Concentrations of Atmospheric Culturable Bioaerosols at Mountain and Seashore Sites
Abstract
1. Introduction
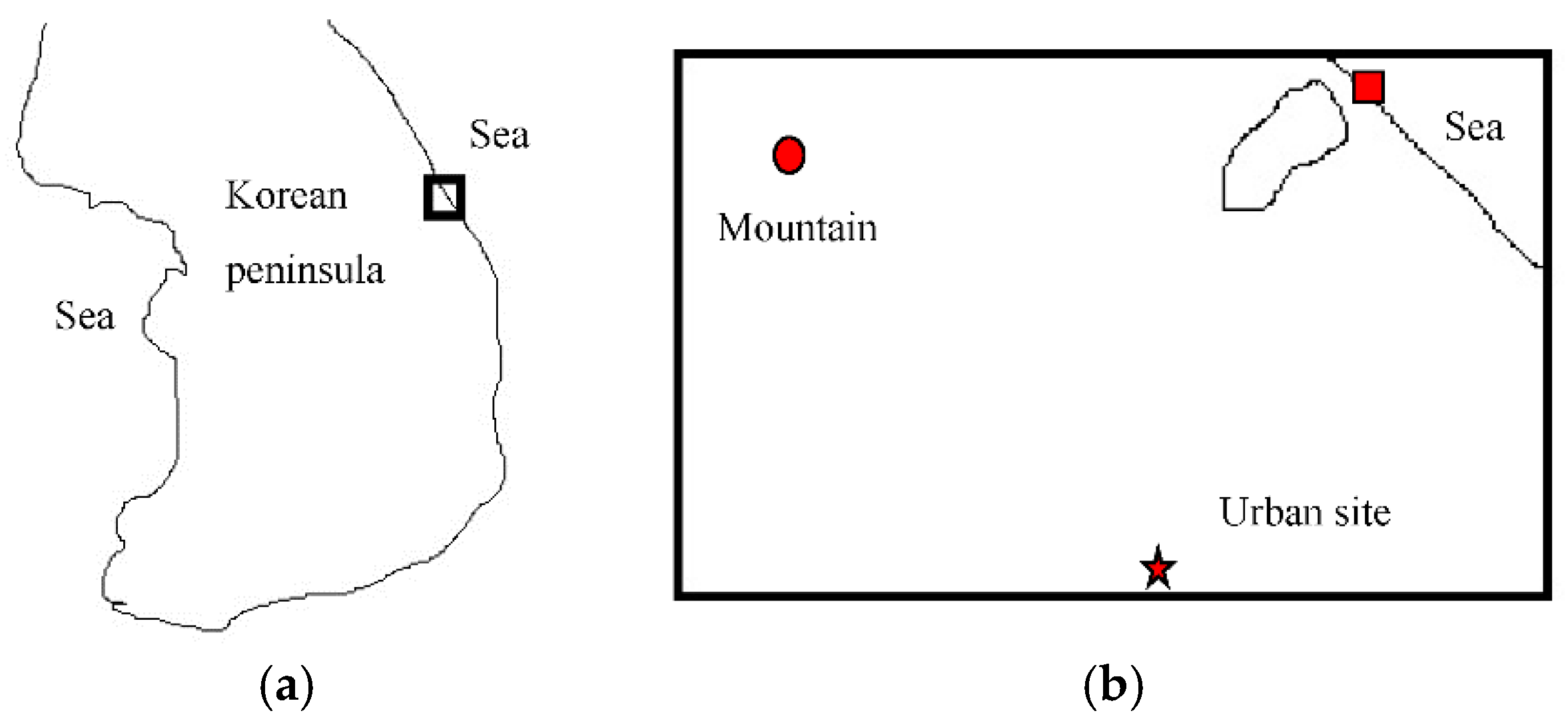
2. Experiments
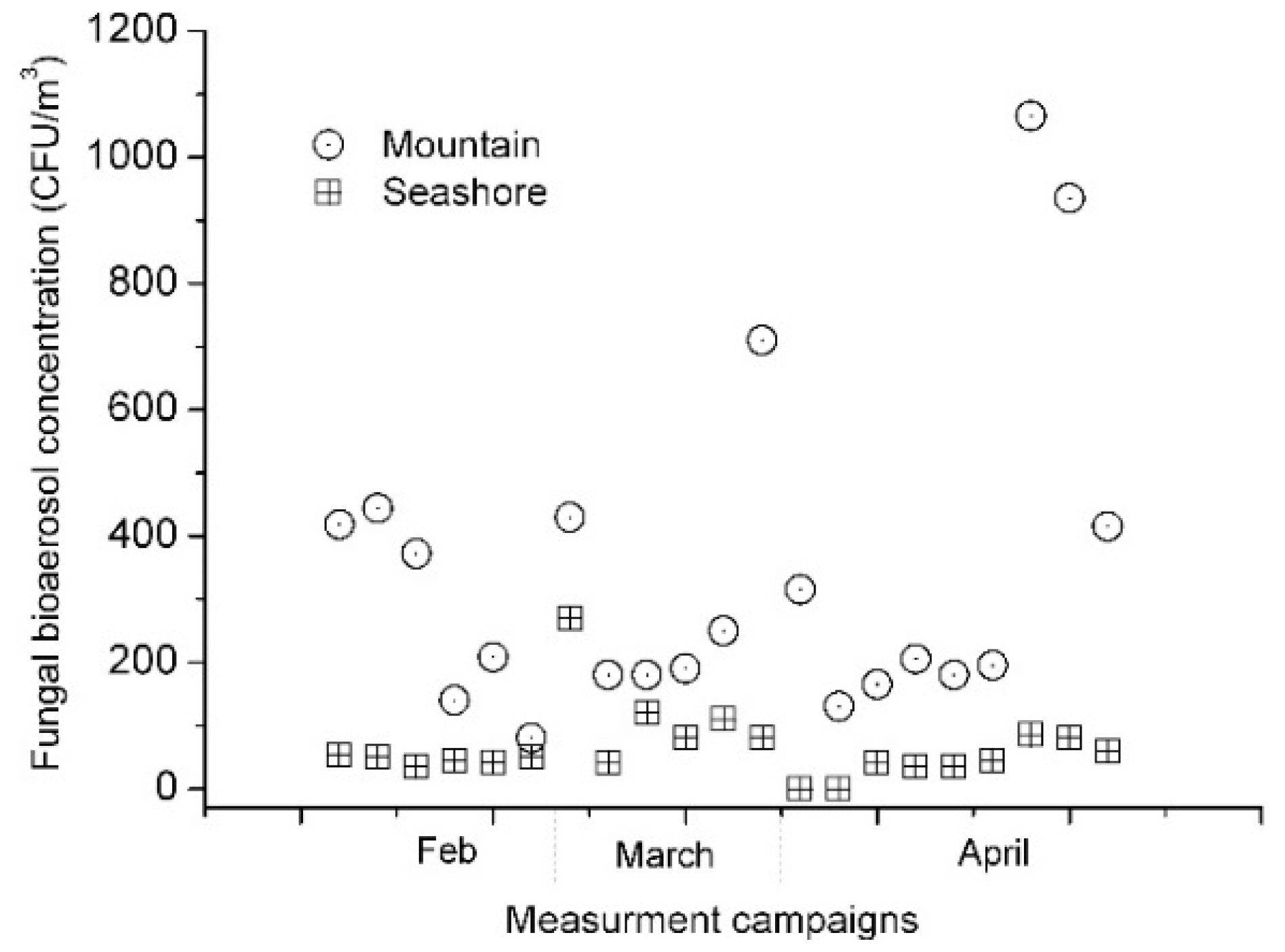
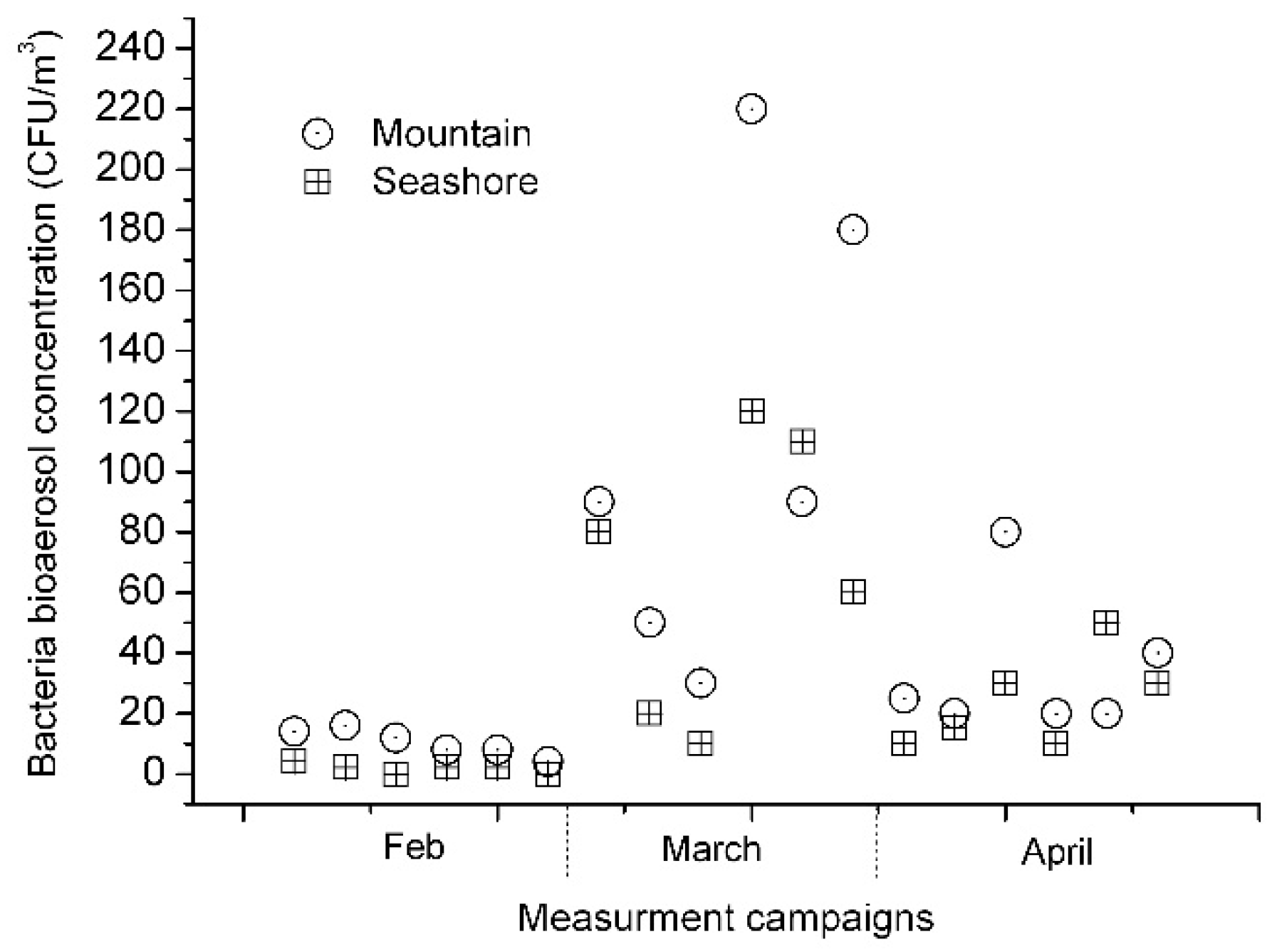
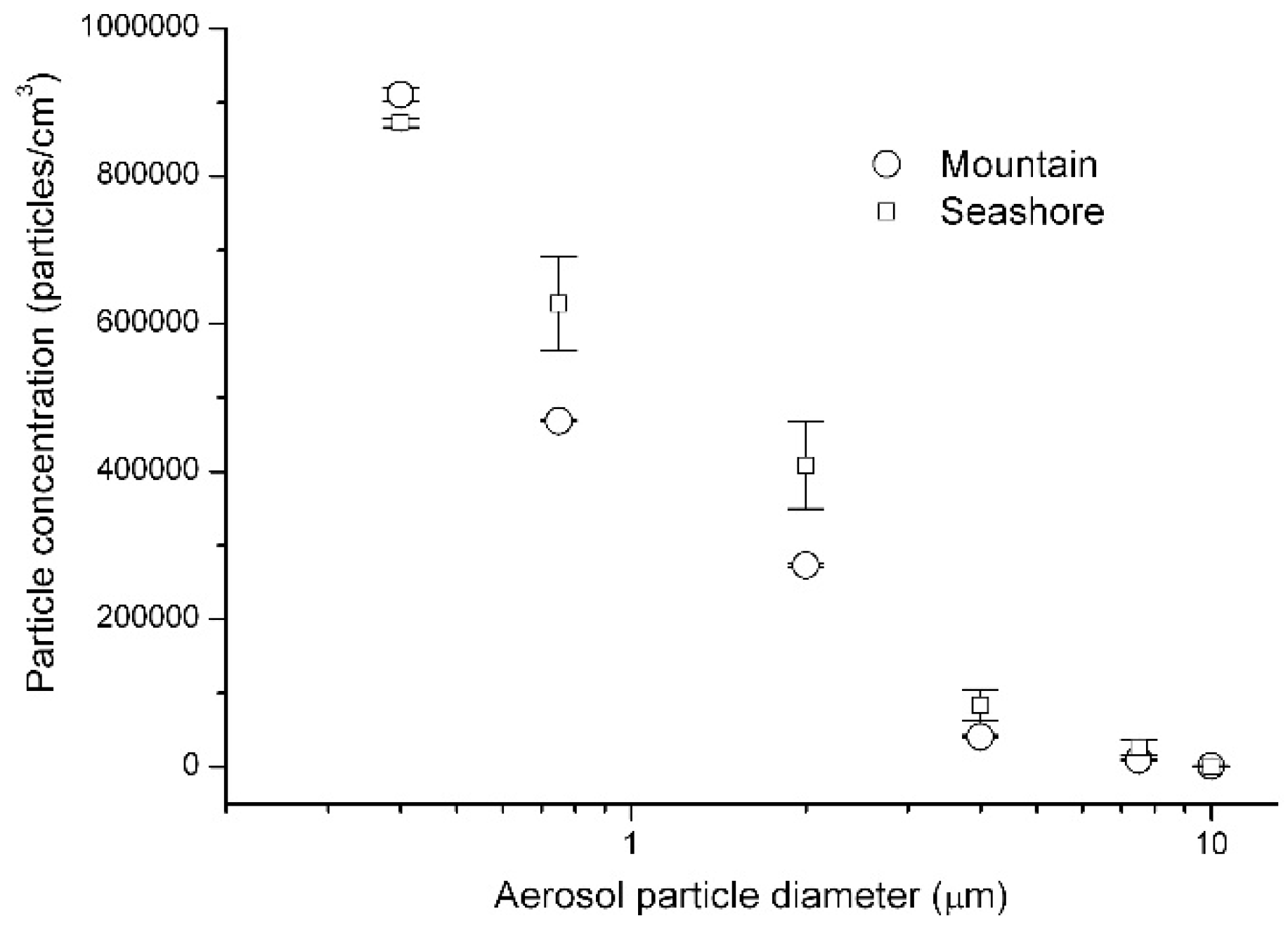
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Arancibia, F.; Bauer, T.T.; Ewig, S.; Mensa, J.; Gonzalez, J.; Niederman, M.S.; Torres, A. Community-acquired pneumonia due to gram-negative bacteria and Pseudomonas aeruginosa: Incidence, risk, and prognosis. Arch. Intern. Med. 2002, 162, 1849–1858. [Google Scholar] [CrossRef] [PubMed]
- LaForce, F.M. Airborne infections and modern building technology. Environ. Int. 1986, 12, 137–146. [Google Scholar] [CrossRef]
- Bush, R.K.; Portnoy, J.M. The role and abatement of fungal allergens in allergic diseases. J. Allergy Clin. Immunol. 2001, 107, S430–S440. [Google Scholar] [CrossRef] [PubMed]
- Cockcroft, D.W.; Ruffin, R.E.; Dolovich, J.; Hargreave, F.E. Allergen-induced increase in non-allergic bronchial reactivity. Clin. Exp. Allergy 1977, 7, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Fiegel, J.; Clarke, R.; Edwards, D.A. Airborne infectious disease and the suppression of pulmonary bioaerosols. Drug Discov. Today 2006, 11, 51–57. [Google Scholar] [CrossRef]
- Fung, F.; Hughson, W.G. Health effects of indoor fungal bioaerosol exposure. Appl. Occup. Environ. Hyg. 2010, 18, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Zuskin, E.; Schachter, E.N.; Kanceljak, B.; Mustajbegovic, J.; Witek, T.J. Immunological and respiratory reactions in workers exposed to organic dusts. Int. Arch. Occup. Environ. Health 1994, 66, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Menetrez, M.Y.; Foarde, K.K.; Esch, R.K.; Dean, T.R.; Betancourt, D.A.; Moore, S.A.; Svendsen, E.R.; Yeatts, K. The measurement of ambient bio-aerosol exposure. Aerosol Sci. Technol. 2007, 41, 884–893. [Google Scholar] [CrossRef]
- Yeo, H.; Kim, J. SPM and fungal spores in the ambient air of west Korea during the Asian dust (Yellow sand) period. Atmos. Environ. 2002, 36, 5437–5442. [Google Scholar] [CrossRef]
- Lee, B.U.; Hong, I.G.; Lee, D.H.; Chong, E.S.; Jung, J.H.; Lee, J.H.; Lee, I.S. Bacterial bio-aerosol concentrations in public restroom environments. Aerosol Air Qual. Res. 2012, 12, 251–255. [Google Scholar] [CrossRef]
- Heo, K.J.; Kim, H.B.; Lee, B.U. Concentration of environmental fungal and bacterial bioaerosols during the monsoon season. J. Aerosol Sci. 2014, 77, 31–37. [Google Scholar] [CrossRef]
- Heo, K.J.; Lee, B.U. Seasonal variation in the concentrations of culturable bacterial and fungal aerosols in underground subway systems. J. Aerosol Sci. 2016, 92, 122–129. [Google Scholar] [CrossRef]
- Hwang, G.B.; Jung, J.H.; Jeong, T.G.; Lee, B.U. Effect of hybrid UV-thermal energy stimuli on inactivation of S. epidermidis and B. subtilis bacterial bioaerosols. Sci. Total Environ. 2010, 408, 5903–5909. [Google Scholar] [CrossRef] [PubMed]
- Macher, J.M. Positive-hole correction of multiple-jet impactors for collecting viable microorganisms. Am. Ind. Hyg. Assoc. J. 1989, 50, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Heo, K.J.; Lim, C.E.; Kim, H.B.; Lee, B.U. Effects of human activities on concentrations of culturable bioaerosols in indoor air environments. J. Aerosol Sci. 2017, 104, 58–65. [Google Scholar] [CrossRef]
- Kang, S.M.; Heo, K.J.; Lee, B.U. Why does rain increase the concentrations of environmental bioaerosols during monsoon? Aerosol Air Qual. Res. 2015, 15, 2320–2324. [Google Scholar] [CrossRef]
- Fröhlich-Nowoisky, J.; Kampf, C.J.; Weber, B.; Huffman, J.A.; Pöhlker, C.; Andreae, M.O.; Lang-Yona, N.; Burrows, S.M.; Gunthe, S.S.; Elbert, W.; et al. Bioaerosols in the Earth system: Climate, health, and ecosystem interactions. Atmos. Res. 2016, 182, 346–376. [Google Scholar] [CrossRef]
- Wijnker, J.J.; Koop, G.; Lipman, L.J.A. Antimicrobial properties of salt (NaCl) used for the preservation of natural casings. Food Microbiol. 2006, 23, 657–662. [Google Scholar] [CrossRef] [PubMed]
| Location | Fungal Bioaerosol Concentration (Temperature, Relative Humidity) | Bacterial Bioaerosol Concentration (Temperature, Relative Humidity) |
|---|---|---|
| Mountain | 805 ± 344 CFU/m3 | 775 ± 361 CFU/m3 |
| 24 °C, 29% | 24 °C, 29% | |
| Seashore | 75 ± 13 CFU/m3 | 150 ± 40 CFU/m3 |
| 26 °C, 20% | 28 °C, 18% | |
| Urban area | 102 ± 11 CFU/m3 | 232 ± 133 CFU/m3 |
| 30 °C, 15% | 29 °C, 18% |
| Location | Fungal Bioaerosol Concentration (Temperature, Relative Humidity) | Bacterial Bioaerosol Concentration (Temperature, Relative Humidity) |
|---|---|---|
| Mountain | 907 ± 825 CFU/m3 | 70 ± 10 CFU/m3 |
| 12 °C, 30% | 12 °C, 30% | |
| Seashore | 230 ± 53 CFU/m3 | 10 ± 10 CFU/m3 |
| 8 °C, 48% | 8 °C, 48% | |
| Urban area | 437 ± 131 CFU/m3 | 20 ± 10 CFU/m3 |
| 10 °C, 25% | 10 °C, 25% |
| Particles | Mountain | Seashore |
|---|---|---|
| 0.5 µm dp < 1.0 µm | 4.7 105 ± 1.4 103 particles/m3 | 6.3 105 ± 6.3 104 particles/m3 |
| 1.0 µm dp < 3.0 µm | 2.7 105 ± 2.7 103 particles/m3 | 4.1 105 ± 5.9 104 particles/m3 |
| 3.0 µm dp < 5.0 µm | 4.1 104 ± 2.0 103 particles/m3 | 8.3 104 ± 2.1 104 particles/m3 |
| Fungal bioaerosol concentration | 411 ±36 CFU/m3 | 46 ± 10 CFU/m3 |
| Bacterial bioaerosol Concentration | 14 ± 2 CFU/m3 | 2 ± 2 CFU/m3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, B.U.; Lee, G.; Heo, K.J.; Jung, J. Concentrations of Atmospheric Culturable Bioaerosols at Mountain and Seashore Sites. Int. J. Environ. Res. Public Health 2019, 16, 4323. https://doi.org/10.3390/ijerph16224323
Lee BU, Lee G, Heo KJ, Jung J. Concentrations of Atmospheric Culturable Bioaerosols at Mountain and Seashore Sites. International Journal of Environmental Research and Public Health. 2019; 16(22):4323. https://doi.org/10.3390/ijerph16224323
Chicago/Turabian StyleLee, Byung Uk, Gunwoong Lee, Ki Joon Heo, and Jaeho Jung. 2019. "Concentrations of Atmospheric Culturable Bioaerosols at Mountain and Seashore Sites" International Journal of Environmental Research and Public Health 16, no. 22: 4323. https://doi.org/10.3390/ijerph16224323
APA StyleLee, B. U., Lee, G., Heo, K. J., & Jung, J. (2019). Concentrations of Atmospheric Culturable Bioaerosols at Mountain and Seashore Sites. International Journal of Environmental Research and Public Health, 16(22), 4323. https://doi.org/10.3390/ijerph16224323





