Urinary and Faecal Incontinence: Point Prevalence and Predictors in a University Hospital
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sanses, T.V.; Kudish, B.; Guralnik, J.M. The relationship between urinary incontinence, mobility limitations, and disability in older women. Curr. Geriatr. Rep. 2017, 6, 74–80. [Google Scholar] [CrossRef]
- Chong, E.; Chan, M.; Lim, W.S.; Ding, Y.Y. Frailty predicts incident urinary incontinence among hospitalized older adults—A 1-year prospective cohort study. J. Am. Med. Dir. Assoc. 2018, 19, 422–427. [Google Scholar] [CrossRef] [PubMed]
- Molinuevo, B.; Batista-Miranda, J.E. Under the tip of the iceberg: Psychological factors in incontinence. Neurourol. Urodyn. 2012, 31, 669–671. [Google Scholar] [CrossRef] [PubMed]
- Fonda, D.; Nickless, R.; Roth, R. A prospective study of the incidence of urinary incontinence in an acute care teaching hospital and its implications on future service development. Aust. Clin. Rev. 1988, 8, 102–107. [Google Scholar] [PubMed]
- Wu, J.M.; Matthews, C.A.; Vaughan, C.P.; Markland, A.D. Urinary, fecal, and dual incontinence in older U S. adults. J. Am. Geriatr. Soc. 2015, 63, 947–953. [Google Scholar] [CrossRef] [PubMed]
- Patton, V.; Parkin, K.; Moore, K.H. A prospective “bottom up” study of the cost of faecal incontinence in ambulatory patients. Neurourol. Urodyn. 2018, 14, 1672–1677. [Google Scholar] [CrossRef] [PubMed]
- Zisberg, A.; Gary, S.; Gur-Yaish, N.; Admi, H.; Shadmi, E. In-hospital use of continence aids and new-onset urinary incontinence in adults aged 70 and older. J. Am. Geriatr. Soc. 2011, 59, 1099–1104. [Google Scholar] [CrossRef]
- Centers for Disease Control. Urinary incontinence among hospitalized persons aged 65 years and older—United States, 1984–1987. MMWR Morb. Mortal Wkly. Rep. 1991, 40, 433–436. [Google Scholar]
- Ostaszkiewicz, J.; O’Connell, B.; Millar, L. Incontinence: Managed or mismanaged in hospital settings? Int. J. Nurs. Pract. 2008, 14, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Halfens, R.J.G.; Meesterberends, E.; van Nie-Visser, N.C.; Lohrmann, C.; Schönherr, S.; Meijers, J.M.M.; Hahn, S.; Vangelooven, C.; Schols, J.M.G.A. International prevalence measurement of care problems: Results. J. Adv. Nurs. 2013, 69, e5–e17. [Google Scholar] [CrossRef]
- Peet, S.M.; Castleden, C.M.; McGrother, C.W. Prevalence of urinary and faecal incontinence in hospitals and residential and nursing homes for older people. BMJ 1995, 311, 1063. [Google Scholar] [CrossRef] [PubMed]
- Toba, K.; Ouchi, Y.; Orimo, H.; Iimura, O.; Sasaki, H.; Nakamura, Y.; Takasaki, M.; Kuzuya, F.; Sekimoto, H.; Yoshioka, H.; et al. Urinary incontinence in elderly inpatients in Japan: A comparison between general and geriatric hospitals. Aging Clin. Exp. Res. 1996, 8, 47–54. [Google Scholar] [CrossRef]
- Bliss, D.Z.; Johnson, S.; Savik, K.; Clabots, C.R.; Gerding, D.N. Fecal incontinence in hospitalized patients who are acutely ill. Nurs. Res. 2000, 49, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Shahin, E.S.; Lohrman, C. Fecal and double fecal and urinary incontinence in hospitalized patients. J. Wound Ostomy Cont. Nurs. 2015, 2, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Stokes, A.L.; Crumley, C.; Taylor-Thompson, K.; Cheng, A.L. Prevalence of fecal incontinence in the acute care setting. J. Wound Ostomy Cont. Nurs. 2016, 43, 517–522. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.A.; Crocker, J.; Wyman, J.F. Breaking the cycle of stigmatization: Managing the stigma of incontinence in social interactions. J. Wound Ostmy Cont. Nurs. 2005, 32, 38–52. [Google Scholar] [CrossRef]
- Cheater, F.M. Retrospective document survey: Identification, assessment and management of urinary incontinence in medical and care of the elderly wards. J. Adv. Nurs. 1993, 18, 1734–1746. [Google Scholar] [CrossRef] [PubMed]
- Kalantar, J.S.; Howell, S.; Talley, N.J. Prevalence of faecal incontinence and associated risk factors. Med. J. Aust. 2002, 176, 54–57. [Google Scholar] [PubMed]
- Zürcher, S.; Saxer, S.; Schwendimann, R. Urinary incontinence in hospitalised elderly patients: Do nurses recognise and manage the problem? Nurs. Res. Pract. 2011, 2011, 671302. [Google Scholar] [CrossRef] [PubMed]
- Roffman, C.E.; Buchanan, J.; Allison, G.T. Charlson comorbidities index. J. Physiother. 2016, 62, 171. [Google Scholar] [CrossRef]
- Rockwood, K.; Song, X.; MacKnight, C.; Bergman, H.; Hogan, D.B.; McDowell, I.; Mitnitski, A. A global clinical measure of fitness and frailty in elderly people. Can. Med. Assoc. J. 2005, 173, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Mahoney, F.I.; Barthel, D.W. Functional evaluation: The barthel index. Md. Stat. Med. J. 1965, 14, 61–65. [Google Scholar]
- Collin, C.; Wade, D.T.; Davies, S.; Horne, V. The barthel ADL index: A reliability study. Int. Disabil. Stud. 1988, 10, 61–63. [Google Scholar] [CrossRef] [PubMed]
- Wallis, S.J.; Wall, J.; Biram, R.W.S.; Romero-Ortuno, R. Association of the clinical frailty scale with hospital outcomes. QJM 2015, 108, 943–949. [Google Scholar] [CrossRef]
- Chan, D.C.; Tsou, H.H.; Chen, C.Y.; Chen, C.Y. Validation of the Chinese-Canadian study of health and aging clinical frailty scale (CSHA-CFS) telephone version. Arch. Gerontol. Geriatr. 2010, 50, 74–80. [Google Scholar] [CrossRef]
- Gregor, J.; Bardini, C.; Combescure, C.; Dällenbach, P. Urinary incontinence as a predictor of death: A systematic review and meta-analysis. PLoS ONE 2016, 11, e0158992. [Google Scholar]
- Junkin, J.; Selekof, J.L. Prevalence of incontinence and associated skin injury in the acute care inpatient. J. Wound Ostomy Cont. Nurs. 2007, 34, 260–269. [Google Scholar] [CrossRef]
- Palmer, M.H.; Baumgarten, M.; Langenberg, P.; Carson, L. Risk factors for hospital-acquired incontinence in elderly female hip fracture patients. J. Gerontol. A Biol. Sci. Med. Sci. 2002, 57, 672–677. [Google Scholar] [CrossRef]
- Wackerbarth, J.; Patel, H.; Singh, D. Estimating the prevalence of haematuria, urinary retention, and incontinence in Nepalese men by using a cluster randomized survey. Eur. Urol. 2016, 69, 181–182. [Google Scholar] [CrossRef]
- Ellis, G.; Whitehead, M.A.; Robinson, D.; O’Neill, D.; Langhorne, P. Comprehensive geriatric assessment for older adults admitted to hospital: Meta-analysis of randomised controlled trials. BMJ 2011, 343, d6553. [Google Scholar] [CrossRef]
| Variable | Total (n = 435 *) | UI (n = 111) | No UI (n = 324) | p = X (OR) | FI (n = 46) | No FI (n = 386) | p = X |
|---|---|---|---|---|---|---|---|
| Age (years) | |||||||
| Median | 72 | 76 | 69 | p < 0.001 | 80 | 70.5 | p < 0.001 |
| (Q3 − Q1 = ±IQR) | (82 − 59 = ±23) | (83 − 67 = ±17) | (80 − 56 = ±24) | (89 − 71 = ±18) | (80 − 57 = ±23) | ||
| Gender | 54% | 57% | 52% | 0.43 | 50% | 54% | 0.59 |
| (% Male) | (233/435) | (63/111) | (169/324) | OR 1.2 | (23/46) | (210/386) | OR 0.85 |
| Charlson Index | |||||||
| Median | 5 | 6 | 5 | p < 0.001 | 6 | 5 | p = 0.015 |
| (Q3 − Q1 = ±IQR) | (7 − 3 = ±4) | (8 − 4 = ±4) | (7 − 3 = ±4) | (8 − 5 = ±3) | (7 − 3 = ±4) | ||
| CFS * | |||||||
| Median | 3 | 5 | 3 | p < 0.001 | 6 | 3 | p < 0.001 |
| (Q3 − Q1 = ±IQR) | (5 − 2 = ±3) | (6 − 5 = ±1) | (4 − 2 = ±2) | (7 − 4 = ±3) | (4 − 2 = ±2) | ||
| Barthel Index * | |||||||
| Median | 20 | 16 | 20 | p < 0.001 | 13.5 | 20 | p < 0.001 |
| (Q − Q1 = ±IQR) | (20 − 17 = ±3) | (20 − 9 = ±11) | (20 − 19 = ±1) | (19 − 5 = ±14) | (20 − 18 = ±2) | ||
| Length of Stay | |||||||
| Median | 9 | 13 | 7 | p = 0.005 | 15 | 8 | p = 0.012 |
| (Q3 − Q1 = ±IQR) | (21 − 3 = ±18) | (42 − 4 = ±38) | (19 − 3 = ±16) | (60 − 4 = ±56) | (19 − 3 = ±16) |
| Variable n = 435 * | Total n | Urinary Incontinence | Prevalence (%) | Faecal Incontinence | Prevalence (%) | |
|---|---|---|---|---|---|---|
| Age | Under 65 | 152 | 23 | 15.1% | 7 | 4.6% |
| 65–74 | 104 | 27 | 26% | 9 | 8.7% | |
| 75–84 | 108 | 36 | 33.3% | 15 | 13.9% | |
| Over 85 | 71 | 25 | 35.2% | 15 | 21.1% | |
| Frailty | Not Frail (CFS 1–3) | 126 | 2 | 1.6% | 0 | 0% |
| Pre-Frail (CFS = 4) | 79 | 6 | 7.6% | 2 | 2.5% | |
| Mild to Moderate Frailty (CFS 5–6) | 131 | 30 | 22.9% | 7 | 5.3% | |
| Severe Frailty (CFS 7–9) | 93 | 68 | 73.1% | 35 | 37.6% | |
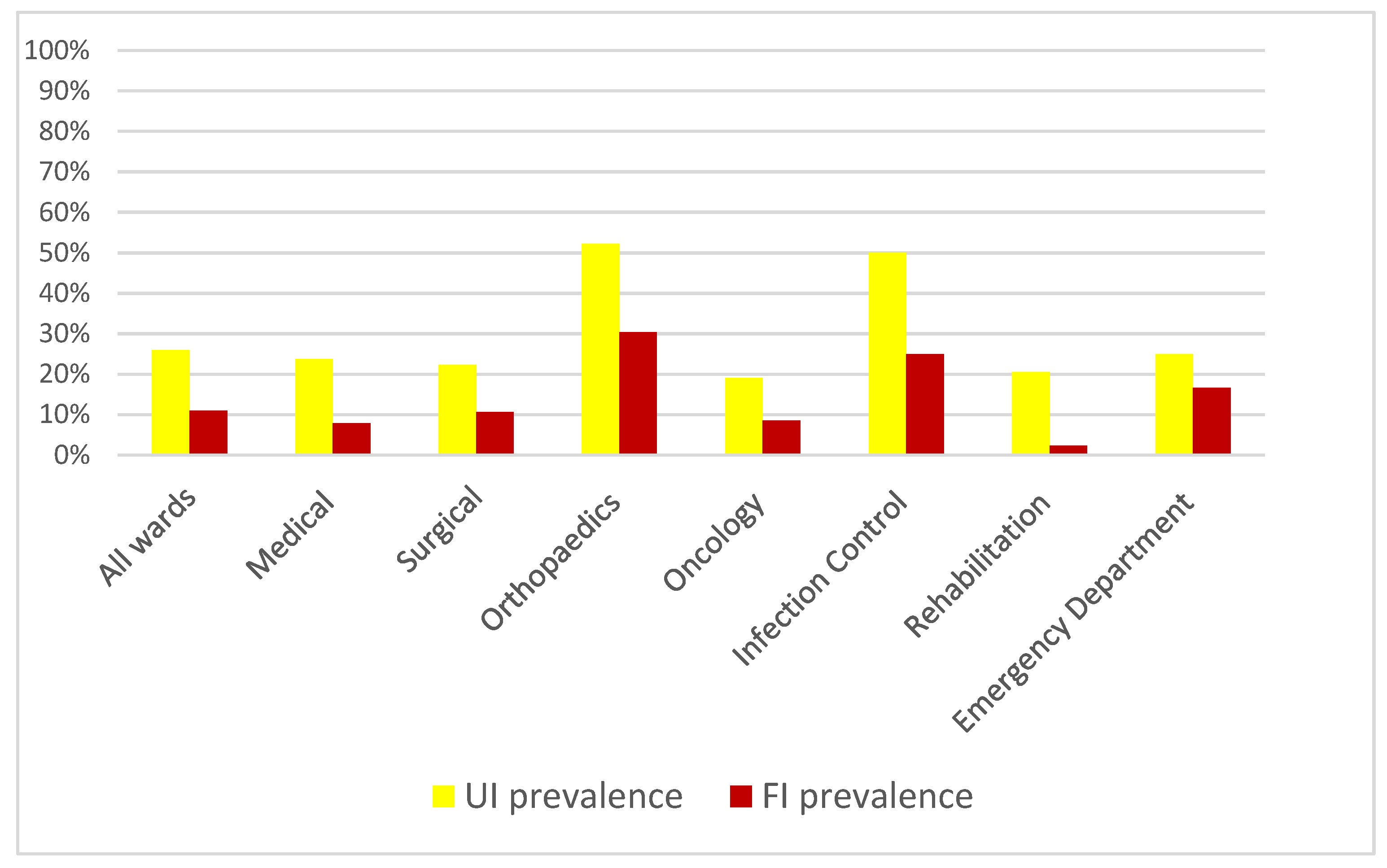
| Ward Type n = 435 * | Total n | Urinary Incontinence n | Prevalence (%) | Faecal Incontinence n | Prevalence (%) |
|---|---|---|---|---|---|
| Medical | 164 | 39 | 23.8% | 13 | 7.9% |
| Surgical | 121 | 27 | 22.3% | 13 | 10.7% |
| Orthopaedic | 23 | 12 | 52.2% | 7 | 30.4% |
| Oncology | 47 | 9 | 19.1% | 4 | 8.5% |
| Infection Control | 24 | 12 | 50% | 6 | 25% |
| Rehabilitation | 44 | 9 | 20.5% | 1 | 2.3% |
| Emergency Department | 12 | 3 | 25% | 2 | 16.7% |
| Variable | B | Exp (B) | 95% Confidence Interval | p = X |
|---|---|---|---|---|
| Urinary Incontinence | ||||
| Age | 0.014 | 1.02 | 0.99–1.04 | 0.26 |
| Gender | −0.540 | 0.58 | 0.33–1.03 | 0.06 |
| Charlson Index | −0.006 | 0.99 | 0.87–1.13 | 0.92 |
| Clinical Frailty Scale | 0.276 | 1.32 | 1.08–1.61 | 0.006 |
| Length of stay | 0.004 | 1.00 | 0.99–1.01 | 0.17 |
| Baseline urinary incontinence | −4.040 | 0.02 | 0.004–0.08 | <0.001 |
| Faecal Incontinence | ||||
| Age | 0.028 | 1.03 | 0.99–1.07 | 0.14 |
| Gender | 0.007 | 1.01 | 0.47–2.18 | 0.99 |
| Charlson Index | −0.056 | 0.95 | 0.79–1.14 | 0.55 |
| Clinical Frailty Scale | 0.285 | 1.33 | 1.02–1.73 | 0.03 |
| Length of stay | 0.001 | 1.00 | 0.99–1.00 | 0.59 |
| Baseline faecal incontinence | 4.374 | 79.3 | 9.36–672 | <0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Condon, M.; Mannion, E.; Molloy, D.W.; O’Caoimh, R. Urinary and Faecal Incontinence: Point Prevalence and Predictors in a University Hospital. Int. J. Environ. Res. Public Health 2019, 16, 194. https://doi.org/10.3390/ijerph16020194
Condon M, Mannion E, Molloy DW, O’Caoimh R. Urinary and Faecal Incontinence: Point Prevalence and Predictors in a University Hospital. International Journal of Environmental Research and Public Health. 2019; 16(2):194. https://doi.org/10.3390/ijerph16020194
Chicago/Turabian StyleCondon, Marie, Edel Mannion, D. William Molloy, and Rónán O’Caoimh. 2019. "Urinary and Faecal Incontinence: Point Prevalence and Predictors in a University Hospital" International Journal of Environmental Research and Public Health 16, no. 2: 194. https://doi.org/10.3390/ijerph16020194
APA StyleCondon, M., Mannion, E., Molloy, D. W., & O’Caoimh, R. (2019). Urinary and Faecal Incontinence: Point Prevalence and Predictors in a University Hospital. International Journal of Environmental Research and Public Health, 16(2), 194. https://doi.org/10.3390/ijerph16020194






