The Sustainability Challenge of Food and Environmental Nanotechnology: Current Status and Imminent Perceptions
Abstract
1. Introduction
2. Nanotechnology and Its Potential Applications in Food Science Technology, Environment, and Human Health
2.1. Nanomaterials in the Food Processing Sector
2.2. Nanomaterials in Food Contact Surfaces
2.3. Nanotechnology in Quality and Safety Management of Food
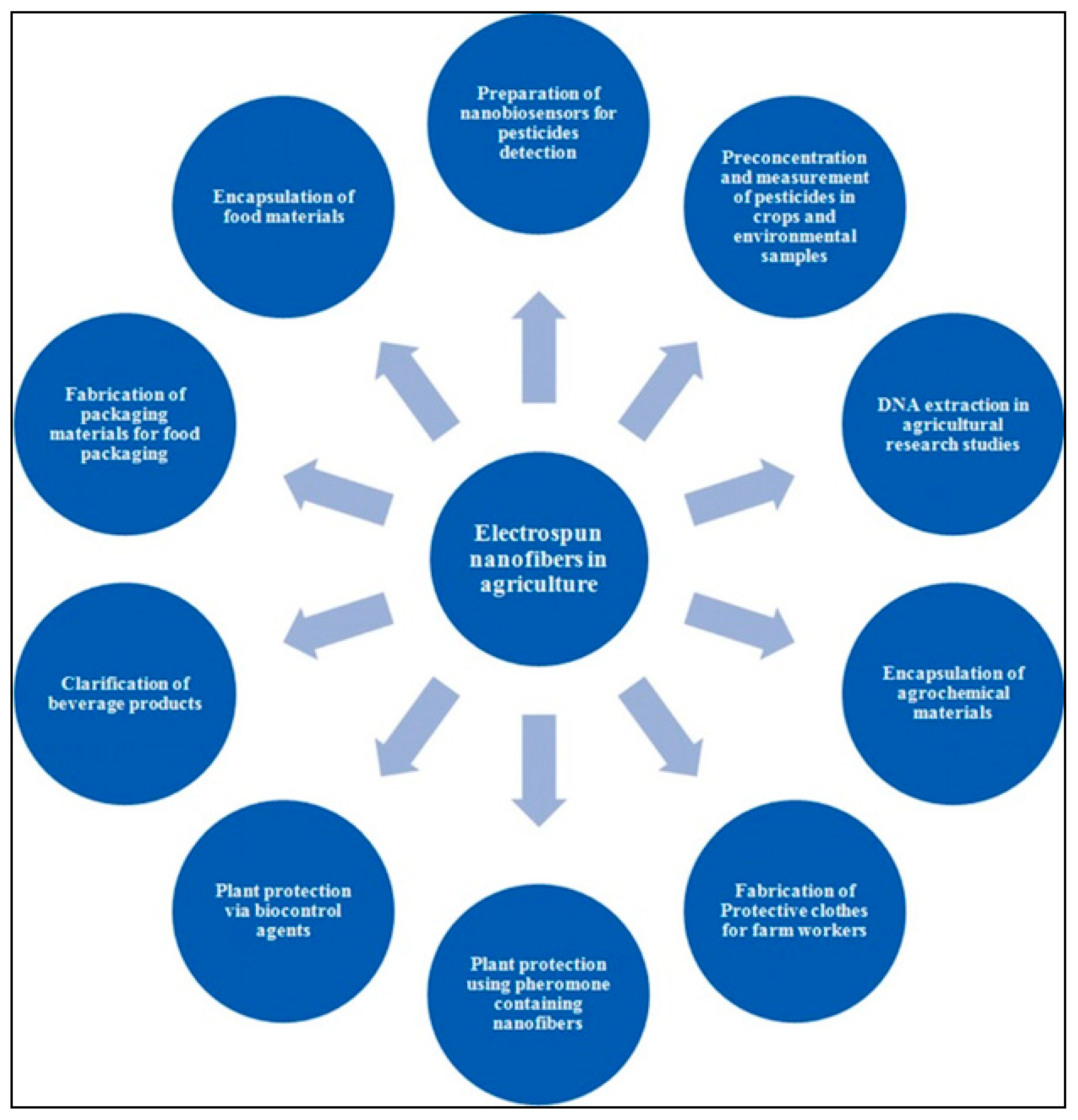
2.4. Electrospun Nanofiber of Food Interest
2.5. Antimicrobial-Rich Nanoparticles in the Food Sector
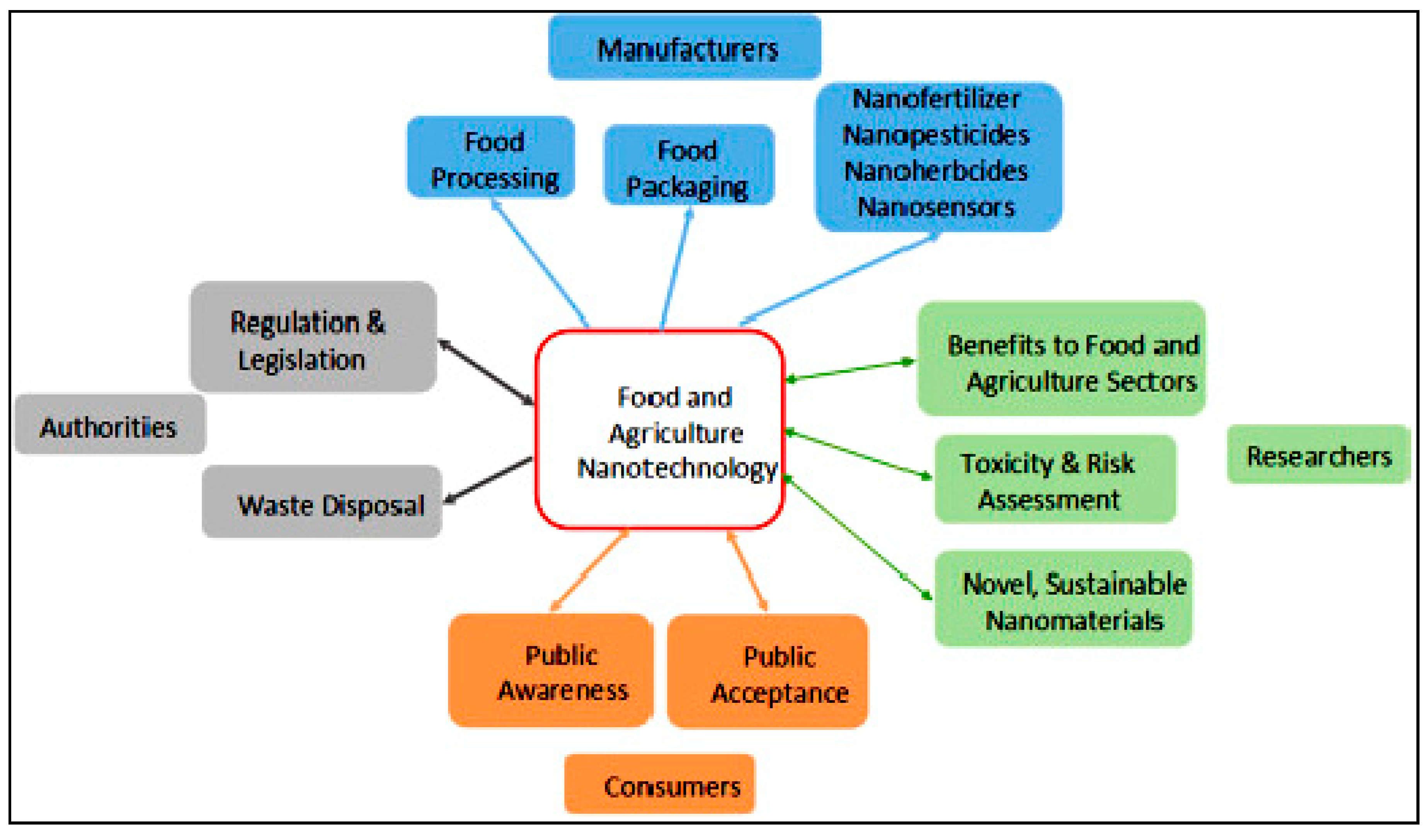
3. Sustainability of Food Nanotechnology
3.1. Nano-Based Sensors
3.2. Nano-Based Control of Pathogens
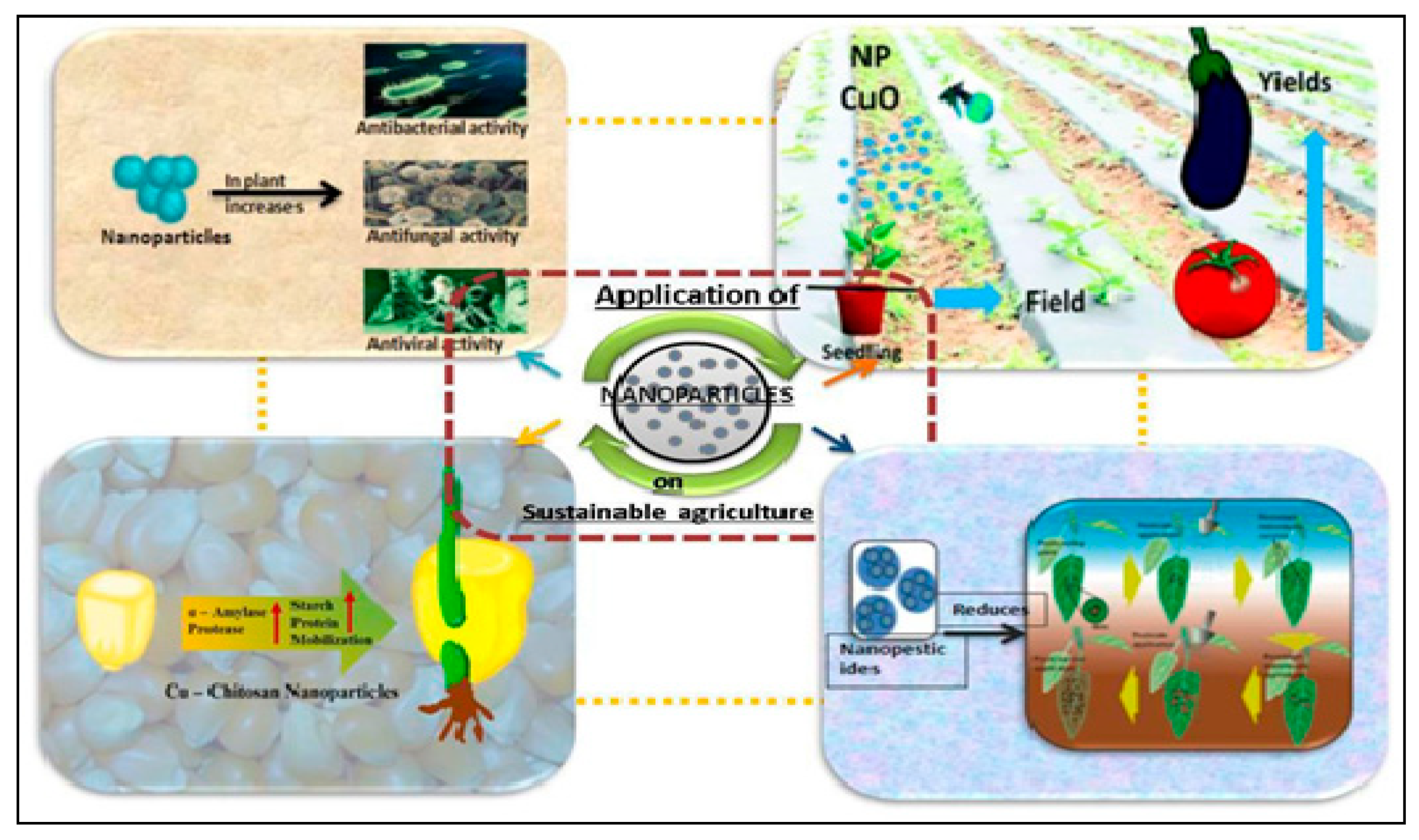
3.3. Nano-Based Fertilizers
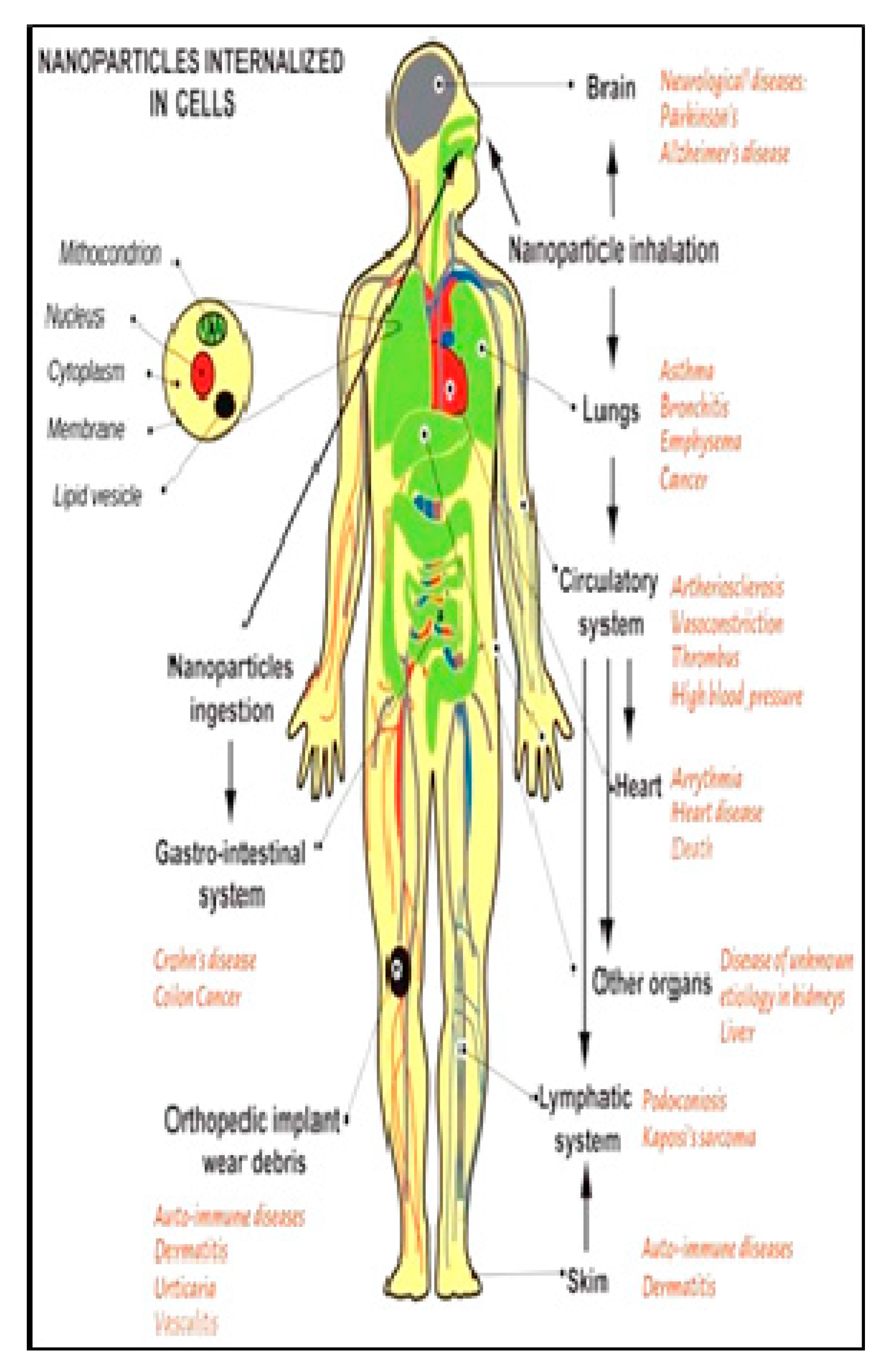
3.4. Nanoparticle Toxicity
3.5. Operational Approaches
3.6. Measurement Issues
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Peters, R.J.B.; Bouwmeester, H.; Gottardo, S.; Amenta, V.; Arena, M.; Brandhoff, P.; Marvin, H.J.P.; Mech, A.; Moniz, F.B.; Pesudo, L.Q.; et al. Nanomaterials for products and application in agriculture, feed and food. Trends Food Sci. Technol. 2016, 54, 155–164. [Google Scholar] [CrossRef]
- King, T.; Osmond-McLeod, M.J.; Duffy, L.L. Nanotechnology in the food sector and potential applications for the poultry industry. Trends Food Sci. Technol. 2018, 72, 62–73. [Google Scholar] [CrossRef]
- Dudefoi, W.; Villares, A.; Peyron, S.; Moreau, C.; Ropers, M.-H.; Gontard, N.; Cathala, B. Nanoscience and nanotechnologies for biobased materials, packaging and food applications: New opportunities and concerns. Innov. Food Sci. Emerg. Technol. 2018, 46, 107–121. [Google Scholar] [CrossRef]
- Chau, C.-F.; Wu, S.-H.; Yen, G.-C. The development of regulations for food nanotechnology. Trends Food Sci. Technol. 2007, 18, 269–280. [Google Scholar] [CrossRef]
- He, X.; Deng, H.; Hwang, H.-M. The current application of nanotechnology in food and agriculture. J. Food Drug Anal. 2019, 27, 1–21. [Google Scholar] [CrossRef]
- Yu, H.; Park, J.-Y.; Kwon, C.W.; Hong, S.-C.; Park, K.-M.; Chang, P.-S. An Overview of Nanotechnology in Food Science: Preparative Methods, Practical Applications, and Safety. J. Chem. 2018, 2018, 10. [Google Scholar] [CrossRef]
- Dwivedi, C.; Pandey, I.; Misra, V.; Giulbudagian, M.; Jungnickel, H.; Laux, P.; Luch, A.; Ramteke, P.; Singh, A. The prospective role of nanobiotechnology in food and food packaging products. Integr. Food Nutr. Metab. IFNM 2018, 5, 1–5. [Google Scholar] [CrossRef]
- Code of Federal Regulations (CFR). Electronic Code of Federal Regulations. Title 21: Food and Drugs. Part 73—Listing of Color Additives Exempt from Certification; The United States Office of the Federal Register (OFR) and The United States Government Publishing Office: Washington, DC, USA, 2018. [Google Scholar]
- U.S. FDA. Color Additive Status List; United States Food & Drug Administration: Washington, DC, USA, 2015. [Google Scholar]
- The European Commission. Commission Regulation (EU) No 10/2011 of 14 January 2011 on plastic materials and articles intended to come into contact with food Text with EEA relevance. Off. J. Eur. Union 2011, 12, 1–89. [Google Scholar]
- U.S. FDA. Inventory of Effective Food Contact Substance (FCS) Notifications; Administration USFaD: Washington, DC, USA, 2018. Available online: https://www.accessdata.fda.gov/scripts/fdcc/?set¼FCN (accessed on 8 August 2018).
- European Commision. Regulation (EC) No. 1333/2008 of the European Parliament and of the Council of 16 December 2008 on Food Additives. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri¼celex%3A32008R1333 (accessed on 16 December 2008).
- Code of Federal Regulations (CFR). Title 21–food and drugs. Chapter i–food and drug administration. Department of health and human services. Subchapter B–food for human consumption (continued). Part 172–food additives permitted for direct addition to food for human consumption. Subpart E–Anticakingagents. Sec. 172.480 Silicon Dioxide. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfCFR/CFRSearch.cfm?fr¼172.480 (accessed on 8 August 2018).
- U.S. FDA. Food Additive Status List; US FDA/CFSAN Office of Food Additive Safety: Washington, DC, USA, 2018. Available online: https://www.fda.gov/Food/IngredientsPackagingLabeling/FoodAdditivesIngredients/ucm091048.htm (accessed on 7 August 2018).
- Sahoo, D.; Mandal, A.; Mitra, T.; Chakraborty, K.; Bardhan, M.; Dasgupta, A.K. Nanosensing of Pesticides by Zinc Oxide Quantum Dot: An Optical and Electrochemical Approach for the Detection of Pesticides in Water. J. Agric. Food Chem. 2018, 66, 414–423. [Google Scholar] [CrossRef]
- Sun, Y.; Fang, L.; Wan, Y.; Gu, Z. Pathogenic detection and phenotype using magnetic nanoparticle-urease nanosensor. Sens. Actuators B Chem. 2018, 259, 428–432. [Google Scholar] [CrossRef]
- Kearns, H.; Goodacre, R.; Jamieson, L.E.; Graham, D.; Faulds, K. SERS detection of multiple antimicrobial-resistant pathogens using nanosensors. Anal. Chem. 2017, 89, 12666–12673. [Google Scholar] [CrossRef] [PubMed]
- Perçin, I.; Idil, N.; Bakhshpour, M.; Yılmaz, E.; Mattiasson, B.; Denizli, A. Microcontact imprinted plasmonic nanosensors: Powerful tools in the detection of Salmonella paratyphi. Sensors 2017, 17, 1375. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, T.; Sulthana, S.; Shelby, T.; Heckert, B.; Jewell, J.; Woody, K.; Karimnia, V.; McAfee, J.; Santra, S. Multiparametric magneto-fluorescent nanosensors for the ultrasensitive detection of Escherichia coli O157: H7. ACS Infect. Dis. 2016, 2, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Sun, A.; Chai, J.; Xiao, T.; Shi, X.; Li, X.; Zhao, Q.; Li, D.; Chen, J. Development of a selective fluorescence nanosensor based on molecularly imprinted-quantum dot optosensing materials for saxitoxin detection in shellfish samples. Sens. Actuators B Chem. 2018, 258, 408–414. [Google Scholar] [CrossRef]
- Zhang, C.-H.; Liu, L.-W.; Liang, P.; Tang, L.-J.; Yu, R.-Q.; Jiang, J.-H. Plasmon coupling enhanced raman scattering nanobeacon for single-step, ultrasensitive detection of cholera toxin. Anal. Chem. 2016, 88, 7447–7452. [Google Scholar] [CrossRef]
- Zhang, W.; Han, Y.; Chen, X.; Luo, X.; Wang, J.; Yue, T.; Li, Z. Surface molecularly imprinted polymer capped Mn-doped ZnS quantum dots as a phosphorescent nanosensor for detecting patulin in apple juice. Food Chem. 2017, 232, 145–154. [Google Scholar] [CrossRef]
- Shi, S.; Wang, W.; Liu, L.; Wu, S.; Wei, Y.; Li, W. Effect of chitosan/nano-silica coating on the physicochemical characteristics of longan fruit under ambient temperature. J. Food Eng. 2013, 118, 125–131. [Google Scholar] [CrossRef]
- Zambrano-Zaragoza, M.; Mercado-Silva, E.; Gutiérrez-Cortez, E.; Cornejo-Villegas, M.; Quintanar-Guerrero, D. The effect of nano-coatings with α-tocopherol and xanthan gum on shelf-life and browning index of fresh-cut “Red Delicious” apples. Innov. Food Sci. Emerg. Technol. 2014, 22, 188–196. [Google Scholar] [CrossRef]
- Robledo, N.; López, L.; Bunger, A.; Tapia, C.; Abugoch, L. Effects of antimicrobial edible coating of thymol nanoemulsion/quinoa protein/chitosan on the safety, sensorial properties, and quality of refrigerated strawberries (Fragaria× ananassa) under commercial storage environment. Food Bioprocess Technol. 2018, 11, 1566–1574. [Google Scholar] [CrossRef]
- Gorrasi, G.; Bugatti, V. Edible bio-nano-hybrid coatings for food protection based on pectins and LDH-salicylate: Preparation and analysis of physical properties. LWT Food Sci. Technol. 2016, 69, 139–145. [Google Scholar] [CrossRef]
- Salvia-Trujillo, L.; Rojas-Graü, M.A.; Soliva-Fortuny, R.; Martín-Belloso, O. Use of antimicrobial nanoemulsions as edible coatings: Impact on safety and quality attributes of fresh-cut Fuji apples. Postharvest Biol. Technol. 2015, 105, 8–16. [Google Scholar] [CrossRef]
- Code of Federal Regulations (CFR). Electronic Code of Federal Regulations. Title 21: Food and Drugs. Part 184—Direct Food Substances Affirmed as Generally Recognized as Safe. Subpart b-Listing of Specific Substances Affirmed as Gras; The United States Office of the Federal Register (OFR) and the United States Government Publishing Office: Washington, DC, USA, 2018. Available online: https://www.ecfr.gov/cgi-bin/text-idx?SID¼79a76b1d7e7a98ae9459d88005ab7058&mc¼true&node¼pt21.1.73&rgn¼div5 (accessed on 8 August 2018).
- Augustin, M.A.; Sanguansri, P. Chapter 5 Nanostructured Materials in the Food Industry. In Advances in Food and Nutrition Research; Academic Press: Cambridge, MA, USA, 2009; Volume 58, pp. 183–213. [Google Scholar]
- Weir, A.; Westerhoff, P.; Fabricius, L.; Hristovski, K.; Von Goetz, N. Titanium dioxide nanoparticles in food and personal care products. Environ. Sci. Technol. 2012, 46, 2242–2250. [Google Scholar] [CrossRef] [PubMed]
- Casado, N.; Pérez-Quintanilla, D.; Morante-Zarcero, S.; Sierra, I. Current development and applications of ordered mesoporous silicas and other sol–gel silica-based materials in food sample preparation for xenobiotics analysis. TrAC Trends Anal. Chem. 2017, 88, 167–184. [Google Scholar] [CrossRef]
- France bans Titanium Dioxide in food products by January 2020|Sanitary/Phytosanitary/Food Safety, SP2-Prevent or Resolve Barriers to Trade that Hinder U.S. Food and Agricultural Exports|Paris|France|May 09, 2019|FR2019-2431. Available online: https://apps.fas.usda.gov/newgainapi/api/report/downloadreportbyfilename?filename=France%20bans%20Titanium%20Dioxide%20in%20food%20products%20by%20January%202020_Paris_France_5-3-2019.pdf (accessed on 9 May 2019).
- Assadpour, E.; Mahdi Jafari, S. A systematic review on nanoencapsulation of food bioactive ingredients and nutraceuticals by various nanocarriers. Crit. Rev. Food Sci. Nutr. 2018, 59, 1–23. [Google Scholar] [CrossRef]
- Rodriguez-Ruiz, V.; Salatti-Dorado, J.; Barzegari, A.; Nicolas-Boluda, A.; Houaoui, A.; Caballo, C.; Caballero-Casero, N.; Sicilia, D.; Bastias Venegas, J.; Pauthe, E. Astaxanthin-Loaded Nanostructured Lipid Carriers for Preservation of Antioxidant Activity. Molecules 2018, 23, 2601. [Google Scholar] [CrossRef]
- Singh, H.; Kumar, C.; Singh, N.; Paul, S.; Jain, S.K. Nanoencapsulation of docosahexaenoic acid (DHA) using a combination of food grade polymeric wall materials and its application for improvement in bioavailability and oxidative stability. Food Funct. 2018, 9, 2213–2227. [Google Scholar] [CrossRef]
- Singh, H. Nanotechnology Applications in Functional Foods; Opportunities and Challenges. Prev. Nutr. Food Sci. 2016, 21, 1–8. [Google Scholar] [CrossRef]
- Shishir, M.R.I.; Xie, L.; Sun, C.; Zheng, X.; Chen, W. Advances in micro and nano-encapsulation of bioactive compounds using biopolymer and lipid-based transporters. Trends Food Sci. Technol. 2018, 78, 34–60. [Google Scholar] [CrossRef]
- De Faria, A.F.; Martinez, D.S.T.; Meira, S.M.M.; de Moraes, A.C.M.; Brandelli, A.; Souza Filho, A.G.; Alves, O.L. Anti-adhesion and antibacterial activity of silver nanoparticles supported on graphene oxide sheets. Colloids Surf. B Biointerfaces 2014, 113, 115–124. [Google Scholar] [CrossRef]
- Weng, X.; van Niekerk, J.; Neethirajan, S.; Warriner, K. Characterization of antimicrobial efficacy of photocatalytic polymers against food-borne biofilms. LWT Food Sci. Technol. 2016, 68, 1–7. [Google Scholar] [CrossRef]
- Otoni, C.G.; de Moura, M.R.; Aouada, F.A.; Camilloto, G.P.; Cruz, R.S.; Lorevice, M.V.; de FF Soares, N.; Mattoso, L.H. Antimicrobial and physical-mechanical properties of pectin/papaya puree/cinnamaldehyde nanoemulsion edible composite films. Food Hydrocoll. 2014, 41, 188–194. [Google Scholar] [CrossRef]
- Donsì, F.; Marchese, E.; Maresca, P.; Pataro, G.; Vu, K.D.; Salmieri, S.; Lacroix, M.; Ferrari, G. Green beans preservation by combination of a modified chitosan based-coating containing nanoemulsion of mandarin essential oil with high pressure or pulsed light processing. Postharvest Biol. Technol. 2015, 106, 21–32. [Google Scholar] [CrossRef]
- Ghanbarzadeh, B.; Oleyaei, S.A.; Almasi, H. Nanostructured materials utilized in biopolymer-based plastics for food packaging applications. Crit. Rev. Food Sci. Nutr. 2015, 55, 1699–1723. [Google Scholar] [CrossRef] [PubMed]
- Pollet, J.; Delport, F.; Janssen, K.; Tran, D.; Wouters, J.; Verbiest, T.; Lammertyn, J. Fast and accurate peanut allergen detection with nanobead enhanced optical fiber SPR biosensor. Talanta 2011, 83, 1436–1441. [Google Scholar] [CrossRef]
- He, L.; Haynes, C.L.; Diez-Gonzalez, F.; Labuza, T.P. Rapid detection of a foreign protein in milk using IMS–SERS. J. Raman Spectrosc. 2011, 42, 1428–1434. [Google Scholar] [CrossRef]
- Godoy-Navajas, J.; Caballos, M.A.; Gómez-Hens, A. Heterogeneous immunoassay for soy protein determination using nile blue-doped silica nanoparticles as labels and front-surface long-wavelength fluorimetry. Anal. Chim. Acta 2011, 701, 194–199. [Google Scholar] [CrossRef]
- Goldman, E.R.; Clapp, A.R.; Anderson, G.P.; Uyeda, H.T.; Mauro, J.M.; Medintz, I.L.; Mattoussi, H. Multiplexed toxin analysis using four colors of quantum dot fluororeagents. Anal. Chem. 2004, 76, 684–688. [Google Scholar] [CrossRef]
- Tang, D.; Tang, J.; Su, B.; Chen, G. Gold nanoparticles-decorated amine-terminated poly (amidoamine) dendrimer for sensitive electrochemical immunoassay of brevetoxins in food samples. Biosens. Bioelectron. 2011, 26, 2090–2096. [Google Scholar] [CrossRef]
- Feng, R.; Zhang, Y.; Li, H.; Wu, D.; Xin, X.; Zhang, S.; Yu, H.; Wei, Q.; Du, B. Ultrasensitive electrochemical immunosensor for zeranol detection based on signal amplification strategy of nanoporous gold films and nano-montmorillonite as labels. Anal. Chim. Acta 2013, 758, 72–79. [Google Scholar] [CrossRef]
- Gan, N.; Zhou, J.; Xiong, P.; Hu, F.; Cao, Y.; Li, T.; Jiang, Q. An ultrasensitive electrochemiluminescent immunoassay for Aflatoxin M1 in milk, based on extraction by magnetic graphene and detection by antibody-labeled CdTe quantumn dots-carbon nanotubes nanocomposite. Toxins 2013, 5, 865–883. [Google Scholar] [CrossRef]
- Zhang, S.P.; Shan, L.G.; Tian, Z.R.; Zheng, Y.; Shi, L.Y.; Zhang, D.S. Study of enzyme biosensor based on carbon nanotubes modified electrode for detection of pesticides residue. Chin. Chem. Lett. 2008, 19, 592–594. [Google Scholar] [CrossRef]
- Mcdonnell, G.E.; Fiorello, A.; Smith, D. Indicator Device Having an Active Agent Encapsulated in an Electrospun Nanofiber. U.S. Patent No. 7,569,359, 4 August 2009. [Google Scholar]
- Torres-Giner, S. 5-Electrospun nanofibers for food packaging applications. In Multifunctional and Nanoreinforced Polymers for Food Packaging; Lagarón, J.-M., Ed.; Woodhead Publishing: Cambridge, UK, 2011; pp. 108–125. [Google Scholar]
- Doyle, J.J.; Choudhari, S.; Ramakrishna, S.; Babu, R.P. Electrospun Nanomaterials: Biotechnology, Food, Water, Environment, and Energy. 2013. Available online: http://dx.doi.org/10.1155/2013/269313 (accessed on 8 August 2019).
- Noruzi, M. Electrospun nanofibres in agriculture and the food industry: A review. J. Sci. Food Agric. 2016, 96, 4663–4678. [Google Scholar] [CrossRef] [PubMed]
- Nikmaram, N.; Roohinejad, S.; Hashemi, S.; Koubaa, M.; Barba, F.J.; Abbaspourrad, A.; Greiner, R. Emulsion-based systems for fabrication of electrospun nanofibers: Food, pharmaceutical and biomedical applications. RSC Adv. 2017, 7, 28951–28964. [Google Scholar] [CrossRef]
- Wang, C.; Wang, J.; Zeng, L.; Qiao, Z.; Liu, X.; Liu, H.; Zhang, J.; Ding, J. Fabrication of electrospun polymer nanofibers with diverse morphologies. Molecules 2019, 24, 834. [Google Scholar] [CrossRef]
- Shankar, A.; Seyam, A.-F.; Hudson, S. Electrospinning of soy protein fibers and their compatibility with synthetic polymers. J. Text. Appar. Technol. Manag. 2013, 8, 1–14. [Google Scholar]
- Sullivan, S.T.; Tang, C.; Kennedy, A.; Talwar, S.; Khan, S.A. Electrospinning and heat treatment of whey protein nanofibers. Food Hydrocoll. 2014, 35, 36–50. [Google Scholar] [CrossRef]
- Kriegel, C.; Arrechi, A.; Kit, K.; McClements, D.; Weiss, J. Fabrication, functionalization, and application of electrospun biopolymer nanofibers. Crit. Rev. Food Sci. Nutr. 2008, 48, 775–797. [Google Scholar] [CrossRef]
- Fabra, M.J.; Lopez-Rubio, A.; Lagaron, J.M. High barrier polyhydroxyalcanoate food packaging film by means of nanostructured electrospun interlayers of zein. Food Hydrocoll. 2013, 32, 106–114. [Google Scholar] [CrossRef]
- Kriegel, C.; Kit, K.M.; McClements, D.J.; Weiss, J. Nanofibers as Carrier Systems for Antimicrobial Microemulsions. Part I: Fabrication and Characterization. Langmuir 2009, 25, 1154–1161. [Google Scholar] [CrossRef]
- Alborzi, S. Encapsulation of Folic Acid in Sodium Alginate-Pectin-Poly (Ethylene Oxide) Electrospun Fibers to Increase Its Stability. Ph.D. Thesis, University of Guelph, Guelph, ON, Canada, 2012. [Google Scholar]
- Ghorani, B.; Tucker, N. Fundamentals of electrospinning as a novel delivery vehicle for bioactive compounds in food nanotechnology. Food Hydrocoll. 2015, 51, 227–240. [Google Scholar] [CrossRef]
- Liu, S.-C.; Li, R.; Tomasula, P.M.; Sousa, A.M.; Liu, L. Electrospun food-grade ultrafine fibers from pectin and pullulan blends. Food Nutr. Sci. 2016, 7, 636. [Google Scholar] [CrossRef]
- Malhotra, B.; Keshwani, A.; Kharkwal, H. Antimicrobial food packaging: Potential and pitfalls. Front. Microbiol. 2015, 6, 611. [Google Scholar] [CrossRef] [PubMed]
- Appendini, P.; Hotchkiss, J.H. Review of antimicrobial food packaging. Innov. Food Sci. Emerg. Technol. 2002, 3, 113–126. [Google Scholar] [CrossRef]
- De Azeredo, H.M. Antimicrobial nanostructures in food packaging. Trends Food Sci. Technol. 2013, 30, 56–69. [Google Scholar] [CrossRef]
- Tunç, S.; Duman, O. Preparation of active antimicrobial methyl cellulose/carvacrol/montmorillonite nanocomposite films and investigation of carvacrol release. LWT Food Sci. Technol. 2011, 44, 465–472. [Google Scholar] [CrossRef]
- Dobrucka, R.; Ankiel, M. Possible applications of metal nanoparticles in antimicrobial food packaging. J. Food Saf. 2019, 39, 12617. [Google Scholar] [CrossRef]
- Huang, T.; Qian, Y.; Wei, J.; Zhou, C. Polymeric antimicrobial food packaging and its applications. Polymers 2019, 11, 560. [Google Scholar] [CrossRef]
- Sadeghizadeh-Yazdi, J.; Habibi, M.; Kamali, A.A.; Banaei, M. Application of Edible and Biodegradable Starch-Based Films in Food Packaging: A Systematic Review and Meta-Analysis. Curr. Res. Nutr. Food Sci. J. 2019, 7. Available online: https://www.foodandnutritionjournal.org/volume7number3/application-of-edible-and-biodegradable-starch-based-films-in-food-packaging-a-systematic-review-and-meta-analysis/ (accessed on 8 August 2019).
- Sofi, S.; Singh, J.; Rafiq, S.; Ashraf, U.; Dar, B.; Nayik, G.A. A Comprehensive Review on Antimicrobial Packaging and its Use in Food Packaging. Curr. Nutr. Food Sci. 2018, 14, 305–312. [Google Scholar] [CrossRef]
- Rodrigues, S.M.; Demokritou, P.; Dokoozlian, N.; Hendren, C.O.; Karn, B.; Mauter, M.S.; Sadik, O.A.; Safarpour, M.; Unrine, J.M.; Viers, J. Nanotechnology for sustainable food production: Promising opportunities and scientific challenges. Environ. Sci. Nano 2017, 4, 767–781. [Google Scholar] [CrossRef]
- Prasad, R.; Bhattacharyya, A.; Nguyen, Q.D. Nanotechnology in Sustainable Agriculture: Recent Developments, Challenges, and Perspectives. Front. Microbiol. 2017, 8, 1014. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, M.; Pastrana, L. Does the future of food pass by using nanotechnologies? Front. Sustain. Food Syst. 2019, 3, 16. [Google Scholar] [CrossRef]
- Committee, E.S. Guidance on the risk assessment of the application of nanoscience and nanotechnologies in the food and feed chain. EFSA J. 2011, 9, 2140. [Google Scholar] [CrossRef]
- OECD Environment Directorate Joint Meeting of the Chemicals Committee and the Working Party on Chemicals, Pesticides and Biotechnology: Alternative Testing Strategies in Risk Assessment of Manufactured Nanomaterials: Current State of Knowledge and Research Needs to Advance Their Use. Available online: http://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?cote=ENV/JM/MONO(2016)63&doclanguage=en (accessed on 19 December 2018).
- Hutchison, J.E. The Road to Sustainable Nanotechnology: Challenges, Progress and Opportunities. ACS Sustain. Chem. Eng 2016, 4, 5907–5914. [Google Scholar] [CrossRef]
- Maksimović, M.; Omanović-Mikličanin, E. Towards green nanotechnology: Maximizing benefits and minimizing harm. In CMBEBIH 2017; IFMBE Proceedings; Badnjevic, A., Ed.; Springer: Singapore, 2017; Volume 62, pp. 164–170. [Google Scholar]
- Sekhon, B.S. Nanotechnology in agri-food production: An overview. Nanotechnol. Sci. Appl. 2014, 7, 31. [Google Scholar] [CrossRef]
- Rai, V.; Acharya, S.; Dey, N. Implications of nanobiosensors in agriculture. J. Biomater. Nanobiotechnol. 2012, 3, 315. [Google Scholar] [CrossRef]
- Neethirajan, S.; Ragavan, V.; Weng, X.; Chand, R. Biosensors for sustainable food engineering: Challenges and perspectives. Biosensors 2018, 8, 23. [Google Scholar] [CrossRef]
- Xiang, K.; Li, Y.; Ford, W.; Land, W.; Schaffer, J.D.; Congdon, R.; Zhang, J.; Sadik, O. Automated analysis of food-borne pathogens using a novel microbial cell culture, sensing and classification system. Analyst 2016, 141, 1472–1482. [Google Scholar] [CrossRef]
- Yazgan, I.; Noah, N.M.; Toure, O.; Zhang, S.; Sadik, O.A. Biosensor for selective detection of E. coli in spinach using the strong affinity of derivatized mannose with fimbrial lectin. Biosens. Bioelectron. 2014, 61, 266–273. [Google Scholar] [CrossRef]
- Farahi, R.H.; Passian, A.; Tetard, L.; Thundat, T. Critical issues in sensor science to aid food and water safety. ACS Nano 2012, 6, 4548–4556. [Google Scholar] [CrossRef]
- Bradley, E.L.; Castle, L.; Chaudhry, Q. Applications of nanomaterials in food packaging with a consideration of opportunities for developing countries. Trends Food Sci. Technol. 2011, 22, 604–610. [Google Scholar] [CrossRef]
- Falguera, V.; Quintero, J.P.; Jiménez, A.; Muñoz, J.A.; Ibarz, A. Edible films and coatings: Structures, active functions and trends in their use. Trends Food Sci. Technol. 2011, 22, 292–303. [Google Scholar] [CrossRef]
- Silvestre, C.; Duraccio, D.; Cimmino, S. Food packaging based on polymer nanomaterials. Prog. Polym. Sci. 2011, 36, 1766–1782. [Google Scholar] [CrossRef]
- Hossain, F.; Perales-Perez, O.J.; Hwang, S.; Roman, F. Antimicrobial nanomaterials as water disinfectant: Applications, limitations and future perspectives. Sci. Total Environ. 2014, 466, 1047–1059. [Google Scholar] [CrossRef]
- Monreal, C.; DeRosa, M.; Mallubhotla, S.; Bindraban, P.; Dimkpa, C. Nanotechnologies for increasing the crop use efficiency of fertilizer-micronutrients. Biol. Fertil. Soils 2016, 52, 423–437. [Google Scholar] [CrossRef]
- Liu, R.; Lal, R. Synthetic apatite nanoparticles as a phosphorus fertilizer for soybean (Glycine max). Sci. Rep. 2014, 4, 5686. [Google Scholar] [CrossRef]
- Bindraban, P.S.; Dimkpa, C.; Nagarajan, L.; Roy, A.; Rabbinge, R. Revisiting fertilisers and fertilisation strategies for improved nutrient uptake by plants. Biol. Fertil. Soils 2015, 51, 897–911. [Google Scholar] [CrossRef]
- Kashyap, P.L.; Xiang, X.; Heiden, P. Chitosan nanoparticle based delivery systems for sustainable agriculture. Int. J. Biol. Macromol. 2015, 77, 36–51. [Google Scholar] [CrossRef]
- Maurer-Jones, M.A.; Gunsolus, I.L.; Murphy, C.J.; Haynes, C.L. Toxicity of engineered nanoparticles in the environment. Anal. Chem. 2013, 85, 3036–3049. [Google Scholar] [CrossRef]
- Mueller, N.C.; Nowack, B. Exposure modeling of engineered nanoparticles in the environment. Environ. Sci. Technol. 2008, 42, 4447–4453. [Google Scholar] [CrossRef]
- Jain, A.; Ranjan, S.; Dasgupta, N.; Ramalingam, C. Nanomaterials in food and agriculture: An overview on their safety concerns and regulatory issues. Crit. Rev. Food Sci. Nutr. 2018, 58, 297–317. [Google Scholar] [CrossRef] [PubMed]
- Bundschuh, M.; Filser, J.; Lüderwald, S.; McKee, M.S.; Metreveli, G.; Schaumann, G.E.; Schulz, R.; Wagner, S. Nanoparticles in the environment: Where do we come from, where do we go to? Environ. Sci. Eur. 2018, 30, 6. [Google Scholar] [CrossRef] [PubMed]
- Buzea, C.; Pacheco, I.I.; Robbie, K. Nanomaterials and nanoparticles: Sources and toxicity. Biointerphases 2007, 2, 17–71. [Google Scholar] [CrossRef] [PubMed]
- Nel, A.; Xia, T.; Mädler, L.; Li, N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–627. [Google Scholar] [CrossRef]
- Gliga, A.R.; Skoglund, S.; Wallinder, I.O.; Fadeel, B.; Karlsson, H.L. Size-dependent cytotoxicity of silver nanoparticles in human lung cells: The role of cellular uptake, agglomeration and Ag release. Part. Fibre Toxicol. 2014, 11, 11. [Google Scholar] [CrossRef]
- Hsiao, I.-L.; Huang, Y.-J. Effects of various physicochemical characteristics on the toxicities of ZnO and TiO2 nanoparticles toward human lung epithelial cells. Sci. Total Environ. 2011, 409, 1219–1228. [Google Scholar] [CrossRef]
- Tarantola, M.; Pietuch, A.; Schneider, D.; Rother, J.; Sunnick, E.; Rosman, C.; Pierrat, S.; Sönnichsen, C.; Wegener, J.; Janshoff, A. Toxicity of gold-nanoparticles: Synergistic effects of shape and surface functionalization on micromotility of epithelial cells. Nanotoxicology 2011, 5, 254–268. [Google Scholar] [CrossRef]
- Sahu, S.C.; Hayes, A.W. Toxicity of nanomaterials found in human environment: A literature review. Toxicol. Res. Appl. 2017, 1. [Google Scholar] [CrossRef]
- Mbeh, D.A.; Akhavan, O.; Javanbakht, T.; Mahmoudi, M.; Yahia, L.H. Cytotoxicity of protein corona-graphene oxide nanoribbons on human epithelial cells. Appl. Surf. Sci. 2014, 320, 596–601. [Google Scholar] [CrossRef]
- Handy, R.D.; Shaw, B.J. Toxic effects of nanoparticles and nanomaterials: Implications for public health, risk assessment and the public perception of nanotechnology. Health Risk Soc. 2007, 9, 125–144. [Google Scholar] [CrossRef]
- Hegde, K.; Brar, S.K.; Verma, M.; Surampalli, R.Y. Current understandings of toxicity, risks and regulations of engineered nanoparticles with respect to environmental microorganisms. Nanotechnol. Environ. Eng. 2016, 1, 5. [Google Scholar] [CrossRef]
- Peters, R.J.B.; Rivera, Z.H.; van Bemmel, G.; Marvin, H.J.P.; Weigel, S.; Bouwmeester, H. Development and validation of single particle ICP-MS for sizing and quantitative determination of nano-silver in chicken meat. Anal. Bioanal. Chem. 2014, 406, 3875–3885. [Google Scholar] [CrossRef] [PubMed]
- Benn, T.M.; Westerhoff, P. Nanoparticle Silver Released into Water from Commercially Available Sock Fabrics. Environ. Sci. Technol. 2008, 42, 4133–4139. [Google Scholar] [CrossRef]
- Navratilova, J.; Praetorius, A.; Gondikas, A.; Fabienke, W.; von der Kammer, F.; Hofmann, T. Detection of Engineered Copper Nanoparticles in Soil Using Single Particle ICP-MS. Int. J. Environ. Res. Public Health 2015, 12, 15756–15768. [Google Scholar] [CrossRef]
- Gondikas, A.P.; Kammer, F.V.D.; Reed, R.B.; Wagner, S.; Ranville, J.F.; Hofmann, T. Release of TiO2 Nanoparticles from Sunscreens into Surface Waters: A One-Year Survey at the Old Danube Recreational Lake. Environ. Sci. Technol. 2014, 48, 5415–5422. [Google Scholar] [CrossRef]
- Donovan, A.R.; Adams, C.D.; Ma, Y.; Stephan, C.; Eichholz, T.; Shi, H. Single particle ICP-MS characterization of titanium dioxide, silver, and gold nanoparticles during drinking water treatment. Chemosphere 2016, 144, 148–153. [Google Scholar] [CrossRef]
- Farré, M.; Pérez, S.; Gajda-Schrantz, K.; Osorio, V.; Kantiani, L.; Ginebreda, A.; Barceló, D. First determination of C60 and C70 fullerenes and N-methylfulleropyrrolidine C60 on the suspended material of wastewater effluents by liquid chromatography hybrid quadrupole linear ion trap tandem mass spectrometry. J. Hydrol. 2010, 383, 44–51. [Google Scholar] [CrossRef]
- Carboni, A.; Helmus, R.; Parsons, J.R.; Kalbitz, K.; de Voogt, P. A method for the determination of fullerenes in soil and sediment matrices using ultra-high performance liquid chromatography coupled with heated electrospray quadrupole time of flight mass spectrometry. J. Chromatogr. A 2016, 1433, 123–130. [Google Scholar] [CrossRef]
- Astefanei, A.; Núñez, O.; Galceran, M.T. Analysis of C60-fullerene derivatives and pristine fullerenes in environmental samples by ultrahigh performance liquid chromatography–atmospheric pressure photoionization-mass spectrometry. J. Chromatogr. A 2014, 1365, 61–71. [Google Scholar] [CrossRef]
- Doudrick, K.; Nosaka, T.; Herckes, P.; Westerhoff, P. Quantification of graphene and graphene oxide in complex organic matrices. Environ. Sci. Nano 2015, 2, 60–67. [Google Scholar] [CrossRef]
- Schierz, A.; Parks, A.N.; Washburn, K.M.; Chandler, G.T.; Ferguson, P.L. Characterization and Quantitative Analysis of Single-Walled Carbon Nanotubes in the Aquatic Environment Using Near-Infrared Fluorescence Spectroscopy. Environ. Sci. Technol. 2012, 46, 12262–12271. [Google Scholar] [CrossRef] [PubMed]
- Dudkiewicz, A.; Boxall, A.B.; Chaudhry, Q.; Mølhave, K.; Tiede, K.; Hofmann, P.; Linsinger, T.P. Uncertainties of size measurements in electron microscopy characterization of nanomaterials in foods. Food Chem. 2015, 176, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Jarzębski, M.; Bellich, B.; Białopiotrowicz, T.; Śliwa, T.; Kościński, J.; Cesàro, A. Particle tracking analysis in food and hydrocolloids investigations. Food Hydrocoll. 2017, 68, 90–101. [Google Scholar] [CrossRef]
- Laborda, F.; Bolea, E.; Cepriá, G.; Gómez, M.T.; Jiménez, M.S.; Pérez-Arantegui, J.; Castillo, J.R. Detection, characterization and quantification of inorganic engineered nanomaterials: A review of techniques and methodological approaches for the analysis of complex samples. Anal. Chim. Acta 2016, 904, 10–32. [Google Scholar] [CrossRef]
- Mattarozzi, M.; Suman, M.; Cascio, C.; Calestani, D.; Weigel, S.; Undas, A.; Peters, R. Analytical approaches for the characterization and quantification of nanoparticles in food and beverages. Anal. Bioanal. Chem. 2017, 409, 63–80. [Google Scholar] [CrossRef]
- Astefanei, A.; Núñez, O.; Galceran, M.T. Characterisation and determination of fullerenes: A critical review. Anal. Chim. Acta 2015, 882, 1–21. [Google Scholar] [CrossRef]
- Petersen, E.J.; Flores-Cervantes, D.X.; Bucheli, T.D.; Elliott, L.C.C.; Fagan, J.A.; Gogos, A.; Hanna, S.; Kägi, R.; Mansfield, E.; Bustos, A.R.M.; et al. Quantification of Carbon Nanotubes in Environmental Matrices: Current Capabilities, Case Studies, and Future Prospects. Environ. Sci. Technol. 2016, 50, 4587–4605. [Google Scholar] [CrossRef]
- Montaño, M.D.; Lowry, G.V.; von der Kammer, F.; Blue, J.; Ranville, J.F. Current status and future direction for examining engineered nanoparticles in natural systems. Environ. Chem. 2014, 11, 351–366. [Google Scholar] [CrossRef]
- Praetorius, A.; Gundlach-Graham, A.; Goldberg, E.; Fabienke, W.; Navratilova, J.; Gondikas, A.; Kaegi, R.; Günther, D.; Hofmann, T.; von der Kammer, F. Single-particle multi-element fingerprinting (spMEF) using inductively-coupled plasma time-of-flight mass spectrometry (ICP-TOFMS) to identify engineered nanoparticles against the elevated natural background in soils. Environ. Sci. Nano 2017, 4, 307–314. [Google Scholar] [CrossRef]
- Szakal, C.; Roberts, S.M.; Westerhoff, P.; Bartholomaeus, A.; Buck, N.; Illuminato, I.; Canady, R.; Rogers, M. Measurement of Nanomaterials in Foods: Integrative Consideration of Challenges and Future Prospects. ACS Nano 2014, 8, 3128–3135. [Google Scholar] [CrossRef]
- Cockburn, A.; Bradford, R.; Buck, N.; Constable, A.; Edwards, G.; Haber, B.; Hepburn, P.; Howlett, J.; Kampers, F.; Klein, C.; et al. Approaches to the safety assessment of engineered nanomaterials (ENM) in food. Food Chem. Toxicol. 2012, 50, 2224–2242. [Google Scholar] [CrossRef] [PubMed]
- Gallocchio, F.; Belluco, S.; Ricci, A. Nanotechnology and Food: Brief Overview of the Current Scenario. Procedia Food Sci. 2015, 5, 85–88. [Google Scholar] [CrossRef]
- Tiede, K.; Boxall, A.B.A.; Tear, S.P.; Lewis, J.; David, H.; Hassellöv, M. Detection and characterization of engineered nanoparticles in food and the environment. Food Addit. Contam. 2008, 25, 795–821. [Google Scholar] [CrossRef] [PubMed]
| Sector | Application | Nanomaterials | Manufacturer | Current Status | Note | Reference |
|---|---|---|---|---|---|---|
| Food processing | Color additives | TiO2 | Exempt from certification | <1% by weight of the food | [8] | |
| Synthetic iron oxide | Exempt from certification | <0.25% (for dogs and cats) and 0.1 (for human) % by weight of the finished food | [8,9] | |||
| Additive or polymer production aid | ZnO, iron oxide, aluminum oxide, silicon dioxide, cobalt oxide, manganese oxide (E530) | Authorized by EC 10/2011 | Authorization based on conventional particle size | [10] | ||
| Titanium nitride | No migration reported. Only to be used in PET bottles up to 20 mg/kg | |||||
| Carbon black | Authorized by EC 10/2011; no longer authorized by the US FDA as additives | <2.5% w/w in the polymer | ||||
| Preservatives | Silver-silica | Nanox Intelligent Materials | FCS Inventory a | FCN No. 1235. <4 ppm by weight of silver as an antimicrobial agent blended into polymers | [11] | |
| Flavor carrier | Silicon dioxide (E551 d) | Authorized by EC1334/2008 | <10,000 mg/kg, excluding foods for infants and young children | [12] | ||
| Marking fruit and vegetables | Silicon dioxide (E551) | Exempt from certification | <2% of the ink solids | [8] | ||
| Anticaking agents | Silicon dioxide (E551) | REG b | <2% by weight of the food | [13] | ||
| Nutritional dietary supplement | Copper oxide, iron oxide | Approved for animal feed | [14] | |||
| ZnO | GRAS c | |||||
| Food contact packaging | Pesticides detection | Zinc Oxide QDs | R&D | [15] | ||
| Pathogens detection | Magnetic nano-sensors | R&D | [16,17] | |||
| Plasmonic nano-sensors | [18] | |||||
| Fluorescent nano-sensors | [19] | |||||
| Toxins detection | Fluorescent nano-sensors | R&D | [20] | |||
| Plasmonic nano-sensors | [21] | |||||
| Phosphorescent QDs | [22] | |||||
| Edible film/coating | Chitosan/nano-silica coating | Tested on longan fruit | [23] | |||
| Poly-ε-caprolactone | Tested on fresh-cut “Red Delicious” apples | [24] | ||||
| Nano-emulsion/quinoa protein/ chitosan | Tested on fresh strawberries | [25] | ||||
| Bio-nano-hybrid pectins and LDH-salicylate | Tested on fresh apricots | [26] | ||||
| Nano-emulsion with lemongrass essential oil | R&D | Tested on fresh-cut Fuji apples | [27] | |||
| Bentonite (Al2O34SiO2nH2O) | GRAS | US FDA 21CFR184.1155 | [28] | |||
| Flame retardation additives, gas barrier, etc. Prevent abrasive wear | Montmorillonite | PolyOne Corporation Nanocor® Inc. | FCS Inventory | FCN No. 1163 | [11] | |
| Montmorillonite chromium (III) oxide | Toyo Seikan Kaisha Limited and Nanocor Incorporated | FCN No. 932 | [26] | |||
| Nano-emulsion with lemongrass essential oil | Oerlikon Balzers Coating AG, Oerlikon Surface Solutions AG | FCN No. 1839. For use at a thickness not to exceed 200 nm, not for use in contact with infant formula and human milk | [27] | |||
| Prevent abrasive wear Heating enhancer in polyethylene terephthalate (PET) polymers | Titanium aluminum nitride | Balzers Aktiengesellschaft | GRAS | FCN No. 302. The maximum thickness of the surface coating shall not exceed 5 mm | [28] | |
| Tin antimony oxide | Nyacol Nano Technologies, Inc. | FCS Inventory | FCN No. 1437. <0.05% by weight of the polymer | [11] |
| Target Nanoparticle (NP) | Matrix | Sample Preparation | Detection/Quantification Method | Comments | Reference |
|---|---|---|---|---|---|
| Ag NPs | chicken meat | sonication followed by proteinase K treatment | SP-ICP-MS | The established method exhibited good performance with respect to trueness, repeatability, reproducibility, and ability to determine Ag NPs transformed into silver sulfide. | [107] |
| Ag NPs | sock fabric | HNO3/H2O2 digestion | ICP-OES | The sock manufacturing process may control silver release; high silver concentration will end with the wastewater treatment facility limiting the disposal of the biosolids as agricultural fertilizers. | [108] |
| Cu NPs | topsoil | colloidal soil suspensions digested by HNO3/HCl/H2O2 and microwaves | ICP-MS | The significance of dwell time, background removal, and sample dilution as methods for optimization and recovery maximization were highlighted. | [109] |
| TiO2 | water suspended particulate matter | filtration | SP-ICP-MS | TiO2 NPs from sunscreens are possibly released into the water but settle into the sediment. | [110] |
| TiO2 NPs, Ag NPs, Au NPs | water | none | SP-ICP-MS | Lime softening followed by alum coagulation collected with powdered activated carbon adsorption resulted in removal of Au and Ag NPs and almost complete of TiO2 NPs in wastewater. | [111] |
| Various fullerenes | wastewater | filtration followed by sonication-assisted toluene extraction and partial evaporation | LC-QqLIT-MS | The established method was characterized as very effective. | [112] |
| C60 and C70 fullerenes | soil and sediment | sonication-assisted toluene extraction and partial evaporation | UHPLC-HRMS | A fast and sensitive method suitable for the analysis of very complex matrices. | [113] |
| Various fullerenes | water and sediment | LLE with toluene (water samples); ultrasound extraction and PLE (sediment samples) | UHPLC-MS/MS | An effective approach for fullerene analysis in biological entities. | [114] |
| Graphene and graphene oxide | wastewater biomass | solubilization followed by thermal digestion and reduction | PTA | The proposed approach provided had promising results. | [115] |
| SWCN | sediment | sonication in the presence of surfactants | NIRF Spectroscopy | The applicability of this tactic was exhibited. | [116] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, G.; Patra, J.K.; Paramithiotis, S.; Shin, H.-S. The Sustainability Challenge of Food and Environmental Nanotechnology: Current Status and Imminent Perceptions. Int. J. Environ. Res. Public Health 2019, 16, 4848. https://doi.org/10.3390/ijerph16234848
Das G, Patra JK, Paramithiotis S, Shin H-S. The Sustainability Challenge of Food and Environmental Nanotechnology: Current Status and Imminent Perceptions. International Journal of Environmental Research and Public Health. 2019; 16(23):4848. https://doi.org/10.3390/ijerph16234848
Chicago/Turabian StyleDas, Gitishree, Jayanta Kumar Patra, Spiros Paramithiotis, and Han-Seung Shin. 2019. "The Sustainability Challenge of Food and Environmental Nanotechnology: Current Status and Imminent Perceptions" International Journal of Environmental Research and Public Health 16, no. 23: 4848. https://doi.org/10.3390/ijerph16234848
APA StyleDas, G., Patra, J. K., Paramithiotis, S., & Shin, H.-S. (2019). The Sustainability Challenge of Food and Environmental Nanotechnology: Current Status and Imminent Perceptions. International Journal of Environmental Research and Public Health, 16(23), 4848. https://doi.org/10.3390/ijerph16234848








