Probiotics for the Treatment of Bacterial Vaginosis: A Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources, Searching Strategy and Eligibility Criteria
2.2. Data Extraction and Bias Assessment
2.3. Outcomes
2.4. Statistical Analysis
2.4.1. Synthesis of Results
2.4.2. Identification of Heterogeneity
2.4.3. Additional Analyses
3. Results
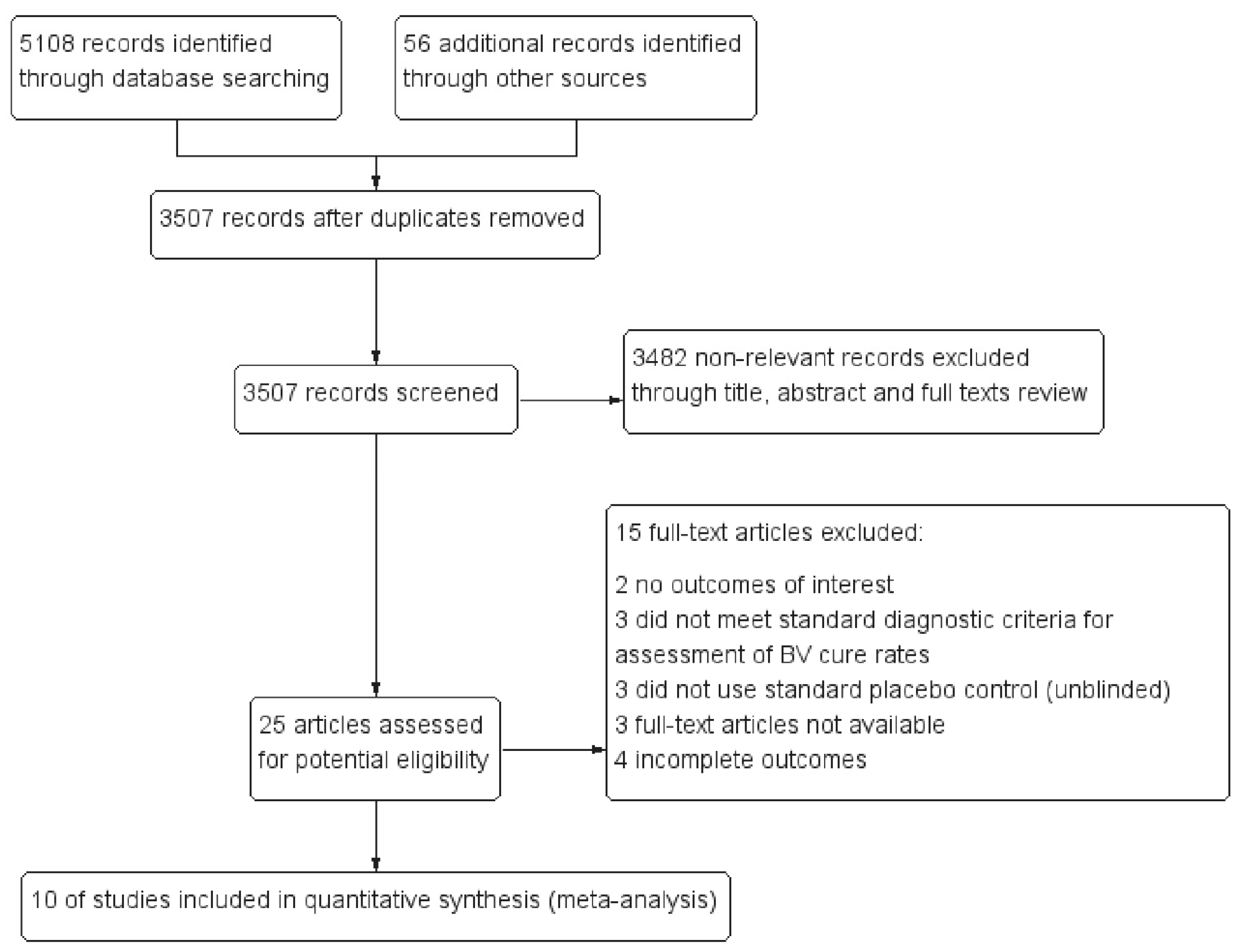
3.1. Study Selection and Characteristics
3.2. Risk of Bias within/across Studies
3.3. Synthesis of Results
3.3.1. Efficacy: Negative Control (Placebo)
Short-Term Effect: after a Normal Menstrual Cycle
Long-Term Effect: More Than Two Menstrual Cycles
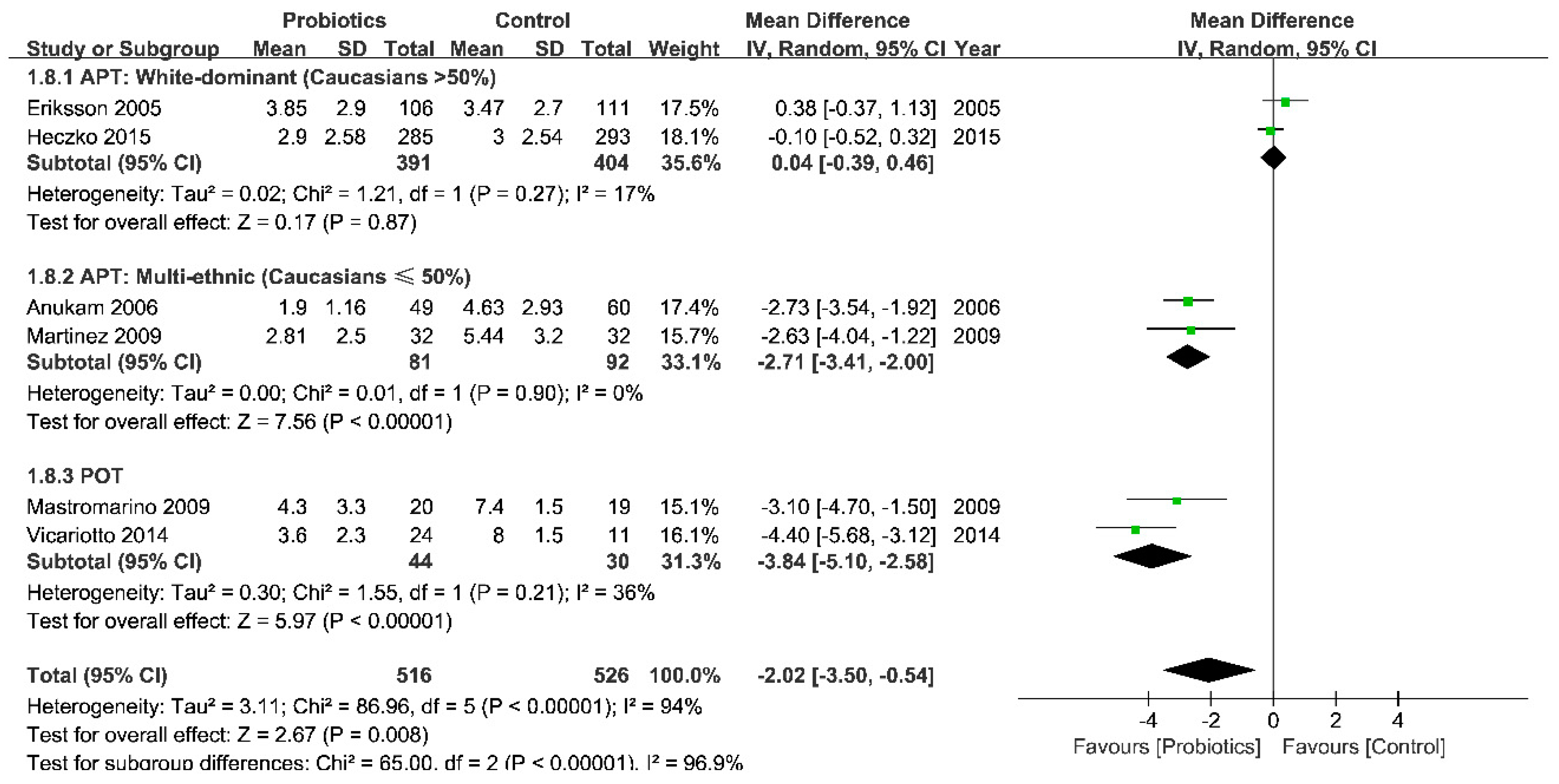
3.3.2. Efficacy: Positive Control (Antibiotics)
3.3.3. Safety
3.4. Additional Analysis
3.4.1. Publication Bias and Selective Reporting
3.4.2. Sensitivity Analyses
3.4.3. Trial Sequential Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ziyadi, S.; Homayouni, A.; Mohammad-Alizadeh-Charandabi, S.; Bastani, P. Chapter 49—Probiotics and Usage in Bacterial Vaginosis. In Probiotics Prebiotics & Synbiotics; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 55–659. [Google Scholar]
- Tang, Y.J.; Samuelson, J.; Dong, Q.S.; Ali, M.M.; Xie, L.; Meng, Y.J.; Cheng, X.Q.; Lan, J.; Cheng, J.; Luo, B. The Prevalence of Sexually Transmitted and Other Lower Reproductive Tract Infections among Rural Women in Sichuan Province, China. Southeast Asian J. Trop. Med. Public Health 2009, 40, 1038–1047. [Google Scholar]
- Goldenberg, R.L.; Klebanoff, M.A.; Nugent, R.; Krohn, M.A.; Hillier, S.; Andrews, W.W. Bacterial colonization of the vagina during pregnancy in four ethnic groups. Vaginal Infections and Prematurity Study Group. Am. J. Obstet. Gynecol. 1996, 174, 1618–1621. [Google Scholar] [CrossRef]
- Kenyon, C.; Colebunders, R.; Crucitti, T. The global epidemiology of bacterial vaginosis: A systematic review. Am. J. Obstet. Gynecol. 2013, 209, 505–523. [Google Scholar] [CrossRef] [PubMed]
- Leitich, H.; Bodner-Adler, B.; Brunbauer, M.; Kaider, A.; Egarter, C.; Husslein, P. Bacterial vaginosis as a risk factor for preterm delivery: A meta-analysis. Am. J. Obstet. Gynecol. 2003, 189, 139–147. [Google Scholar] [CrossRef]
- Hillier, S.L.; Nugent, R.P.; Eschenbach, D.A.; Krohn, M.A.; Gibbs, R.S.; Martin, D.H.; Cotch, M.F.; Edelman, R.; Pastorek, J.G.; Rao, A.V.; et al. Association between Bacterial Vaginosis and Preterm Delivery of a Low-Birth-Weight Infant. N. Engl. J. Med. 1995, 333, 1737–1742. [Google Scholar] [CrossRef]
- Ralph, S.G.; Rutherford, A.J.; Wilson, J.D. Influence of bacterial vaginosis on conception and miscarriage in the first trimester: Cohort study. BMJ 1999, 319, 220–223. [Google Scholar] [CrossRef]
- Balkus, J.E.; Srinivasan, S.; Anzala, O.; Kimani, J.; Andac, C.; Schwebke, J.; Fredricks, D.N.; McClelland, R.S. Impact of periodic presumptive treatment for bacterial vaginosis on the vaginal microbiome among women participating in the Preventing Vaginal Infections trial. J. Infect. Dis. 2017, 215, jiw622. [Google Scholar] [CrossRef]
- Onderdonk, A.B.; Delaney, M.L.; Fichorova, R.N. The Human Microbiome during Bacterial Vaginosis. Clin. Microbiol. Rev. 2016, 29, 223–238. [Google Scholar] [CrossRef]
- Ravel, J.; Gajer, P.; Abdo, Z.; Schneider, G.M.; Koenig, S.S.K.; McCulle, S.L.; Karlebach, S.; Gorle, R.; Russell, J.; Tacket, C.O.; et al. Vaginal microbiome of reproductive-age women. Proc. Natl. Acad. Sci. USA 2011, 108, 4680–4687. [Google Scholar] [CrossRef]
- FAO/WHO Working Group. Report of a Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria, Córdoba, Argentina; FAO/WHO Working Group: Roma, Italy, 2001. [Google Scholar]
- Metchnikoff, E. The Prolongation of Life; GP Putman’s Sons: New York, NY, USA, 1907. [Google Scholar]
- Newman, D. The treatment of cystitis by intravesical injections of lactic bacillus cultures. Lancet 1915, 2, 330–332. [Google Scholar] [CrossRef]
- Löser, A. Zentralblatt für Gynäkologie; Gynaecological University Clinic: Berlin, Germany, 1920; p. 417. [Google Scholar]
- Sartor, R.B. Therapeutic manipulation of the enteric microflora in inflammatory bowel diseases: Antibiotics, probiotics, and prebiotics. Gastroenterology 2004, 126, 1620–1633. [Google Scholar] [CrossRef] [PubMed]
- Martinez, R.C.; Franceschini, S.A.; Patta, M.C.; Quintana, S.M.; Gomes, B.C.; De Martinis, E.C.; Reid, G. Improved cure of bacterial vaginosis with single dose of tinidazole (2 g), Lactobacillus rhamnosus GR-1, and Lactobacillus reuteri RC-14: A randomized, double-blind, placebo-controlled trial. Can. J. Microbiol. 2009, 55, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, C.S.; Pirotta, M.; De Guingand, D.; Hocking, J.S.; Morton, A.N.; Garland, S.M.; Fehler, G.; Morrow, A.; Walker, S.; Vodstrcil, L.A.; et al. Efficacy of Oral Metronidazole with Vaginal Clindamycin or Vaginal Probiotic for Bacterial Vaginosis: Randomised Placebo-Controlled Double-Blind Trial. PLoS ONE 2012, 7, e34540. [Google Scholar] [CrossRef] [PubMed]
- Heczko, P.B.; Tomusiak, A.; Adamski, P.; Jakimiuk, A.J.; StefaD ski, G.; MikoB ajczyk-CichoD ska, A.; Suda-Szczurek, M.; Strus, M. Supplementation of standard antibiotic therapy with oral probiotics for bacterial vaginosis and aerobic vaginitis: A randomised, double-blind, placebo-controlled trial. BMC Women’s Health 2015, 15, 115. [Google Scholar] [CrossRef] [PubMed]
- Senok, A.C.; Verstraelen, H.; Temmerman, M.; Botta, G.A. Probiotics for the treatment of bacterial vaginosis. Cochrane Database Syst. Rev. 2009. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Fu, Y.; Yang, C.; Ma, J. Effects of metronidazole combined probiotics over metronidazole alone for the treatment of bacterial vaginosis: A meta-analysis of randomized clinical trials. Arch. Gynecol. Obstet. 2017, 295, 1331–1339. [Google Scholar] [CrossRef]
- Huang, H.; Song, L.; Zhao, W. Effects of probiotics for the treatment of bacterial vaginosis in adult women: A meta-analysis of randomized clinical trials. Arch. Gynecol. Obstet. 2014, 289, 1225–1234. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Huang, X.; Lin, J.; Demner-Fushman, D. Evaluation of PICO as a knowledge representation for clinical questions. In Proceedings of the AMIA Annual Symposium, Washington, DC, USA, 11–15 November 2006; pp. 359–363. [Google Scholar]
- Van, S.J.; Yudin, M.H.; Yudin, M.H.; Allen, V.; Bouchard, C.; Boucher, M.; Boucoiran, I.; Caddy, S.; Castillo, E.; Kennedy, V.L.; et al. Vulvovaginitis: Screening for and Management of Trichomoniasis, Vulvovaginal Candidiasis, and Bacterial Vaginosis. J. Obstet. Gynaecol. Can. 2015, 37, 266–274. [Google Scholar]
- Workowski, K.A.; Bolan, G.A. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm. Rep. 2015, 64, 1–137. [Google Scholar]
- Nugent, R.P.; Krohn, M.A.; Hillier, S.L. Reliability of diagnosing bacterial vaginosis is improved by a standardized method of gram stain interpretation. J. Clin. Microbiol. 1991, 29, 297–301. [Google Scholar] [PubMed]
- Amsel, R.; Totten, P.A.; Spiegel, C.A.; Chen, K.C.S.; Eschenbach, D.; Holmes, K.K. Nonspecific vaginitis. Am. J. Med. 1983, 74, 14–22. [Google Scholar] [CrossRef]
- Ison, C.A.; Hay, P.E. Validation of a simplified grading of Gram stained vaginal smears for use in genitourinary medicine clinics. Sex. Transm. Infect. 2002, 78, 413–415. [Google Scholar] [CrossRef] [PubMed]
- US Dept of Health and Human Services; F.D.A. Center for Drug Evaluation and Research. Bacterial Vaginosis: Developing Drugs for Treatment Guidance for Industry. Available online: http://www. fda. gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM510948.pdf (accessed on 19 April 2017).
- Gille, C.; Böer, B.; Marschal, M.; Urschitz, M.S.; Heinecke, V.; Hund, V.; Speidel, S.; Tarnow, I.; Mylonas, I.; Franz, A.; et al. Effect of probiotics on vaginal health in pregnancy. EFFPRO, a randomized controlled trial. Am. J. Obstet. Gynecol. 2016, 215, e601–e608. [Google Scholar] [CrossRef] [PubMed]
- Larsson, P.-G.; Stray-Pedersen, B.; Ryttig, K.R.; Larsen, S. Human lactobacilli as supplementation of clindamycin to patients with bacterial vaginosis reduce the recurrence rate; a 6-month, double-blind, randomized, placebo-controlled study. BMC Women’s Health 2008, 8, 3. [Google Scholar] [CrossRef] [PubMed]
- Larsson, P.G.; Carlsson, B.; Fahraeus, L.; Jakobsson, T.; Forsum, U. Diagnosis of bacterial vaginosis: Need for validation of microscopic image area used for scoring bacterial morphotypes. Sex. Transm. Infect. 2004, 80, 63–67. [Google Scholar] [CrossRef]
- Thulkar, J.; Kriplani, A.; Agarwal, N. Probiotic and metronidazole treatment for recurrent bacterial vaginosis. Int. J. Gynecol. Obstet. 2010, 108, 251–252. [Google Scholar] [CrossRef]
- Huedo-Medina, T.B.; Sánchez-Meca, J.; Marín-Martínez, F.; Botella, J. Assessing heterogeneity in meta-analysis: Q statistic or I² index? Psychol. Methods 2006, 11, 193–206. [Google Scholar] [CrossRef]
- Copenhagen Trial Unit Centre for Clinical Intervention Research. User Manual for the Trial Sequential Meta-Analysis (TSA) Software. 2011. Available online: http://www.ctu.dk/tsa/files/tsa_manual.pdf (accessed on 19 April 2017).
- Anukam, K.; Osazuwa, E.; Ahonkhai, I.; Ngwu, M.; Osemene, G.; Bruce, A.W.; Reid, G. Augmentation of antimicrobial metronidazole therapy of bacterial vaginosis with oral probiotic Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14: Randomized, double-blind, placebo controlled trial. Microbes Infect. 2006, 8, 1450–1454. [Google Scholar] [CrossRef]
- Eriksson, K.; Carlsson, B.; Forsum, U.; Larsson, P.-G. A Double-blind Treatment Study of Bacterial Vaginosis with Normal Vaginal Lactobacilli after an Open Treatment with Vaginal Clindamycin Ovules. Acta Derm. Venereol. 2005, 85, 42–46. [Google Scholar] [CrossRef]
- Mastromarino, P.; Macchia, S.; Meggiorini, L.; Trinchieri, V.; Mosca, L.; Perluigi, M.; Midulla, C. Effectiveness of Lactobacillus-containing vaginal tablets in the treatment of symptomatic bacterial vaginosis. Clin. Microbiol. Infect. 2009, 15, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Hemmerling, A.; Harrison, W.; Schroeder, A.; Park, J.; Korn, A.; Shiboski, S.; Foster-Rosales, A.; Cohen, C.R. Phase 2a Study Assessing Colonization Efficiency, Safety, and Acceptability of Lactobacillus crispatus CTV-05 in Women With Bacterial Vaginosis. Sex. Transm. Dis. 2010, 37, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Vujic, G.; Jajac Knez, A.; Despot Stefanovic, V.; Kuzmic Vrbanovic, V. Efficacy of orally applied probiotic capsules for bacterial vaginosis and other vaginal infections: A double-blind, randomized, placebo-controlled study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 168, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Vicariotto, F.; Mogna, L.; Piano, M.D. Effectiveness of the Two Microorganisms Lactobacillus fermentum LF15 and Lactobacillus plantarum LP01, Formulated in Slow-release Vaginal Tablets, in Women Affected by Bacterial Vaginosis. J. Clin. Gastroenterol. 2014, 48, S106–S112. [Google Scholar] [CrossRef]
- Marcone, V.; Calzolari, E.; Bertini, M. Effectiveness of vaginal administration of Lactobacillus rhamnosus following conventional metronidazole therapy: How to lower the rate of bacterial vaginosis recurrences. New Microbiol. 2008, 31, 429–433. [Google Scholar]
- Marcone, V.; Rocca, G.; Lichtner, M.; Calzolari, E. Long-term vaginal administration ofLactobacillus rhamnosus as a complementary approach to management of bacterial vaginosis. Int. J. Gynecol. Obstet. 2010, 110, 223–226. [Google Scholar] [CrossRef]
- Hallen, A.; Jarstrand, C.; Pahlson, C. Treatment of bacterial vaginosis with lactobacilli. Sex. Transm. Dis. 1992, 19, 146–148. [Google Scholar]
- Petricevic, L.; Witt, A. The role of Lactobacillus casei rhamnosus Lcr35 in restoring the normal vaginal flora after antibiotic treatment of bacterial vaginosis. BJOG 2008, 115, 1369–1374. [Google Scholar] [CrossRef]
- Parent, D.; Bossens, M.; Bayot, D.; Kirkpatrick, C.; Graf, F.; Wilkinson, F.E.; Kaiser, R.R. Therapy of bacterial vaginosis using exogenously-applied Lactobacilli acidophili and a low dose of estriol: A placebo-controlled multicentric clinical trial. Arzneimittelforschung 1996, 46, 68–73. [Google Scholar]
- Swidsinski, A.; Loening-Baucke, V.; Mendling, W.; Dorffel, Y.; Schilling, J.; Halwani, Z.; Jiang, X.F.; Verstraelen, H.; Swidsinski, S. Infection through structured polymicrobial Gardnerella biofilms (StPM-GB). Histol. Histopathol. 2014, 29, 567–587. [Google Scholar]
- Deng, Z.L.; Gottschick, C.; Bhuju, S.; Masur, C.; Abels, C.; Wagner-Dobler, I. Metatranscriptome Analysis of the Vaginal Microbiota Reveals Potential Mechanisms for Protection against Metronidazole in Bacterial Vaginosis. mSphere 2018, 3, e00262-18. [Google Scholar] [CrossRef] [PubMed]
- Fettweis, J.M.; Brooks, J.P.; Serrano, M.G.; Sheth, N.U.; Girerd, P.H.; Edwards, D.J.; Strauss, J.F.; Jefferson, K.K.; Buck, G.A. Differences in vaginal microbiome in African American women versus women of European ancestry. Microbiology 2014, 160, 2272–2282. [Google Scholar] [CrossRef] [PubMed]
- Foxman, B.; Wen, A.; Srinivasan, U.; Goldberg, D.; Marrs, C.F.; Owen, J.; Wing, D.A.; Misra, D. Mycoplasma, bacterial vaginosis associated bacteria BVAB3, race, and risk of preterm birth in a high-risk cohort. Am. J. Obstet. Gynecol. 2014, 210, 226.e1–226.e7. [Google Scholar] [CrossRef] [PubMed]
- Suez, J.; Zmora, N.; Zilberman-Schapira, G.; Mor, U.; Dori-Bachash, M.; Bashiardes, S.; Zur, M.; Regev-Lehavi, D.; Ben-Zeev Brik, R.; Federici, S.; et al. Post-Antibiotic Gut Mucosal Microbiome Reconstitution Is Impaired by Probiotics and Improved by Autologous FMT. Cell 2018, 174, 1406–1423.e1416. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Hansmann, M.A.; Davis, C.C.; Suzuki, H.; Brown, C.J.; Schütte, U.; Pierson, J.D.; Forney, L.J. The vaginal bacterial communities of Japanese women resemble those of women in other racial groups. FEMS Immunol. Med. Microbiol. 2010, 58, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Fu, R.; Gartlehner, G.; Grant, M.; Shamliyan, T.; Sedrakyan, A.; Wilt, T.J.; Griffith, L.; Oremus, M.; Raina, P.; Ismaila, A.; et al. Conducting quantitative synthesis when comparing medical interventions: AHRQ and the Effective Health Care Program. J. Clin. Epidemiol. 2011, 64, 1187–1197. [Google Scholar] [CrossRef]
- Luchiari, H.R.; Ferreira, C.S.; Golim, M.A.; Silva, M.G.; Marconi, C. Cervicovaginal bacterial count and failure of metronidazole therapy for bacterial vaginosis. Int. J. Gynaecol. Obstet. 2016, 132, 297–301. [Google Scholar] [CrossRef]
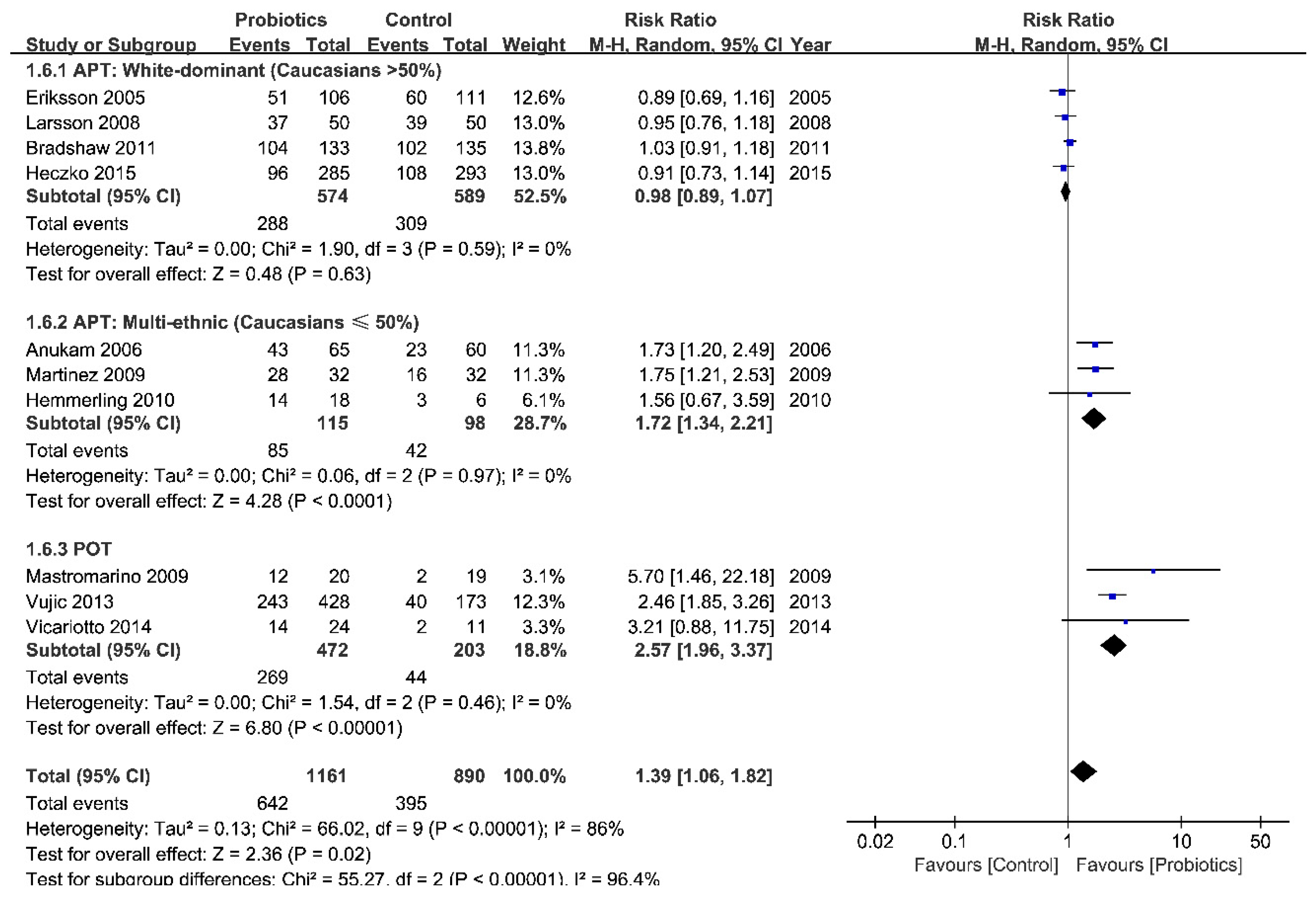
| Groups | No. of Studies | n Total | RR (95% CI) | p (Overall Effect) | I2, % | p (Heterogeneity) |
|---|---|---|---|---|---|---|
| All studies | 10 | 2047 | 1.39 (1.05 to 1.83) | 0.02 | 90 | <0.00001 |
| Type of intervention | ||||||
| APT | 7 | 1376 | 1.11 (0.94 to 1.31) | 0.22 | 69 | 0.004 |
| POT | 3 | 675 | 2.57 (1.96 to 3.37) | <0.00001 | 0 | 0.46 |
| Diagnostic Standard (APT) | ||||||
| Amsel | 5 | 983 | 1.07 (0.85 to 1.35) | 0.58 | 65 | 0.02 |
| Nugent | 2 | 393 | 1.30 (0.76 to 2.22) | 0.33 | 87 | 0.006 |
| Ethics of participants (APT) | ||||||
| White-dominant | 4 | 1163 | 0.98 (0.89 to 1.07) | 0.63 | 0 | 0.59 |
| Multi-ethnic | 3 | 213 | 1.72 (1.34 to 2.21) | <0.0001 | 0 | 0.97 |
| Route of intervention (APT) | ||||||
| Vaginally | 4 | 609 | 1.00 (0.90 to 1.11) | 0.97 | 0 | 0.50 |
| Orally | 3 | 767 | 1.38 (0.85 to 2.23) | 0.19 | 86 | 0.0009 |
| Number of strains (APT) | ||||||
| 1 | 2 | 292 | 1.05 (0.87 to 1.27) | 0.59 | 8 | 0.30 |
| 2 | 3 | 289 | 1.39 (0.87 to 2.23) | 0.17 | 85 | 0.001 |
| 3 | 2 | 795 | 0.92 (0.77 to 1.09) | 0.32 | 0 | 0.97 |
| Dosage per capsule (APT) | ||||||
| ≤1 × 108 CFU | 2 | 485 | 1.01 (0.91 to 1.13) | 0.81 | 20 | 0.26 |
| (1–10) × 108 CFU | 3 | 803 | 1.11 (0.80 to 1.53) | 0.54 | 79 | 0.009 |
| >10 × 108 CFU | 2 | 88 | 1.72 (1.22 to 2.41) | 0.002 | 0 | 0.80 |
| Dosage in total (APT) | ||||||
| ≤1 × 109 CFU | 2 | 485 | 1.00 (0.91 to 1.10) | 0.94 | 20 | 0.26 |
| (1–10) × 109 CFU | 2 | 703 | 1.06 (0.88 to 1.28) | 0.53 | 88 | 0.003 |
| >10 × 109 CFU | 3 | 188 | 1.21 (1.00 to 1.46) | 0.05 | 77 | 0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Z.; He, Y.; Zheng, Y. Probiotics for the Treatment of Bacterial Vaginosis: A Meta-Analysis. Int. J. Environ. Res. Public Health 2019, 16, 3859. https://doi.org/10.3390/ijerph16203859
Wang Z, He Y, Zheng Y. Probiotics for the Treatment of Bacterial Vaginosis: A Meta-Analysis. International Journal of Environmental Research and Public Health. 2019; 16(20):3859. https://doi.org/10.3390/ijerph16203859
Chicago/Turabian StyleWang, Ziyue, Yining He, and Yingjie Zheng. 2019. "Probiotics for the Treatment of Bacterial Vaginosis: A Meta-Analysis" International Journal of Environmental Research and Public Health 16, no. 20: 3859. https://doi.org/10.3390/ijerph16203859
APA StyleWang, Z., He, Y., & Zheng, Y. (2019). Probiotics for the Treatment of Bacterial Vaginosis: A Meta-Analysis. International Journal of Environmental Research and Public Health, 16(20), 3859. https://doi.org/10.3390/ijerph16203859





