Evaluating Successful Aging in Older People Who Participated in Computerized or Paper-and-Pencil Memory Training: The Memoria Mejor Program
Abstract
1. Introduction
2. Method
2.1. Paricipants
2.2. Process
2.2.1. Memoria Mejor Training Program
Therapeutic training
2.2.2. Measuring Instruments
2.2.3. Word List from the Wechsler Memory Scale
2.2.4. EEG Recording and Analysis
2.2.5. Source Localization
Statistical Analysis
3. Results
4. Discussion
4.1. Psychological Well-Being
4.1.1. Cognitive Processes
4.1.2. Brain Band Activity
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Proyer, R.T.; Gander, F.; Wellenzohn, S.; Ruch, W. Positive psychology interventions in people aged 50–79 years: Long-term effects of placebo-controlled online interventions on well-being and depression. Aging Ment. Health 2014, 18, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ballesteros, R.; Robine, J.M.; Walker, A.; Kalache, A. Active aging: A global goal. Curr. Gerontol. Geriatr. Res. 2013, 2013, 298012. [Google Scholar] [CrossRef] [PubMed]
- Steverink, N.; Westerhof, G.J.; Bode, C.; Dittmann-Kohli, F. The Personal Experience of Aging, Individual Resources, and Subjective Well-Being. J. Gerontol. Psychol. Sci. Copyr. 2001, 56, 364–373. [Google Scholar] [CrossRef]
- Seeman, T.E.; Lusignolo, T.M.; Albert, M.; Berkman, L. Social relationships, social support, and patterns of cognitive aging in healthy, high-functioning older adults: MacArthur studies of successful aging. Health Psychol. 2001, 20, 243. [Google Scholar] [CrossRef] [PubMed]
- Zeeb, H.; Rothgang, H.; Darmann-Finck, I. Ageing, Health and Equity—Broad Perspectives Are Needed to Understand and Tackle Health Challenges of Ageing Societies. Int. J. Environ. Res. Public Health 2018, 18, 457. [Google Scholar] [CrossRef]
- Bowling, A.; Dieppe, P. What is successful ageing and who should define it? BMJ 2005, 331, 1548–1551. [Google Scholar] [CrossRef]
- Steptoe, A.; Deaton, A.; Stone, A.A. Subjective wellbeing, health, and ageing. Lancet 2015, 385, 640–648. [Google Scholar] [CrossRef]
- Martela, F.; Ryan, R.M.; Steger, M.F. Meaningfulness as satisfaction of autonomy, competence, relatedness, and beneficence: Comparing the four satisfactions and positive affect as predictors of meaning in life. J. Happiness Stud. 2018, 19, 1261–1282. [Google Scholar] [CrossRef]
- Villar, F.; Triadó, C.; Celdrán, M.; Solé, C. Measuring well-being among Spanish older adults: Development of a simplified version of Ryff’s Scales of Psychological Well-Being. Psychol. Rep. 2010, 107, 265–280. [Google Scholar] [CrossRef]
- Farkas, P.; Schrenk, M.; Hlauschek, W. Senior Social Platform—An Application Aimed to Reduce the Social and Digital Isolation of Seniors; Real Corp Publ. Proceedings/Tagungsband: Vienna, Austria, 2010; pp. 1247–1252. [Google Scholar]
- Mather, M.; Carstensen, L.L. Aging and motivated cognition: The positivity effect in attention and memory. Trends Cogn. Sci. 2005, 9, 496–502. [Google Scholar] [CrossRef]
- Rebok, G.W.; Ball, K.; Guey, L.T.; Jones, R.N.; Kim, H.Y.; King, J.W.; Marsiske, M.; Morris, J.N.; Tennstedt, S.L.; Unverzagt, F.W.; et al. Ten-Year Effects of the Advanced Cognitive Training for Independent and Vital Elderly Cognitive Training Trial on Cognition and Everyday Functioning in Older Adults. J. Am. Geriatr. Soc. 2014, 62, 16–24. [Google Scholar] [CrossRef]
- Requena, C.; Turrero, A.; Ortiz, T. Six-year training improves everyday memory in healthy older people. Randomized controlled trial. Front. Aging Neurosci. 2016, 8, 135. [Google Scholar] [CrossRef] [PubMed]
- Tzuang, M.; Parisi, J.; Rebok, G.W. Efficacy and Acceptability of Web-Based Memory Training for Older Adults: ACTIVE Memory Works; University Press: Oxford, UK, 2018. [Google Scholar]
- Van Hoof, J.; Kazak, J.; Perek-Białas, J.; Peek, S. The Challenges of Urban Ageing: Making Cities Age-Friendly in Europe. Int. J. Environ. Res. Public Health 2018, 15, 2473. [Google Scholar] [CrossRef]
- Schulz, R.; Wahl, H.W.; Matthews, J.T.; De Vito Dabbs, A.; Beach, S.R.; Czaja, S.J. Advancing the Aging and Technology Agenda in Gerontology. Gerontologist 2015, 55, 724–734. [Google Scholar] [CrossRef] [PubMed]
- Wild, K.; Howieson, D.; Webbe, F.; Seelye, A.; Dementia, J.K. Status of computerized cognitive testing in aging: A systematic review. Alzheimer’s Dementia 2008, 4, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Barban, F.; Annicchiarico, R.; Pantelopoulos, S.; Federici, A.; Perri, R.; Fadda, L.; Carlesimo, G.A.; Ricci, C.; Giuli, S.; Scalici, F.; et al. Protecting cognition from aging and Alzheimer’s disease: A computerized cognitive training combined with reminiscence therapy. Int. J. Geriatr. Psychiatry 2016, 31, 340–348. [Google Scholar] [CrossRef]
- Marusic, U.; Giordani, B.; Moffat, S.D.; Petrič, M.; Dolenc, P.; Pišot, R.; Kavcic, V. Computerized cognitive training during physical inactivity improves executive functioning in older adults. Aging Neuropsychol. Cogn. 2018, 25, 49–69. [Google Scholar] [CrossRef]
- Merzenich, M.M.; Van Vleet, T.M.; Nahum, M. Brain plasticity-based therapeutics. Front. Hum. Neurosci. 2014, 8, 385. [Google Scholar] [CrossRef]
- Ahissar, M.; Nahum, M.; Nelken, I. Reverse hierarchies and sensory learning. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 285–299. [Google Scholar] [CrossRef]
- Nahum, M.; Lee, H.; Merzenich, M.M. Principles of neuroplasticity-based rehabilitation. Prog. Brain Res. 2013, 207, 141–171. [Google Scholar]
- Kueider, A.M.; Parisi, J.M.; Gross, A.L.; Rebok, G.W. Computerized cognitive training with older adults: A systematic review. PLoS ONE 2012, 7, e40588. [Google Scholar] [CrossRef] [PubMed]
- Buckner, R.L.; Andrews-Hanna, J.R.; Schacter, D.L. The Brain’s Default Network Anatomy, Function, and Relevance to Disease. Ann. N. Y. Acad. Sci. 2008, 1124, 1–38. [Google Scholar] [CrossRef] [PubMed]
- Sperazza, L.J.; Banerjee, P. Baby Boomers and Seniors: Understanding Their Leisure Values Enhances Programs. Act. Adapt. Aging 2010, 34, 196–215. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatry Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Ryff, C.D.; Singer, B.H. Positive health: Connecting well-being with biology. Philos. Trans. R. Soc. B Biol. Sci. 2004, 359, 1383. [Google Scholar] [CrossRef]
- Requena, C. Manual memoria mejor. In Ejercicios de Entrenamiento de Memoria III; Universidad de León, Secretariado de Publicaciones: León, Spain, 2005. [Google Scholar]
- Requena, C. Memoria mejor: Ejercicios de entrenamiento la memoria/II. Universidad de León, Secretariado de Publicaciones, 2002. Available online: http://envejecimientoentodaslasedades.unileon.es/primera-generacion.html (accessed on 13 November 2018).
- Díaz, D.; Rodríguez, R.; Blanco, A.; Moreno, B.; Gallardo, I.; Valle, C. Adaptación española de las escalas de bienestar psicológico de Ryff. Psicothema 2006, 18, 572–577. [Google Scholar]
- Keyes, C.L.; Shmotkin, D.; Ryff, C.D. Optimizing well-being: The empirical encounter of two traditions. J. Pers. Soc. Psychol. 2002, 82, 1007. [Google Scholar] [CrossRef]
- Korte, J.; Bohlmeijer, E.T.; Cappeliez, P.; Smit, F. Life review therapy for older adults with moderate depressive symptomatology: A pragmatic randomized controlled trial. Psychol. Med. 2012, 42, 1163–1173. [Google Scholar] [CrossRef]
- Golden, C.J.; Freshwater, S.M. Stroop Color and Word Test; Stoetling: Chicago, IL, USA, 1978. [Google Scholar]
- Kurtz, M.M. Rivermead Behavioral Memory Test. In Encyclopedia of Clinical Neuropsychology; Springer: New York, NY, USA, 2011. [Google Scholar]
- Cockburn, J. The pragmatic and ecological perspective: Evaluation of memory disorders. Eur. Rev. Appl. Psychol. 1996, 46, 201–205. [Google Scholar]
- Wechsler, D. Wechsler Preschool and Primary Scale of Intelligence, 4th ed.; Psychological Corporation: San Antonio, TX, USA, 2012. [Google Scholar]
- Gomez-Pilar, J.; Poza, J.; Bachiller, A.; Gómez, C.; Molina, V.; Hornero, R. Neural network reorganization analysis during an auditory oddball task in schizophrenia using wavelet entropy. Entropy 2015, 17, 5241–5256. [Google Scholar] [CrossRef]
- Makeig, S.; Debener, S.; Onton, J.; Delorme, A. Mining event-related brain dynamics. Trends Cogn. Sci. 2004, 8, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Thatcher, R.; North, D.; Biver, C. LORETA EEG phase reset of the default mode network. Front. Hum. Neurosci. 2014, 8, 529. [Google Scholar] [CrossRef] [PubMed]
- Pascual-Marqui, R.D.; Biscay, R.J.; Bosch-Bayard, J.; Lehmann, D.; Kochi, K.; Kinoshita, T.; Yamada, N.; Sadato, N. Assessing direct paths of intracortical causal information flow of oscillatory activity with the isolated effective coherence (iCoh). Front. Hum. Neurosci. 2014, 8, 448. [Google Scholar] [CrossRef] [PubMed]
- Mazziotta, J.; Toga, A.; Evans, A.; Fox, P.; Lancaster, J.; Zilles, K.; Woods, R.; Paus, T.; Simpson, G.; Pike, B.; et al. A probabilistic atlas and reference system for the human brain: International Consortium for Brain Mapping (ICBM). Philos. Trans. R. Soc. Lond. B Biol. Sci. 2001, 356, 1293–1322. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, J.L.; Woldorff, M.G.; Parsons, L.M.; Liotti, M.; Freitas, C.S.; Rainey, L.; Kochunov, P.V.; Nickerson, D.; Mikiten, S.A.; Fox, P.T. Automated Talairach Atlas labels for functional brain mapping. Hum. Brain Mapp. 2000, 10, 120–131. [Google Scholar] [CrossRef]
- De Villers-Sidani, E.; Merzenich, M.M. Lifelong plasticity in the rat auditory cortex. Basic mechanisms and role of sensory experience. Prog. Brain Res. 2011, 191, 119–131. [Google Scholar]
- Ryff, C.D. Psychological well-being in adult life. Curr. Dir. Psychol. Sci. 1995, 4, 99–104. [Google Scholar] [CrossRef]
- Chou, C.; Hwang, C.; Wu, Y. Effect of exercise on physical function, daily living activities, and quality of life in the frail older adults: A meta-analysis. Arch. Phys. Med. Rehabil. 2012, 93, 237–244. [Google Scholar] [CrossRef]
- Hermida, P.; Stefani, D. La jubilación como un factor de estrés psicosocial. Un análisis de los trabajos científicos de las últimas décadas. Perspect. Psicol. 2011, 8, 101–107. [Google Scholar]
- Escobar, M.A.; Puga, D.; Martín, M. Asociaciones entre la red social y la discapacidad al comienzo de la vejez en las ciudades de Madrid y Barcelona en 2005. Rev. Esp. Salud. Publ. 2008, 82, 637–651. [Google Scholar] [CrossRef]
- Oerlemans, W.G.; Bakker, A.B.; Veenhoven, R. Finding the key to happy aging: A day reconstruction study of happiness. J. Gerontol. Ser. B Psychol. Sci. Soc. Sci. 2011, 66, 665–674. [Google Scholar] [CrossRef]
- Gale, C.R.; Cooper, C.; Deary, I.J.; Aihie Sayer, A. Psychological well-being and incident frailty in men and women: The English Longitudinal Study of Ageing. Psychol. Med. 2014, 44, 697–706. [Google Scholar] [CrossRef]
- Findlay, R.A. Interventions to reduce social isolation amongst older people: Where is the evidence? Ageing Soc. 2003, 23, 647–658. [Google Scholar] [CrossRef]
- Lampit, A.; Hallock, H.; Valenzuela, M. Computerized cognitive training in cognitively healthy older adults: A systematic review and meta-analysis of effect modifiers. PLoS Med. 2014, 11, e1001756. [Google Scholar] [CrossRef] [PubMed]
- Old, S.R.; Naveh-Benjamin, M. Differential effects of age on item and associative measures of memory: A meta-analysis. Psychol. Aging 2008, 23, 104. [Google Scholar] [CrossRef]
- Reijnders, J.; van Heugten, C.; van Boxtel, M. Cognitive interventions in healthy older adults and people with mild cognitive impairment: A systematic review. Ageing Res. Rev. 2013, 12, 263–275. [Google Scholar] [CrossRef]
- Niedermeyer, E. Ultrafast EEG activities and their significance. Clin. EEG Neurosci. 2005, 36, 257. [Google Scholar] [PubMed]
- Bouchard, J.; Villeda, S.A. Aging and brain rejuvenation as systemic events. J. Neurochem. 2015, 132, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Gooding, A.L.; Choi, J.; Fiszdon, J.M.; Wilkins, K.; Kirwin, P.D.; van Dyck, C.H.; Devanand, D.; Bell, M.D.; Rivera Mindt, M. Comparing three methods of computerised cognitive training for older adults with subclinical cognitive decline. Neuropsychol. Rehabil. 2016, 26, 810–821. [Google Scholar] [CrossRef]
- Nouchi, R.; Taki, Y.; Takeuchi, H.; Hashizume, H.; Nozawa, T.; Kambara, T.; Sekiguchi, A.; Miyauchi, C.M.; Kotozaki, Y.; Nouchi, H.; et al. Brain training game boosts executive functions, working memory and processing speed in the young adults: A randomized controlled trial. PLoS ONE 2013, 7, e29676. [Google Scholar] [CrossRef]
| Subjects | Group 1 | Group 2 |
|---|---|---|
| N = 54 | 26 | 28 |
| Age | 73.61 (±3.32) | 74.25 (±1.9) |
| Sex | ||
| Male | 10 | 11 |
| Female | 16 | 17 |
| Living arrangement | ||
| Alone | 10 | 11 |
| Accompanied | 16 | 17 |
| Education level | ||
| University | 3 | 0 |
| High school | 4 | 6 |
| Primary school | 19 | 22 |
| Job | ||
| Civil servant | 6 | 7 |
| Businessman | 3 | 5 |
| Farmer | 4 | 4 |
| Housewife | 13 | 12 |
| Dimensions | Group 1 | SD | Group 2 | SD | ||
|---|---|---|---|---|---|---|
| Self-Acceptance | 27.53 | ± | 4.37 | 26.13 | ± | 2.57 |
| Positive Relations | 34.71 | ± | 3.00 | 32.00 | ± | 5.33 |
| Alone | 32.98 | ± | 3.58 | 30.22 | ± | 3.80 |
| Accompanied | 35.78 | ± | 2.52 | 33.14 | ± | 2.35 |
| Autonomy | 25.29 | ± | 4.22 | 24.47 | ± | 5.80 |
| Environmental Mastery | 28.94 | ± | 3.38 | 26.00 | ± | 4.47 |
| Personal Growth | 31.06 | ± | 3.40 | 30.60 | ± | 4.32 |
| Purpose in Life | 28.00 | ± | 3.55 | 26.67 | ± | 3.95 |
| Cognitve Test | Group 1 | Group 2 | Group 1 vs. Group 2 | ||||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | P | |||
| Atention | 3.22 | ± | 4.35 | −2.90 | ± | 5.47 | p = 0.001 |
| Everyday memory | 11.23 | ± | 0.24 | −9.45 | ± | 1.13 | p = 0.002 |
| Word List Recall | 6.56 | ± | 1.42 | 5.12 | ± | 1.38 | p = 0.014 |
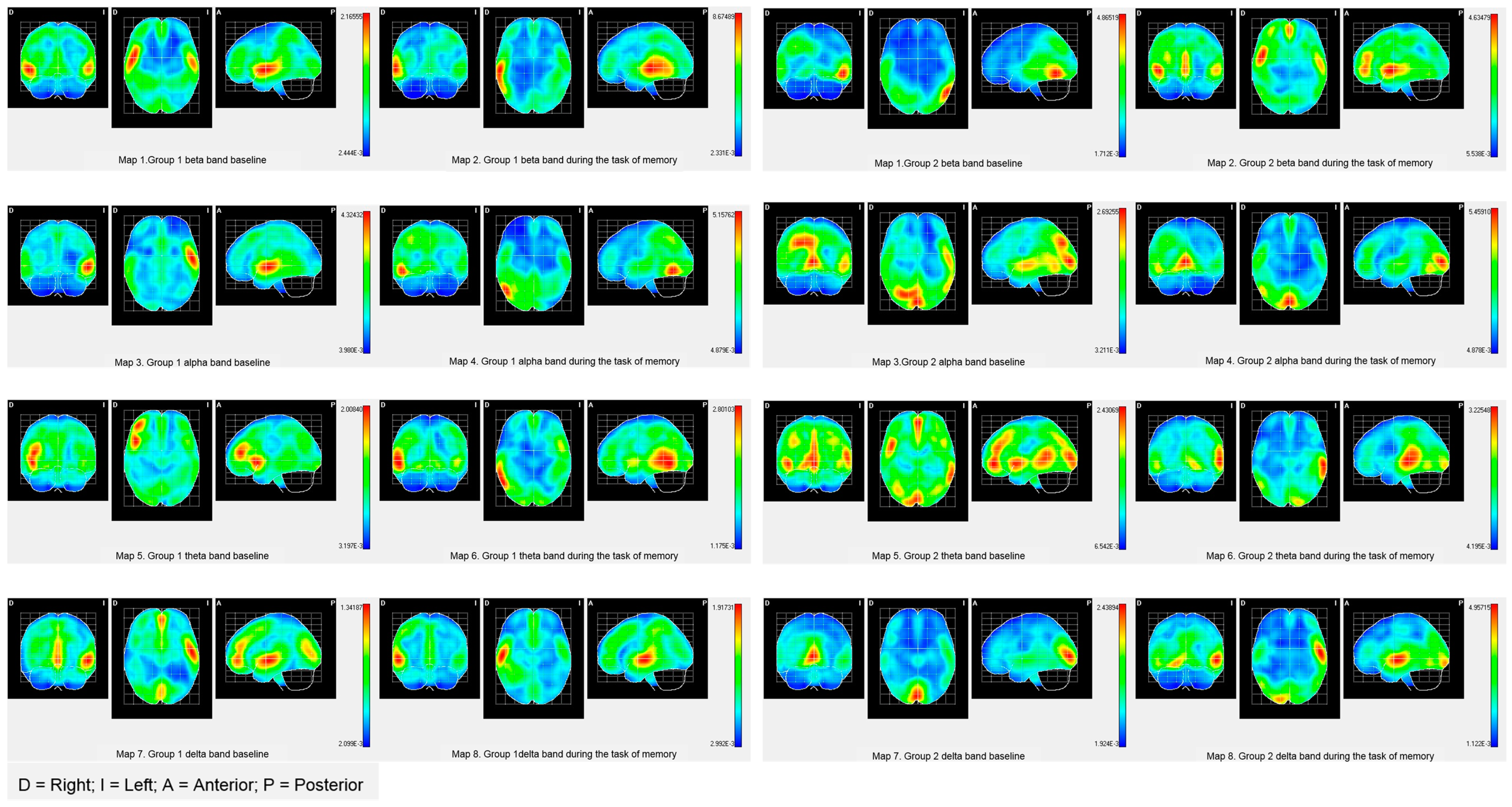
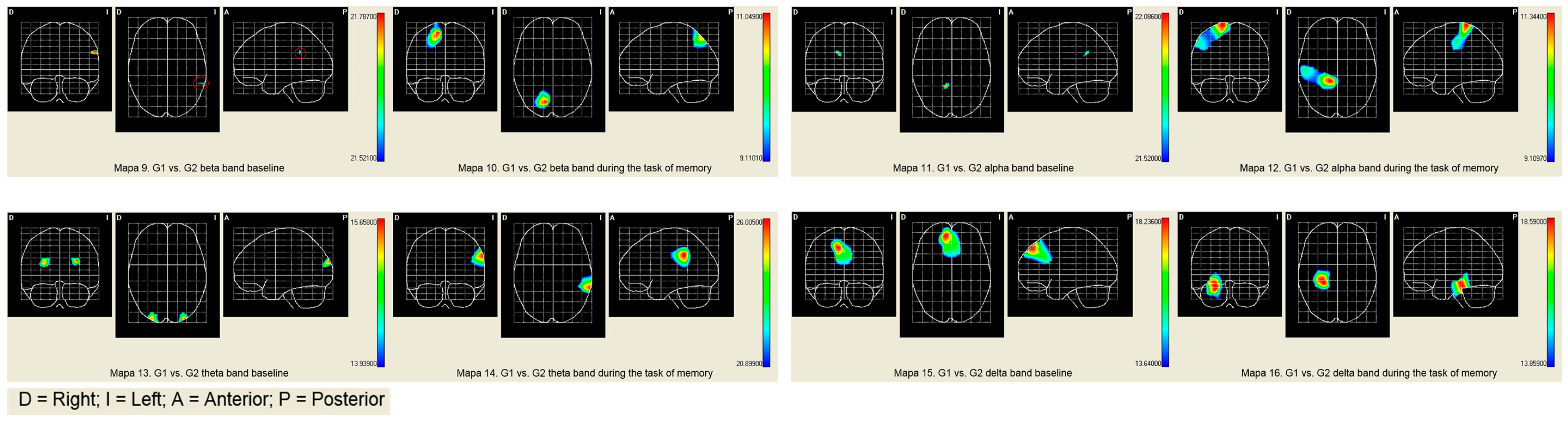
| Brain Activities | PALA | BA | X | Y | Z | T2 Hotelling | Mean | |
|---|---|---|---|---|---|---|---|---|
| G1 | G2 | |||||||
| Beta band | ||||||||
| Baseline | Left Angular gyrus | 39 | 140 | 104 | 144 | 9.1357 ** | 0.3452 | 0.3662 |
| Task of memory | Right superior Parietal Lobe | 7 | 76 | 126 | 164 | 11.0486 * | 0.0688 | 0.2250 |
| Alpha band | ||||||||
| Baseline | Right Cingulate Region | 7 | 76 | 100 | 136 | 22.0856 *** | 0.1019 | 0.1119 |
| Task of memory | Postcentral area right | 3 | 76 | 148 | 128 | 11.3437 * | 0.2848 | 07802 |
| Theta band | ||||||||
| Baseline | Superior left occipital area | 19 | 120 | 96 | 184 | 15.6583 ** | 0.1442 | 0.4200 |
| Task of memory | Left Occipital Pole | 18 | 108 | 52 | 192 | 9.6772 * | 0.4046 | 0.4883 |
| Occipitotemporal area Lateral left | 18 | 108 | 56 | 168 | 9.1443 * | 0.7235 | 0.7850 | |
| Lingual Area Left | 18 | 108 | 60 | 180 | 9.39411 * | 20.845 | 25.436 | |
| Delta band | ||||||||
| Baseline | Right Lateral Orbitofrontal Region | 11 | 72 | 48 | 76 | 11.37077 * | 0.0926 | 0.1901 |
| Task of memory | Right parahipocampal area | 30 | 64 | 52 | 116 | 18.3420 ** | 0.4071 | 0.4257 |
| Right lateral Occipitotemporal area | 30 | 64 | 52 | 120 | 18.5904 ** | 0.3481 | 0.3735 | |
| Hippocampal area right | 30 | 64 | 52 | 112 | 17.9080 ** | 0.4427 | 0.4594 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Requena, C.; Rebok, G.W. Evaluating Successful Aging in Older People Who Participated in Computerized or Paper-and-Pencil Memory Training: The Memoria Mejor Program. Int. J. Environ. Res. Public Health 2019, 16, 191. https://doi.org/10.3390/ijerph16020191
Requena C, Rebok GW. Evaluating Successful Aging in Older People Who Participated in Computerized or Paper-and-Pencil Memory Training: The Memoria Mejor Program. International Journal of Environmental Research and Public Health. 2019; 16(2):191. https://doi.org/10.3390/ijerph16020191
Chicago/Turabian StyleRequena, Carmen, and George W. Rebok. 2019. "Evaluating Successful Aging in Older People Who Participated in Computerized or Paper-and-Pencil Memory Training: The Memoria Mejor Program" International Journal of Environmental Research and Public Health 16, no. 2: 191. https://doi.org/10.3390/ijerph16020191
APA StyleRequena, C., & Rebok, G. W. (2019). Evaluating Successful Aging in Older People Who Participated in Computerized or Paper-and-Pencil Memory Training: The Memoria Mejor Program. International Journal of Environmental Research and Public Health, 16(2), 191. https://doi.org/10.3390/ijerph16020191





