Association of Low Sputum Smear Positivity among Tuberculosis Patients with Interferon-Gamma Release Assay Outcomes of Close Contacts in Japan
Abstract
1. Introduction
2. Materials and Methods
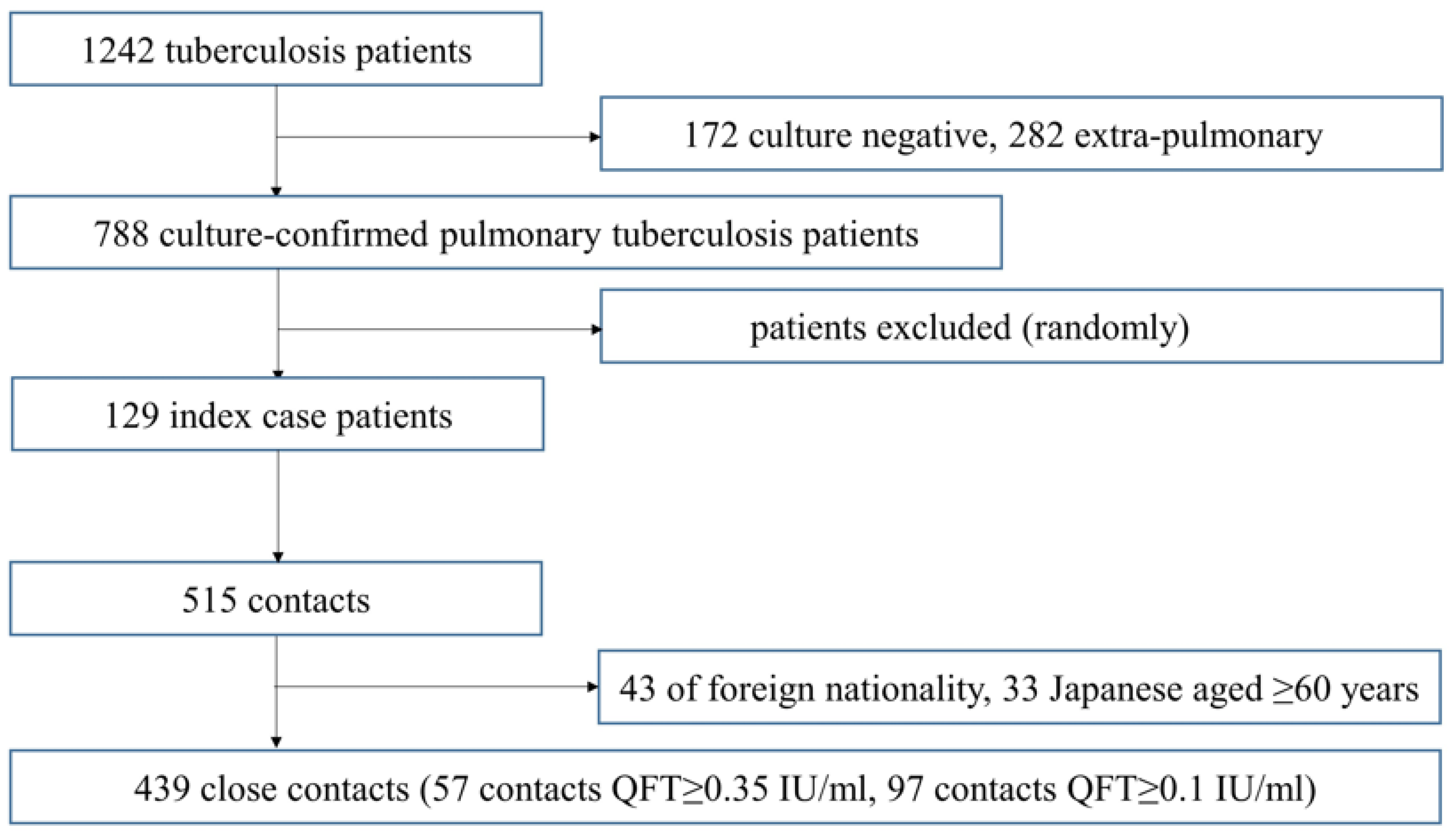
2.1. Study Design and Setting
2.2. Index TB Patients and Exposure
2.3. Subject Contacts
2.4. Outcome
2.5. Statistical Analysis
2.6. Ethical Approval
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Tuberculosis 2018 Report; WHO/CDS/TB: Paris, France, 2018. [Google Scholar]
- Capewell, S.; Leitch, A.G. The value of contact procedures for tuberculosis in Edinburgh. Br. J. Dis. Chest 1984, 78, 317–329. [Google Scholar] [CrossRef]
- Marais, B.J.; Gie, R.P.; Schaaf, H.S.; Hesseling, A.C.; Obihara, C.C.; Nelson, L.J.; Enarson, D.A.; Donald, P.R.; Beyers, N. The clinical epidemiology of childhood pulmonary tuberculosis: A critical review of literature from the prechemotherapy era. Int. J. Tuberc. Lung Dis. 2004, 8, 278–285. [Google Scholar] [PubMed]
- World Health Organization. Recommendations for Investigating Contacts of Persons with Infectious Tuberculosis in Low- and Middle-Income Countries; WHO/HTM/TB: Geneva, Switzerland, 2012. [Google Scholar]
- World Health Organization. Latent Tuberculosis Infection: Updated and Consolidated Guidelines for Programmatic Management; WHO/CDS/TB: Geneva, Switzerland, 2018. [Google Scholar]
- Kenyon, T.A.; Creek, T.; Laserson, K.; Makhoa, M.; Chimidza, N.; Mwasekaga, M.; Tappero, J.; Lockman, S.; Moeti, T.; Binkin, N. Risk factors for transmission of Mycobacterium tuberculosis from HIV-infected tuberculosis patients, Botswana. Int. J. Tuberc. Lung Dis. 2002, 6, 843–850. [Google Scholar] [PubMed]
- Sinfield, R.; Nyirenda, M.; Haves, S.; Molyneux, E.M.; Graham, S.M. Risk factors for TB infection and disease in young childhood contacts in Malawi. Ann. Trop. Paediatr. 2006, 26, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Aissa, K.; Madhi, F.; Ronsin, N.; Delarocque, F.; Lecuyer, A.; Decludt, B.; Remus, N.; Abel, L.; Poirier, C.; Delacourt, C.; et al. Evaluation of a model for efficient screening of tuberculosis contact subjects. Am. J. Respir. Crit. Care Med. 2008, 177, 1041–1047. [Google Scholar] [CrossRef]
- Ma, N.; Zalwango, S.; Malone, L.L.; Nsereko, M.; Wampande, E.M.; Thiel, B.A.; Okware, B.; Igo, R.P.; Joloba, M.L.; Mupere, E.; et al. Tuberculosis Research Unit (TBRU). Clinical and epidemiological characteristics of individuals resistant to M. tuberculosis infection in a longitudinal TB household contact study in Kampala, Uganda. Infect. Dis. 2014, 14, 352–362. [Google Scholar]
- Ibaraki Prefectural Government. Tuberculosis Satistics in Ibaraki Prefectural Government. Available online: http://www.pref.ibaraki.jp/hokenfukushi/yobo/kiki/yobo/kansen/idwr/information/kekkaku/documents/2012.pdf (accessed on 1 August 2019).
- Isikawa, N.; Ahiko, T.; Inuzuka, K.; Inagaki, T.; Kato, S.; Kawabe, Y.; Kobayashi, N.; Sasaki, Y.; Suzuki, K.; Takamatsu, I.; et al. Kekkaku no Sesshokusha Kenko Shindan no Tebiki (Guidelines on Tuberculosis Contact Investigation Based on the Infectious Disease Control Law); Japan Anti-Tuberculosis Association: Tokyo, Japan, 2010. [Google Scholar]
- Seto, J.; Ahiko, T. Effectiveness of interferon-gamma release assays in the tuberculosis contact investigation of elderly people. Kekkaku 2014, 89, 503–508. [Google Scholar]
- Harada, N.; Nakajima, Y.; Higuchi, K.; Sekiya, Y.; Rothel, J.; Mori, T. Screening for Tuberculosis Infection Using Whole-Blood Interferon-γ and Mantoux Testing Among Japanese Healthcare Workers. Infect. Control Hosp. Epidemiol. 2006, 27, 442–448. [Google Scholar] [CrossRef]
- Nienhaus, A.; Schablon, A.; Diel, R. Interferon-Gamma Release Assay for the Diagnosis of Latent TB Infection–Analysis of Discordant Results, when Compared to the Tuberculin Skin Test. PLoS ONE 2008, 3. [Google Scholar] [CrossRef]
- Radhakrishna, S.; Frieden, T.R.; Subramani, R.; Santha, T.; Narayanan, P.R. Indian Council of Medical Research. Additional risk of developing TB for household members with a TB case at home at intake: A 15–year study. Int. J. Tuberc. Lung Dis. 2007, 11, 282–288. [Google Scholar]
- Triasih, R.; Robertson, C.; Duke, T.; Graham, S.M. Risk of infection and disease with Mycobacterium tuberculosis among children identified through prospective community–based contact screening in Indonesia. Trop. Med. Int. Health 2015, 20, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Pagaoa, M.A.; Royce, R.A.; Chen, M.P.; Golub, J.E.; Davidow, A.L.; Hirsch-Moverman, Y.; Marks, S.M.; Teeter, L.D.; Thickstun, P.M.; Katz, D.J. Risk factors for transmission of tuberculosis among United States-born African Americans and Whites. Int. J. Tuberc. Lung Dis. 2015, 19, 1485–1492. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cavany, S.M.; Sumner, T.; Vynnycky, E.; Flach, C.; White, R.G.; Thomas, H.L.; Maguire, H.; Anderson, C. An evaluation of tuberculosis contact investigations against national standards. Thorax 2017, 72, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Martinez, L.; Shen, Y.; Mupere, E.; Kizza, A.; Hill, P.C.; Whalen, C.C. Transmission of Mycobacterium Tuberculosis in Households and the Community: A Systematic Review and Meta-Analysis. Am. J. Epidemiol. 2017, 185, 1327–1339. [Google Scholar] [CrossRef] [PubMed]
- Eom, J.S.; Kim, I.; Kim, W.Y.; Jo, E.J.; Mok, J.; Kim, M.H.; Lee, K.; Kim, K.U.; Park, H.K.; Lee, M.K. Household tuberculosis contact investigation in a tuberculosis-prevalent country: Are the tuberculin skin test and interferon-gamma release assay enough in elderly contacts? Medicine 2018, 97, 9681–9687. [Google Scholar] [CrossRef] [PubMed]
- Rathi, S.K.; Akhtar, S.; Rahbar, M.H.; Azam, S.I. Prevalence and risk factors associated with tuberculin skin test positivity among household contacts of smear-positive pulmonary tuberculosis cases in Umerkot, Pakistan. Int. J. Tuberc. Lung Dis. 2002, 6, 851–857. [Google Scholar] [PubMed]
- Tornee, S.; Kaewkungwa, J.; Fungladda, W.; Silachamroon, U.; Akarasewi, P.; Sunakorn, P. Risk factors for tuberculosis infection among household contacts in Bangkok, Thailand. Southeast Asian J. Trop. Med. Public Health 2004, 35, 375–383. [Google Scholar]
- Nguyen, T.H.; Odermatt, P.; Slesak, G.; Barennes, H. Risk of latent tuberculosis infection in children living in households with tuberculosis patients: A cross sectional survey in remote northern Lao People’s Democratic Republic. Infect. Dis. 2009, 9, 96–105. [Google Scholar] [CrossRef]
- Rutherford, M.E.; Hill, P.C.; Maharani, W.; Apriani, L.; Sampurno, H.; van Crevel, R.; Ruslami, R. Risk factors for Mycobacterium tuberculosis infection in Indonesian children living with a sputum smear-positive case. Int. J. Tuberc. Lung Dis. 2012, 16, 1594–1599. [Google Scholar] [CrossRef]
- Acuña-Villaorduña, C.; Schmidt-Castellani, L.G.; Marques-Rodrigues, P.; White, L.F.; Hadad, D.J.; Gaeddert, M.; Ellner, J.J.; Fennelly, K.P.; Palaci, M.; Dietze, R.; et al. Cough-aerosol cultures of Mycobacterium tuberculosis in the prediction of outcomes after exposure. A household contact study in Brazil. PLoS ONE 2018, 13, e0206384. [Google Scholar] [CrossRef]
- Diel, R.; Loddenkemper, R.; Nienhaus, A. Predictive Value of Interferon-γ Release Assays and Tuberculin Skin Testing for Progression From Latent TB Infection to Disease State. Chest 2012, 142, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Pai, M.; Denkinger, C.M.; Kik, S.V.; Rangaka, M.X.; Zwerling, A.; Oxlade, O.; Metcalfe, J.Z.; Cattamanchi, A.; Dowdy, D.W.; Dheda, K.; et al. Gamma Interferon Release Assays for Detection of Mycobacterium tuberculosis Infection. Clin Microbiol Rev 2014, 27. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Systematic Screening for Active Tuberculosis: Principles and Recommendations; WHO/HTM/TB: Geneva, Switzerland.
- World Health Organization. Guidelines on the management of latent tuberculosis infection; WHO/HTM/TB: Geneva, Switzerland, 2015; ISBN 978 92 4 154890 8. [Google Scholar]
- Julie, L.; Dixie, E.S.; Tanja, P.; Steven, L.S.; Jay, M.B.; Maria, S.P.; Mary, L.L.; Suzanne, M.H.; Teresa, F.R.; Jeffrey, D.S.; et al. Guidelines for the Investigation of Contacts of Persons with Infectious Tuberculosis. Recomm. Rep. 2005, 54, 1–47. [Google Scholar]
- TB CARE 1. International Standards for Tuberculosis Care; TB CARE 1: The Hague, The Netherlands, 2014. [Google Scholar]
| Variables | Close Contact (N = 439) |
|---|---|
| Age | |
| 0–19 | 70 (16%) |
| 20–29 | 74 (17%) |
| 30–39 | 105 (24%) |
| 40–49 | 98 (22%) |
| 50–59 | 92 (21%) |
| Sex | |
| Male | 220 (50%) |
| Female | 219 (50%) |
| Cough in index patient | |
| + | 243 (55%) |
| − | 196 (45%) |
| Cavitation on radiographs of index patient | |
| + | 221 (50%) |
| − | 218 (50%) |
| Sputum smear AFB of index patient | |
| − | 103 (23%) |
| Scanty | 41 (9%) |
| 1+ | 115 (26%) |
| 2+ | 132 (30%) |
| 3+ | 48 (11%) |
| Variables | Close Contacts of Index TB Patient | |||
|---|---|---|---|---|
| QFT ≥ 0.35 IU/mL | QFT < 0.35 IU/mL | Univariate Analysis | Multivariate Analysis | |
| (%) | (%) | OR (95% CI) | aOR (95% CI) | |
| N | 57 (13%) | 382 (87%) | ||
| Index TB patient | ||||
| Cough | ||||
| + | 41 (17%) | 202 (83%) | 2.28 (1.2–4.2) | 1.80 (0.94–3.5) |
| − | 16 (8%) | 180 (92%) | 1 | 1 |
| Cavitation on radiographs | ||||
| + | 31 (14%) | 190 (86%) | 1.20 (0.69–2.1) | 0.95 (0.49–1.8) |
| − | 26 (12%) | 192 (88%) | 1 | 1 |
| Sputum smear | ||||
| − | 10 (10%) | 93 (90%) | 1 | 1 |
| Scanty | 3 (7%) | 38 (93%) | 0.73 (0.19–2.8) | 0.68 (0.17–2.8) |
| 1+ | 11 (10%) | 104 (90%) | 0.98 (0.40–2.4) | 1.12 (0.45–2.8) |
| 2+ | 16 (12%) | 116 (88%) | 1.28 (0.56–3.0) | 1.20 (0.48–3.0) |
| 3+ | 17 (35%) | 31 (65%) | 5.10 (2.1–12.3) | 4.96 (1.9–12.9) |
| Close contacts of index TB patients | ||||
| Age | ||||
| 0–19 | 11 (16%) | 59 (84%) | 1 | 1 |
| 20–29 | 7 (9%) | 67 (91%) | 0.56 (0.20–1.5) | 0.49 (0.17–1.4) |
| 30–39 | 10 (10%) | 95 (90%) | 0.56 (0.23–1.4) | 0.58 (0.22–1.5) |
| 40–49 | 15 (15%) | 83 (85%) | 0.97 (0.42–2.3) | 0.91 (0.37–2.2) |
| 50–59 | 14 (15%) | 78 (85%) | 0.96 (0.41–2.3) | 1.09 (0.44–2.7) |
| Sex | ||||
| Male | 30 (14%) | 190 (86%) | 1.12 (0.64–2.0) | 0.97 (0.54–1.7) |
| Female | 27 (12%) | 192 (88%) | 1 | 1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogata, T.; Nagasu, N.; Uehara, R.; Ito, K. Association of Low Sputum Smear Positivity among Tuberculosis Patients with Interferon-Gamma Release Assay Outcomes of Close Contacts in Japan. Int. J. Environ. Res. Public Health 2019, 16, 3713. https://doi.org/10.3390/ijerph16193713
Ogata T, Nagasu N, Uehara R, Ito K. Association of Low Sputum Smear Positivity among Tuberculosis Patients with Interferon-Gamma Release Assay Outcomes of Close Contacts in Japan. International Journal of Environmental Research and Public Health. 2019; 16(19):3713. https://doi.org/10.3390/ijerph16193713
Chicago/Turabian StyleOgata, Tsuyoshi, Natsuki Nagasu, Ritei Uehara, and Kunihiko Ito. 2019. "Association of Low Sputum Smear Positivity among Tuberculosis Patients with Interferon-Gamma Release Assay Outcomes of Close Contacts in Japan" International Journal of Environmental Research and Public Health 16, no. 19: 3713. https://doi.org/10.3390/ijerph16193713
APA StyleOgata, T., Nagasu, N., Uehara, R., & Ito, K. (2019). Association of Low Sputum Smear Positivity among Tuberculosis Patients with Interferon-Gamma Release Assay Outcomes of Close Contacts in Japan. International Journal of Environmental Research and Public Health, 16(19), 3713. https://doi.org/10.3390/ijerph16193713





