Measuring Patient Enablement (German Version)" International Journal of Environmental Research and Public Health 16, no. 23: 4867. https://doi.org/10.3390/ijerph16234867
APA StyleSiegel, A., Ehmann, A. T., Meyer, I., Gröne, O., Niebling, W., Martus, P., & Rieger, M. A. (2019). PEN-13: A New Generic 13-Item Questionnaire for Measuring Patient Enablement (German Version). International Journal of Environmental Research and Public Health, 16(23), 4867. https://doi.org/10.3390/ijerph16234867






