Modification of the Association between Visual Impairment and Mortality by Physical Activity: A Cohort Study among the Korean National Health Examinees
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Visual Impairment
2.3. Physical Activity
2.4. All-Cause and Cause-Specific Mortality
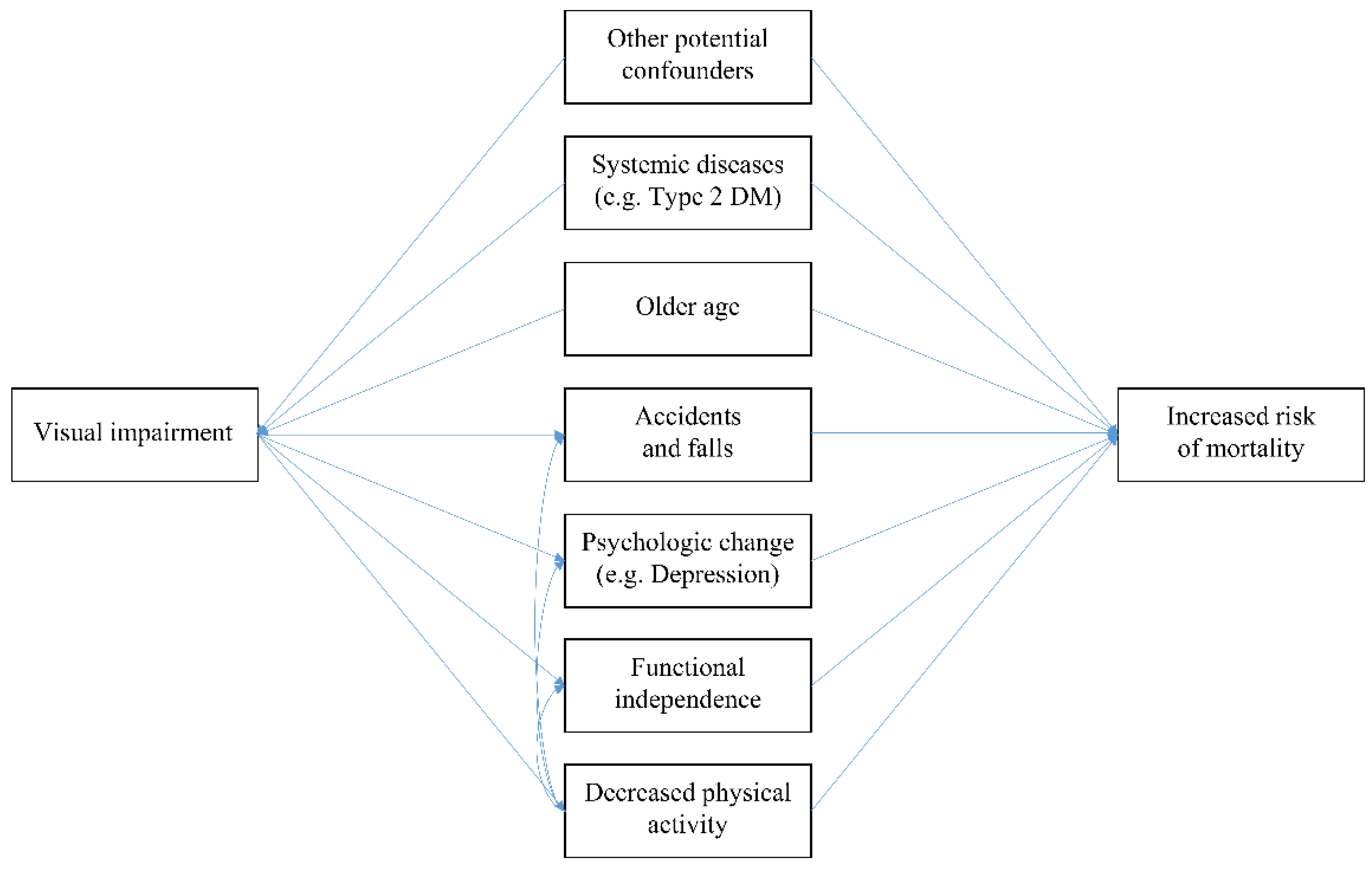
2.5. Assumed Causal Pathway and Adjusted Covariates
2.6. Statistical Analysis
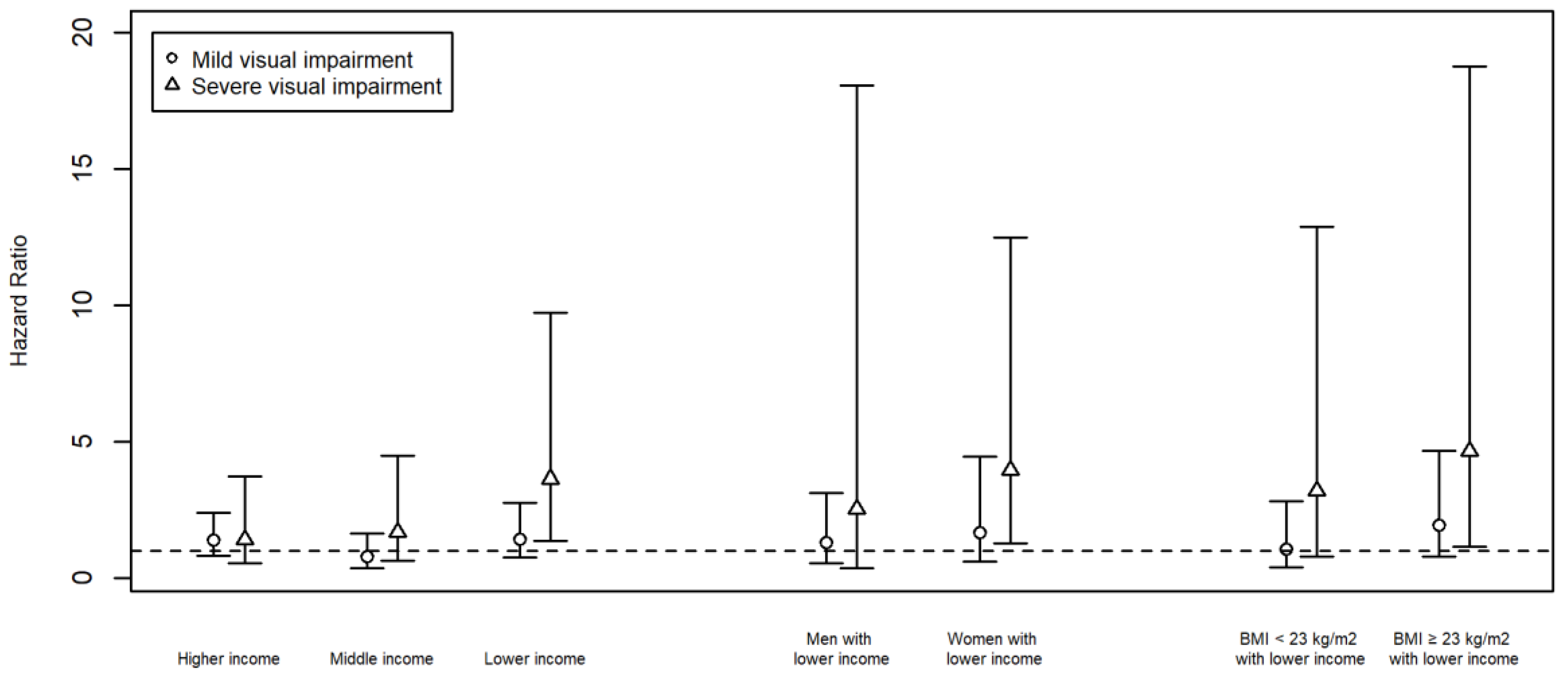
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Park, S.J.; Ahn, S.; Woo, S.J.; Park, K.H. Extent of Exacerbation of Chronic Health Conditions by Visual Impairment in Terms of Health-Related Quality of Life. JAMA Ophthalmol. 2015, 133, 1267–1275. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Ahn, S.; Park, K.H. Burden of Visual Impairment and Chronic Diseases. JAMA Ophthalmol. 2016, 134, 778–784. [Google Scholar] [CrossRef] [PubMed]
- Köberlein, J.; Beifus, K.; Schaffert, C.; Finger, R.P. The economic burden of visual impairment and blindness: A systematic review. BMJ Open 2013, 3, e003471. [Google Scholar] [CrossRef] [PubMed]
- Bourne, R.R.A.; Flaxman, S.R.; Braithwaite, T.; Cicinelli, M.V.; Das, A.; Jonas, J.B.; Keeffe, J.; Kempen, J.H.; Leasher, J.; Limburg, H.; et al. Magnitude, temporal trends, and projections of the global prevalence of blindness and distance and near vision impairment: A systematic review and meta-analysis. Lancet Glob. Health 2017, 5, e888–e897. [Google Scholar] [CrossRef]
- Brunes, A.; Flanders, W.D.; Augestad, L.B. Self-reported visual impairment, physical activity and all-cause mortality: The HUNT Study. Scand. J. Public Health 2017, 45, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Age-Related Eye Disease Study 2 Research Group; Papudesu, C.; Clemons, T.E.; Agrón, E.; Chew, E.Y. Association of Mortality with Ocular Diseases and Visual Impairment in the Age-Related Eye Disease Study 2: Age-Related Eye Disease Study 2 Report Number 13. Ophthalmology 2018, 125, 512–521. [Google Scholar] [CrossRef]
- Ng, S.K.; Kahawita, S.; Andrew, N.H.; Henderson, T.; Craig, J.E.; Landers, J. Association of Visual Impairment and All-Cause 10-Year Mortality Among Indigenous Australian Individuals Within Central Australia: The Central Australian Ocular Health Study. JAMA Ophthalmol. 2018, 136, 534–537. [Google Scholar] [CrossRef]
- Hong, Y.A.; Kim, S.Y.; Kim, S.H.; Kim, Y.O.; Jin, D.C.; Song, H.C.; Choi, E.J.; Kim, Y.L.; Kim, Y.S.; Kang, S.W.; et al. The Association of Visual Impairment with Clinical Outcomes in Hemodialysis Patients. Medicine 2016, 95, e3591. [Google Scholar] [CrossRef]
- Siantar, R.G.; Cheng, C.Y.; Gemmy Cheung, C.M.; Lamoureux, E.L.; Ong, P.G.; Chow, K.Y.; Mitchell, P.; Aung, T.; Wong, T.Y.; Cheung, C.Y. Impact of Visual Impairment and Eye diseases on Mortality: The Singapore Malay Eye Study (SiMES). Sci. Rep. 2015, 5, 16304. [Google Scholar] [CrossRef]
- Thomas, B.J.; Sanders, D.S.; Oliva, M.S.; Orrs, M.S.; Glick, P.; Ruit, S.; Chen, W.; Luoto, J.; Tasfaw, A.K.; Tabin, G.C. Blindness, cataract surgery and mortality in Ethiopia. Br. J. Ophthalmol. 2016, 100, 1157–1162. [Google Scholar] [CrossRef]
- Liu, E.; Ng, S.K.; Kahawita, S.; Andrew, N.H.; Henderson, T.; Craig, J.E.; Landers, J. Ten-year all-cause mortality and its association with vision among Indigenous Australians within Central Australia: The Central Australian Ocular Health Study. Clin. Experiment. Ophthalmol. 2017, 45, 348–356. [Google Scholar] [CrossRef]
- Zhu, Z.; Wang, L.; Young, C.A.; Huang, S.; Chang, B.H.W.; He, M. Cataract-Related Visual Impairment Corrected by Cataract Surgery and 10-Year Mortality: The Liwan Eye Study. Investig. Ophthalmol. Vis. Sci. 2016, 57, 2290–2295. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schubert, C.R.; Fischer, M.E.; Pinto, A.A.; Klein, B.E.K.; Klein, R.; Tweed, T.S.; Cruickshanks, K.J. Sensory Impairments and Risk of Mortality in Older Adults. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Kuang, T.M.; Tsai, S.Y.; Liu, C.J.L.; Lee, S.M.; Hsu, W.M.; Chou, P. The association of visual impairment and 3-year mortality among the elderly in Taiwan: The Shihpai Eye Study. J. Chin. Med. Assoc. 2015, 78, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Mitoku, K.; Masaki, N.; Ogata, Y.; Okamoto, K. Vision and hearing impairments, cognitive impairment and mortality among long-term care recipients: A population-based cohort study. BMC Geriatr. 2016, 16, 112. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Denkinger, M.D.; Onder, G.; van der Roest, H.G.; Finne-Soveri, H.; Bernabei, R.; Topinkova, E. Joint Associations of Dual Sensory Impairment and No-Activity Involvement with 1-Year Mortality in Nursing Homes: Results from the SHELTER Study. J. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Seong, S.C.; Kim, Y.Y.; Park, S.K.; Khang, Y.H.; Kim, H.C.; Park, J.H.; Kang, H.J.; Do, C.H.; Song, J.S.; Lee, E.J.; et al. Cohort profile: The National Health Insurance Service-National Health Screening Cohort (NHIS-HEALS) in Korea. BMJ Open 2017, 7, e016640. [Google Scholar] [CrossRef]
- Song, S.J.; Han, K.; Choi, K.S.; Ko, S.H.; Rhee, E.J.; Park, C.Y.; Park, J.Y.; Lee, K.U.; Ko, K.S. Task Force Team for Diabetes Fact Sheet of the Korean Diabetes Association Trends in diabetic retinopathy and related medical practices among type 2 diabetes patients: Results from the National Insurance Service Survey 2006–2013. J. Diabetes. Investig. 2018, 9, 173–178. [Google Scholar] [CrossRef]
- Williams, G.; Aggio, D.; Stubbs, B.; Pardhan, S.; Gardner, B.; Smith, L. Physical activity levels in children with sensory problems: Cross-sectional analyses from the Millennium Cohort Study. Disabil. Health J. 2018, 11, 58–61. [Google Scholar] [CrossRef]
- Estevez, J.; Kaidonis, G.; Henderson, T.; Craig, J.E.; Landers, J. Association of disease-specific causes of visual impairment and 10-year mortality amongst Indigenous Australians: The Central Australian Ocular Health Study. Clin. Experiment. Ophthalmol. 2018, 46, 18–24. [Google Scholar] [CrossRef]
- Korpelainen, R.; Lämsä, J.; Kaikkonen, K.M.; Korpelainen, J.; Laukkanen, J.; Palatsi, I.; Takala, T.E.; Ikäheimo, T.M.; Hautala, A.J. Exercise capacity and mortality—A follow-up study of 3033 subjects referred to clinical exercise testing. Ann. Med. 2016, 48, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.G. Visual impairment and mortality: Are they related? J. Chin. Med. Assoc. 2015, 78, 137–138. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Andersson, T.; Alfredsson, L.; Källberg, H.; Zdravkovic, S.; Ahlbom, A. Calculating measures of biological interaction. Eur. J. Epidemiol. 2005, 20, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Knol, M.J.; VanderWeele, T.J.; Groenwold, R.H.H.; Klungel, O.H.; Rovers, M.M.; Grobbee, D.E. Estimating measures of interaction on an additive scale for preventive exposures. Eur. J. Epidemiol. 2011, 26, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Skalska, A.; Wizner, B.; Piotrowicz, K.; Klich-Rączka, A.; Klimek, E.; Mossakowska, M.; Rowiński, R.; Kozak-Szkopek, E.; Jóźwiak, A.; Gąsowski, J.; et al. The prevalence of falls and their relation to visual and hearing impairments among a nation-wide cohort of older Poles. Exp. Gerontol. 2013, 48, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.D.; Christ, S.L.; Lam, B.L.; Arheart, K.L.; Galor, A.; Lee, D.J. Increased mortality risk among the visually impaired: The roles of mental well-being and preventive care practices. Investig. Ophthalmol. Vis. Sci. 2012, 53, 2685–2692. [Google Scholar] [CrossRef]
- Christ, S.L.; Zheng, D.D.; Swenor, B.K.; Lam, B.L.; West, S.K.; Tannenbaum, S.L.; Muñoz, B.E.; Lee, D.J. Longitudinal relationships among visual acuity, daily functional status, and mortality: The Salisbury Eye Evaluation Study. JAMA Ophthalmol. 2014, 132, 1400–1406. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Joyner, C. Accelerometer-determined physical activity and mortality in a national prospective cohort study: Considerations by visual acuity. Prev. Med. 2016, 87, 18–21. [Google Scholar] [CrossRef]
- Lear, S.A.; Hu, W.; Rangarajan, S.; Gasevic, D.; Leong, D.; Iqbal, R.; Casanova, A.; Swaminathan, S.; Anjana, R.M.; Kumar, R.; et al. The effect of physical activity on mortality and cardiovascular disease in 130000 people from 17 high-income, middle-income, and low-income countries: The PURE study. Lancet 2017, 390, 2643–2654. [Google Scholar] [CrossRef]
- Kikuchi, H.; Inoue, S.; Lee, I.M.; Odagiri, Y.; Sawada, N.; Inoue, M.; Tsugane, S. Impact of Moderate-Intensity and Vigorous-Intensity Physical Activity on Mortality. Med. Sci. Sports Exerc. 2018, 50, 715–721. [Google Scholar] [CrossRef]
- Barbosa Porcellis da Silva, R.; Marques, A.C.; Reichert, F.F. Objectively measured physical activity in Brazilians with visual impairment: Description and associated factors. Disabil. Rehabil. 2018, 40, 2131–2137. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.R.; Crowston, J.G.; Loprinzi, P.D.; Ramulu, P.Y. Physical activity, visual impairment, and eye disease. Eye 2018, 32, 1296–1303. [Google Scholar] [CrossRef] [PubMed]
- Kulmala, J.; Era, P.; Törmäkangas, T.; Pärssinen, O.; Rantanen, T.; Heikkinen, E. Visual acuity and mortality in older people and factors on the pathway. Ophthalmic Epidemiol. 2008, 15, 128–134. [Google Scholar] [CrossRef]
- Rim, T.H.T.; Nam, J.S.; Choi, M.; Lee, S.C.; Lee, C.S. Prevalence and risk factors of visual impairment and blindness in Korea: The Fourth Korea National Health and Nutrition Examination Survey in 2008–2010. Acta Ophthalmol. 2014, 92, e317–e325. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Jiang, W.; Song, X.; Zhang, D. The association between visual impairment and the risk of mortality: A meta-analysis of prospective studies. J. Epidemiol. Community Health 2016, 70, 836–842. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | No Visual Impairment (n = 359,523) | Mild Visual Impairment (n = 328) | Severe Visual Impairment (n = 133) | p-Value a |
|---|---|---|---|---|
| Sex | ||||
| Men | 194,785 (54.2) | 197 (60.1) | 70 (52.6) | 0.10 |
| Women | 164,738 (45.8) | 131 (39.9) | 63 (47.4) | |
| Household income (deciles) | ||||
| 0–3 | 73,251 (20.4) | 80 (24.4) | 25 (18.8) | 0.41 |
| 4–7 | 115,402 (32.1) | 104 (31.7) | 46 (34.6) | |
| 8–10 | 170,870 (47.5) | 144 (43.9) | 62 (46.6) | |
| Body mass index (kg/m2) | ||||
| <18.5 | 7848 (2.2) | 19 (5.8) | 7 (5.3) | <0.01 |
| 18.5–22.9 | 125,809 (35.0) | 114 (34.8) | 48 (36.1) | |
| 23–24.9 | 100,866 (28.1) | 93 (28.4) | 32 (24.1) | |
| ≥25 | 124,894 (34.8) | 102 (31.1) | 46 (34.6) | |
| Smoking status | ||||
| Non-smoker | 227,957 (64.5) | 229 (70.0) | 89 (68.5) | 0.04 |
| Ex-smoker | 65,591 (18.5) | 63 (19.3) | 21 (16.2) | |
| Smoker | 60,142 (17.0) | 35 (10.7) | 20 (15.4) | |
| Alcohol consumption | ||||
| None | 215,080 (60.3) | 234 (71.6) | 99 (74.4) | <0.01 |
| 1–2 times/week | 91,879 (25.7) | 47 (14.4) | 20 (15.0) | |
| 3–4 times/week | 32,086 (9.0) | 21 (6.4) | 8 (6.0) | |
| ≥5 times/week | 17,876 (5.0) | 25 (7.7) | 6 (4.5) | |
| Physical activity | ||||
| None | 211,833 (59.3) | 258 (79.1) | 101 (76.5) | <0.01 |
| 1–2 times/week | 78,179 (21.9) | 36 (11.0) | 20 (15.2) | |
| 3–4 times/week | 39,323 (11.0) | 7 (2.2) | 3 (2.3) | |
| ≥5 times/week | 28,044 (7.9) | 25 (7.7) | 8 (6.1) | |
| History of stroke | ||||
| No | 253,662 (98.2) | 251 (93.7) | 97 (93.3) | <0.01 |
| Yes | 4638 (1.8) | 17 (6.3) | 7 (6.7) | |
| History of heart disease | ||||
| No | 246,268 (95.3) | 246 (92.1) | 94 (90.4) | <0.01 |
| Yes | 12,137 (4.7) | 21 (7.9) | 10 (9.6) | |
| History of hypertension | ||||
| No | 164,856 (63.5) | 133 (49.4) | 49 (46.7) | <0.01 |
| Yes | 94,633 (36.5) | 136 (50.6) | 56 (53.3) | |
| History of type 2 diabetes | ||||
| No | 227,279 (87.9) | 212 (79.1) | 82 (78.1) | <0.01 |
| Yes | 31,401 (12.1) | 56 (20.9) | 23 (21.9) | |
| History of dyslipidemia | ||||
| No | 242,507 (93.8) | 251 (94.0) | 98 (92.5) | 0.84 |
| Yes | 16,087 (6.2) | 16 (6.0) | 8 (7.6) | |
| History of other diseases including cancer | ||||
| No | 229,876 (88.9) | 219 (82.0) | 88 (84.6) | <0.01 |
| Yes | 28,576 (11.1) | 48 (18.0) | 16 (15.4) | |
| Age (years) | 59.0 ± 8.8 | 72.5 ± 8.8 | 68.9 ± 9.2 | <0.01 |
| Waist circumference (cm) | 82.0 ± 8.2 | 83.0 ± 8.9 | 82.9 ± 8.5 | 0.05 |
| Systolic blood pressure (mmHg) | 125.3 ± 15.2 | 130.5 ± 16.3 | 129.1 ± 16.3 | <0.01 |
| Diastolic blood pressure (mmHg) | 77.6 ± 9.9 | 78.6 ± 9.9 | 77.9 ± 10.8 | 0.18 |
| Fasting serum glucose level (mg/dL) | 101.2 ± 25.3 | 106.7 ± 32.1 | 104.4 ± 26.2 | <0.01 |
| Serum creatinine level (mg/dL) | 1.1 ± 1.2 | 1.1 ± 1.0 | 1.0 ± 1.0 | 0.94 |
| AST level (U/L) | 26.5 ± 18.4 | 25.9 ± 10.0 | 27.4 ± 14.3 | 0.71 |
| ALT level (U/L) | 25.1 ± 19.1 | 21.9 ± 12.1 | 23.2 ± 15.6 | <0.01 |
| γ-GT level (U/L) | 38.2 ± 53.6 | 30.9 ± 23.8 | 36.1 ± 36.6 | 0.04 |
| Visual Function | No. of Deaths | HR | 95% CI | p-Value for Trend |
|---|---|---|---|---|
| Analyses not adjusted for physical activity a | ||||
| All-cause mortality | ||||
| No visual impairment | 8211 | Ref. | Ref. | 0.03 |
| Mild visual impairment | 34 | 1.17 | 0.81, 1.69 | |
| Severe visual impairment | 16 | 1.90 | 1.08, 3.35 | |
| Mortality due to cardiovascular diseases | ||||
| No visual impairment | 1202 | Ref. | Ref. | 0.02 |
| Mild visual impairment | 6 | 1.22 | 0.85, 1.76 | |
| Severe visual impairment | 5 | 1.84 | 1.04, 3.23 | |
| Mortality due to cancers | ||||
| No visual impairment | 3415 | Ref. | Ref. | 0.34 |
| Mild visual impairment | 9 | 0.83 | 0.39, 1.74 | |
| Severe visual impairment | 2 | 0.44 | 0.06, 3.10 | |
| Mortality due to other diseases | ||||
| No visual impairment | 3504 | Ref. | Ref. | 0.01 |
| Mild visual impairment | 19 | 1.60 | 1.00, 2.54 | |
| Severe visual impairment | 9 | 2.20 | 0.99, 4.90 | |
| Analyses further adjusted for physical activityb | ||||
| All-cause mortality | ||||
| No visual impairment | 8211 | Ref. | Ref. | 0.03 |
| Mild visual impairment | 34 | 1.16 | 0.81, 1.67 | |
| Severe visual impairment | 16 | 1.87 | 1.06, 3.29 | |
| Mortality due to cardiovascular diseases | ||||
| No visual impairment | 1202 | Ref. | Ref. | 0.03 |
| Mild visual impairment | 6 | 1.21 | 0.84, 1.75 | |
| Severe visual impairment | 5 | 1.80 | 1.02, 3.18 | |
| Mortality due to cancers | ||||
| No visual impairment | 3415 | Ref. | Ref. | 0.33 |
| Mild visual impairment | 9 | 0.83 | 0.39, 1.74 | |
| Severe visual impairment | 2 | 0.43 | 0.06, 3.07 | |
| Mortality due to other diseases | ||||
| No visual impairment | 3504 | Ref. | Ref. | 0.01 |
| Mild visual impairment | 19 | 1.58 | 0.99, 2.51 | |
| Severe visual impairment | 9 | 2.15 | 0.96, 4.79 | |
| Visual Function | No. of Death | HR | 95% CI | p-Value for Trend |
|---|---|---|---|---|
| Not engaging in physical activity (None) | ||||
| No visual impairment | 5921 | Ref. | Ref. | 0.01 |
| Mild visual impairment | 31 | 1.28 | 0.88, 1.86 | |
| Severe visual impairment | 15 | 1.96 | 1.09, 3.55 | |
| Engaging in physical activity (≥once/week) | ||||
| No visual impairment | 2234 | Ref. | Ref. | 0.41 |
| Mild visual impairment | 3 | 0.29 | 0.04, 2.09 | |
| Severe visual impairment | 1 | 1.11 | 0.16, 7.88 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, K.-N.; Park, S.J.; Kim, W.; Joo, J.; Kim, H.; Kim, K.H.; Sohn, J.H.; Kwon, Y.J. Modification of the Association between Visual Impairment and Mortality by Physical Activity: A Cohort Study among the Korean National Health Examinees. Int. J. Environ. Res. Public Health 2019, 16, 4386. https://doi.org/10.3390/ijerph16224386
Kim K-N, Park SJ, Kim W, Joo J, Kim H, Kim KH, Sohn JH, Kwon YJ. Modification of the Association between Visual Impairment and Mortality by Physical Activity: A Cohort Study among the Korean National Health Examinees. International Journal of Environmental Research and Public Health. 2019; 16(22):4386. https://doi.org/10.3390/ijerph16224386
Chicago/Turabian StyleKim, Kyoung-Nam, Sang Jun Park, Woosung Kim, Jungmin Joo, Haebin Kim, Kyae Hyung Kim, Ji Hoon Sohn, and Yong Jin Kwon. 2019. "Modification of the Association between Visual Impairment and Mortality by Physical Activity: A Cohort Study among the Korean National Health Examinees" International Journal of Environmental Research and Public Health 16, no. 22: 4386. https://doi.org/10.3390/ijerph16224386
APA StyleKim, K.-N., Park, S. J., Kim, W., Joo, J., Kim, H., Kim, K. H., Sohn, J. H., & Kwon, Y. J. (2019). Modification of the Association between Visual Impairment and Mortality by Physical Activity: A Cohort Study among the Korean National Health Examinees. International Journal of Environmental Research and Public Health, 16(22), 4386. https://doi.org/10.3390/ijerph16224386





