Persistent Organic Pollutants in Food: Contamination Sources, Health Effects and Detection Methods
Abstract
1. Introduction
2. Sources of POP Contamination in Food
2.1. OCPs
2.2. PCB/BDE
2.3. Dioxin and Furan
2.4. PAHs and Per- and Polyfluoroalkyl Substances (PFASs)
3. Health Effects
3.1. Endocrine Disruption and Cancers
3.2. Cardiovascular and Metabolic Diseases
4. Detection Methods
4.1. Extraction
4.2. Separation and Detection
4.3. Quality Control and Assurance
5. Efforts to Control POP Food Contamination
5.1. Monitoring of POPs in Food
5.2. Removal Methods
5.3. Dietary Make-Up
6. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Ashraf, M.A. Persistent organic pollutants (POPs): A global issue, a global challenge. Environ. Sci. Pollut. Res. Int. 2017, 24, 4223–4227. [Google Scholar] [CrossRef] [PubMed]
- Gaur, N.; Narasimhulu, K.; PydiSetty, P. Recent advances in the bio-remediation of persistent organic pollutants and its effect on environment. J. Clean. Prod. 2018, 198, 1602–1631. [Google Scholar] [CrossRef]
- El-Shahawi, M.S.; Hamza, A.; Bashammakh, A.S.; Al-Saggaf, W.T. An overview on the accumulation, distribution, transformations, toxicity and analytical methods for the monitoring of persistent organic pollutants. Talanta 2010, 80, 1587–1597. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.C.; de Voogt, P. Persistent organic pollutants (POPs): State of the science. Environ. Pollut. 1999, 100, 209–221. [Google Scholar] [CrossRef]
- Loganathan, B.G.; Masunaga, S. Chapter 18—PCBs, Dioxins, and Furans: Human Exposure and Health Effects. In Handbook of Toxicology of Chemical Warfare Agents; Gupta, R.C., Ed.; Academic Press: San Diego, CA, USA, 2009; pp. 245–253. [Google Scholar] [CrossRef]
- UNEP. Stockholm Convention on Persistent Organic Pollutants; Secretariat of the Stockholm Convention Report: Geneva, Switzerland, 2001; p. 43. [Google Scholar]
- UNECE. Protocol to the 1979 Convention on Long-Range Transboundary Air Pollution on Persistent Organic Pollutants; UNECE: Aarhus, Denmark, 1998; Available online: http://www.unece.org/env/lrtap/pops_h1.html (accessed on 31 October 2019).
- Rodríguez-Hernández, Á.; Camacho, M.; Boada, L.D.; Ruiz-Suarez, N.; Almeida-González, M.; Henríquez-Hernández, L.A.; Zumbado, M.; Luzardo, O.P. Daily intake of anthropogenic pollutants through yogurt consumption in the Spanish population. J. Appl. Anim. Res. 2015, 43, 373–383. [Google Scholar] [CrossRef]
- Abdel-Shafy, H.I.; Mansour, M.S.M. A review on polycyclic aromatic hydrocarbons: Source, environmental impact, effect on human health and remediation. Egypt. J. Pet. 2016, 25, 107–123. [Google Scholar] [CrossRef]
- Viktor, S.; Knut, B.; Torunn, B.; Dutchak, S.; Pacyna, J. Persistent Organic Pollutants. In EMEP Assessment Report Part1: European Perspective; Lovblad, G., Tarrason, L., Torseth, K., Dutchak, S., Eds.; Norwegian Meteorological Institute: Oslo, Norway, 2004; pp. 129–150. [Google Scholar]
- Alharbi, O.M.L.; Basheer, A.A.; Khattab, R.A.; Ali, I. Health and environmental effects of persistent organic pollutants. J. Mol. Liq. 2018, 263, 442–453. [Google Scholar] [CrossRef]
- Ang, E.L.; Zhao, H.; Obbard, J.P. Recent advances in the bioremediation of persistent organic pollutants via biomolecular engineering. Enzym. Microb. Technol. 2005, 37, 487–496. [Google Scholar] [CrossRef]
- Archer, J.C.; Jenkins, R.G. Automated milk fat extraction for the analyses of persistent organic pollutants. J. Chromatogr. B 2017, 1041, 70–76. [Google Scholar] [CrossRef]
- Focant, J.-F.; Eppe, G.; Scippo, M.-L.; Massart, A.-C.; Pirard, C.; Maghuin-Rogister, G.; De Pauw, E. Comprehensive two-dimensional gas chromatography with isotope dilution time-of-flight mass spectrometry for the measurement of dioxins and polychlorinated biphenyls in foodstuffs. J. Chromatogr. A 2005, 1086, 45–60. [Google Scholar] [CrossRef]
- Bernard, A.; Hermans, C.; Broeckaert, F.; De Poorter, G.; De Cock, A.; Houins, G. Food contamination by PCBs and dioxins. Nature 1999, 401, 231–232. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bermejo, A.; Abalos, M.; Saulo, J.; Abad, E.; Gonzalez, M.J.; Gomara, B. Triple quadrupole tandem mass spectrometry: A real alternative to high resolution magnetic sector instrument for the analysis of polychlorinated dibenzo-p-dioxins, furans and dioxin-like polychlorinated biphenyls. Anal. Chim. Acta 2015, 889, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Huwe, J.K.; Archer, J.C. Dioxin congener patterns in commercial catfish from the United States and the indication of mineral clays as the potential source. Food Addit. Contam. A 2013, 30, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Kedikoglou, K.; Costopoulou, D.; Vassiliadou, I.; Bakeas, E.; Leondiadis, L. An effective and low cost carbon based clean-up method for PCDD/Fs and PCBs analysis in food. Chemosphere 2018, 206, 531–538. [Google Scholar] [CrossRef] [PubMed]
- L’Homme, B.; Scholl, G.; Eppe, G.; Focant, J.F. Validation of a gas chromatography-triple quadrupole mass spectrometry method for confirmatory analysis of dioxins and dioxin-like polychlorobiphenyls in feed following new EU Regulation 709/2014. J. Chromatogr. A 2015, 1376, 149–158. [Google Scholar] [CrossRef]
- Portoles, T.; Sales, C.; Abalos, M.; Saulo, J.; Abad, E. Evaluation of the capabilities of atmospheric pressure chemical ionization source coupled to tandem mass spectrometry for the determination of dioxin-like polychlorobiphenyls in complex-matrix food samples. Anal. Chim. Acta 2016, 937, 96–105. [Google Scholar] [CrossRef]
- Chan, H.M.; Khoury, M.E.; Sedgemore, M.; Sedgemore, S.; Kuhnlein, H.V. Organochlorine pesticides and polychlorinated biphenyl congeners in ooligan grease: A traditional food fat of British Columbia first nations. J. Food Compos. Anal. 1996, 9, 32–42. [Google Scholar] [CrossRef]
- Cajka, T.; Hajslova, J.; Kazda, R.; Poustka, J. Challenges of gas chromatography-high-resolution time-of-flight mass spectrometry for simultaneous analysis of polybrominated diphenyl ethers and other halogenated persistent organic pollutants in environmental samples. J. Sep. Sci. 2005, 28, 601–611. [Google Scholar] [CrossRef]
- Gerig, B.S.; Chaloner, D.T.; Janetski, D.J.; Rediske, R.R.; O’Keefe, J.P.; Moerke, A.H.; Lamberti, G.A. Congener patterns of persistent organic pollutants establish the extent of contaminant biotransport by pacific salmon in the great lakes. Environ. Sci. Technol. 2016, 50, 554–563. [Google Scholar] [CrossRef]
- Son, M.-H.; Kim, J.-T.; Park, H.; Kim, M.; Paek, O.-J.; Chang, Y.-S. Assessment of the daily intake of 62 polychlorinated biphenyls from dietary exposure in South Korea. Chemosphere 2012, 89, 957–963. [Google Scholar] [CrossRef]
- Megson, D.; Reiner, E.J.; Jobst, K.J.; Dorman, F.L.; Robson, M.; Focant, J.-F. A review of the determination of persistent organic pollutants for environmental forensics investigations. Anal. Chim. Acta 2016, 941, 10–25. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.P.-O. Recent development in analysis of persistent organic pollutants under the Stockholm Convention. TrAC Trends Anal. Chem. 2013, 45, 48–66. [Google Scholar] [CrossRef]
- Weber, R.; Herold, C.; Hollert, H.; Kamphues, J.; Blepp, M.; Ballschmiter, K. Reviewing the relevance of dioxin and PCB sources for food from animal origin and the need for their inventory, control and management. Environ. Sci. Eur. 2018, 30, 42. [Google Scholar] [CrossRef] [PubMed]
- Domingo, J.L. Concentrations of environmental organic contaminants in meat and meat products and human dietary exposure: A review. Food Chem. Toxicol. 2017, 107, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.W.C.; Chen, B.L.S. Determination of organochlorine pesticide residues in fatty foods: A critical review on the analytical methods and their testing capabilities. J. Chromatogr. A 2011, 1218, 5555–5567. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, M.D.; Martin, M.; Tjeerdema, R.S. Long-term trends in DDT, polychlorinated biphenyls, and chlordane in California Mussels. Arch. Environ. Contam. Toxicol. 1995, 28, 443–450. [Google Scholar] [CrossRef]
- Patel, K.; Fussell, R.J.; Hetmanski, M.; Goodall, D.M.; Keely, B.J. Evaluation of gas chromatography-tandem quadrupole mass spectrometry for the determination of organochlorine pesticides in fats and oils. J. Chromatogr. A 2005, 1068, 289–296. [Google Scholar] [CrossRef]
- Stefanelli, P.; Santilio, A.; Cataldi, L.; Dommarco, R. Multiresidue analysis of organochlorine and pyrethroid pesticides in ground beef meat by gas chromatography-mass spectrometry. J. Environ. Sci. Health B 2009, 44, 350–356. [Google Scholar] [CrossRef][Green Version]
- Ericson, I.; Martí-Cid, R.; Nadal, M.; Van Bavel, B.; Lindström, G.; Domingo, J.L. Human exposure to perfluorinated chemicals through the diet: Intake of perfluorinated compounds in foods from the Catalan (Spain) market. J. Agric. Food. Chem. 2008, 56, 1787–1794. [Google Scholar] [CrossRef]
- Shitamichi, O.; Matsui, T.; Hui, Y.; Chen, W.; Imasaka, T. Determination of persistent organic pollutants by gas chromatography/laser multiphoton ionization/time-of-flight mass spectrometry. Front. Environ. Sci. Eng. 2011, 6, 26–31. [Google Scholar] [CrossRef]
- Rosenfeld, P.E.; Feng, L.G.H. 15- Bioaccumulation of Dioxins, PCBs, and PAHs. In Risks of Hazardous Wastes; Rosenfeld, P.E., Feng, L.G.H., Eds.; William Andrew Publishing: Boston, MA, USA, 2011; pp. 201–213. [Google Scholar] [CrossRef]
- Carlsson, P.; Breivik, K.; Brorström-Lundén, E.; Cousins, I.; Christensen, J.; Grimalt, J.O.; Halsall, C.; Kallenborn, R.; Abass, K.; Lammel, G.; et al. Polychlorinated biphenyls (PCBs) as sentinels for the elucidation of Arctic environmental change processes: A comprehensive review combined with ArcRisk project results. Environ. Sci. Pollut. Res. Int. 2018, 25, 22499–22528. [Google Scholar] [CrossRef] [PubMed]
- Nost, T.H.; Halse, A.K.; Schlabach, M.; Backlund, A.; Eckhardt, S.; Breivik, K. Low concentrations of persistent organic pollutants (POPs) in air at Cape Verde. Sci. Total Environ. 2018, 612, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Vorkamp, K.; Rigét, F.F. A review of new and current-use contaminants in the Arctic environment: Evidence of long-range transport and indications of bioaccumulation. Chemosphere 2014, 111, 379–395. [Google Scholar] [CrossRef] [PubMed]
- Kogevinas, M. Human health effects of dioxins: Cancer, reproductive and endocrine system effects. Hum. Reprod. Update 2001, 7, 331–339. [Google Scholar] [CrossRef]
- Li, Q.; Loganath, A.; Seng Chong, Y.; Tan, J.; Philip Obbard, J. Persistent organic pollutants and adverse health effects in humans. J. Toxicol. Environ. Health A 2006, 69, 1987–2005. [Google Scholar] [CrossRef]
- Çok, I.; Donmez, M.K.; Uner, M.; Demirkaya, E.; Henkelmann, B.; Shen, H.; Kotalik, J.; Schramm, K.-W. Polychlorinated dibenzo-p-dioxins, dibenzofurans and polychlorinated biphenyls levels in human breast milk from different regions of Turkey. Chemosphere 2009, 76, 1563–1571. [Google Scholar] [CrossRef]
- Polder, A.; Savinova, T.N.; Tkachev, A.; Løken, K.B.; Odland, J.O.; Skaare, J.U. Levels and patterns of Persistent Organic Pollutants (POPS) in selected food items from Northwest Russia (1998–2002) and implications for dietary exposure. Sci. Total Environ. 2010, 408, 5352–5361. [Google Scholar] [CrossRef]
- Bedi, J.S.; Singh, V.; Gupta, A.; Gill, J.P.S.; Aulakh, R.S. Persistent organic pollutants (POPs) in fresh water farm fish species from Punjab (India) and evaluation of their dietary intake for human risk assessment. Hum. Ecol. Risk Assess. Int. J. 2018, 24, 1659–1672. [Google Scholar] [CrossRef]
- Fair, P.A.; White, N.D.; Wolf, B.; Arnott, S.A.; Kannan, K.; Karthikraj, R.; Vena, J.E. Persistent organic pollutants in fish from Charleston Harbor and tributaries, South Carolina, United States: A risk assessment. Environ. Res. 2018, 167, 598–613. [Google Scholar] [CrossRef]
- Ábalos, M.; Barceló, D.; Parera, J.; Farré, M.l.; Llorca, M.; Eljarrat, E.; Giulivo, M.; Capri, E.; Paunović, M.; Milačič, R.; et al. Levels of regulated POPs in fish samples from the Sava River Basin. Comparison to legislated quality standard values. Sci. Total Environ. 2019, 647, 20–28. [Google Scholar] [CrossRef]
- Schafer, K.S.; Kegley, S.E. Persistent toxic chemicals in the US food supply*. J. Epidemiol. Community Health 2002, 56, 813. [Google Scholar] [CrossRef] [PubMed]
- Kalachova, K.; Pulkrabova, J.; Cajka, T.; Drabova, L.; Hajslova, J. Implementation of comprehensive two-dimensional gas chromatography–time-of-flight mass spectrometry for the simultaneous determination of halogenated contaminants and polycyclic aromatic hydrocarbons in fish. Anal. Bioanal. Chem. 2012, 403, 2813–2824. [Google Scholar] [CrossRef] [PubMed]
- Dopico, M.; Gómez, A. Review of the current state and main sources of dioxins around the world. J. Air Waste Manag. Assoc. 2015, 65, 1033–1049. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, M.; Kypke, K.; Kotz, A.; Tritscher, A.; Lee, S.Y.; Magulova, K.; Fiedler, H.; Malisch, R. WHO/UNEP global surveys of PCDDs, PCDFs, PCBs and DDTs in human milk and benefit-risk evaluation of breastfeeding. Arch. Toxicol. 2017, 91, 83–96. [Google Scholar] [CrossRef]
- Adlard, B.; Davis, K.; Liang, C.L.; Curren, M.S.; Rodríguez-Dozal, S.; Riojas-Rodríguez, H.; Hernández-Ávila, M.; Foster, W.; Needham, L.; Wong, L.-Y.; et al. Persistent organic pollutants (POPs) and metals in primiparous women: A comparison from Canada and Mexico. Sci. Total Environ. 2014, 500, 302–313. [Google Scholar] [CrossRef]
- Zhang, L.; Li, J.; Liu, X.; Zhao, Y.; Li, X.; Wen, S.; Wu, Y. Dietary intake of PCDD/Fs and dioxin-like PCBs from the Chinese total diet study in 2007. Chemosphere 2013, 90, 1625–1630. [Google Scholar] [CrossRef]
- Costopoulou, D.; Vassiliadou, I.; Leondiadis, L. Infant dietary exposure to dioxins and dioxin-like compounds in Greece. Food Chem. Toxicol. 2013, 59, 316–324. [Google Scholar] [CrossRef]
- Stubleski, J.; Lind, L.; Salihovic, S.; Lind, P.M.; Karrman, A. Longitudinal changes in persistent organic pollutants (POPs) from 2001 to 2009 in a sample of elderly Swedish men and women. Environ. Res. 2018, 165, 193–200. [Google Scholar] [CrossRef]
- Mortimer, D.N. Persistent organic pollutants in foods: Science, policy and regulation. In Persistent Organic Pollutants and Toxic Metals in Foods; Rose, M., Fernandes, A., Eds.; Woodhead Publishing: Cambridge, UK, 2013; pp. 3–19. [Google Scholar] [CrossRef]
- Stenberg, M.; Hamers, T.; Machala, M.; Fonnum, F.; Stenius, U.; Lauy, A.-A.; van Duursen, M.B.M.; Westerink, R.H.S.; Fernandes, E.C.A.; Andersson, P.L. Multivariate toxicity profiles and QSAR modeling of non-dioxin-like PCBs—An investigation of in vitro screening data from ultra-pure congeners. Chemosphere 2011, 85, 1423–1429. [Google Scholar] [CrossRef]
- Zhou, H.; Wu, H.; Liao, C.; Diao, X.; Zhen, J.; Chen, L.; Xue, Q. Toxicology mechanism of the persistent organic pollutants (POPs) in fish through AhR pathway. Toxicol. Mech. Methods 2010, 20, 279–286. [Google Scholar] [CrossRef]
- Darnerud, P.O. Toxic effects of brominated flame retardants in man and in wildlife. Environ. Int. 2003, 29, 841–853. [Google Scholar] [CrossRef]
- Henríquez-Hernández, L.A.; Luzardo, O.P.; Zumbado, M.; Camacho, M.; Serra-Majem, L.; Álvarez-León, E.E.; Boada, L.D. Blood pressure in relation to contamination by polychlorobiphenyls and organochlorine pesticides: Results from a population-based study in the Canary Islands (Spain). Environ. Res. 2014, 135, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Shoiful, A.; Fujita, H.; Watanabe, I.; Honda, K. Concentrations of organochlorine pesticides (OCPs) residues in foodstuffs collected from traditional markets in Indonesia. Chemosphere 2013, 90, 1742–1750. [Google Scholar] [CrossRef] [PubMed]
- Varjani, S.J.; Gnansounou, E.; Pandey, A. Comprehensive review on toxicity of persistent organic pollutants from petroleum refinery waste and their degradation by microorganisms. Chemosphere 2017, 188, 280–291. [Google Scholar] [CrossRef] [PubMed]
- Martorell, I.; Perelló, G.; Martí-Cid, R.; Castell, V.; Llobet, J.M.; Domingo, J.L. Polycyclic aromatic hydrocarbons (PAH) in foods and estimated PAH intake by the population of Catalonia, Spain: Temporal trend. Environ. Int. 2010, 36, 424–432. [Google Scholar] [CrossRef]
- Tian, W.; Xie, H.Q.; Fu, H.; Pei, X.; Zhao, B. Immunoanalysis methods for the detection of dioxins and related chemicals. Sensors 2012, 12, 16710–16731. [Google Scholar] [CrossRef]
- Sosa-Ferrera, Z.; Mahugo-Santana, C.; Santana-Rodríguez, J.J. Analytical methodologies for the determination of endocrine disrupting compounds in biological and environmental samples. Biomed. Res. Int. 2013, 2013, 674838. [Google Scholar] [CrossRef]
- Archer, J.C.; Moore, M.; Guo, W.; Bruce, J.; McLain, M.; Fairchild, R.; Hong, H. Quality control algorithm for determining data acceptability. Organohalogen Compd. 2018, 80, 269–272. [Google Scholar]
- Dimpe, K.M.; Nomngongo, P.N. Current sample preparation methodologies for analysis of emerging pollutants in different environmental matrices. TrAC Trends Anal. Chem. 2016, 82, 199–207. [Google Scholar] [CrossRef]
- Capriotti, A.L.; Cavaliere, C.; Colapicchioni, V.; Piovesana, S.; Samperi, R.; Lagana, A. Analytical strategies based on chromatography-mass spectrometry for the determination of estrogen-mimicking compounds in food. J. Chromatogr. A 2013, 1313, 62–77. [Google Scholar] [CrossRef]
- Xu, W.; Wang, X.; Cai, Z. Analytical chemistry of the persistent organic pollutants identified in the Stockholm Convention: A review. Anal. Chim. Acta 2013, 790, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Farré, M.; Barceló, D.; Barceló, D. Analysis of emerging contaminants in food. TrAC Trends Anal. Chem. 2013, 43, 240–253. [Google Scholar] [CrossRef]
- Campo, J.; Picó, Y. Emerging Contaminants. In Comprehensive Analytical Chemistry; Picó, Y., Ed.; Elsevier: Valencia, Spain, 2015; Volume 68, pp. 515–578. [Google Scholar]
- Zhao, R.; Chu, S.; Zhao, R.; Xu, X.; Liu, X. Ultrasonic extraction followed by sulfuric acid silica gel cleanup for the determination of α-hexachlorocyclohexane enantiomers in biota samples. Anal. Bioanal. Chem. 2005, 381, 1248–1252. [Google Scholar] [CrossRef] [PubMed]
- Thompson, L.A.; Darwish, W.S. Environmental chemical contaminants in food: Review of a global problem. J. Toxicol. 2019, 2019, 14. [Google Scholar] [CrossRef] [PubMed]
- Jensen, E.; Bolger, P.M. Exposure assessment of dioxins/furans consumed in dairy foods and fish. Food Addit. Contam. 2001, 18, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Lu, Y.; Han, J.; Luo, W.; Shi, Y.; Wang, T.; Sun, Y. Hexachlorobenzene sources, levels and human exposure in the environment of China. Environ. Int. 2010, 36, 122–130. [Google Scholar] [CrossRef]
- Wang, H.-S.; Sthiannopkao, S.; Du, J.; Chen, Z.-J.; Kim, K.-W.; Mohamed Yasin, M.S.; Hashim, J.H.; Wong, C.K.-C.; Wong, M.-H. Daily intake and human risk assessment of organochlorine pesticides (OCPs) based on Cambodian market basket data. J. Hazard. Mater. 2011, 192, 1441–1449. [Google Scholar] [CrossRef]
- Usydus, Z.; Szlinder-Richert, J.; Polak-Juszczak, L.; Komar, K.; Adamczyk, M.; Malesa-Ciecwierz, M.; Ruczynska, W. Fish products available in Polish market—Assessment of the nutritive value and human exposure to dioxins and other contaminants. Chemosphere 2009, 74, 1420–1428. [Google Scholar] [CrossRef]
- Tadeo, J.L.; Sanchez-Brunete, C.; Albero, B.; Garcia-Valcarcel, A.I. Application of ultrasound-assisted extraction to the determination of contaminants in food and soil samples. J. Chromatogr. A 2010, 1217, 2415–2440. [Google Scholar] [CrossRef]
- Páez-Osuna, F.; Ruiz-Fernández, A.C.; Botello, A.V.; Ponce-Vélez, G.; Osuna-López, J.I.; Frías-Espericueta, M.G.; López-López, G.; Zazueta-Padilla, H.M. Concentrations of selected trace metals (Cu, Pb, Zn), organochlorines (PCBs, HCB) and total PAHs in mangrove oysters from the Pacific Coast of Mexico: An overview. Mar. Pollut. Bull. 2002, 44, 1303–1308. [Google Scholar] [CrossRef]
- Barber, J.L.; Sweetman, A.J.; van Wijk, D.; Jones, K.C. Hexachlorobenzene in the global environment: Emissions, levels, distribution, trends and processes. Sci. Total Environ. 2005, 349, 1–44. [Google Scholar] [CrossRef] [PubMed]
- Rogan, W.J.; Chen, A. Health risks and benefits of bis(4-chlorophenyl)-1,1,1-trichloroethane (DDT). Lancet 2005, 366, 763–773. [Google Scholar] [CrossRef]
- Thompson, L.A.; Darwish, W.S.; Ikenaka, Y.; Nakayama, S.M.M.; Mizukawa, H.; Ishizuka, M. Organochlorine pesticide contamination of foods in Africa: Incidence and public health significance. J. Vet. Med. Sci. 2017, 79, 751–764. [Google Scholar] [CrossRef] [PubMed]
- Bouwman, H.; Bornman, R.; van Dyk, C.; Barnhoorn, I. First report of the concentrations and implications of DDT residues in chicken eggs from a malaria-controlled area. Chemosphere 2015, 137, 174–177. [Google Scholar] [CrossRef][Green Version]
- Van Dyk, J.C.; Bouwman, H.; Barnhoorn, I.E.J.; Bornman, M.S. DDT contamination from indoor residual spraying for malaria control. Sci. Total Environ. 2010, 408, 2745–2752. [Google Scholar] [CrossRef]
- Sharma, B.M.; Bharat, G.K.; Tayal, S.; Nizzetto, L.; Cupr, P.; Larssen, T. Environment and human exposure to persistent organic pollutants (POPs) in India: A systematic review of recent and historical data. Environ. Int. 2014, 66, 48–64. [Google Scholar] [CrossRef]
- Breivik, K.; Sweetman, A.; Pacyna, J.M.; Jones, K.C. Towards a global historical emission inventory for selected PCB congeners—A mass balance approach: 3. An update. Sci. Total Environ. 2007, 377, 296–307. [Google Scholar] [CrossRef]
- Lavandier, R.; Quinete, N.; Hauser-Davis, R.A.; Dias, P.S.; Taniguchi, S.; Montone, R.; Moreira, I. Polychlorinated biphenyls (PCBs) and Polybrominated Diphenyl ethers (PBDEs) in three fish species from an estuary in the southeastern coast of Brazil. Chemosphere 2013, 90, 2435–2443. [Google Scholar] [CrossRef]
- Devanathan, G.; Subramanian, A.; Sudaryanto, A.; Takahashi, S.; Isobe, T.; Tanabe, S. Brominated flame retardants and polychlorinated biphenyls in human breast milk from several locations in India: Potential contaminant sources in a municipal dumping site. Environ. Int. 2012, 39, 87–95. [Google Scholar] [CrossRef]
- Ng, C.A.; von Goetz, N. The global food system as a transport pathway for hazardous chemicals: The missing link between emissions and exposure. Environ. Health Perspect. 2017, 125, 1–7. [Google Scholar] [CrossRef]
- Hernández, Á.R.; Boada, L.D.; Mendoza, Z.; Ruiz-Suárez, N.; Valerón, P.F.; Camacho, M.; Zumbado, M.; Almeida-González, M.; Henríquez-Hernández, L.A.; Luzardo, O.P. Consumption of organic meat does not diminish the carcinogenic potential associated with the intake of persistent organic pollutants (POPs). Environ. Sci. Pollut. Res. Int. 2017, 24, 4261–4273. [Google Scholar] [CrossRef] [PubMed]
- Ten Dam, G.; Pussente, I.C.; Scholl, G.; Eppe, G.; Schaechtele, A.; van Leeuwen, S. The performance of atmospheric pressure gas chromatography—Tandem mass spectrometry compared to gas chromatography—High resolution mass spectrometry for the analysis of polychlorinated dioxins and polychlorinated biphenyls in food and feed samples. J. Chromatogr. A 2016, 1477, 76–90. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Wu, N.; Han, J.; Shen, H.; Tan, Y.; Ding, G.; Xiang, J.; Tao, H.; Jin, S. Levels of PCDD/Fs and DL-PCBs in selected foods and estimated dietary intake for the local residents of Luqiao and Yuhang in Zhejiang, China. Chemosphere 2011, 85, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Polder, A.; Müller, M.B.; Brynildsrud, O.B.; de Boer, J.; Hamers, T.; Kamstra, J.H.; Lie, E.; Mdegela, R.H.; Moberg, H.; Nonga, H.E.; et al. Dioxins, PCBs, chlorinated pesticides and brominated flame retardants in free-range chicken eggs from peri-urban areas in Arusha, Tanzania: Levels and implications for human health. Sci. Total Environ. 2016, 551, 656–667. [Google Scholar] [CrossRef]
- Qin, Y.Y.; Leung, C.K.M.; Leung, A.O.W.; Zheng, J.S.; Wong, M.H. Persistent organic pollutants in food items collected in Hong Kong. Chemosphere 2011, 82, 1329–1336. [Google Scholar] [CrossRef]
- Kohler, M.; Tremp, J.; Zennegg, M.; Seiler, C.; Minder-Kohler, S.; Beck, M.; Lienemann, P.; Wegmann, L.; Schmid, P. Joint sealants: An overlooked diffuse source of polychlorinated biphenyls in buildings. Environ. Sci. Technol. 2005, 39, 1967–1973. [Google Scholar] [CrossRef]
- Ruiz-Fernández, A.C.; Ontiveros-Cuadras, J.F.; Sericano, J.L.; Sanchez-Cabeza, J.-A.; Liong Wee Kwong, L.; Dunbar, R.B.; Mucciarone, D.A.; Pérez-Bernal, L.H.; Páez-Osuna, F. Long-range atmospheric transport of persistent organic pollutants to remote lacustrine environments. Sci. Total Environ. 2014, 493, 505–520. [Google Scholar] [CrossRef]
- Ljunggren, S.A.; Helmfrid, I.; Salihovic, S.; van Bavel, B.; Wingren, G.; Lindahl, M.; Karlsson, H. Persistent organic pollutants distribution in lipoprotein fractions in relation to cardiovascular disease and cancer. Environ. Int. 2014, 65, 93–99. [Google Scholar] [CrossRef]
- Kanan, S.; Samara, F. Dioxins and furans: A review from chemical and environmental perspectives. Trends Environ. Anal. Chem. 2018, 17, 1–13. [Google Scholar] [CrossRef]
- Fiedler, H. National PCDD/PCDF release inventories under the stockholm convention on persistent organic pollutants. Chemosphere 2007, 67, S96–S108. [Google Scholar] [CrossRef]
- Adamse, P.; Schoss, S.; Theelen, R.M.; Hoogenboom, R.L. Levels of dioxins and dioxin-like PCBs in food of animal origin in the Netherlands during the period 2001–2011. Food Addit. Contam. A 2017, 34, 78–92. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, T. Yusho in Japan. Ind. Health 2003, 41, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.-T.; Ma, C.-I.; Hsu, S.K.-H.; Wu, S.-S.; Hsu, N.H.-M.; Yeh, C.-C. Discovery and epidemiology of PCB poisoning in Taiwan. Am. J. Ind. Med. 1984, 5, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Masuda, Y. Approach to risk assessment of chlorinated dioxins from Yusho PCB poisoning. Chemosphere 1996, 32, 583–594. [Google Scholar] [CrossRef]
- Bernard, A.; Broeckaert, F.; De Poorter, G.; De Cock, A.; Hermans, C.; Saegerman, C.; Houins, G. The Belgian PCB/dioxin incident: Analysis of the food chain contamination and health risk evaluation. Environ. Res. 2002, 88, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Pratt, I.S.; Anderson, W.A.; Crowley, D.; Daly, S.F.; Evans, R.I.; Fernandes, A.R.; Fitzgerald, M.; Geary, M.P.; Keane, D.P.; Malisch, R.; et al. Polychlorinated dibenzo-p-dioxins (PCDDs), polychlorinated dibenzofurans (PCDFs) and polychlorinated biphenyls (PCBs) in breast milk of first-time Irish mothers: Impact of the 2008 dioxin incident in Ireland. Chemosphere 2012, 88, 865–872. [Google Scholar] [CrossRef]
- Debacker, N.; Sasse, A.; van Wouwe, N.; Goeyens, L.; Sartor, F.; van Oyen, H. PCDD/F levels in plasma of a belgian population before and after the 1999 belgian PCB/DIOXIN incident. Chemosphere 2007, 67, S217–S223. [Google Scholar] [CrossRef]
- Tuomisto, J.; Airaksinen, R.; Kiviranta, H.; Tukiainen, E.; Pekkanen, J.; Tuomisto, J.T. A pharmacokinetic analysis and dietary information are necessary to confirm or reject the hypothesis on persistent organic pollutants causing type 2 diabetes. Toxicol. Lett. 2016, 261, 41–48. [Google Scholar] [CrossRef]
- Hayward, D.G.; Nortrup, D.; Gardner, A.; Clower, M., Jr. Elevated TCDD in chicken eggs and farm-raised catfish fed a diet with ball clay from a Southern United States mine. Environ. Res. 1999, 81, 248–256. [Google Scholar] [CrossRef]
- Ábalos, M.; Cojocariu, C.I.; Silcock, P.; Roberts, D.; Pemberthy, D.M.; Sauló, J.; Abad, E. Meeting the European Commission performance criteria for the use of triple quadrupole GC-MS/MS as a confirmatory method for PCDD/Fs and dl-PCBs in food and feed samples. Anal. Bioanal. Chem. 2016, 408, 3511–3525. [Google Scholar] [CrossRef]
- Dennis, M.J.; Massey, R.C.; McWeeny, D.J.; Knowles, M.E.; Watson, D. Analysis of polycyclic aromatic hydrocarbons in UK total diets. Food Chem. Toxicol. 1983, 21, 569–574. [Google Scholar] [CrossRef]
- Dennis, M.J.; Massey, R.C.; Cripps, G.; Venn, I.; Howarth, N.; Lee, G. Factors affecting the polycyclic aromatic hydrocarbon content of cereals, fats and other food products. Food Addit. Contam. 1991, 8, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Domingo, J.L.; Nadal, M. Human dietary exposure to polycyclic aromatic hydrocarbons: A review of the scientific literature. Food Chem. Toxicol. 2015, 86, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Mastrantonio, M.; Bai, E.; Uccelli, R.; Cordiano, V.; Screpanti, A.; Crosignani, P. Drinking water contamination from perfluoroalkyl substances (PFAS): An ecological mortality study in the Veneto Region, Italy. Eur. J. Public Health 2017, 28, 180–185. [Google Scholar] [CrossRef]
- Domingo, J.L.; Nadal, M. Per- and Polyfluoroalkyl Substances (PFASs) in food and human dietary Intake: A review of the recent scientific literature. J. Agric. Food Chem. 2017, 65, 533–543. [Google Scholar] [CrossRef]
- Chen, W.-L.; Bai, F.-Y.; Chang, Y.-C.; Chen, P.-C.; Chen, C.-Y. Concentrations of perfluoroalkyl substances in foods and the dietary exposure among Taiwan general population and pregnant women. J. Food Drug Anal. 2018, 26, 994–1004. [Google Scholar] [CrossRef]
- Boronow, K.E.; Brody, J.G.; Schaider, L.A.; Peaslee, G.F.; Havas, L.; Cohn, B.A. Serum concentrations of PFASs and exposure-related behaviors in African American and non-Hispanic white women. J. Expo. Sci. Environ. Epidemiol. 2019, 29, 206–217. [Google Scholar] [CrossRef]
- Begley, T.H.; White, K.; Honigfort, P.; Twaroski, M.L.; Neches, R.; Walker, R.A. Perfluorochemicals: Potential sources of and migration from food packaging. Food Addit. Contam. 2005, 22, 1023–1031. [Google Scholar] [CrossRef]
- Malinsky, M.D.; Jacoby, C.B.; Reagen, W.K. Determination of perfluorinated compounds in fish fillet homogenates: Method validation and application to fillet homogenates from the Mississippi River. Anal. Chim. Acta 2011, 683, 248–257. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, Y.; Li, J.; Zhao, Y.; Guo, F.; Liu, J.; Cai, Z. Perfluorinated compounds in seafood from coastal areas in China. Environ. Int. 2012, 42, 67–71. [Google Scholar] [CrossRef]
- Schuetze, A.; Heberer, T.; Effkemann, S.; Juergensen, S. Occurrence and assessment of perfluorinated chemicals in wild fish from Northern Germany. Chemosphere 2010, 78, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Van Asselt, E.D.; Kowalczyk, J.; van Eijkeren, J.C.H.; Zeilmaker, M.J.; Ehlers, S.; Fürst, P.; Lahrssen-Wiederholt, M.; van der Fels-Klerx, H.J. Transfer of perfluorooctane sulfonic acid (PFOS) from contaminated feed to dairy milk. Food Chem. 2013, 141, 1489–1495. [Google Scholar] [CrossRef] [PubMed]
- Schaider, L.A.; Balan, S.A.; Blum, A.; Andrews, D.Q.; Strynar, M.J.; Dickinson, M.E.; Lunderberg, D.M.; Lang, J.R.; Peaslee, G.F. Fluorinated compounds in U.S. fast food packaging. Environ. Sci. Technol. Lett. 2017, 4, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Zabaleta, I.; Bizkarguenaga, E.; Bilbao, D.; Etxebarria, N.; Prieto, A.; Zuloaga, O. Fast and simple determination of perfluorinated compounds and their potential precursors in different packaging materials. Talanta 2016, 152, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Jin, Y.; Cheng, Y.; Leaderer, B.; Lin, S.; Holford, T.R.; Qiu, J.; Zhang, Y.; Shi, K.; Zhu, Y.; et al. Prenatal exposure to organochlorine pesticides and infant birth weight in China. Chemosphere 2014, 110, 1–7. [Google Scholar] [CrossRef]
- Cabrera-Rodríguez, R.; Luzardo, O.P.; Almeida-González, M.; Boada, L.D.; Zumbado, M.; Acosta-Dacal, A.; Rial-Berriel, C.; Henríquez-Hernández, L.A. Association between prenatal exposure to multiple persistent organic pollutants (POPs) and growth indicators in newborns. Environ. Res. 2019, 171, 285–292. [Google Scholar] [CrossRef]
- Vafeiadi, M.; Georgiou, V.; Chalkiadaki, G.; Rantakokko, P.; Kiviranta, H.; Karachaliou, M.; Fthenou, E.; Venihaki, M.; Sarri, K.; Vassilaki, M.; et al. Association of prenatal exposure to persistent organic pollutants with obesity and cardiometabolic traits in early childhood: The Rhea mother-child cohort (Crete, Greece). Environ. Health Perspect. 2015, 123, 1015–1021. [Google Scholar] [CrossRef]
- Papadopoulou, E.; Vafeiadi, M.; Agramunt, S.; Mathianaki, K.; Karakosta, P.; Spanaki, A.; Besselink, H.; Kiviranta, H.; Rantakokko, P.; Koutis, A.; et al. Maternal diet, prenatal exposure to dioxins and other persistent organic pollutants and anogenital distance in children. Sci. Total Environ. 2013, 461, 222–229. [Google Scholar] [CrossRef]
- Hertz-Picciotto, I.; Park, H.-Y.; Dostal, M.; Kocan, A.; Trnovec, T.; Sram, R. Prenatal Exposures to persistent and non-persistent organic compounds and effects on immune system development. Basic Clin. Pharmacol. Toxicol. 2008, 102, 146–154. [Google Scholar] [CrossRef]
- Zong, G.; Valvi, D.; Coull, B.; Göen, T.; Hu, F.B.; Nielsen, F.; Grandjean, P.; Sun, Q. Persistent organic pollutants and risk of type 2 diabetes: A prospective investigation among middle-aged women in Nurses’ Health Study II. Environ. Int. 2018, 114, 334–342. [Google Scholar] [CrossRef]
- Multigner, L.; Kadhel, P.; Rouget, F.; Blanchet, P.; Cordier, S. Chlordecone exposure and adverse effects in French West Indies populations. Environ. Sci. Pollut. Res. Int. 2016, 23, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.-L.; Tsai, P.-C.; Yang, C.-Y.; Leon Guo, Y. Increased risk of diabetes and polychlorinated biphenyls and dioxins: A 24-year follow-up study of the Yucheng cohort. Diabetes Care 2008, 31, 1574–1579. [Google Scholar] [CrossRef] [PubMed]
- Lyche, J.L.; Rosseland, C.; Berge, G.; Polder, A. Human health risk associated with brominated flame-retardants (BFRs). Environ. Int. 2015, 74, 170–180. [Google Scholar] [CrossRef] [PubMed]
- Gregoraszczuk, E.L.; Ptak, A. Endocrine-Disrupting chemicals: Some actions of POPs on female reproduction. J. Endocrinol. 2013, 2013, 828532. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, N.; Ma, M.; Giesy, J.P.; Wang, Z. In vitro profiling of the endocrine disrupting potency of organochlorine pesticides. Toxicol. Lett. 2008, 183, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.T.; Lee, H.K. Childhood obesity and endocrine disrupting chemicals. Ann. Pediatr. Endocrinol. Metab. 2017, 22, 219–225. [Google Scholar] [CrossRef]
- Vandelac, L. Endocrine disruption agents: Environment, health, public policies, and the precautionary principle. Bull. Acad. Natl. Med. 2000, 184, 1477–1486. [Google Scholar]
- Sanderson, J.T. The steroid hormone biosynthesis pathway as a target for endocrine-disrupting chemicals. Toxicol. Sci. 2006, 94, 3–21. [Google Scholar] [CrossRef]
- Vallack, H.W.; Bakker, D.J.; Brandt, I.; Brostrom-Lunden, E.; Brouwer, A.; Bull, K.R.; Gough, C.; Guardans, R.; Holoubek, I.; Jansson, B.; et al. Controlling persistent organic pollutants—what next? Environ. Toxicol. Pharmacol. 1998, 6, 143–175. [Google Scholar] [CrossRef]
- Kumar, S. Occupational exposure associated with reproductive dysfunction. J. Occup. Health 2004, 46, 1–19. [Google Scholar] [CrossRef]
- Yu, H.-Y.; Guo, Y.; Zeng, E.Y. Dietary intake of persistent organic pollutants and potential health risks via consumption of global aquatic products. Environ. Toxicol. Chem. 2010, 29, 2135–2142. [Google Scholar] [CrossRef] [PubMed]
- Mathur, V.; Bhatnagar, P.; Sharma, R.G.; Acharya, V.; Sexana, R. Breast cancer incidence and exposure to pesticides among women originating from Jaipur. Environ. Int. 2002, 28, 331–336. [Google Scholar] [CrossRef]
- Arrebola, J.P.; Belhassen, H.; Artacho-Cordón, F.; Ghali, R.; Ghorbel, H.; Boussen, H.; Perez-Carrascosa, F.M.; Expósito, J.; Hedhili, A.; Olea, N. Risk of female breast cancer and serum concentrations of organochlorine pesticides and polychlorinated biphenyls: A case-Control study in Tunisia. Sci. Total Environ. 2015, 520, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Weiderpass, E.; Adami, H.-O.; Baron, J.A.; Wicklund-Glynn, A.; Aune, M.; Atuma, S.; Persson, I. Organochlorines and endometrial cancer risk. Cancer Epidemiol. Biomark. Prev. 2000, 9, 487. [Google Scholar]
- López-Carrillo, L.; Blair, A.; López-Cervantes, M.; Cebrián, M.; Rueda, C.; Reyes, R.; Mohar, A.; Bravo, J. Dichlorodiphenyltrichloroethane serum levels and breast cancer risk: A case-control study from Mexico. Cancer Res. 1997, 57, 3728. [Google Scholar]
- Moysich, K.B.; Ambrosone, C.B.; Vena, J.E.; Shields, P.G.; Mendola, P.; Kostyniak, P.; Greizerstein, H.; Graham, S.; Marshall, J.R.; Schisterman, E.F.; et al. Environmental organochlorine exposure and postmenopausal breast cancer risk. Cancer Epidemiol. Biomark. Prev. 1998, 7, 181. [Google Scholar]
- Krieger, N.; Wolff, M.S.; Hiatt, R.A.; Rivera, M.; Vogelman, J.; Orentreich, N. Breast cancer and serum organochlorines: A prospective study among white, black, and Asian women. J. Natl. Cancer Inst. 1994, 86, 589–599. [Google Scholar] [CrossRef]
- IARC. Some Non-Heterocyclic polycyclic aromatic hydrocarbons and some related exposures. IARC Monogr. Eval. Carcinog. Risks Hum. 2010, 92, 765–771. [Google Scholar]
- US EPA. Polycyclic Organic Matter; US EPA: Washington, DC, USA, 2002. Available online: https://www.epa.gov/sites/production/files/2016-09/documents/polycyclic-organic-matter.pdf (accessed on 31 October 2019).
- Lind, P.M.; van Bavel, B.; Salihovic, S.; Lind, L. Circulating levels of persistent organic pollutants (POPs) and carotid atherosclerosis in the elderly. Environ. Health Perspect. 2012, 120, 38–43. [Google Scholar] [CrossRef]
- Færch, K.; Højlund, K.; Vind, B.F.; Vaag, A.; Dalgård, C.; Nielsen, F.; Grandjean, P. Increased serum concentrations of persistent organic pollutants among prediabetic individuals: Potential role of altered substrate oxidation patterns. J. Clin. Endocrinol. Metab. 2012, 97, E1705–E1713. [Google Scholar] [CrossRef]
- Lee, D.-H.; Steffes, M.W.; Sjödin, A.; Jones, R.S.; Needham, L.L.; Jacobs, D.R., Jr. Low dose organochlorine pesticides and polychlorinated biphenyls predict obesity, dyslipidemia, and insulin resistance among people free of diabetes. PLoS ONE 2011, 6, e15977. [Google Scholar] [CrossRef] [PubMed]
- Pesatori, A.C.; Zocchetti, C.; Guercilena, S.; Consonni, D.; Turrini, D.; Bertazzi, P.A. Dioxin exposure and non-malignant health effects: A mortality study. Occup. Environ. Med. 1998, 55, 126. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Lessner, L.; Carpenter, D.O. Exposure to persistent organic pollutants and hypertensive disease. Environ. Res. 2006, 102, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Valera, B.; Ayotte, P.; Poirier, P.; Dewailly, É. Associations between plasma persistent organic pollutant levels and blood pressure in Inuit adults from Nunavik. Environ. Int. 2013, 59, 282–289. [Google Scholar] [CrossRef]
- Vafeiadi, M.; Roumeliotaki, T.; Chalkiadaki, G.; Rantakokko, P.; Kiviranta, H.; Fthenou, E.; Kyrtopoulos, S.A.; Kogevinas, M.; Chatzi, L. Persistent organic pollutants in early pregnancy and risk of gestational diabetes mellitus. Environ. Int. 2017, 98, 89–95. [Google Scholar] [CrossRef]
- Carpenter, D.O. Environmental contaminants as risk factors for developing diabetes. Rev. Environ. Health 2008, 23, 59–74. [Google Scholar] [CrossRef]
- Uemura, H.; Arisawa, K.; Hiyoshi, M.; Kitayama, A.; Takami, H.; Sawachika, F.; Dakeshita, S.; Nii, K.; Satoh, H.; Sumiyoshi, Y.; et al. Prevalence of metabolic syndrome associated with body burden levels of dioxin and related compounds among Japan’s general population. Environ. Health Perspect. 2009, 117, 568–573. [Google Scholar] [CrossRef]
- Ochiai, N.; Ieda, T.; Sasamoto, K.; Takazawa, Y.; Hashimoto, S.; Fushimi, A.; Tanabe, K. Stir bar sorptive extraction and comprehensive two-dimensional gas chromatography coupled to high-resolution time-of-flight mass spectrometry for ultra-trace analysis of organochlorine pesticides in river water. J. Chromatogr. A 2011, 1218, 6851–6860. [Google Scholar] [CrossRef]
- Wilkowska, A.M.; Biziuk, M. Rapid method for the determination of organochlorine pesticides and PCBs in fish muscle samples by microwave-assisted extraction and analysis of extracts by GC-ECD. J. AOAC Int. 2010, 93, 1987–1994. [Google Scholar]
- Muscalu, A.M.; Górecki, T. Comprehensive two-dimensional gas chromatography in environmental analysis. TrAC Trends Anal. Chem. 2018, 106, 225–245. [Google Scholar] [CrossRef]
- Fiddler, W.; Pensabene, J.W.; Gates, R.A.; Donoghue, D.J. Supercritical fluid extraction of organochlorine pesticides in eggs. J. Agric. Food. Chem. 1999, 47, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Hopper, M.L. Automated one-step supercritical fluid extraction and clean-up system for the analysis of pesticide residues in fatty matrices. J. Chromatogr. A 1999, 840, 93–105. [Google Scholar] [CrossRef]
- Weichbrodt, M.; Vetter, W.; Luckas, B. Microwave-assisted extraction and accelerated solvent extraction with ethyl acetate-cyclohexane before determination of organochlorines in fish tissue by gas chromatography with electron-capture detection. J. AOAC Int. 2000, 83, 1334–1343. [Google Scholar] [PubMed]
- Pare, J.R.; Matni, G.; Belanger, J.M.; Li, K.; Rule, C.; Thibert, B.; Yaylayan, V.; Liu, Z.; Mathe, D.; Jacquault, P. Use of the Microwave-Assisted Process in extraction of fat from meat, dairy, and egg products under atmospheric pressure conditions. J. AOAC Int. 1997, 80, 928–933. [Google Scholar] [PubMed]
- Vetter, W.; Weichbrodt, M.; Hummert, K.; Glotz, D.; Luckas, B. Combined microwave-assisted extraction and gel permeation chromatography for the determination of chlorinated hydrocarbons in seal blubber and cod livers. Chemosphere 1998, 37, 2439–2449. [Google Scholar] [CrossRef]
- Labadie, P.; Alliot, F.; Bourges, C.; Desportes, A.; Chevreuil, M. Determination of polybrominated diphenyl ethers in fish tissues by matrix solid-phase dispersion and gas chromatography coupled to triple quadrupole mass spectrometry: Case study on European eel (Anguilla anguilla) from Mediterranean coastal lagoons. Anal. Chim. Acta 2010, 675, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Schenck, F.J.; Donoghue, D.J. Determination of organochlorine and organophosphorus pesticide residues in eggs using a solid phase extraction cleanup. J. Agric. Food. Chem. 2000, 48, 6412–6415. [Google Scholar] [CrossRef]
- De Fátima Alpendurada, M. Solid-phase microextraction: A promising technique for sample preparation in environmental analysis. J. Chromatogr. A 2000, 889, 3–14. [Google Scholar] [CrossRef]
- Fernandes, A.R.; Rose, M.; Mortimer, D.; Carr, M.; Panton, S.; Smith, F. Mixed brominated/chlorinated dibenzo-p-dioxins, dibenzofurans and biphenyls: Simultaneous congener-selective determination in food. J. Chromatogr. A 2011, 1218, 9279–9287. [Google Scholar] [CrossRef]
- Souza Tette, P.A.; Rocha Guidi, L.; de Abreu Glória, M.B.; Fernandes, C. Pesticides in honey: A review on chromatographic analytical methods. Talanta 2016, 149, 124–141. [Google Scholar] [CrossRef]
- Montory, M.; Habit, E.; Fernandez, P.; Grimalt, J.O.; Barra, R. PCBs and PBDEs in wild Chinook salmon (Oncorhynchus tshawytscha) in the Northern Patagonia, Chile. Chemosphere 2010, 78, 1193–1199. [Google Scholar] [CrossRef] [PubMed]
- Halbert, M.K.; Archer, J.C. Dioxin and furan contamination of deodorizer distillates and natural vitamin E supplements. J. Food Compos. Anal. 2007, 20, 506–514. [Google Scholar] [CrossRef]
- Voorspoels, S.; Covaci, A.; Schepens, P. Polybrominated Diphenyl ethers in marine species from the Belgian North Sea and the Western Scheldt Estuary: Levels, profiles, and distribution. Environ. Sci. Technol. 2003, 37, 4348–4357. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Reiner, E.J.; Bhavsar, S.P.; Helm, P.A.; Mabury, S.A.; Braekevelt, E.; Tittlemier, S.A. Determination of polyfluoroalkyl phosphoric acid diesters, perfluoroalkyl phosphonic acids, perfluoroalkyl phosphinic acids, perfluoroalkyl carboxylic acids, and perfluoroalkane sulfonic acids in lake trout from the Great Lakes region. Anal. Bioanal. Chem. 2012, 404, 2699–2709. [Google Scholar] [CrossRef] [PubMed]
- Ghidini, S.; Zanardi, E.; Battaglia, A.; Varisco, G.; Ferretti, E.; Campanini, G.; Chizzolini, R. Comparison of contaminant and residue levels in organic and conventional milk and meat products from northern Italy. Food Addit. Contam. 2005, 22, 9–14. [Google Scholar] [CrossRef]
- Wei, S.; Leong, M.; Li, Y.; Huang, S. Development of liquid phase microextraction based on manual shaking and ultrasound-assisted emulsification method for analysis of organochlorine pesticides in aqueous samples. J. Chromatogr. A 2011, 1218, 9142–9148. [Google Scholar] [CrossRef]
- Zgoła-Grześkowiak, A.; Grześkowiak, T. Dispersive liquid-liquid microextraction. TrAC Trends Anal. Chem. 2011, 30, 1382–1399. [Google Scholar] [CrossRef]
- Andrade-Eiroa, A.; Canle, M.; Leroy-Cancellieri, V.; Cerdà, V. Solid-phase extraction of organic compounds: A critical review (Part I). TrAC Trends Anal. Chem. 2016, 80, 641–654. [Google Scholar] [CrossRef]
- Hennion, M.-C. Solid-phase extraction: Method development, sorbents, and coupling with liquid chromatography. J. Chromatogr. A 1999, 856, 3–54. [Google Scholar] [CrossRef]
- Hawthorne, S.B.; Grabanski, C.B.; Miller, D.J. Solid-phase-microextraction measurement of 62 polychlorinated biphenyl congeners in milliliter sediment pore water samples and determination of K(DOC) values. Anal. Chem. 2009, 81, 6936–6943. [Google Scholar] [CrossRef]
- Camino-Sanchez, F.J.; Zafra-Gomez, A.; Cantarero-Malagon, S.; Vilchez, J.L. Validation of a method for the analysis of 77 priority persistent organic pollutants in river water by stir bar sorptive extraction in compliance with the European Water Framework Directive. Talanta 2012, 89, 322–334. [Google Scholar] [CrossRef] [PubMed]
- Camino-Sanchez, F.J.; Zafra-Gomez, A.; Perez-Trujillo, J.P.; Conde-Gonzalez, J.E.; Marques, J.C.; Vilchez, J.L. Validation of a GC-MS/MS method for simultaneous determination of 86 persistent organic pollutants in marine sediments by pressurized liquid extraction followed by stir bar sorptive extraction. Chemosphere 2011, 84, 869–881. [Google Scholar] [CrossRef] [PubMed]
- Farre, M.; Kantiani, L.; Petrovic, M.; Perez, S.; Barcelo, D. Achievements and future trends in the analysis of emerging organic contaminants in environmental samples by mass spectrometry and bioanalytical techniques. J. Chromatogr. A 2012, 1259, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Juhler, R.K. Supercritical fluid extraction of pesticides from meat: A systematic approach for optimisation. Analyst 1998, 123, 1551–1556. [Google Scholar] [CrossRef] [PubMed]
- Carabias-Martínez, R.; Rodríguez-Gonzalo, E.; Revilla-Ruiz, P.; Hernández-Méndez, J. Pressurized liquid extraction in the analysis of food and biological samples. J. Chromatogr. A 2005, 1089, 1–17. [Google Scholar] [CrossRef]
- Gilbert-López, B.; García-Reyes, J.F.; Molina-Díaz, A. Sample treatment and determination of pesticide residues in fatty vegetable matrices: A review. Talanta 2009, 79, 109–128. [Google Scholar] [CrossRef]
- LeDoux, M. Analytical methods applied to the determination of pesticide residues in foods of animal origin. A review of the past two decades. J. Chromatogr. A 2011, 1218, 1021–1036. [Google Scholar] [CrossRef]
- Geng, D.; Kukucka, P.; Jogsten, I.E. Analysis of brominated flame retardants and their derivatives by atmospheric pressure chemical ionization using gas chromatography coupled to tandem quadrupole mass spectrometry. Talanta 2017, 162, 618–624. [Google Scholar] [CrossRef]
- Rivera-Austrui, J.; Martinez, K.; Abalos, M.; Sales, C.; Portoles, T.; Beltran, J.; Saulo, J.; Aristizabal, B.H.; Abad, E. Analysis of polychlorinated dibenzo-p-dioxins and dibenzofurans in stack gas emissions by gas chromatography-atmospheric pressure chemical ionization-triple-quadrupole mass spectrometry. J. Chromatogr. A 2017, 1513, 245–249. [Google Scholar] [CrossRef]
- Geng, D.; Jogsten, I.E.; Dunstan, J.; Hagberg, J.; Wang, T.; Ruzzin, J.; Rabasa-Lhoret, R.; van Bavel, B. Gas chromatography/atmospheric pressure chemical ionization/mass spectrometry for the analysis of organochlorine pesticides and polychlorinated biphenyls in human serum. J. Chromatogr. A 2016, 1453, 88–98. [Google Scholar] [CrossRef]
- Hagberg, J. Analysis of brominated dioxins and furans by high resolution gas chromatography/high resolution mass spectrometry. J. Chromatogr. A 2009, 1216, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Alder, L.; Greulich, K.; Kempe, G.; Vieth, B. Residue analysis of 500 high priority pesticides: Better by GC–MS or LC–MS/MS? Mass Spectrom. Rev. 2006, 25, 838–865. [Google Scholar] [CrossRef] [PubMed]
- Garrido Frenich, A.; Martínez Vidal, J.L.; Moreno Frías, M.; Olea-Serrano, F.; Olea, N.; Cuadros Rodriguez, L. Determination of organochlorine pesticides by GC-ECD and GC-MS-MS techniques including an evaluation of the uncertainty associated with the results. Chromatographia 2003, 57, 213–220. [Google Scholar] [CrossRef]
- Zrostlíková, J.; Lehotay, S.J.; Hajšlová, J. Simultaneous analysis of organophosphorus and organochlorine pesticides in animal fat by gas chromatography with pulsed flame photometric and micro-electron capture detectors. J. Sep. Sci. 2002, 25, 527–537. [Google Scholar] [CrossRef]
- Focant, J.-F.; Pirard, C.; Eppe, G.; De Pauw, E. Recent advances in mass spectrometric measurement of dioxins. J. Chromatogr. A 2005, 1067, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Archer, J.; Moore, M.; Bruce, J.; McLain, M.; Shojaee, S.; Zou, W.; Benjamin, L.A.; Adeuya, A.; Fairchild, R.; et al. QUICK: Quality and usability investigation and control kit for mass spectrometric data from detection of persistent organic pollutants. Int. J. Environ. Res. Public Health 2019, 16, 4203. [Google Scholar] [CrossRef] [PubMed]
- Abelkop, A.D.K.; Graham, J.D.; Royer, T.V. Persistent, Bioaccumulative, and Toxic (PBT) Chemicals: Technical Aspects, Policies, and Practices; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar] [CrossRef]
- Matthies, M.; Solomon, K.; Vighi, M.; Gilman, A.; Tarazona, J.V. The origin and evolution of assessment criteria for persistent, bioaccumulative and toxic (PBT) chemicals and persistent organic pollutants (POPs). Environ. Sci. Process. Impacts 2016, 18, 1114–1128. [Google Scholar] [CrossRef]
- European Community. Regulation (EU) No 253/2011 of 15 March 2011 Amending Regulation (EC) No 1907/2006 of the European Parliament and of the Council on the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) as Regards Annex XIII. Off. J. Eur. Community 2011, 54, 7–12. [Google Scholar]
- EP&C. Regulation (EC) No 1107/2009 of the European Parliament and of the Council of 21 October 2009 Concerning the Placing of Plant Protection Products on the Market and Repealing Council Directives 79/117/EEC and 91/414/EEC (91/414/EEC). Off. J. Eur. Union 2009, 52, 1–50. [Google Scholar]
- North American Agreement on Environmental Cooperation (NAAEC)-Commission for Environmental Cooperation (CEC). Process for Identifying Candidate Substances for Regional Action Under the Sound Management of Chemicals Initiative: Report to the North American Working Group on the Sound Management of Chemicals by the Task Force on Criteria; CEC Report No. NAAEC-CEC: Montreal, QC, Canada, 1997; Available online: http://www3.cec.org/islandora/en/item/1750-process-identifying-candidate-substances-regional-action-en.pdf (accessed on 31 October 2019).
- Environment Canada. Toxic Substances Management Policy; Environment Canada Report No. En 40-499/1-1995; Government of Canada: Ottawa, ON, Canada, 1995; p. 18.
- US EPA. Category for persistent, bioaccumulative, and toxic new chemical substances. Fed. Regist. 1999, 64, 20194–60204. [Google Scholar]
- Sharma, B.M.; Bharat, G.K.; Tayal, S.; Nizzetto, L.; Larssen, T. The legal framework to manage chemical pollution in India and the lesson from the Persistent Organic Pollutants (POPs). Sci. Total Environ. 2014, 490, 733–747. [Google Scholar] [CrossRef] [PubMed]
- Lau, M.H.Y.; Leung, K.M.Y.; Wong, S.W.Y.; Wang, H.; Yan, Z.-G. Environmental policy, legislation and management of persistent organic pollutants (POPs) in China. Environ. Pollut. 2012, 165, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.; MacLeod, M.; Hung, H.; Cousins, I.T. Statistical analysis of long-term monitoring data for persistent organic pollutants in the atmosphere at 20 monitoring stations broadly indicates declining concentrations. Environ. Sci. Technol. 2014, 48, 12492–12499. [Google Scholar] [CrossRef] [PubMed]
- Vigh, É.; Colombo, A.; Benfenati, E.; Håkansson, H.; Berglund, M.; Bódis, J.; Garai, J. Individual breast milk consumption and exposure to PCBs and PCDD/Fs in Hungarian infants: A time-course analysis of the first three months of lactation. Sci. Total Environ. 2013, 449, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Vogt, R.; Bennett, D.; Cassady, D.; Frost, J.; Ritz, B.; Hertz-Picciotto, I. Cancer and non-cancer health effects from food contaminant exposures for children and adults in California: A risk assessment. Environ. Health 2012, 11, 83. [Google Scholar] [CrossRef]
- Van Leeuwen, F.X.R.; Feeley, M.; Schrenk, D.; Larsen, J.C.; Farland, W.; Younes, M. Dioxins: WHO’s tolerable daily intake (TDI) revisited. Chemosphere 2000, 40, 1095–1101. [Google Scholar] [CrossRef]
- EC. Commission Regulation (EU) No 1259/2011 of 2 December 2011 amending Regulation (EC) No 1881/2006 as regards maximum levels for dioxins, dioxin-like PCBs and non dioxin-like PCBs in foodstuffs. Off. J. Eur. Union 2011, 320, 18–23. [Google Scholar]
- Perelo, L.W. Review: In situ and bioremediation of organic pollutants in aquatic sediments. J. Hazard. Mater. 2010, 177, 81–89. [Google Scholar] [CrossRef]
- Maftoonazad, N.; Badii, F. Use of edible films and coatings to extend the shelf life of food products. Recent Pat. Food Nutr. Agric. 2009, 1, 162–170. [Google Scholar] [CrossRef]
- Wang, R.Y.; Needham, L.L. Environmental chemicals: From the environment to food, to breast milk, to the infant. J. Toxicol. Env. Heal. B 2007, 10, 597–609. [Google Scholar] [CrossRef]
- Muir, D.C.G.; Howard, P.H. Are there other persistent organic pollutants? A challenge for environmental chemists. Environ. Sci. Technol. 2006, 40, 7157–7166. [Google Scholar] [CrossRef] [PubMed]
- Ruzzin, J. Public health concern behind the exposure to persistent organic pollutants and the risk of metabolic diseases. BMC Public Health 2012, 12, 298. [Google Scholar] [CrossRef] [PubMed]
- WHO (World Health Organisation). Consultation on assessment of the health risk of dioxins; re-evaluation of the tolerable daily intake (TDI). Food Addit. Contam. 1998, 17, 223–240. [Google Scholar]
- EFSA. Risk for animal and human health related to the presence of dioxins and dioxin-like PCBs in feed and food. EFSA J. 2018, 16, e05333. [Google Scholar] [CrossRef]
- JECFA. Evaluation of certain food additives and contaminants. In Proceedings of the Summary of the fifty-seventh meeting of the Joint FAO/WHO Expert Committee on Food Additives, Rome, Italy, 5–14 June 2001; pp. 24–40. Available online: https://apps.who.int/iris/handle/10665/42578 (accessed on 31 October 2019).
- EU. Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, 364, 324–365. [Google Scholar]
- The International Programme on Chemical Safety. Inventory of IPCS and Other WHO Pesticide Evaluations and Summary of Toxicological Evaluations Performed by the Joint Meeting on Pesticide Residues (JMPR) through 2009; World Health Organization: Geneva, Switzerland, 2009; Available online: https://www.who.int/ipcs/publications/jmpr/pesticide_inventory_edition10.pdf (accessed on 31 October 2019).
- EU. No 835/2011 of 19 August 2011 amending Regulation (EC) No 1881/2006 as regards maximum levels for polycyclic aromatic hydrocarbons in foodstuffs. Off. J. Eur. Union 2011, 215, 1–5. [Google Scholar]
| POPs Class | POPs | Structure | Reference |
|---|---|---|---|
| Chlorodibenzo-p- dioxin (CDD) | 2,3,7,8-tetraCDD; 1,2,3,7,8-pentaCDD; 1,2,3,4,7,8-hexaCDD; 1,2,3,6,7,8-hexaCDD; 1,2,3,7,8,9-hexaCDD; 1,2,3,4,6,7,8-heptaCDD; octaCDD | 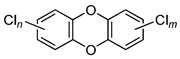 | [13,14,15,16,17,18,19,20] |
| Chlorodibenzo furan (CDF) | 2,3,7,8-tetraCDF; 1,2,3,7,8-pentaCDF; 2,3,4,7,8-pentaCDF; 1,2,3,4,7,8-hexaCDF; 1,2,3,6,7,8-hexaCDF; 2,3,4,6,7,8-hexaCDF; 1,2,3,7,8,9-hexaCDF; 1,2,3,4,6,7,8-heptaCDF; 1,2,3,4,7,8,9-heptaCDF; octaCDF | 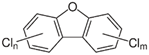 | [13,14,16,17,18,19,20] |
| Polychlorinated biphenyls (PCBs) | PCB-28; PCB-52; PCB-70; PCB-77; PCB-81; PCB-101; PCB-105; PCB-114; PCB-118; PCB-123; PCB-126; PCB-138; PCB-153; PCB-156; PCB-157; PCB-167; PCB-169; PCB-170; PCB-180; PCB-189 | 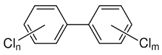 | [13,15,16,18,19,20,21,22,23,24] |
| Polybrominated diphenyl ethers (PBDEs) | pentaBDE; decaBDE; heptaBDE | 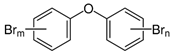 | [22,23] |
| Hexabromocyclododecanes (HBCDs) | α-HBCD β-HBCD γ-HBCD | 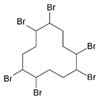 | [25] |
| Hexabromobiphenyl | hexabromobiphenyl | 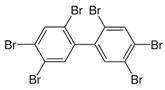 | [26] |
| Hexachlorobutadiene (HCBD) | HCBD | 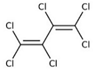 | [27] |
| Polychlorinated naphthalenes (PCNs) | PCN | 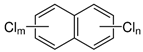 | [28] |
| Short-chain chlorinated paraffins (SCCPs) | SCCPs(C10–13) | 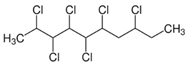 | [27] |
| Organochlorine pesticide (OCPs) | p,p′-dichlorodiphenyltrichloroethane (DDT) | 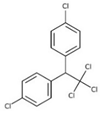 | [8,21,29,30,31,32] |
| o, p′-DDT |  | ||
| p, p′-dichlorodiphenyldichloroethylene | 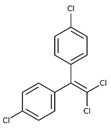 | ||
| p, p′-dichlorodiphenyldichloroethane |  | ||
| o,p′-dichlorodiphenyldichloroethane |  | ||
| o,p′-dichlorodiphenyldichloroethylene |  | ||
| p,p′-1-chloro-2,2-(bis-(4-chlorophenyl)ethylene | 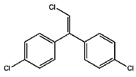 | ||
| p,p′-1-chloro-2,2-bis(p-chlorophenyl)ethane | 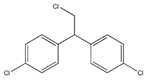 | ||
| cis-chlordane(α-chlordane); trans-chlordane(γ-chlordane) | 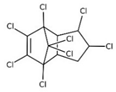 | ||
| cis nonachlor; trans-nonachlor |  | ||
| oxychlordane |  | ||
| heptachlor |  | ||
| aldrin | 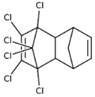 | ||
| α-endosulfan; β-endosulfan | 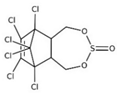 | ||
| endosulfan sulfate |  | ||
| α-hexachlorocyclohexane (α-HCH); β-hexachlorocyclohexane (β-HCH); γ-hexachlorocyclohexane (lindane) |  | ||
| Hexachlorobenzene (HCB) | 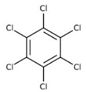 | ||
| dieldrin; endrin | 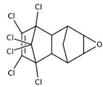 | ||
| mirex |  | ||
| chlordecone | 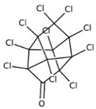 | ||
| pentachlorophenol (PCP) |  | ||
| pentachlorobenzene (PeCB) | 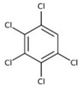 | ||
| toxaphene | 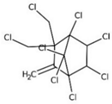 | ||
| Perfluorinated compounds | perfluorooctanesulfonate (PFOS) | 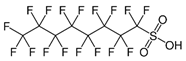 | [33] |
| Perfluorooctanoic acid (PFOA) | 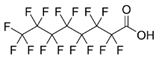 | ||
| Polyaromatic hydrocarbons (PAHs) | anthracene | 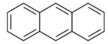 | [34] |
| pyrene | 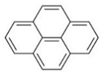 | ||
| benzo(a)anthracene | 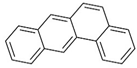 | ||
| benzo(k)fluoranthene | 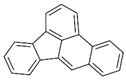 | ||
| benzo(a)pyrene | 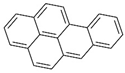 | ||
| indeno(1,2,3cd)pyrene | 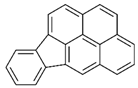 | ||
| dibenzo(a,h)anthracene | 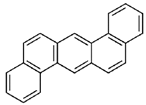 | ||
| benzo(g,h,i)perylene | 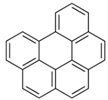 |
| Foodstuff | POPs | Reference |
|---|---|---|
| Egg | Dioxins/furans, PCBs, OCPs, PFCs and HBCDs | [15,20,23,28,29,52,70] |
| Dairy product (milk, butter, cheese, cream, yogurt, ice cream, etc.) | Dioxins/furans, PCBs, OCPs and PAHs | [13,15,20,29,42,71,72] |
| Meat and meat product (pork, chicken, beef, sausage, etc.) | Dioxins/furans, PCBs, OCPs, HCBD and PCN | [15,20,28,31,32,52,70] |
| Grain, flour and bran | PAHs | [71] |
| Rice, Fruit and vegetable (cabbage, carrot, potato, etc.) | OCPs, PCBs and PAHs | [3,42,73,74] |
| Honey | OCPs | [71] |
| Oil (vegetable oil, olive oil, etc.) | Dioxins/furans, PCBs, OCPs and HBCDs | [18,31,52] |
| Fish | OCPs, PCBs, PBDEs, PFOS, Dioxins/furans and HBCDs | [20,21,22,23,25,26,27,28,29,59,72,75] |
| Mussel | OCPs, PCBs and PBDEs | [26,30,76] |
| Oyster | PAHs | [71] |
| Water | PFOS, OCPs, PCBs and PAHs | [3,26] |
| POP | Possible Hazards | Reference |
|---|---|---|
| PAHs | Mutagenicity and carcinogenicity, DNA damage, oxidative stress, impaired male fertility, respiratory diseases, cognitive dysfunction among children and cancer (breast cancer) | [11,28,71,88] |
| OCPs | Neurological symptoms, endocrine disruption, infertility and fetal malformation, diabetes, cancer (breast cancer, testicular, prostate and kidney cancer), reproductive problems, cardiovascular problems, high blood pressure, glucose intolerance and obesity | [11,28,40,127,128] |
| Dioxins/furans | Language delay, disturbances in mental and motor development, cancer, diabetes, endocrine disruption, high blood pressure, glucose intolerance and cardiovascular problems | [11,28,129] |
| PCBs | Endocrine disruption, neurological disorders, liver injury, diabetes, cancer (breast, prostate, testicular, kidney, ovarian and uterine cancers), cardiovascular problems and obesity | [11,28,129] |
| PBDE | Reproductive problems, cancer(testicular), diabetes, obesity and cardiovascular problems | [11] |
| PFOS and PFOA | Breast cancer | [11] |
| HBCD | Endocrine disruption, reproductive problems and behavioral effects | [130] |
| PCN | Cancers | [28] |
| PCDE | Cancers | [28] |
| Type | Method | Description | Reference |
|---|---|---|---|
| Extraction | Soxhlet extraction (SOX) | Suitable for solid samples; efficient but time consuming and possible low analyte recovery | [18,22,26,29] |
| Solid–liquid extraction (SLE) | Suitable for solid samples; expensive and uses large volumes of organic solvents | [29] | |
| Pressurized liquid extraction (PLE) | Suitable for solid samples; highly automated but need expensive specialized equipment | [29] | |
| Supercritical fluid extraction (SFE) | Suitable for solid matrices; high efficiency, selectivity and low solvent volume, but need clean-up step | [29,159,160] | |
| Microwave-assisted extraction (MAE) | Suitable for solid samples; high efficiency but need clean-up step | [29,157,161,162,163] | |
| Ultrasonic-assisted extraction (UAE) | Suitable for solid samples; require low solvent volumes but need to optimize different operating factors | [70] | |
| Matrix solid-phase dispersion (MSPD) | Suitable for solid, semi-solid and viscous sample matrices; combines extraction and cleanup within a single step but need trials and errors to pick the right sorbent | [164] | |
| Liquid–liquid extraction (LLE) | Suitable for liquid/aqueous sample; high efficiency and selectivity but tedious and requires large amounts of organic solvents | [29] | |
| Solid-phase extraction (SPE) | Suitable for aqueous/liquid samples; requires large sample volumes | [26,165] | |
| Stir bar sorptive extraction (SBSE) | Suitable for liquid/aqueous samples; simple and solvent-less, but not suitable for polar compounds | [65,156] | |
| Solid-phase microextraction (SPME) | Suitable for liquid/aqueous samples; simple, solvent-less, less sample loss and contamination, but may need a clean-up process | [26,166] | |
| Separation | Gas chromatography (GC) | Good separation potential but restricted to use on more volatile compounds, e.g., high-resolution gas chromatography (HRGC), Atmospheric Pressure Gas Chromatography (APGC) | [3,18,59,167] |
| Liquid chromatography (LC) | Good for polar water-soluble class of chemicals; poor separation potential, e.g., High-Pressure Liquid Chromatography (HPLC) | [4,168] | |
| GC×GC | Good separation potential but restricted on more volatile compounds | [14,164] | |
| Detection | Electron capture detector | Most commonly used detection method with low detection limits | [29,168,169] |
| Mass spectrometry (MS) in the negative chemical ionization mode | Better sensitivity but restricted on non-polar POPs | [169] | |
| MS in the electron ionization mode | Better sensitivity and selectivity due to abundant fragmentation but restricted on non-polar POPs | [18,170] | |
| MS in the selected ion monitoring mode | Better sensitivity but the selected ion window may need to be monitored | [32,171] | |
| High-resolution mass spectrometry (HRMS) | High sensitivity but expensive | [14,18,59] | |
| MS/MS | Improves sensitivity and selectivity compared to single quadrupole MS, e.g., ion trap MS/MS; triple quadrupole MS/MS | [14,88,107,164,172] | |
| Time-of-flight TOF–MS | Wide mass analysis range but poor instrument limits of detection | [14,47] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, W.; Pan, B.; Sakkiah, S.; Yavas, G.; Ge, W.; Zou, W.; Tong, W.; Hong, H. Persistent Organic Pollutants in Food: Contamination Sources, Health Effects and Detection Methods. Int. J. Environ. Res. Public Health 2019, 16, 4361. https://doi.org/10.3390/ijerph16224361
Guo W, Pan B, Sakkiah S, Yavas G, Ge W, Zou W, Tong W, Hong H. Persistent Organic Pollutants in Food: Contamination Sources, Health Effects and Detection Methods. International Journal of Environmental Research and Public Health. 2019; 16(22):4361. https://doi.org/10.3390/ijerph16224361
Chicago/Turabian StyleGuo, Wenjing, Bohu Pan, Sugunadevi Sakkiah, Gokhan Yavas, Weigong Ge, Wen Zou, Weida Tong, and Huixiao Hong. 2019. "Persistent Organic Pollutants in Food: Contamination Sources, Health Effects and Detection Methods" International Journal of Environmental Research and Public Health 16, no. 22: 4361. https://doi.org/10.3390/ijerph16224361
APA StyleGuo, W., Pan, B., Sakkiah, S., Yavas, G., Ge, W., Zou, W., Tong, W., & Hong, H. (2019). Persistent Organic Pollutants in Food: Contamination Sources, Health Effects and Detection Methods. International Journal of Environmental Research and Public Health, 16(22), 4361. https://doi.org/10.3390/ijerph16224361






