Impact of Resistance Therapy on Motor Function in Children with Cerebral Palsy: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
Data Analysis
3. Results
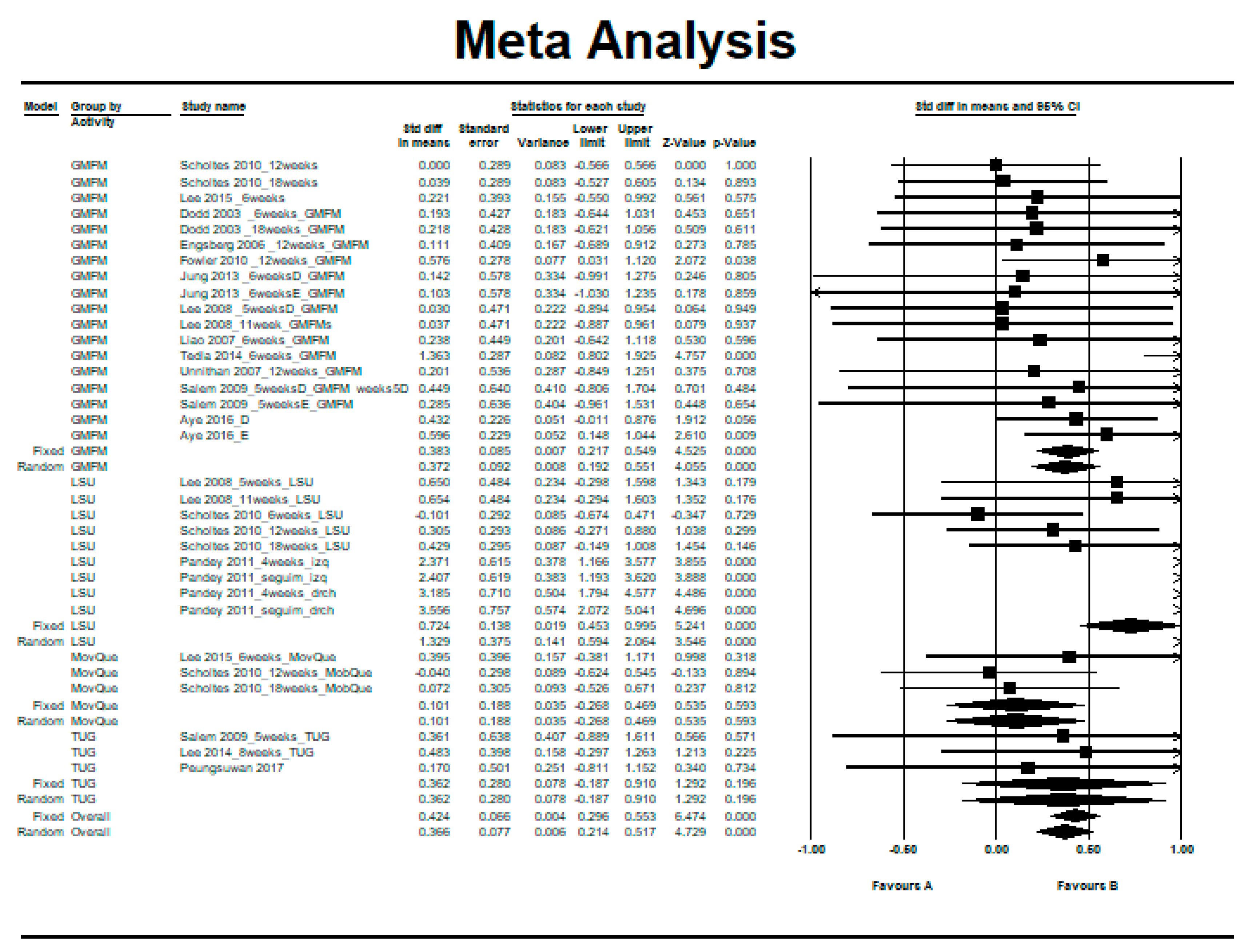
3.1. Intra-Group Pre–Post Difference in the Intervention Group
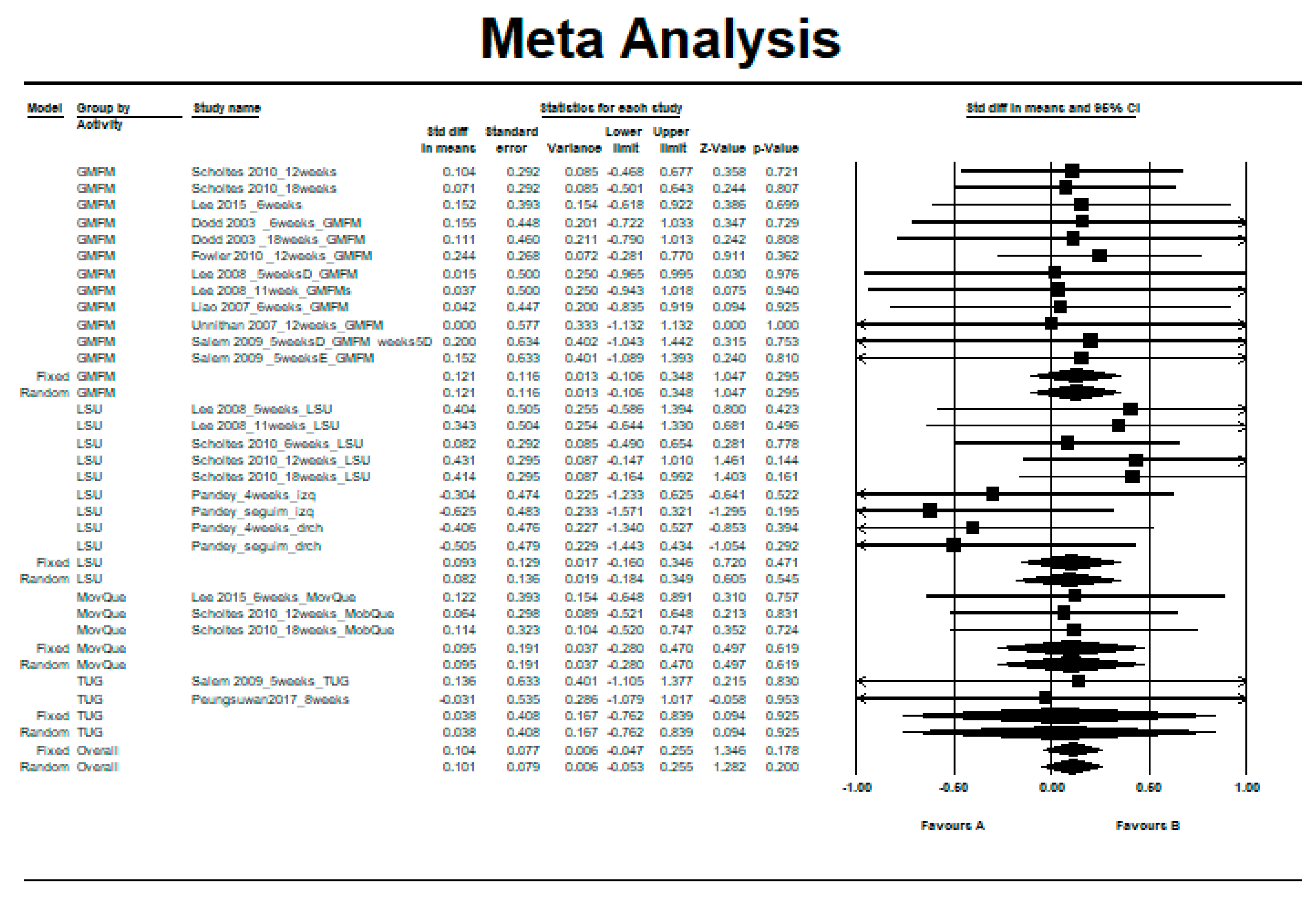
3.2. Intra-Group Difference in the Control Group
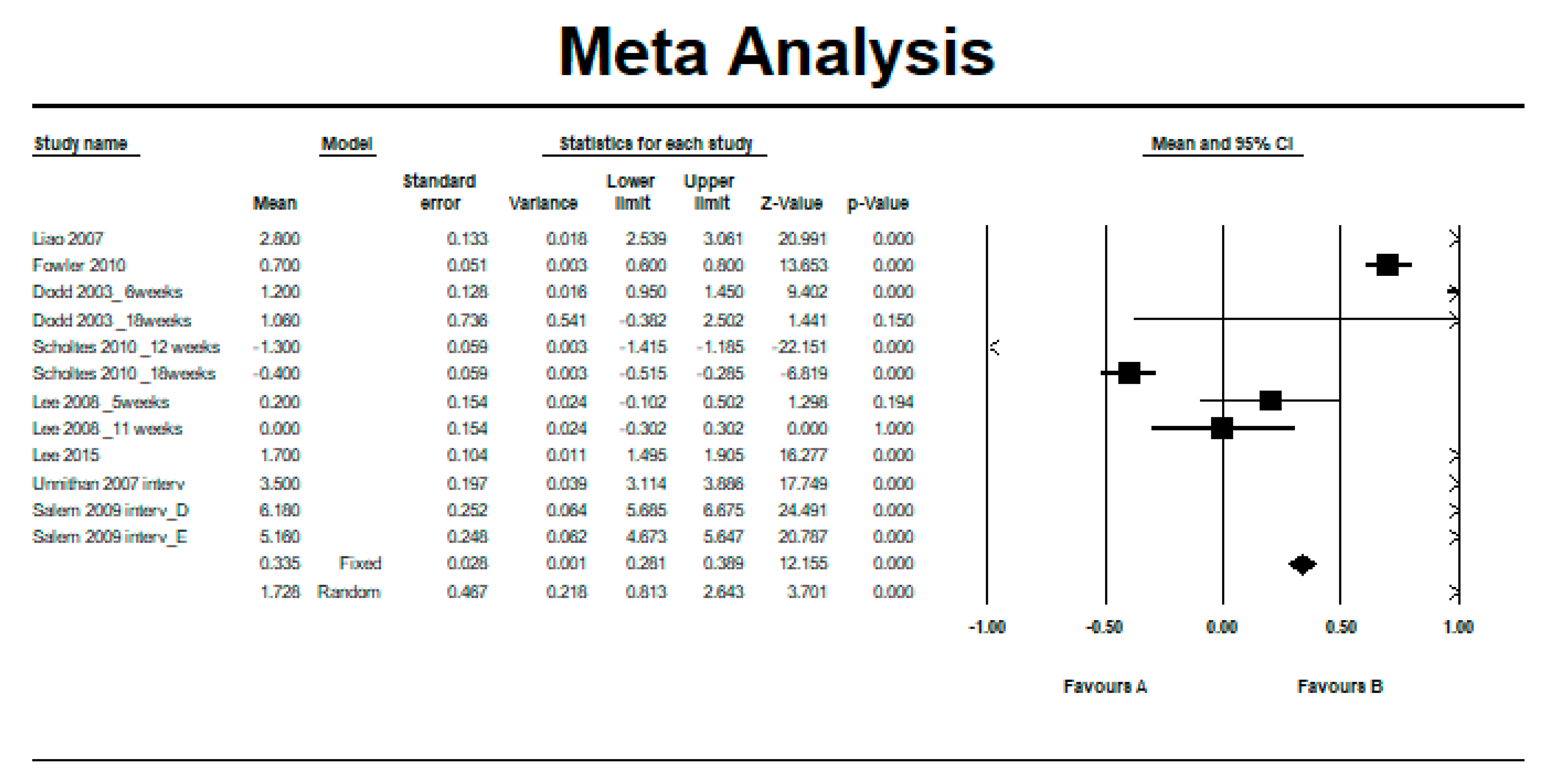
3.3. Between-Group Difference
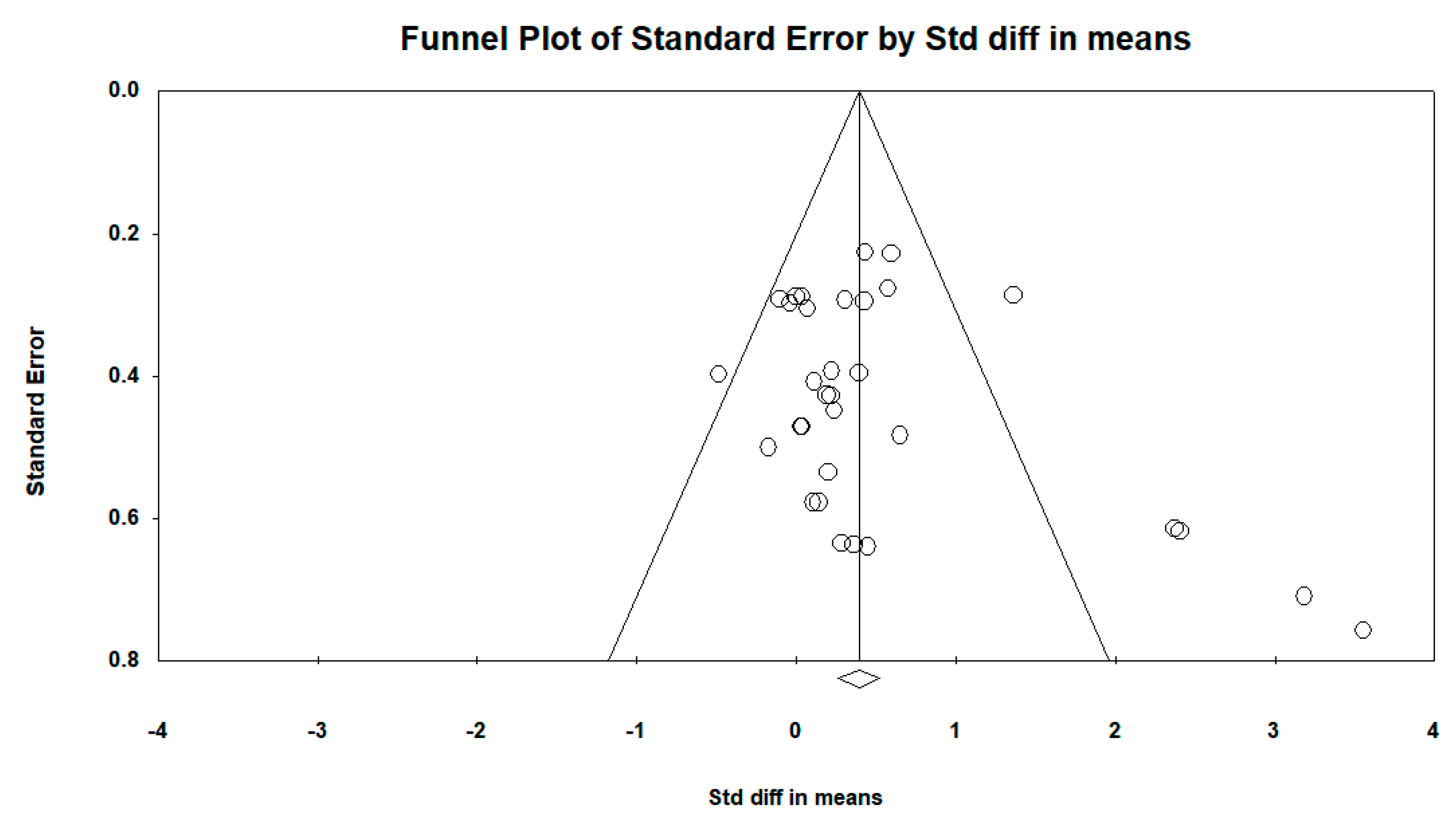
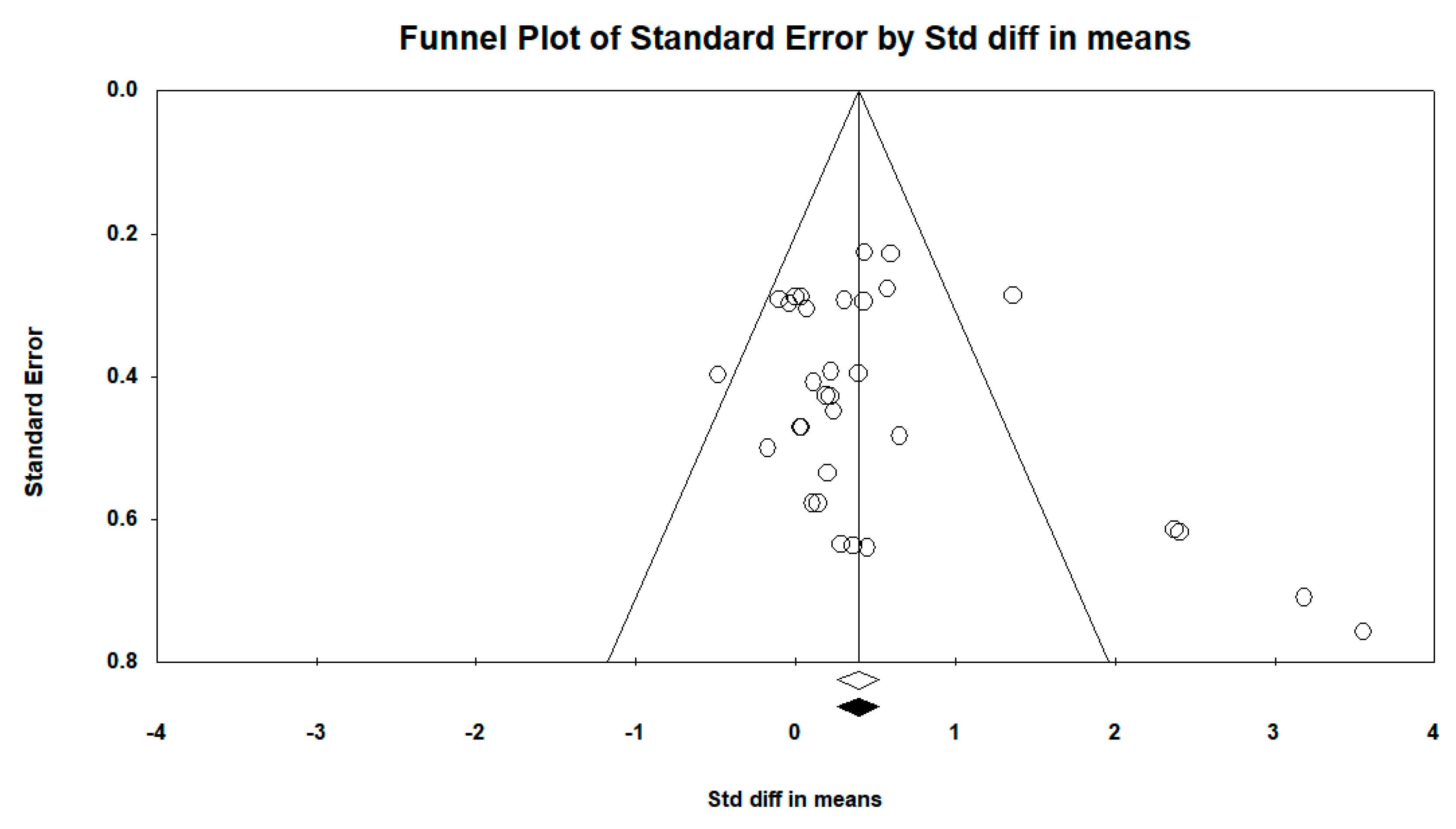
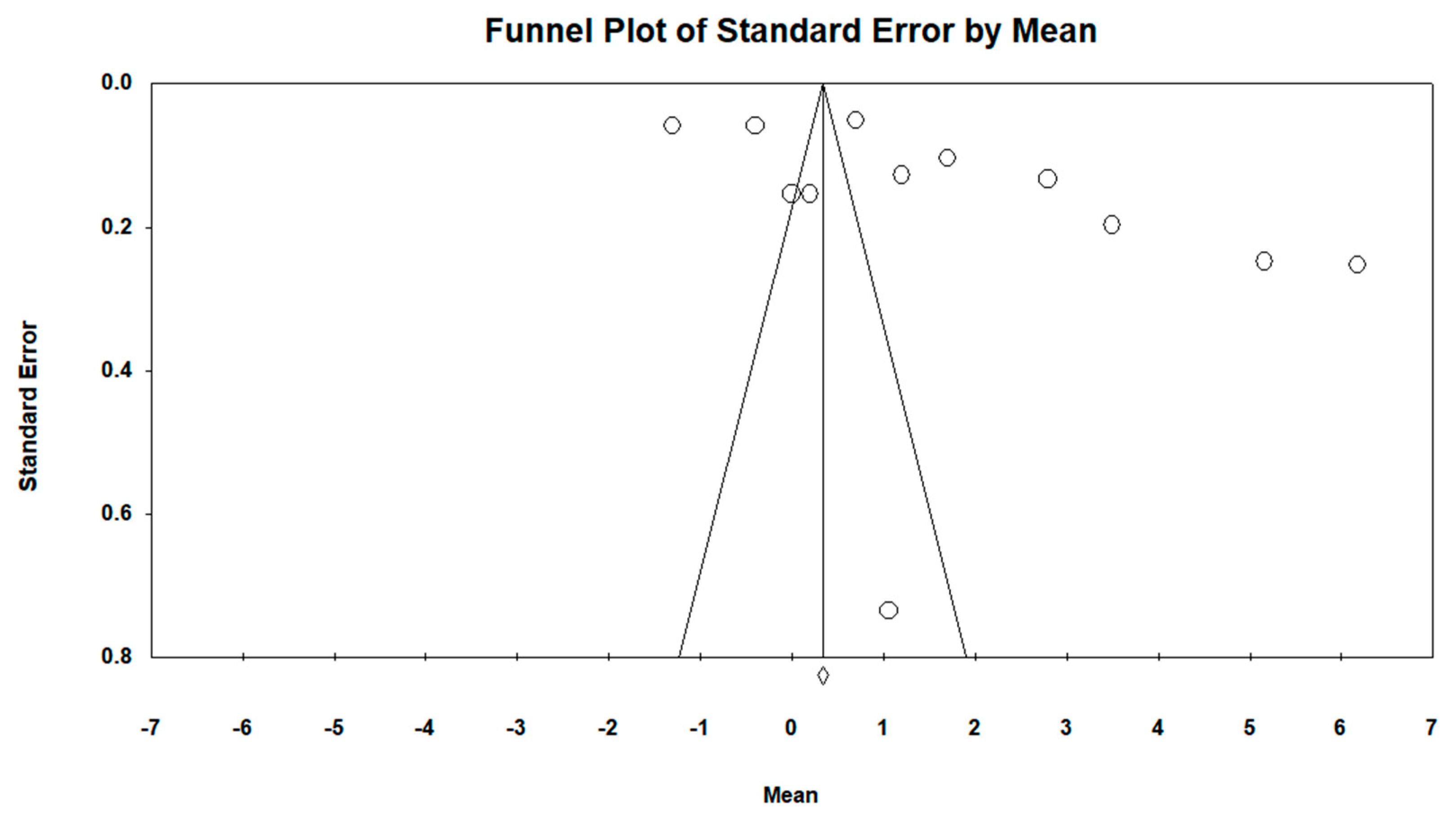
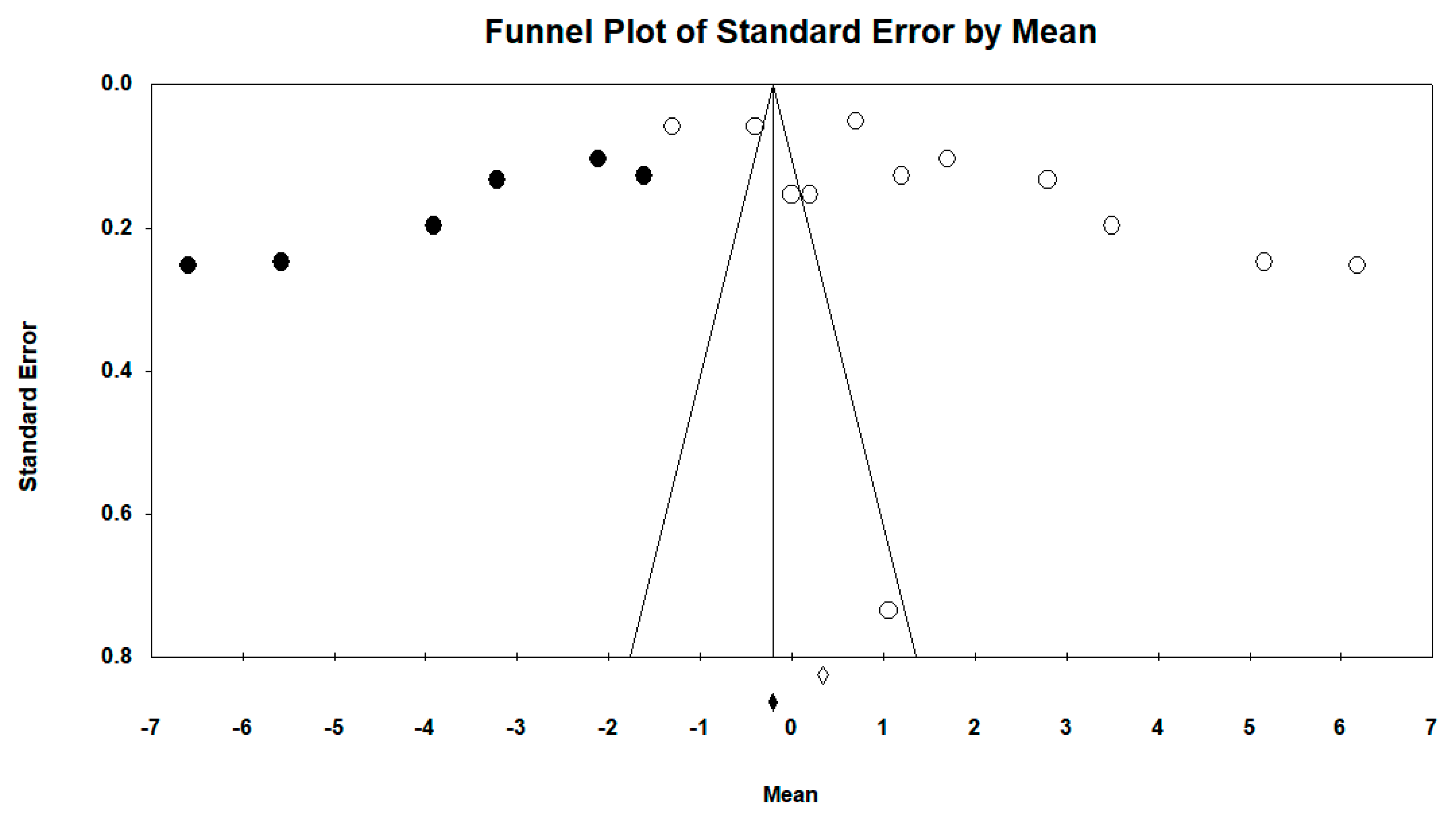
3.4. Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
References
- Odding, E.; Roebroeck, M.E.; Stam, H.J. The epidemiology of cerebral palsy: Incidence, impairments and risk factors. Disabil. Rehabil. 2006, 28, 183–191. [Google Scholar] [CrossRef]
- Rhimmer, J.H. Physical fitness levels of persons with cerebral palsy. Dev. Med. Child Neurol. 2001, 43, 208–212. [Google Scholar] [CrossRef]
- Andersson, C.; Grooten, W.; Hellsten, M.; Kaping, K.; Mattsson, E. Adults with cerebral palsy: Walking ability after progressive strength training. Dev. Med. Child Neurol. 2003, 45, 220–228. [Google Scholar] [CrossRef]
- Dodd, K.J.; Taylor, N.F.; Damiano, D.L. A systematic review of the effectiveness of strength-training programs for people with cerebral palsy. Arch. Phys. Med. Rehabil. 2002, 83, 1157–1164. [Google Scholar] [CrossRef]
- Scianni, A.; Butler, J.M.; Ada, L.; Teixeira-Salmela, L.F. Muscle strengthening is not effective in children and adolescents with cerebral palsy: A systematic review. Aust. J. Physiother. 2009, 55, 81–87. [Google Scholar] [CrossRef]
- Park, E.Y.; Kim, W.H. Meta-Analysis of the effect of strengthening interventions in individuals with cerebral palsy. Res. Dev. Disabil. 2014, 35, 239–249. [Google Scholar] [CrossRef]
- Ryan, J.M.; Cassidy, E.E.; Noorduyn, S.G.; O’Connell, N.E. Exercise interventions for cerebral palsy. Cochrane Database Syst. Rev. 2017, 11, 6. [Google Scholar] [CrossRef]
- Dodd, K.J.; Taylor, N.F.; Graham, H.K. A randomized clinical trial of strength training in young people with cerebral palsy. Dev. Med. Child Neurol. 2003, 45, 652–657. [Google Scholar] [CrossRef]
- Engsberg, J.R.; Ross, S.A.; Collins, D.R. Increasing ankle strength to improve gait and function in children with cerebral palsy: A pilot study. Pediatr. Phys. Ther. 2006, 18, 266–275. [Google Scholar] [CrossRef]
- Liao, H.F.; Liu, Y.C.; Liu, W.Y.; Lin, Y.T. Effectiveness of loaded sit-to-stand resistance exercise for children with mild spastic diplegia: A randomized clinical trial. Arch. Phys. Med. Rehabil. 2007, 88, 25–31. [Google Scholar] [CrossRef]
- Seniorou, M.; Thompson, N.; Harrington, M.; Theologis, T. Recovery of muscle strength following multi-level orthopaedic surgery in diplegic cerebral palsy. Gait Posture 2007, 26, 475–481. [Google Scholar] [CrossRef]
- Lee, J.H.; Sung, I.Y.; Yoo, J.Y. Therapeutic effects of strengthening exercise on gait function of cerebral palsy. Disabil. Rehabil. 2008, 30, 1439–1444. [Google Scholar] [CrossRef]
- Scholtes, V.A.; Becher, J.G.; Comuth, A.; Dekkers, H.; Van Dijk, L.; Dallmeijer, A.J. Effectiveness of functional progressive resistance exercise strength training on muscle strength and mobility in children with cerebral palsy: A randomized controlled trial. Dev. Med. Child Neurol. 2010, 52, e107–e113. [Google Scholar] [CrossRef]
- Lee, M.; Ko, Y.; Shin, M.M.S.; Lee, W. The effects of progressive functional training on lower limb muscle architecture and motor function in children with spastic cerebral palsy. J. Phys. Ther. Sci. 2015, 27, 1581–1584. [Google Scholar] [CrossRef]
- Mockford, M.; Caulton, J.M. Systematic review of progressive strength training in children and adolescent who are ambulatory. Pediatr. Phys. Ther. 2008, 20, 318–333. [Google Scholar] [CrossRef]
- Franki, I.; Desloovere, K.; Cat, J.; Feys, H.; Molenaers, G.; Calders, P.; Vanderstraeten, G.; Himpens, E.; Van den Broeck, C. The evidence-base for basic physical therapy techniques targeting lower limb function in children with cerebral palsy: A systematic review using the International Classification of Functioning, Disability and Health as a conceptual framework. J. Rehabil. Med. 2012, 44, 385–395. [Google Scholar] [CrossRef]
- Romera-De Francisco, L.; Jiménez-Del Barrio, S. Efectividad de la electroestimulación funcional en pacientes con ictus: Revisión sistemática. Rev. Neurol. 2016, 63, 109–118. [Google Scholar] [CrossRef]
- Verhagen, A.P.; de Vet, H.C.; de Bie, R.A.; Kessels, A.G.; Boers, M.; Bouter, L.M.; Knipschild, P.G. The Delphi list: A criteria list for quality assessment of randomized clinical trials for conducting systematic reviews developed by Delplhi consensus. J. Clin. Epidemiol. 1998, 51, 1235–1241. [Google Scholar] [CrossRef]
- Maher, C.G.; Sherrington, C.; Herbert, R.D.; Moseley, A.M.; Elkins, M. Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys. Ther. 2003, 83, 713–721. [Google Scholar]
- PEDro (Physiotherapy Evidence Database) Centre for Evidence-Based Physiotherapy. Sidney 1999. Available online: https://www.pedro.org.au/spanish/downloads/pedro-scale/ (accessed on 3 February 2017).
- Chalmers, T.C. Problems induced by meta-analyses. Stat. Med. 1991, 10, 971–979. [Google Scholar] [CrossRef]
- Santibanez, M.; Bolumar, F.; Garcia, A.M. Occupational risk factors in Alzheimer’s disease: A review assessing the quality of published epidemiological studies. Occup. Environ. Med. 2007, 64, 723–732. [Google Scholar] [CrossRef]
- Almeida, K.M.; Fonseca, S.T.; Figueiredo, P.R.P.; Aquino, A.A.; Mancini, M.C. Effects of interventions with therapeutic suits (clothing) on impairments and functional limitations of children with cerebral palsy: A systematic review. Braz. J. Phys. Ther. 2017, 21, 307–320. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- DerSimonian, R.; Kacker, R. Random-effects model for meta-analysis of clinical trials: An update. Contemp. Clin. Trials 2007, 28, 105–114. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Higgins, J.P.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.; Rothstein, H.R. Introduction to Meta-Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.; Rothstein, H.R. Comprehensive Meta-Analysis Version 2, Biostat; National Institutes of Health: Englewood, NJ, USA, 2005. [Google Scholar]
- Fowler, E.G.; Knutson, L.M.; Demuth, S.K.; Siebert, K.L.; Simms, V.D.; Sugi, M.H.; Souza, R.B.; Karim, R.; Azen, S.P. Physical Therapy Clinical Research Network (PTClinResNet). Pediatric endurance and limb strengthening (PEDALS) for children with cerebral palsy using stationary cycling: A randomized controlled trial. Phys. Ther. 2010, 90, 367–381. [Google Scholar] [CrossRef]
- Salem, Y.; Godwin, E.M. Effects of task-oriented training on mobility function in children with cerebral palsy. NeuroRehabilitation 2009, 24, 307–313. [Google Scholar]
- Tedla, J.S. Strength training effects on balance in spastic diplegia subjects: A randomized controlled trial. J. Pediatr. Neurol. 2014, 12, 15–28. [Google Scholar] [CrossRef]
- Unnithan, V.B.; Katsimanis, G.; Evengelinou, C.; Kosmas, C.; Kandrali, I.; Kellis, E. Effect of strength and aerobic training in children with cerebral palsy. Med. Sci. Sports Exerc. 2007, 39, 1902–1909. [Google Scholar] [CrossRef]
- Pandey, D.P.; Tyagi, V. Effect of functional strength training on functional motor performance in young children with cerebral palsy. Indian J. Phys. Occup. Ther. 2011, 5, 52–55. [Google Scholar]
- Peungsuwan, P.; Parasin, P.; Siritaratiwat, W.; Prasertnu, J.; Yamauchi, J. Effects of combined exercise training on functional performance in children with cerebral palsy: A randomized-controlled study. Pediatr. Phys. Ther. 2017, 29, 39–46. [Google Scholar] [CrossRef]
- Aye, T.; Thein, S.; Hlaing, T. Effects of strength training program on hip extensors and knee extensors strength of lower limb in children with spastic diplegic cerebral palsy. J. Phys. Ther. Sci. 2016, 28, 671–676. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, W.; Park, J. The effect of exercise using a sliding rehabilitation machine on the gait function of children with cerebral palsy. J. Phys. Ther. Sci. 2014, 26, 1667–1669. [Google Scholar] [CrossRef]
- Jung, J.W.; Her, J.G.; Ko, J. Effect of strength training of ankle plantarflexors on selective voluntary motor control, gait parameters, and Gross Motor Function of children with cerebral palsy. J. Phys. Ther. Sci. 2013, 25, 1259–1263. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioural Sciences; Routledge: New York, NY, USA, 1977; p. 7. [Google Scholar]
- Verschuren, O.; Ada, L.; Maltais, D.B.; Gorter, J.W.; Scianni, A.; Ketelaar, M. Muscle strengthening in children and adolescents with spastic cerebral palsy: Considerations for future resistance training protocols. Phys. Ther. 2011, 91, 1130–1139. [Google Scholar] [CrossRef]
- Stackhouse, S.K.; Binder-Macleod, S.A.; Lee, S.C. Voluntary muscle activation, contractile properties, and fatigability in children with and without cerebral palsy. Muscle Nerve 2005, 31, 594–601. [Google Scholar] [CrossRef]
- Kerr, C.; McDowel, B.; Cosgrove, A.; Walsh, D.; Bradbury, I.; McDonough, S. Electrical stimulation in cerebral palsy: A randomized controlled trial. Dev. Med. Child Neurol. 2006, 48, 870–876. [Google Scholar] [CrossRef]
- Van der Linden, M.L.; Hazlewood, M.E.; Aitchison, A.M.; Hillman, S.J.; Robb, J.E. Electrical stimulation of gluteus maximus in children with cerebral palsy. Dev. Med. Child Neurol. 1988, 40, 100–107. [Google Scholar]
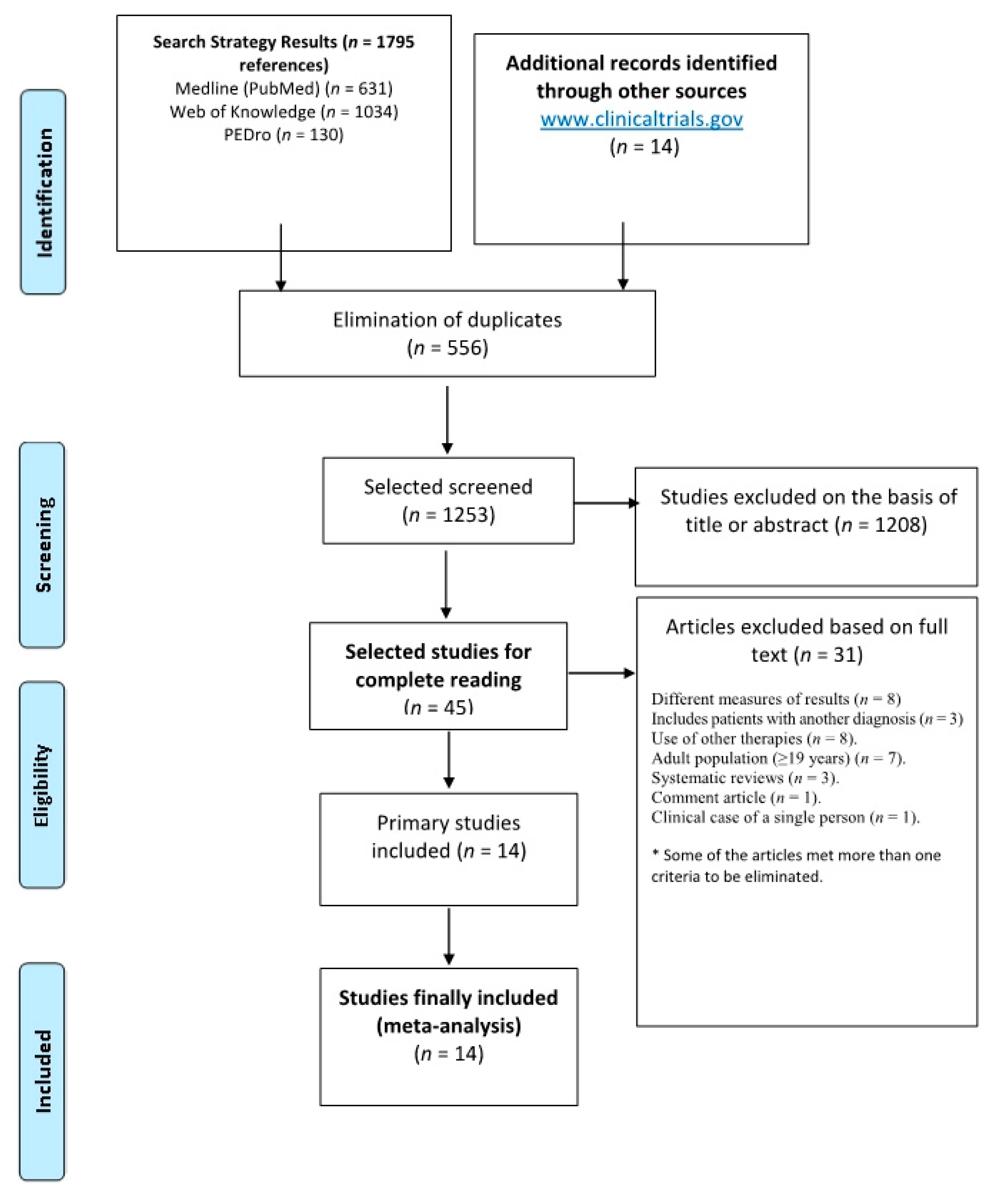
| Inclusion Criteria |
|
| Exclusion Criteria |
|
| First Author. Publication Year | Country | Study Design | Study Population | Duration | Procedure | Main Results | Other Results |
|---|---|---|---|---|---|---|---|
| Aye et al., 2016 [37] | Myanmar | Non-randomized controlled trial | 40 CP a children (I–II GMFCS b) Average age: 6 years | 6 weeks 3 times/week 45 min/session | Progressive strength training | GMFM c D (p = 0.056): Pre: 28.4 ± 11.1 Post: 33.2 ± 11.1 GMFM E (p = 0.009): Pre: 42.4 ± 19.3 Post: 54.9 ± 22.5 | |
| Dodd et al., 2003 [8] | Australia | Randomized controlled trial | 21 CP children (I–III GMFCS) Average age: 13 years | 6 weeks 3 times/week 20–30 min/session | Intervention group (n = 11): Functional strength training/conventional therapy Control group (n = 10): Conventional therapy | GMFM: intervention group (p = 0.651): Pre: 64.2 ± 27.8 Post: 69 ± 21.4 Control group (p = 0.729): Pre: 71.7 ± 24.9 Post: 75.3 ± 21.3 | Gait speed (cm/s): Intervention group (p = 0.950): Pre: 79 ± 38.83 Post: 80 ± 35.33 Control group: Pre: 82.5 ± 40.83 Post: 84.16 ± 34.66 |
| Engsberg et al., 2006 [9] | USA | Randomized controlled trial | 12 CP children (I–III GMFCS) Average age: 9.9 years | 12 weeks 3 times/week | Intervention group (n = 9): Progressive strength training. Control group (n = 3): Non-therapy | GMFM: Intervention group (p = 0.785): Pre: 65.8 ± 30.8 Post: 69.1 ± 28.4 | Gait speed (cm/s): Intervention group (p = 0.743): Pre: 85.9 ± 31.1 Post: 91 ± 34.6 Control group: Pre: 80.1 ± 23.4 Post: 78.6 ± 31.3 Cadence (steps/min): Intervention group (p = 0.813): Pre: 120.3 ± 36.3 Post: 124.4 ± 37.2 Control group: Pre: 121.7 ± 17.9 Post: 123.1 ± 12.9 Step length (cm): Intervention group (p = 0.826): Pre: 82.6 ± 21 Post: 84.8 ± 21.4 Control group: Pre: 80.6 ± 14.8 Post: 77.7 ± 25.8 |
| Fowler et al., 2010 [31] | USA | Randomized controlled trial | 62 CP children (I–III GMFCS) Age: 7–18 years | 12 weeks 3 times/week 60 min/session | Intervention group (n = 31): Resistance training on bicycle Control group (n = 31): Non-therapy | GMFM: Intervention group (p = 0.038): Pre: 69.6 ± 2.1 Post: 70.8 ± 2.07 Control group (p = 0.382): Pre: 68.8 ± 2.12 Post: 69.3 ± 1.97 | Gait speed (cm/s): Intervention group (p = 0.282): Pre: 111.5 ± 6.67 Post: 113.33 ± 6.2 Control group: Pre: 97.83 ± 6.05 Post: 103.5 ± 6.26 |
| Jung et al., 2013 [39] | South Korea | Non-randomized controlled trial | 6 CP children (I GMFCS) Age: 4–10 years | 6 weeks 3 times/week 30 min/session | Progressive strength training | GMFM D (p = 0.805): Pre: 88.5 ± 15 Post: 90.6 ± 14.5 GMFM E (p = 0.859): Pre: 78 ± 21.9 Post: 80.3 ± 22.9 | Gait speed (cm/s) (p = 0.512): Pre: 81.4 ± 19 Post: 88.7 ± 19.2 Cadence (steps/min) (p = 0.039): Pre: 117.7 ± 10.7 Post: 129.6 ± 7.1 Step length (cm) (p = 0.607): Pre: 84 ± 15.9 Post: 88.9 ± 16.9 |
| Lee et al., 2008 [12] | Korea | Randomized controlled trial | 17 CP children (II–III GMFCS) Age: 4–12 years | 5 weeks 3 times/week 60 min/session | Intervention group (n = 9): Table of resistance exercises Control group (n = 8): Conventional neurodevelopmental therapy | GMFM: Intervention group (p = 0.949): Pre: 86.5 ± 13.3 Post: 86.9 ± 13.4 Control group (p = 0.976): Pre: 85.2 ± 13.4 Post: 85.4 ± 13.5 | Gait speed (cm/s): Intervention group (p = 0.236): Pre: 54.7 ± 30.7 Post: 74.6 ± 38.7 Control group: Pre: 69.8 ± 43 Post: 68.2 ± 42.9 Cadence (steps/min): Intervention group (p = 0.848): Pre: 106.8 ± 37.1 Post: 109.7 ± 26 Control group: Pre: 107.9 ± 48.4 Post: 101.1 ± 47.4 Step length (cm): Intervention group (p = 0.137): Pre: 62.5 ± 21.8 Post: 80 ± 26.4 Control group: Pre: 70 ± 32.1 Post: 68.2 ± 42.9 |
| Lee et al., 2014 [38] | Korea | Non-randomized controlled trial | 13 CP children (I–II GMFCS) Age: 6–18 years | 8 weeks 2 times/week 30 min/session | Table of resistance exercises | TUG d (p = 0.225): Pre: 25.17 ± 7.53 Post: 21.79 ± 6.43 | |
| Lee et al., 2015 [14] | Korea | Randomized controlled trial | 26 CP children (I–III GMFCS) Age: 5–10 years | 6 weeks 3 times/week 50 min/session | Intervention group (n = 13): Functional strength training/conventional neurodevelopmental therapy Control group (n = 13): Conventional neurodevelopmental therapy | GMFM: Intervention group (p = 0.575): Pre: 78 ± 19.1 Post: 81.9 ± 16.1 Control group (p = 0.699): Pre: 79.1 ± 14.7 Post: 81.3 ± 14.3 MobQue e: Intervention group (p = 0.318): Pre: 55.7 ± 29.9 Post: 66.9 ± 26.7 Control group (p = 0.757): Pre: 48.3 ± 26.9 Post: 51.5 ± 25.7 | |
| Liao et al., 2007 [10] | China | Randomized controlled trial | 20 CP children (I–II GMFCS) Age: 5–12 years | 6 weeks 3 times/week 90 min/session | Intervention group (n = 10): Table of resistance exercises/conventional neurodevelopmental therapy Control group (n = 10): Conventional neurodevelopmental therapy | GMFM: Intervention group (p = 0.596): Pre: 76.6 ± 13.91 Post: 79.8 ± 12.96 Control group (p = 0.925): Pre: 83.1 ± 10.11 Post: 83.5 ± 8.85 | |
| Pandey et al., 2011 [35] | India | Randomized controlled trial | 18 CP children Age: 5–10 years | 4 weeks 2 times/week 60 min/session | Intervention group (n = 9): Functional strength training Control group (n = 9): Non-therapy | Lateral step up (left): Intervention group (p < 0.001): Pre: 6.22 ± 1.44 Post: 11.8 ± 3 Control group (p = 0.522): Pre: 6.44 ± 1.66 Post: 6 ± 1.2 Lateral step up (right): Intervention group (p = 0.000): Pre: 6.4 ± 1.6 Post: 12.6 ± 2.24 Control group (p = 0.394): Pre: 5.8 ± 1.2 Post: 5.34 ± 1.06 | Gait speed: Intervention group (p = 0.001): Pre: 54 ± 8 Post: 70 ± 10 Control group: Pre: 59 ± 9 Post: 60 ± 10 Cadence: Intervention group (p = 0.004): Pre: 111 ± 10 Post: 127 ± 11 Control group: Pre: 125 ± 27 Post: 127 ± 26 Step length: Intervention group (p = 1.000): Pre: 63 ± 16 Post: 63 ± 10 Control group: Pre: 58 ± 14 Post: 60 ± 10 |
| Peungsuwan et al., 2017 [36] | Thailand | Randomized controlled trial | 15 CP children (I–III GMFCS) Age: 7–16 years | 8 weeks 3 times/week 70 min/session | Intervention group (n = 8): Functional strength training/conventional therapy Control group (n = 7): Conventional therapy | TUG: Intervention group (p = 0.734): Pre: 10.1 ± 3.1 Post: 9.5 ± 3.9 Control group (p = 0.953): Pre: 11.6 ± 3 Post: 11.7 ± 3.4 | Gait speed (cm/s): Intervention group (p = 0.280): Pre: 100 ± 20 Post: 111 ± 20 Control group: Pre: 111 ± 20 Post: 99 ± 20 |
| Salem et al., 2009 [32] | USA | Randomized controlled trial | 10 CP children (I–III GMFCS) Average age: 8 years | 5 weeks 2 times/week | Intervention group (n = 5): Functional strength training Control group (n = 5): Conventional therapy | GMFM D: Intervention group (p = 0.484): Pre: 62.02 ± 25.24 Post: 73.3 ± 25.04 Control group (p = 0.753): Pre: 64.6 ± 26.07 Post: 69.7 ± 24.97 GMFM E: Intervention group (p = 0.654): Pre: 45.62 ± 31.57 Post: 54.66 ± 31.9 Control group (p = 0.810): Pre: 49.42 ± 25.42 Post: 53.3 ± 25.62 TUG: Intervention group (p = 0.571): Pre: 19.8 ± 11.32 Post: 15.8 ± 10.83 Control group (p = 0.830): Pre: 23.6 ± 13.09 Post: 11.7 ± 3.4 | |
| Scholtes et al., 2010 [13] | The Netherlands | Randomized controlled trial | 51 CP children (I–III GMFCS) Age:6–13 years | 12 weeks 3 times/week 60 min/session | Intervention group (n = 26): Functional strength training Control group (n = 25): Conventional therapy | GMFM: Intervention group (p = 1.000): Pre: 76.1 ± 12.8 Post: 76.1 ± 11.8 Control group (p = 0.721): Pre: 71.8 ± 12.5 Post: 73.1 ± 12.4 Lateral step up: Intervention group (p = 0.299): Pre: 15.6 ± 4 Post: 17 ± 5.1 Control group (p = 0.144): Pre: 13.3 ± 5.4 Post: 15.4 ± 4.3 MobQue: Intervention group (p = 0.894): Pre: 68.42 ± 20.93 Post: 67.51 ± 24.5 Control group (p = 0.831): Pre: 64.77 ± 26.26 Post: 66.4 ± 25.93 | |
| Tedla et al., 2014 [33] | Saudi Arabia | Randomized controlled trial | 60 CP children (I–V GMFCS) Age: 5–14 years | 6 weeks 3 times/week 60–90 min/session | Intervention group (n = 30): Progressive strength training. Control group (n = 30): Conventional therapy | GMFM (p < 0.001): Intervention group: Pre: 69.02 ± 7.64 Post: 78.84 ± 6.45 | |
| Unnithan et al., 2007 [34] | United Kingdom and Greece | Randomized controlled trial | 13 CP children (II–III GMFCS) Age:14–18 years | 12 weeks 3 times/week 70 min/session | Intervention group (n = 7): Progressive strength training/conventional neurodevelopmental therapy Control group (n = 6): Conventional neurodevelopmental therapy | GMFM: Intervention group (p = 0.708): Pre: 30.35 ± 16.95 Post: 33.85 ± 17.87 Control group (p = 1.000): Pre: 30.76 ± 12.52 Post: 30.76 ± 12.52 |
| Criteria | Aye | Dodd | Engsberg | Fowler | Jung | Lee | Lee | Lee | Pandey | Peungsuwan | Liao | Salem | Scholtes | Tedla | Unnithan |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2016 | 2003 | 2006 | 2010 | 2013 | 2008 | 2014 | 2015 | 2011 | 2017 | 2007 | 2009 | 2010 | 2014 | 2007 | |
| Eligibility criteria were specified a | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 |
| Random allocation | n.a. b | 1 | 1 | 1 | n.a. | 1 | n.a. | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Cancelled allocation | n.a. | 1 | 0 | 0 | n.a. | 1 | n.a. | 0 | 1 | 1 | 0 | 1 | 1 | 0 | 0 |
| Groups similar at baseline | n.a. | 0 | 0 | 1 | n.a. | 0 | n.a. | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 1 |
| Subject blinding | n.a. | 0 | 0 | 0 | n.a. | 0 | n.a. | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Therapist blinding | n.a. | 0 | 0 | 0 | n.a. | 0 | n.a. | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Assessor blinding | n.a. | 1 | 0 | 1 | n.a. | 0 | n.a. | 1 | 1 | 1 | 1 | 1 | 1 | 0 | 0 |
| <15% dropout | 0 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 0 | 1 | 1 | 1 | 1 |
| Intention-to-treat analysis | n.a. | 0 | 0 | 0 | n.a. | 1 | n.a. | 1 | 0 | 1 | 0 | 1 | 0 | 0 | 1 |
| Between-group statistical comparisons | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Point measures and variability data | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Total score (0/10) | 2 | 6 | 3 | 6 | 3 | 6 | 2 | 6 | 6 | 8 | 4 | 7 | 6 | 4 | 6 |
| Scales | N of Determinations | Heterogeneity | |||||
|---|---|---|---|---|---|---|---|
| Q | df | p (χ2) | I2 (%) | τ2 | τ | ||
| All scales and all follow-ups | 33 | 79.64 | 32 | 0.000 | 59.82 | 0.21 | 0.46 |
| GMFM A | 18 | 19.00 | 17 | 0.328 | 10.54 | 0.02 | 0.13 |
| LSU B | 9 | 51.64 | 8 | 0.000 | 84.51 | 1.00 | 1.00 |
| MobQue C | 3 | 0.78 | 2 | 0.676 | 0.00 | 0.00 | 0.00 |
| TUG D | 3 | 0.24 | 2 | 0.888 | 0.00 | 0.00 | 0.00 |
| Scales | N of Determinations | Heterogeneity | |||||
|---|---|---|---|---|---|---|---|
| Q | df | p (χ2) | I2 (%) | τ2 | τ | ||
| All scales and all follow-ups | 26 | 9.23 | 25 | 0.998 | 0.00 | 0.00 | 0.00 |
| GMFM A | 12 | 0.42 | 11 | 1.000 | 0.00 | 0.00 | 0.00 |
| LSU B | 9 | 8.69 | 8 | 0.369 | 7.97 | 0.01 | 0.12 |
| MobQue C | 3 | 0.02 | 2 | 0.990 | 0.00 | 0.00 | 0.00 |
| TUG D | 2 | 0.04 | 1 | 0.840 | 0.00 | 0.00 | 0.00 |
| Difference between Groups Using GMFM A | N of Determinations | Heterogeneity | |||||
|---|---|---|---|---|---|---|---|
| Q | df | P (χ2) | I2 (%) | τ2 | τ | ||
| All follow-ups | 12 | 2720.45 | 11 | 0 | 99.60 | 2.56 | 1.60 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Collado-Garrido, L.; Parás-Bravo, P.; Calvo-Martín, P.; Santibáñez-Margüello, M. Impact of Resistance Therapy on Motor Function in Children with Cerebral Palsy: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2019, 16, 4513. https://doi.org/10.3390/ijerph16224513
Collado-Garrido L, Parás-Bravo P, Calvo-Martín P, Santibáñez-Margüello M. Impact of Resistance Therapy on Motor Function in Children with Cerebral Palsy: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2019; 16(22):4513. https://doi.org/10.3390/ijerph16224513
Chicago/Turabian StyleCollado-Garrido, Luisa, Paula Parás-Bravo, Pilar Calvo-Martín, and Miguel Santibáñez-Margüello. 2019. "Impact of Resistance Therapy on Motor Function in Children with Cerebral Palsy: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 16, no. 22: 4513. https://doi.org/10.3390/ijerph16224513
APA StyleCollado-Garrido, L., Parás-Bravo, P., Calvo-Martín, P., & Santibáñez-Margüello, M. (2019). Impact of Resistance Therapy on Motor Function in Children with Cerebral Palsy: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 16(22), 4513. https://doi.org/10.3390/ijerph16224513






