The Association between Autism Spectrum Disorder and Pre- and Postnatal Antibiotic Exposure in Childhood—A Systematic Review with Meta-Analysis
Abstract
1. Introduction
2. Methods
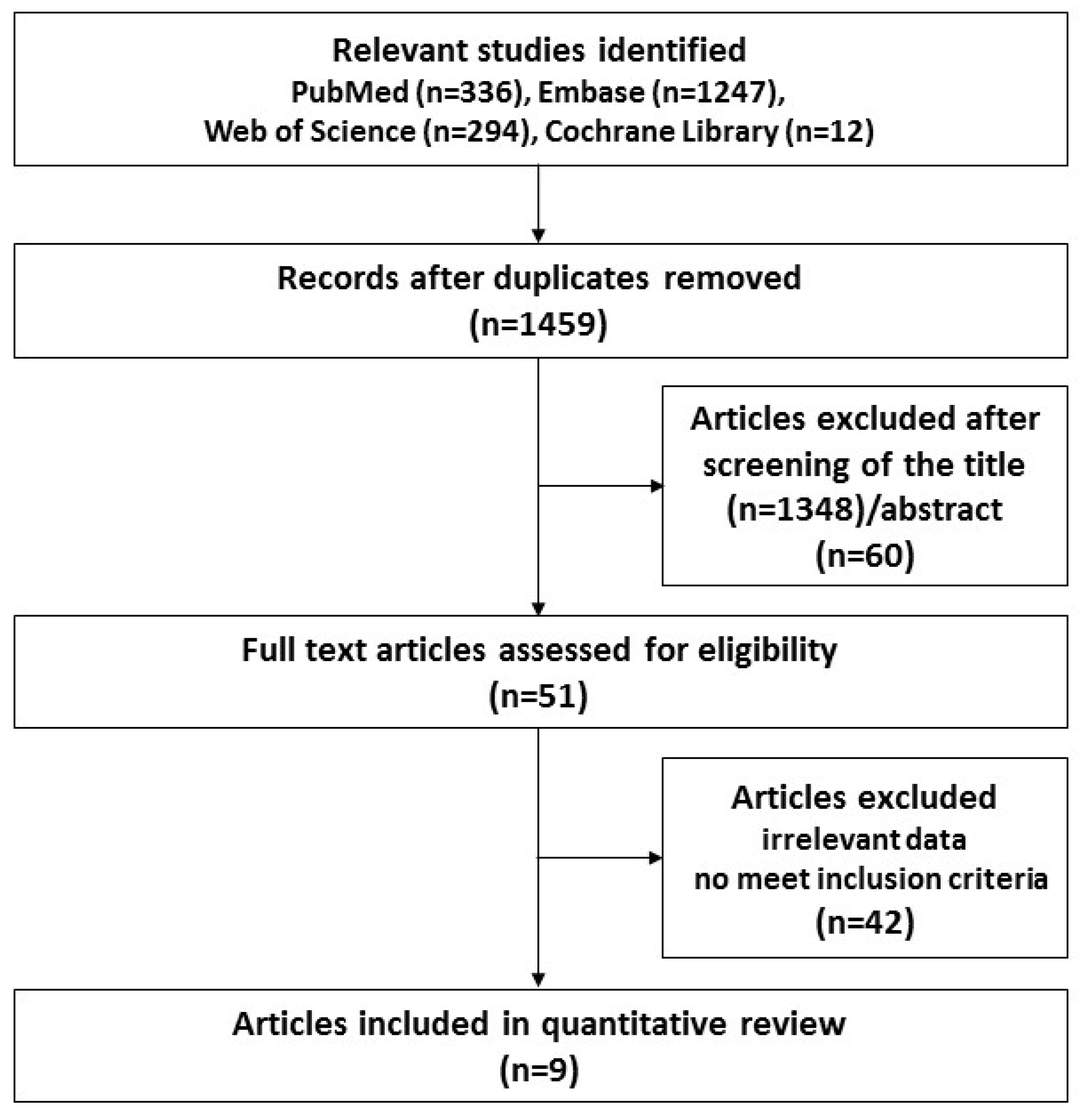
2.1. Search Strategy and Selection Criteria
2.2. Data Extraction and Analysis
3. Results
3.1. Characteristics of the Studies
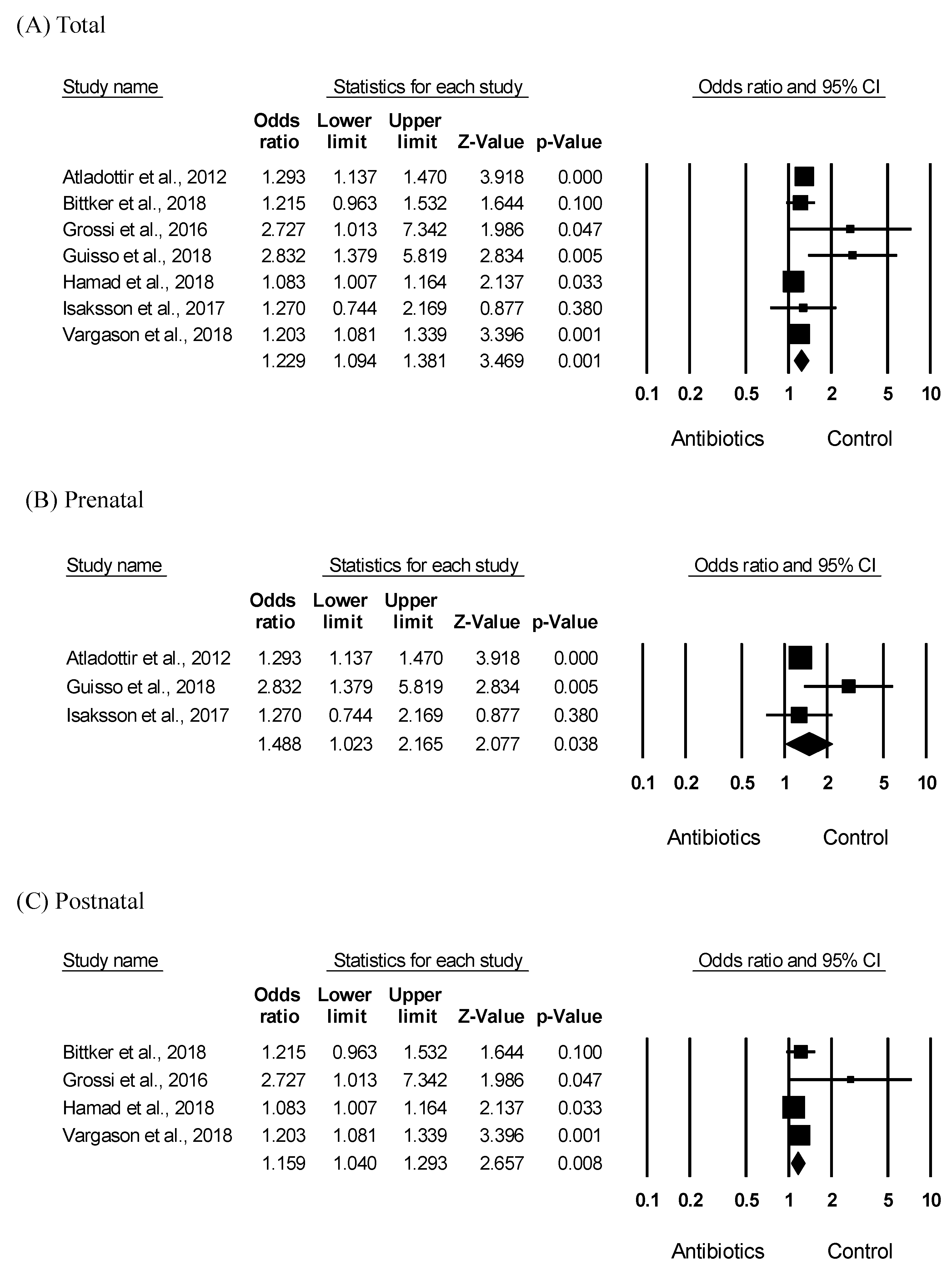
3.2. Meta-Analysis Result Relating the ASD Risk in Antibiotic Exposure
3.3. Meta-Analysis Results Related to Antibiotic Exposure in ASD
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5, 5th ed.; Arlington, V.A., Ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Baxter, A.J.; Brugha, T.S.; Erskine, H.E.; Scheurer, R.W.; Vos, T.; Scott, J.G. The epidemiology and global burden of autism spectrum disorders. Psychol. Med. 2015, 45, 601–613. [Google Scholar] [CrossRef] [PubMed]
- GBD 2016 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1211–1259. [Google Scholar] [CrossRef]
- Christensen, D.L.; Maenner, M.J.; Bilder, D.; Constantino, J.N.; Daniels, J.; Durkin, M.S.; Fitzgerald, R.T.; Kurzius-Spencer, M.; Pettygrove, S.D.; Robinson, C.; et al. Prevalence and characteristics of autism spectrum disorder among children aged 4 years—Early autism and developmental disabilities monitoring network, seven sites, United States, 2010, 2012, and 2014. MMWR Surveill. Summ. 2019, 68, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Taylor, B.; Jick, H.; Maclaughlin, D. Prevalence and incidence rates of autism in the UK: Time trend from 2004–2010 in children aged 8 years. BMJ Open 2013, 3, e003219. [Google Scholar] [CrossRef]
- Geschwind, D.H. Genetics of autism spectrum disorders. Trends Cogn. Sci. 2011, 15, 409–416. [Google Scholar] [CrossRef]
- Hallmayer, J.; Cleveland, S.; Torres, A.; Phillips, J.; Cohen, B.; Torigoe, T.; Miller, J.; Fedele, A.; Collins, J.; Smith, K.; et al. Genetic heritability and shared environmental factors among twin pairs with autism. Arch. Gen. Psychiatry 2011, 68, 1095–1102. [Google Scholar] [CrossRef]
- Bernard, S.; Enayati, A.; Redwood, L.; Roger, H.; Binstock, T. Autism: A novel form of mercury poisoning. Med. Hypotheses 2001, 56, 462–471. [Google Scholar] [CrossRef]
- Mahaffey, K.R.; Clickner, R.P.; Bodurow, C.C. Blood organic mercury and dietary mercury intake: National Health and Nutrition Examination Survey, 1999 and 2000. Environ. Health Perspect. 2004, 112, 562–570. [Google Scholar] [CrossRef]
- Hamad, A.F.; Alessi-Severini, S.; Mahmud, S.M.; Brownell, M.; Kuo, I.F. Prenatal antibiotics exposure and the risk of autism spectrum disorders: A population-based cohort study. PLoS ONE 2019, 14, e0221921. [Google Scholar] [CrossRef]
- Liew, Z.; Ritz, B.; Virk, J.; Olsen, J. Maternal use of acetaminophen during pregnancy and risk of autism spectrum disorders in childhood: A Danish national birth cohort study. Autism Res. 2016, 9, 951–958. [Google Scholar] [CrossRef]
- Atladottir, H.O.; Henriksen, T.B.; Schendel, D.E.; Parner, E.T. Autism after infection, febrile episodes, and antibiotic use during pregnancy: An exploratory study. Pediatrics 2012, 130, e1447–e1454. [Google Scholar] [CrossRef] [PubMed]
- Schulfer, A.F.; Battaglia, T.; Alvarez, Y.; Bijnens, L.; Ruiz, V.E.; Ho, M.; Robinson, S.; Ward, T.; Cox, L.M.; Rogers, A.B.; et al. Intergenerational transfer of antibiotic-perturbed microbiota enhances colitis in susceptible mice. Nat. Microbiol. 2018, 3, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Schultz, S.T.; Klonoff-Cohen, H.S.; Wingard, D.L.; Akshoomoff, N.A.; Macera, C.A.; Ji, M. Acetaminophen (paracetamol) use, measles-mumps-rubella vaccination, and autistic disorder: The results of a parent survey. Autism 2008, 12, 293–307. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.B.; Romdalvik, J.; Ramanujam, V.M.; Legator, M.S. Mercury, lead, and zinc in baby teeth of children with autism versus controls. J. Toxicol. Environ. Health A 2007, 70, 1046–1051. [Google Scholar] [CrossRef]
- Bittker, S.S.; Bell, K.R. Acetaminophen, antibiotics, ear infection, breastfeeding, vitamin D drops, and autism: An epidemiological study. Neuropsychiatr. Dis. Treat. 2018, 14, 1399–1414. [Google Scholar] [CrossRef]
- Borre, Y.E.; O’Keeffe, G.W.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Microbiota and neurodevelopmental windows: Implications for brain disorders. Trends Mol. Med. 2014, 20, 509–518. [Google Scholar] [CrossRef]
- Petra, A.I.; Panagiotidou, S.; Hatziagelaki, E.; Stewart, J.M.; Conti, P.; Theoharides, T.C. Gut-microbiota-brain axis and its effect on neuropsychiatric disorders with suspected immune dysregulation. Clin. Ther. 2015, 37, 984–995. [Google Scholar] [CrossRef]
- Mezzelani, A.; Landini, M.; Facchiano, F.; Raggi, M.E.; Villa, L.; Molteni, M.; De Santis, B.; Brera, C.; Caroli, A.M.; Milanesi, L.; et al. Environment, dysbiosis, immunity and sex-specific susceptibility: A translational hypothesis for regressive autism pathogenesis. Nutr. Neurosci. 2015, 18, 145–161. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. J. Clin. Epidemiol. 2009, 62, 1006–1012. [Google Scholar] [CrossRef]
- Bae, J.M. A suggestion for quality assessment in systematic reviews of observational studies in nutritional epidemiology. Epidemiol. Health 2016, 38, e2016014. [Google Scholar] [CrossRef]
- Well, G.A.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 26 June 2019).
- Grossi, E.; Veggo, F.; Narzisi, A.; Compare, A.; Muratori, F. Pregnancy risk factors in autism: A pilot study with artificial neural networks. Pediatric Res. 2016, 79, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Guisso, D.R.; Saadeh, F.S.; Saab, D.; El Deek, J.; Chamseddine, S.; Abou-El-Hassan, H.; Majari, G.; Boustany, R.M. Association of autism with maternal infections, perinatal and other risk factors: A case-control study. J. Autism Dev. Disord. 2018, 48, 2010–2021. [Google Scholar] [CrossRef] [PubMed]
- Hamad, A.F.; Alessi-Severini, S.; Mahmud, S.M.; Brownell, M.; Kuo, I.F. Early childhood antibiotics use and autism spectrum disorders: A population-based cohort study. Int. J. Epidemiol. 2018, 47, 1497–1506. [Google Scholar] [CrossRef] [PubMed]
- House, S.A.; Goodman, D.C.; Weinstein, S.J.; Chang, C.H.; Wasserman, J.R.; Morden, N.E. Prescription use among children with autism spectrum disorders in northern New England: Intensity and small area variation. J. Pediatrics 2016, 169, 277–283.e2. [Google Scholar] [CrossRef] [PubMed]
- Isaksson, J.; Pettersson, E.; Kostrzewa, E.; Diaz Heijtz, R.; Bolte, S. Brief report: Association between autism spectrum disorder, gastrointestinal problems and perinatal risk factors within sibling pairs. J. Autism Dev. Disord. 2017, 47, 2621–2627. [Google Scholar] [CrossRef]
- Vargason, T.; McGuinness, D.L.; Hahn, J. Gastrointestinal symptoms and oral antibiotic use in children with autism spectrum disorder: Retrospective analysis of a privately insured U.S. population. J. Autism Dev. Disord. 2019, 49, 647–659. [Google Scholar] [CrossRef]
- Tanaka, K.; Miyake, Y.; Furukawa, S.; Arakawa, M. Pre-and postnatal smoking exposure and risk of atopic eczema in young Japanese children: A prospective prebirth cohort study. Nicotine Tob. Res. 2017, 19, 804–809. [Google Scholar] [CrossRef]
- Xiao, Z.; Qiu, T.; Ke, X.; Xiao, X.; Xiao, T.; Liang, F.; Zou, B.; Huang, H.; Fang, H.; Chu, K.; et al. Autism spectrum disorder as early neurodevelopmental disorder: Evidence from the brain imaging abnormalities in 2–3 years old toddlers. J. Autism Dev. Disord. 2014, 44, 1633–1640. [Google Scholar] [CrossRef]
- Bellinger, D.C. Prenatal exposures to environmental chemicals and children’s neurodevelopment: An update. Saf. Health Work 2013, 4, 1–11. [Google Scholar] [CrossRef]
- Walhovd, K.B.; Moe, V.; Slinning, K.; Siqveland, T.; Fjell, A.M.; Bjornebekk, A.; Smith, L. Effects of prenatal opiate exposure on brain development—A call for attention. Nat. Rev. Neurosci. 2009, 10, 390. [Google Scholar] [CrossRef]
- Fang, S.Y.; Wang, S.; Huang, N.; Yeh, H.H.; Chen, C.Y. Prenatal infection and autism spectrum disorders in childhood: A population-based case-control study in Taiwan. Paediatr. Perinat. Epidemiol. 2015, 29, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Zerbo, O.; Qian, Y.; Yoshida, C.; Grether, J.K.; Van de Water, J.; Croen, L.A. Maternal infection during pregnancy and autism spectrum disorders. J. Autism Dev. Disord. 2015, 45, 4015–4025. [Google Scholar] [CrossRef] [PubMed]

| Study | Study Type | Participants | Location | Age | Outcome | Quality * |
|---|---|---|---|---|---|---|
| Adams et al., 2007 [15] | Case-control | 27 | USA | 3–8 years | Children with autism showed higher oral antibiotic usage in various time periods. | 5 |
| Atladóttir et al., 2012 [12] | Cohort | 976 | Denmark | 8–14 years | The use of various antibiotics showed an increased risk of ASD/infantile autism. | 7 |
| Bittker et al., 2018 [16] | Case-control | 1515 | USA | 3–12 years | Postnatal antibiotic use was associated with an increased ASD risk. | 6 |
| Grossi et al., 2016 [23] | Case-control | 113 | Italy | 5–16 years | Regarding potential risk factors, the frequency of early antibiotic use was higher in the autism group than in the typical group. | 7 |
| Guisso et al., 2018 [24] | Case-control | 314 | Lebanon | 2–18 years | In the multivariable analysis, antibiotics were negatively associated with ASD. | 7 |
| Hamad et al., 2018 [25] | Cohort | 214,834 | Canada | 2–8 years | Children who received antibiotics had a reduced ASD risk. | 7 |
| House et al., 2016 [26] | Cross-sectional | 949,821 | USA | 0–17 years | The rate of antibiotic use among children diagnosed with ASD was more than 2-fold that of the general population. | 8 |
| Isaksson et al., 2017 [27] | Case-control | 415 | Sweden | 4–9 years | An environmental risk index based on the use of antibiotics during pregnancy and/or the breastfeeding period was associated with ASD. | 7 |
| Vargason et al., 2019 [28] | Cohort | 281,623 | USA | 0–5 years | Greater numbers of oral antibiotic fills during the first 3 years of enrollment were found to significantly increase the hazard rate of having a later GI-related diagnosis in both children with and without ASD. | 7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, E.; Cho, J.; Kim, K.Y. The Association between Autism Spectrum Disorder and Pre- and Postnatal Antibiotic Exposure in Childhood—A Systematic Review with Meta-Analysis. Int. J. Environ. Res. Public Health 2019, 16, 4042. https://doi.org/10.3390/ijerph16204042
Lee E, Cho J, Kim KY. The Association between Autism Spectrum Disorder and Pre- and Postnatal Antibiotic Exposure in Childhood—A Systematic Review with Meta-Analysis. International Journal of Environmental Research and Public Health. 2019; 16(20):4042. https://doi.org/10.3390/ijerph16204042
Chicago/Turabian StyleLee, Eunmi, Jeonghyun Cho, and Ka Young Kim. 2019. "The Association between Autism Spectrum Disorder and Pre- and Postnatal Antibiotic Exposure in Childhood—A Systematic Review with Meta-Analysis" International Journal of Environmental Research and Public Health 16, no. 20: 4042. https://doi.org/10.3390/ijerph16204042
APA StyleLee, E., Cho, J., & Kim, K. Y. (2019). The Association between Autism Spectrum Disorder and Pre- and Postnatal Antibiotic Exposure in Childhood—A Systematic Review with Meta-Analysis. International Journal of Environmental Research and Public Health, 16(20), 4042. https://doi.org/10.3390/ijerph16204042





