Epidermal Growth Factor is Effective in the Treatment of Diabetic Foot Ulcers: Meta-Analysis and Systematic Review
Abstract
1. Introduction
2. Methodology
2.1. Information Source and Search Strategy
2.2. Eligibility, Study Selection, and Data Extraction
2.3. Data Synthesis and Analysis
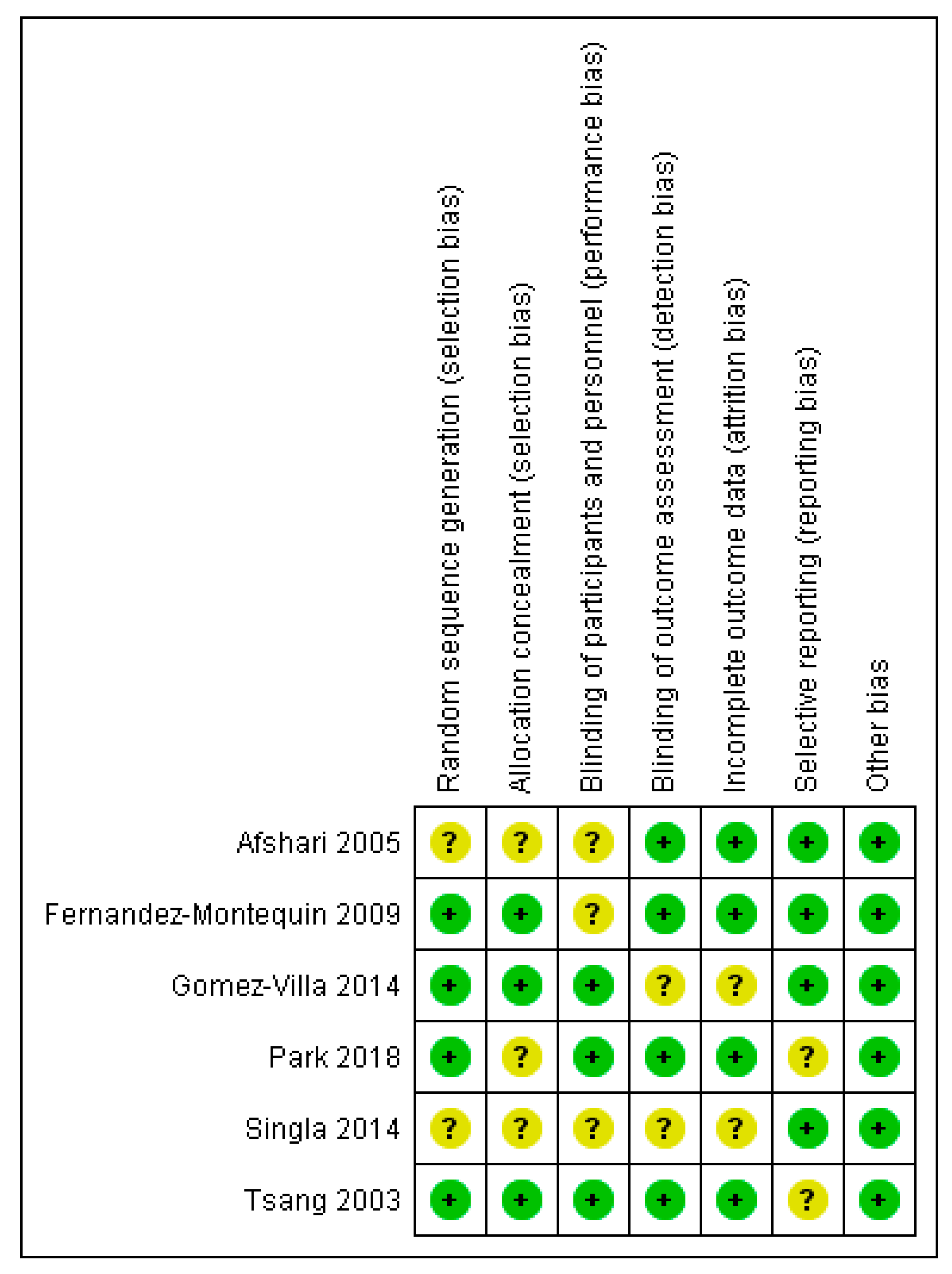
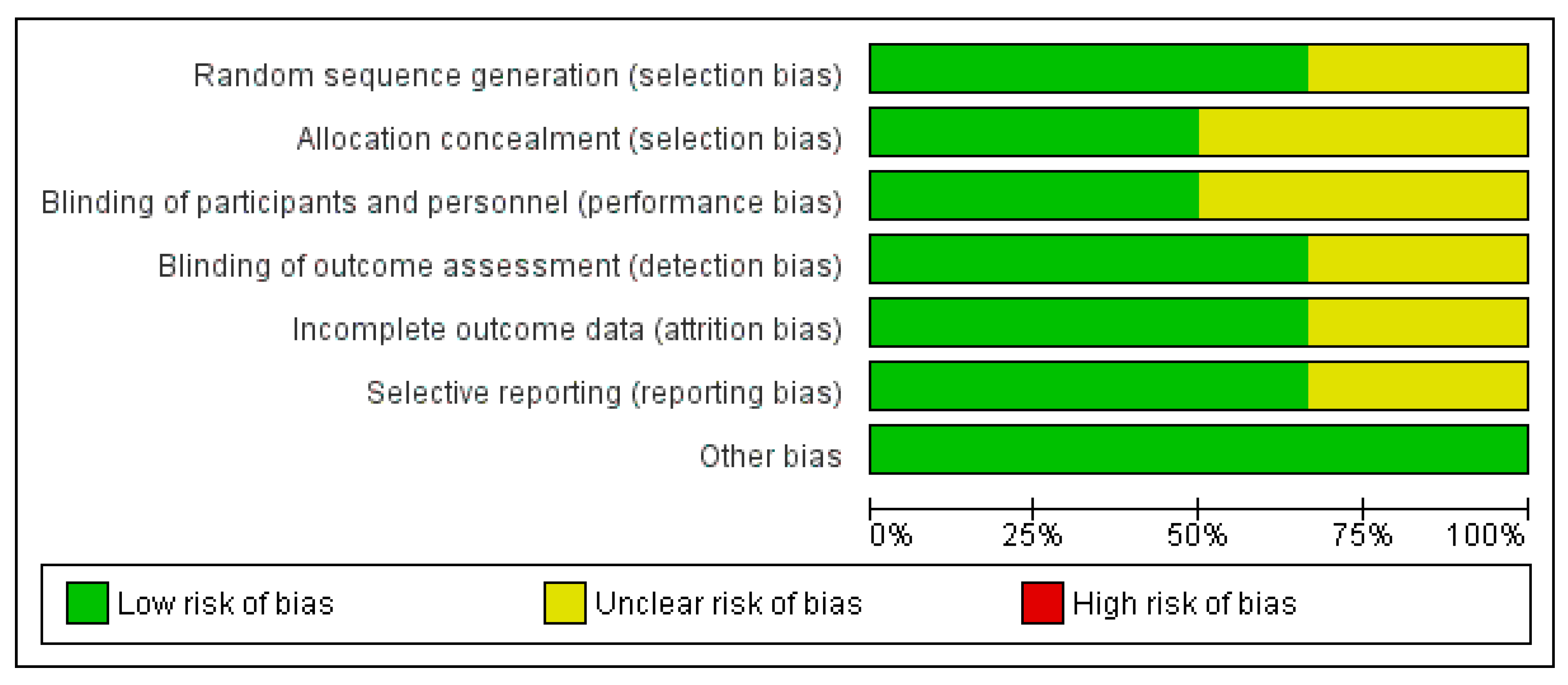
2.4. Risk of Bias
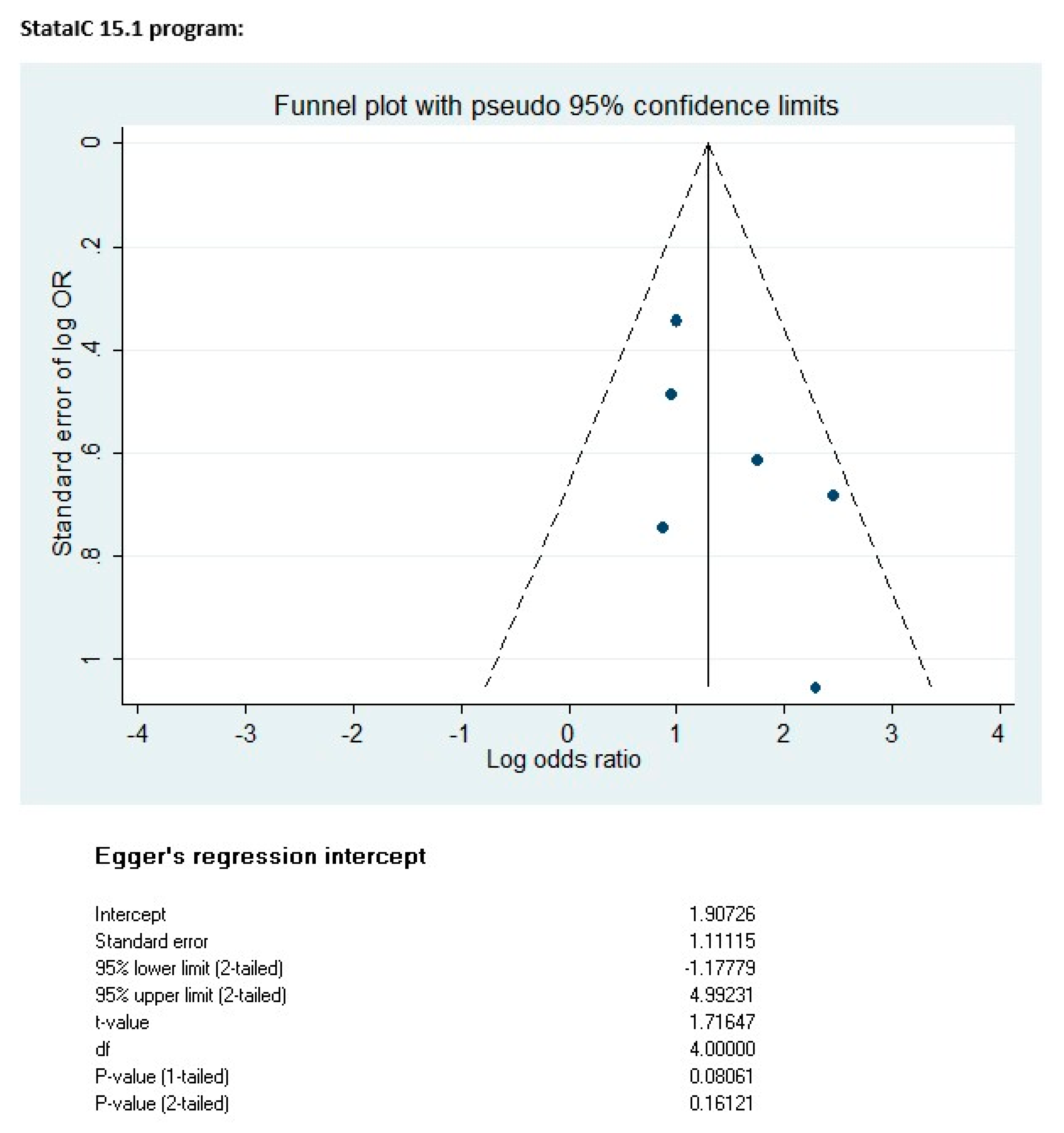
2.5. Publication Bias
2.6. Quality of Evidence
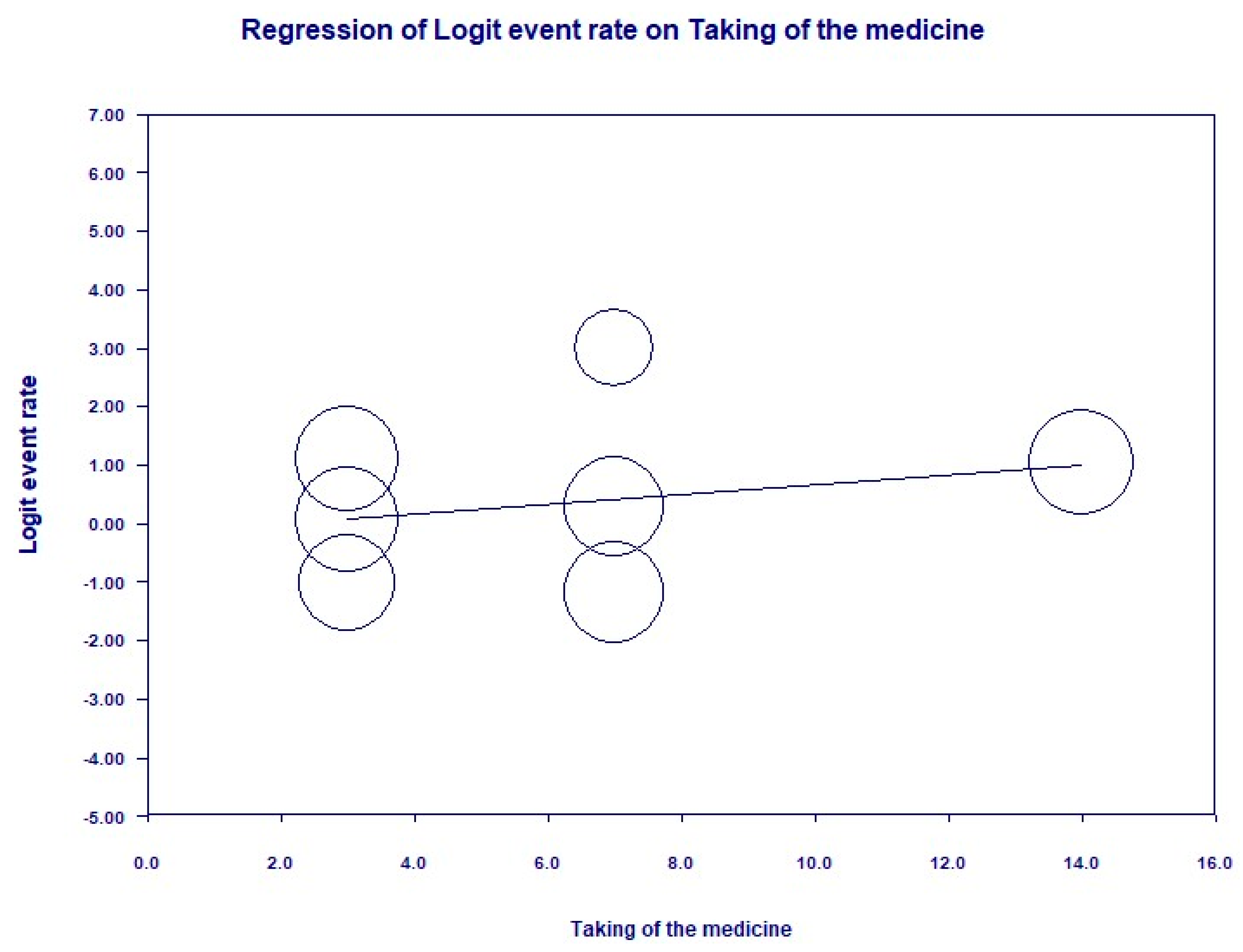
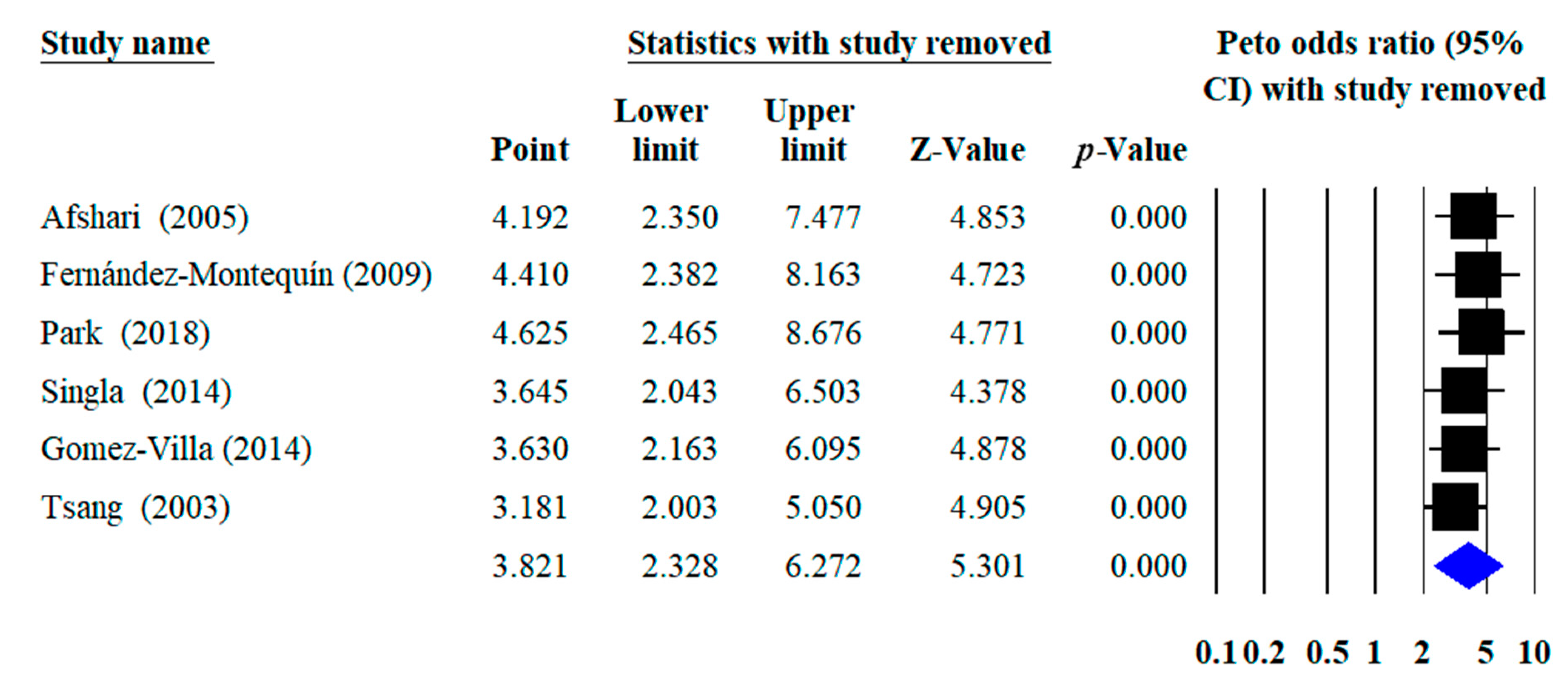
2.7. Sensitivity Analysis
3. Results
3.1. Study Selection
3.2. Study Characteristic
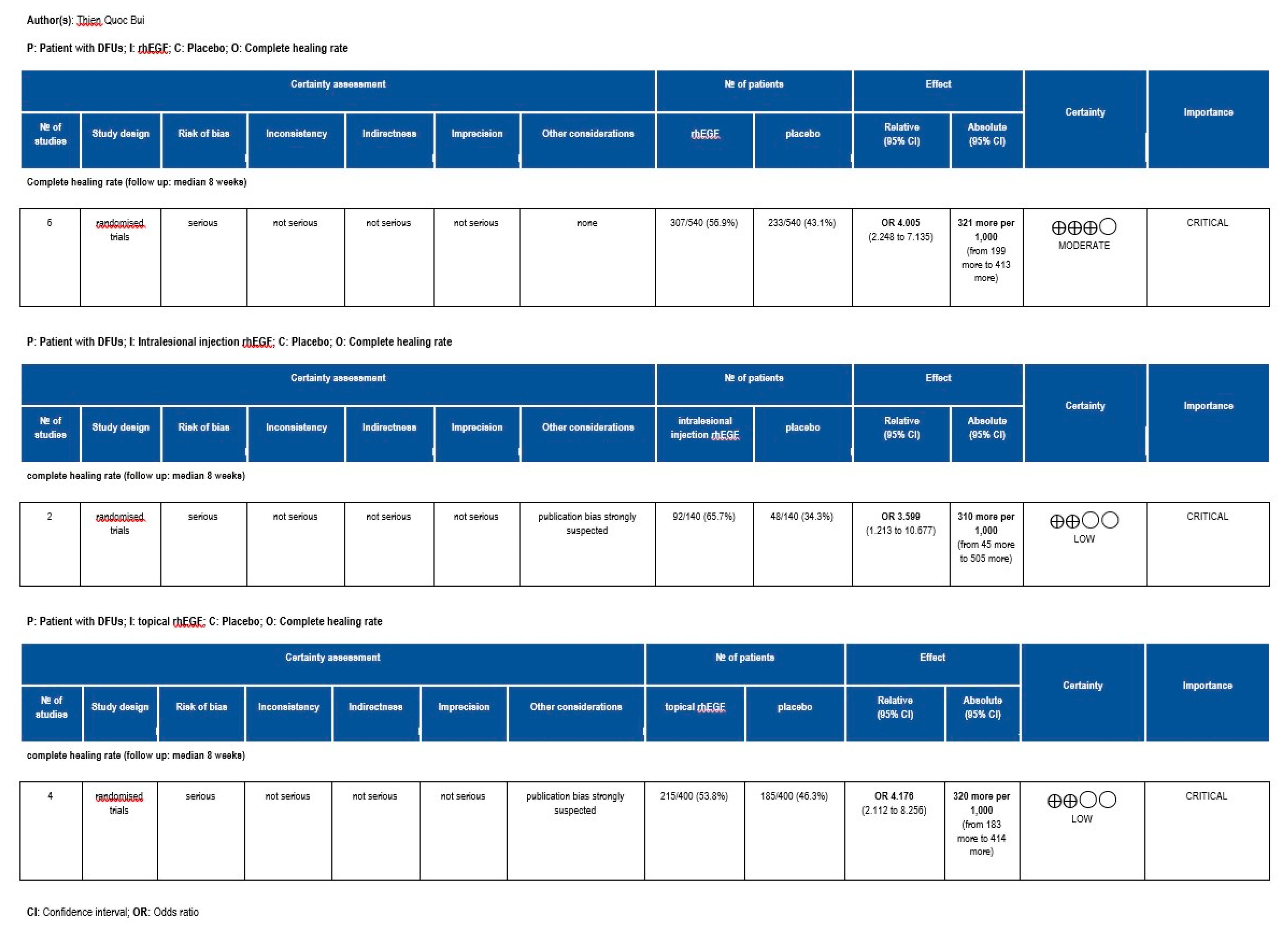
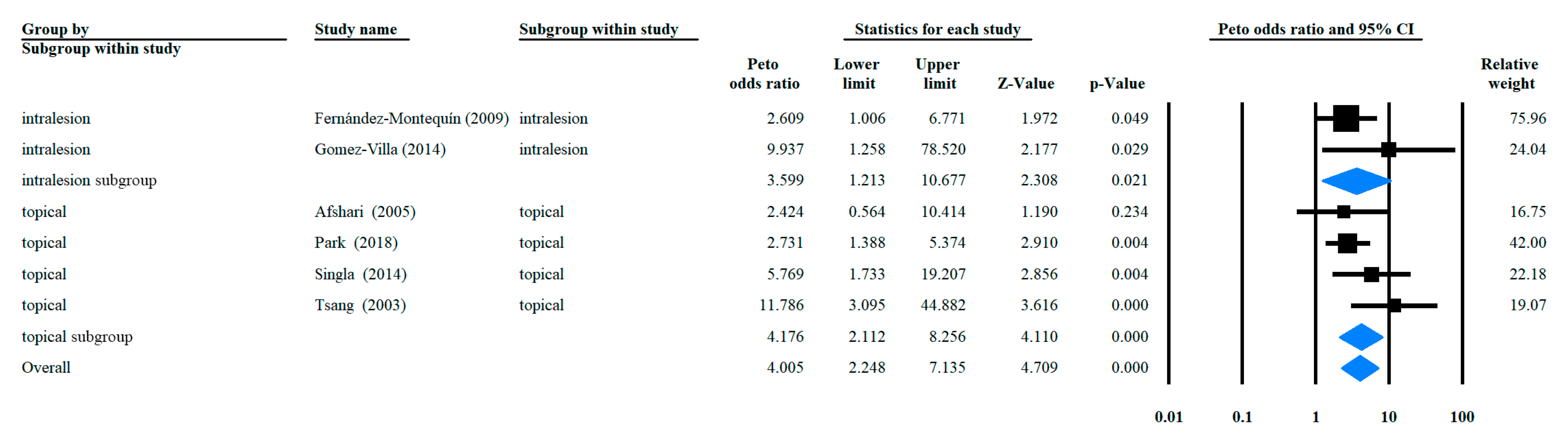
3.3. Odds Ratio of Complete Healing Rate with rhEGF Versus Placebo
3.4. Quality of Evidence
3.5. Risk of Bias Assessment
3.6. Publication Bias
3.7. Sensitivity Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Papatheodorou, K.; Banach, M.; Bekiari, E.; Rizzo, M.; Edmonds, M. Complications of Diabetes 2017. J. Diabetes Res. 2018, 2018, 3086167. [Google Scholar] [CrossRef] [PubMed]
- W.H.O. Diabetes Programme. Available online: https://www.who.int/diabetes/action_online/basics/en/index3.html (accessed on 16 July 2019).
- Zhang, P.; Lu, J.; Jing, Y.; Tang, S.; Zhu, D.; Bi, Y. Global epidemiology of diabetic foot ulceration: A systematic review and meta-analysis (dagger). Ann. Med. 2017, 49, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- The National Institute for Health and Care Excellence. Diabetic Foot Problems: Prevention and Management; The National Institute for Health and Care Excellence: London, UK, 2016. [Google Scholar]
- Lavery, L.A.; Davis, K.E.; Berriman, S.J.; Braun, L.; Nichols, A.; Kim, P.J.; Margolis, D.; Peters, E.J.; Attinger, C. WHS guidelines update: Diabetic foot ulcer treatment guidelines. Wound Repair Regen. 2016, 24, 112–126. [Google Scholar] [CrossRef] [PubMed]
- Martí-Carvajal, A.J.; Gluud, C.; Nicola, S.; Simancas-Racines, D.; Reveiz, L.; Oliva, P.; Cedeño-Taborda, J. Growth factors for treating diabetic foot ulcers. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Oka, Y.; Orth, D.N. Human plasma epidermal growth factor/beta-urogastrone is associated with blood platelets. J. Clin. Investig. 1983, 72, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Goodlad, R.A.; Raja, K.B.; Peters, T.J.; Wright, N.A. Effects of urogastrone-epidermal growth factor on intestinal brush border enzymes and mitotic activity. Gut 1991, 32, 994–998. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Tsang, M.W.; Wong, W.K.; Hung, C.S.; Lai, K.M.; Tang, W.; Cheung, E.Y.; Kam, G.; Leung, L.; Chan, C.W.; Chu, C.M.; et al. Human epidermal growth factor enhances healing of diabetic foot ulcers. Diabetes Care 2003, 26, 1856–1861. [Google Scholar] [CrossRef] [PubMed]
- Afshari, M.; Larijani, B.; Fadayee, M.; Darvishzadeh, F.; Ghahary, A.; Pajouhi, M.; Bastanhagh, M.H.; Baradar-Jalili, R.; Vassigh, A.R. Efficacy of topical epidermal growth factor in healing diabetic foot ulcers. Therapy 2005, 2, 759–765. [Google Scholar] [CrossRef]
- Fernandez-Montequin, J.I.; Valenzuela-Silva, C.M.; Diaz, O.G.; Savigne, W.; Sancho-Soutelo, N.; Rivero-Fernandez, F.; Sanchez-Penton, P.; Morejon-Vega, L.; Artaza-Sanz, H.; Garcia-Herrera, A.; et al. Intra-lesional injections of recombinant human epidermal growth factor promote granulation and healing in advanced diabetic foot ulcers: Multicenter, randomised, placebo-controlled, double-blind study. Int. Wound J. 2009, 6, 432–443. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Villa, R.; Aguilar-Rebolledo, F.; Lozano-Platonoff, A.; Teran-Soto, J.M.; Fabian-Victoriano, M.R.; Kresch-Tronik, N.S.; Garrido-Espindola, X.; Garcia-Solis, A.; Bondani-Guasti, A.; Bierzwinsky-Sneider, G.; et al. Efficacy of intralesional recombinant human epidermal growth factor in diabetic foot ulcers in Mexican patients: A randomized double-blinded controlled trial. Wound Repair Regen. 2014, 22, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Singla, S.; Garg, R.; Kumar, A.; Gill, C. Efficacy of topical application of beta urogastrone (recombinant human epidermal growth factor) in Wagner’s Grade 1 and 2 diabetic foot ulcers: Comparative analysis of 50 patients. J. Nat. Sci. Biol. Med. 2014, 5, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Park, K.H.; Han, S.H.; Hong, J.P.; Han, S.K.; Lee, D.H.; Kim, B.S.; Ahn, J.H.; Lee, J.W. Topical epidermal growth factor spray for the treatment of chronic diabetic foot ulcers: A phase III multicenter, double-blind, randomized, placebo-controlled trial. Diabetes Res. Clin. Pract. 2018, 142, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Min, D.; Guo, G.; Liao, X.; Fu, Z. Experimental study of epidermal growth factor and acidic fibroblast growth factor in the treatment of diabetic foot wounds. Exp. Ther. Med. 2018, 15, 5365–5370. [Google Scholar] [CrossRef] [PubMed]
- Sutton, A.J.; Duval, S.J.; Tweedie, R.L.; Abrams, K.R.; Jones, D.R. Empirical assessment of effect of publication bias on meta-analyses. BMJ 2000, 320, 1574–1577. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Geng, Z.; Ma, K.; Sun, X.; Fu, X. Efficacy of Topical Recombinant Human Epidermal Growth Factor for Treatment of Diabetic Foot Ulcer: A Systematic Review and Meta-Analysis. Int. J. Low. Extrem. Wounds 2016, 15, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Berlanga-Acosta, J.; Fernandez-Montequin, J.; Valdes-Perez, C.; Savigne-Gutierrez, W.; Mendoza-Mari, Y.; Garcia-Ojalvo, A.; Falcon-Cama, V.; Garcia Del Barco-Herrera, D.; Fernandez-Mayola, M.; Perez-Saad, H.; et al. Diabetic Foot Ulcers and Epidermal Growth Factor: Revisiting the Local Delivery Route for a Successful Outcome. BioMed Res. Int. 2017, 2017, 2923759. [Google Scholar] [CrossRef] [PubMed]
- Laimer, M. MAPK14 as candidate for genetic susceptibility to diabetic foot ulcer. Br. J. Dermatol. 2017, 177, 1482–1483. [Google Scholar] [CrossRef] [PubMed]

| Author | Year of Publication | Study Design | Demography | Intervention Type | Route | Apply Frequency | Treatment Duration (weeks) | Patient Data | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Country | Number of Centers | Number of Patients | Age ± SD (Years) | Type of DM | Duration of Diabetes ± SD (Years) | |||||||
| Tsang | 2003 | RCT | China | 1 | Actovecgin % | Topical | Once a day | 12 | 19 | 64.37 ± 11.67 | 1 or 2 | 10.11 ± 8.29 |
| 0.02 wt% rhEGF | 21 | 68.76 ± 10.34 | 9.85 ± 7.79 | |||||||||
| 0.04 wt% rhEGF | 21 | 62.24 ± 13.68 | 9.005 ± 6.19 | |||||||||
| Afshari | 2005 | RCT | Iran | 1 | 1000 mg 1% sulfadiazine | Topical | Once a day | 8 | 20 | 55.84 | 1 or 2 | NA |
| 1 mg rhEGF in 1000 mg 1% sulfadiazine | 30 | 58.8 | NA | |||||||||
| Fernández-Montequín | 2009 | RCT | Cuba | 20 | Placebo | Intralesional injection | Three times a week on alternate days | 8 | 32 | NA | 1 or 2 | 15 |
| 25 µ/g | 33 | NA | 15 | |||||||||
| 75 µ/g | 44 | NA | 19.5 | |||||||||
| Gomez-Villa | 2014 | RCT | Mexico | 2 | Placebo | Intralesional injection | Three times a week on alternate days | 8 | 16 | 55.1 ± 10.6 | 1 or 2 | 15.3 ± 8.4 |
| 75 µ/g | 15 | 62.1 ± 12.8 | 17.3 ± 10 | |||||||||
| Singla | 2014 | RCT | India | 1 | Betadine | Topical | Once every two weeks | 8 | 25 | 55.84 | 1 or 2 | NA |
| Urogastrone (rhEGF) gel 15 g | 24 | 58.8 | NA | |||||||||
| Park | 2018 | RCT | Korean | 6 | Saline | Topical | Twice a day | 12 | 72 | 59.31 ± 12.64 | 1 or 2 | NA |
| 0.005% rhEGF spray | 69 | 56.52 ± 12.71 | NA | |||||||||
| Xu | 2018 | RCT | China | 1 | Saline | Topical | Once a day | 8 | 49 | 63 ± 4.56 | 1 or 2 | NA |
| 40 IU/cm2 | 50 | 65 ± 3.65 | NA | |||||||||
| Author | Intervention Type | Route | Apply Frequency | Treatment Duration (weeks) | Ulcer Data | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Location | Severity | Ischemia Level | Infection | Ulcer Area ± SD (cm2) | Ulcer Duration ± SD (Weeks) | Complete Healing Rate (%) | |||||
| Tsang 2003 | Actovecgin % | Topical | Once a day | 12 | Foot | Wagner 1 or 2 | ABI ≥ 0.7 | NA | 3.48 ± 0.82 | 12 ± 15.47 | 42.1 |
| 0.02 wt% rhEGF | Foot | 3.4 ± 1.1 | 11.48 ± 14.68 | 57.14 | |||||||
| 0.04 wt% rhEGF | Foot | 2.78 ± 0.82 | 8.24 ± 5.55 | 95.3 | |||||||
| Afshari 2005 | 1000 mg 1%sulfadiazine | Topical | Once a day | 8 | Foot | Wagner 1 or 2 | ABI < 1: 49% participants | NA | 103.4 ± 147.8 | 59.7 ± 55.5 | 10 |
| 1 mg rhEGF in 1000 mg 1% sulfadiazine | Foot | ABI < 1: 50% participants | 87.5 ± 103.2 | 42.9 ± 38.4 | 23.3 | ||||||
| Fernández-Montequín 2009 | placebo | Intralesional injection | Three times a week on alternate days | 8 | Foot | Wagner 3 or 4 | Hemoglobin ≥100 g/L | NA | 21.8 | 4.9 | 52.1 |
| 25 µ/g | Foot | 4.3 | 20.1 | 52.1 | |||||||
| 75 µ/g | Foot | 4.3 | 28.5 | 75.5 | |||||||
| Gomez-Villa 2014 | Placebo | Intralesional injection | Three times a week on alternate days | 8 | Foot | Texas 1, 2, and 3 | ABI > 0.6 | NA | 11.9 ± 11.8 | 15.3 ± 8.4 | 0 |
| 75 µ/g | Foot | 19.2 ± 15.7 | 25.8 ± 44 | 23.5 | |||||||
| Singla 2014 | Betadine | Topical | Once every two weeks | 8 | Foot | Wagner 1 or 2 | ABI ≥ 0.75 | NA | NA | NA | 12 |
| Urogastrone (rhEGF) gel 15 g | Foot | NA | NA | 48 | |||||||
| Park 2018 | Saline | Topical | Twice a day | 12 | Foot | Wagner 1 or 2 | TcPO2 ≥ 30 mmHg or palpable dorsalis pedis artery or posterior tibial artery | NA | 2.35 ± 2.69 | 29.6 ± 60.2 | 50.6 |
| 0.005% rhEGF spray | Foot | 2.8 ± 3.72 | 38.48 ± 70.24 | 73.2 | |||||||
| Xu 2018 | Saline | Topical | Once a day | 8 | Foot | Wagner 1 or 2 | NA | NA | 2.35 ± 2.69 | 29.6 ± 60.2 | NA |
| 40 IU/cm2 | Foot | 2.8 ± 3.72 | 70.24 ± 38.48 | NA | |||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bui, T.Q.; Bui, Q.V.P.; Németh, D.; Hegyi, P.; Szakács, Z.; Rumbus, Z.; Tóth, B.; Emri, G.; Párniczky, A.; Sarlós, P.; et al. Epidermal Growth Factor is Effective in the Treatment of Diabetic Foot Ulcers: Meta-Analysis and Systematic Review. Int. J. Environ. Res. Public Health 2019, 16, 2584. https://doi.org/10.3390/ijerph16142584
Bui TQ, Bui QVP, Németh D, Hegyi P, Szakács Z, Rumbus Z, Tóth B, Emri G, Párniczky A, Sarlós P, et al. Epidermal Growth Factor is Effective in the Treatment of Diabetic Foot Ulcers: Meta-Analysis and Systematic Review. International Journal of Environmental Research and Public Health. 2019; 16(14):2584. https://doi.org/10.3390/ijerph16142584
Chicago/Turabian StyleBui, Thien Quoc, Quoc Van Phu Bui, Dávid Németh, Péter Hegyi, Zsolt Szakács, Zoltán Rumbus, Barbara Tóth, Gabriella Emri, Andrea Párniczky, Patricia Sarlós, and et al. 2019. "Epidermal Growth Factor is Effective in the Treatment of Diabetic Foot Ulcers: Meta-Analysis and Systematic Review" International Journal of Environmental Research and Public Health 16, no. 14: 2584. https://doi.org/10.3390/ijerph16142584
APA StyleBui, T. Q., Bui, Q. V. P., Németh, D., Hegyi, P., Szakács, Z., Rumbus, Z., Tóth, B., Emri, G., Párniczky, A., Sarlós, P., & Varga, O. (2019). Epidermal Growth Factor is Effective in the Treatment of Diabetic Foot Ulcers: Meta-Analysis and Systematic Review. International Journal of Environmental Research and Public Health, 16(14), 2584. https://doi.org/10.3390/ijerph16142584






