Neonatal Bisphenol A Exposure Affects the IgM Humoral Immune Response to 4T1 Breast Carcinoma Cells in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Neonatal BPA Exposure
2.3. Mammary Tumour Induction
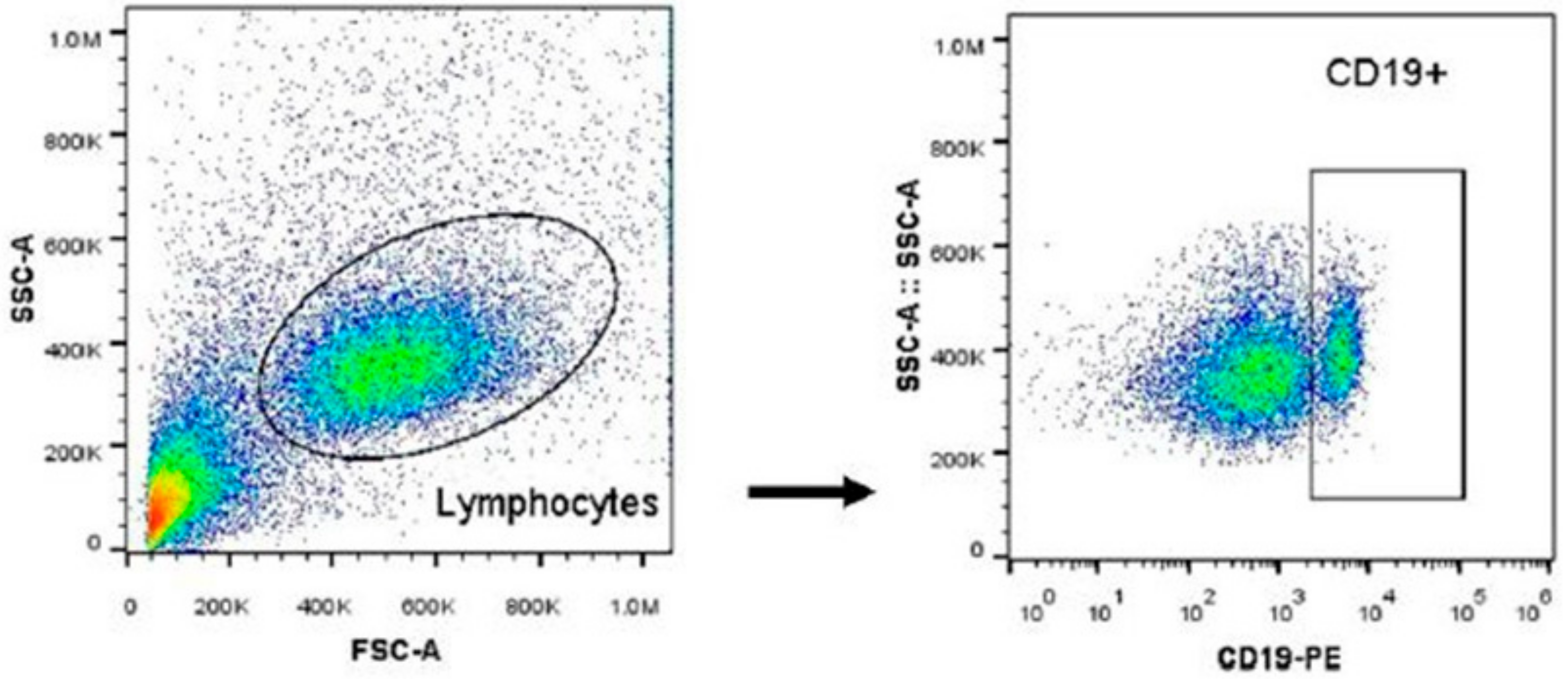
2.4. Cell Culture and Flow Cytometry
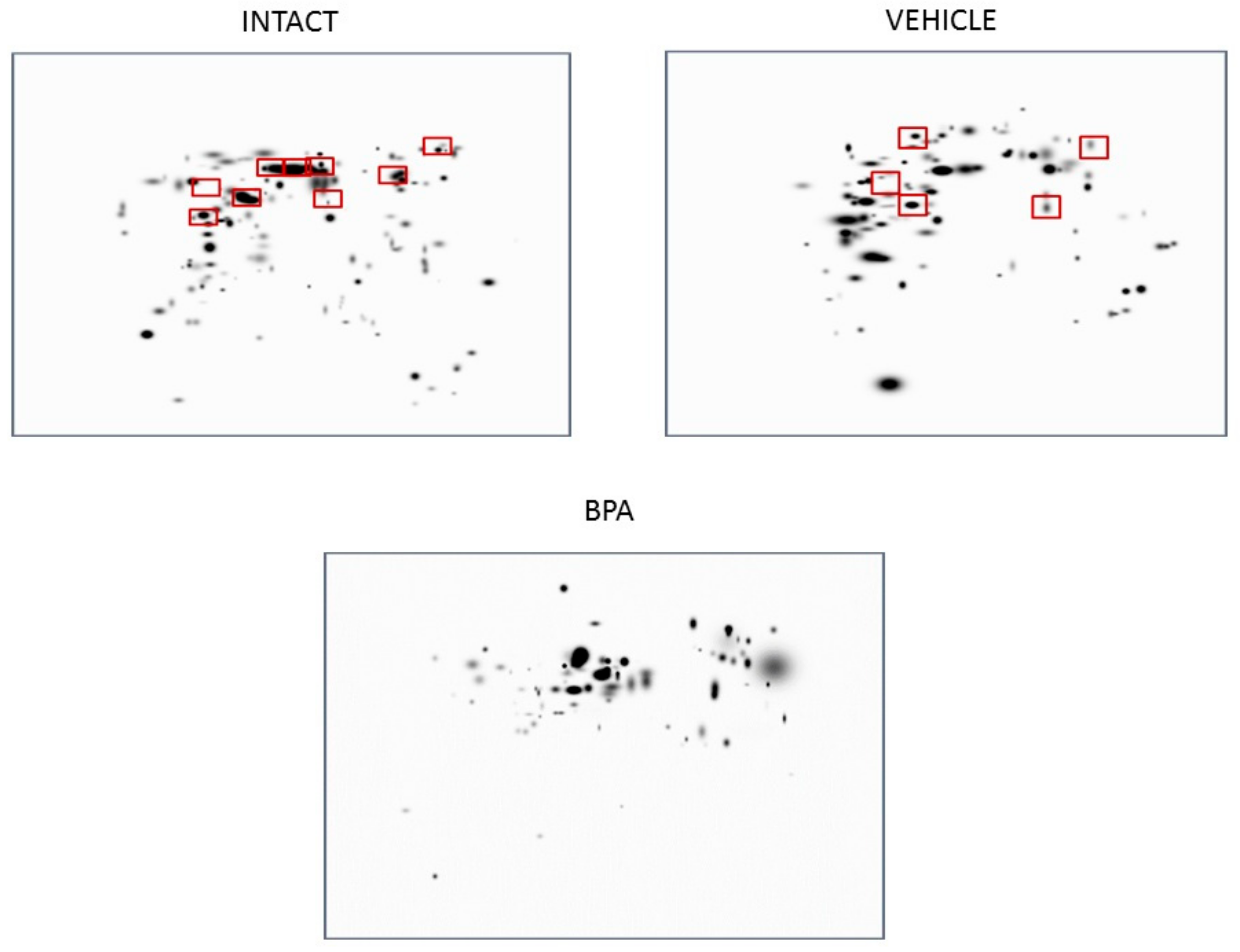
2.5. Two-Dimensional Immunoblot and Image Processing
2.6. Statistical Analysis
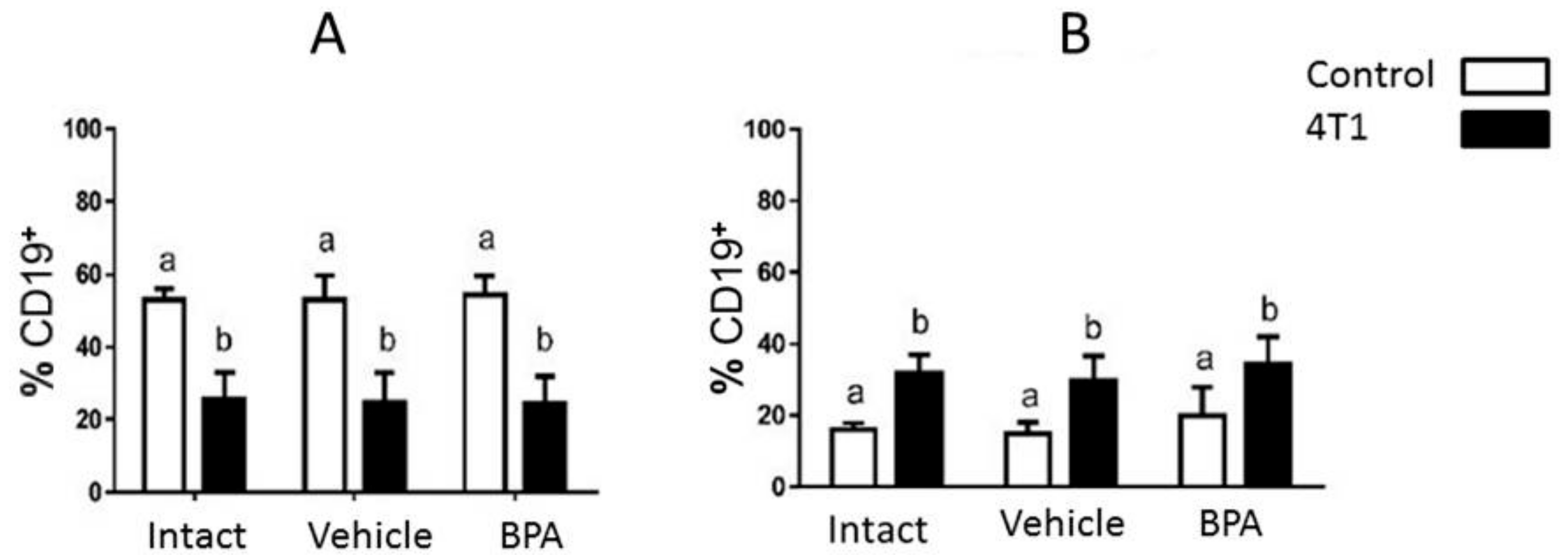
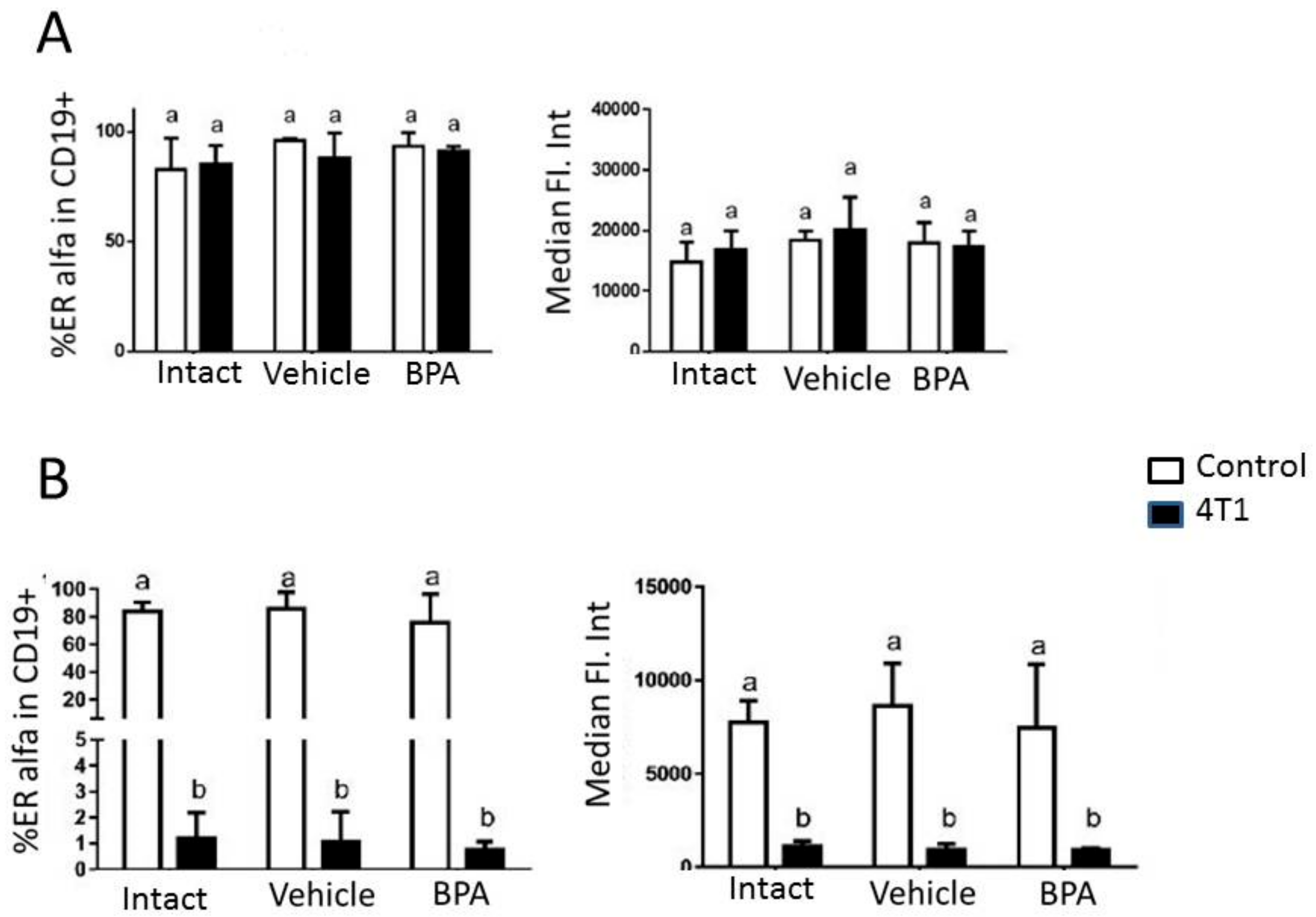
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stefanidou, M.; Maravelias, C.; Spiliopoulou, C. Human Exposure to Endocrine Disruptors and Breast Milk. Endocr. Metab. Immune Disord. Drug Targets 2009, 9, 269–276. [Google Scholar] [CrossRef]
- Welshons, W.V.; Nagel, S.C.; vom Saal, F.S. Large effects from small exposures. III. Endocrine mechanisms mediating effects of bisphenol A at levels of human exposure. Endocrinology 2006, 147, S56–S69. [Google Scholar] [CrossRef]
- Amaral Mendes, J.J. The endocrine disrupters: A major medical challenge. Food Chem. Toxicol. 2002, 40, 781–788. [Google Scholar] [CrossRef]
- Soto, A.M.; Vandenberg, L.N.; Maffini, M.V.; Sonnenschein, C. Does breast cancer start in the womb? Basic Clin. Pharmacol. Toxicol. 2008, 102, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Maffini, M.V.; Rubin, B.S.; Sonnenschein, C.; Soto, A.M. Endocrine disruptors and reproductive health: The case of bisphenol A. Mol. Cell Endocrinol. 2006, 254–255, 179–186. [Google Scholar] [CrossRef]
- Rubin, B.S. Bisphenol A: An endocrine disruptor with widespread exposure and multiple effects. J. Steroid Biochem. Mol. Biol. 2011, 127, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Zalko, D.; Jacques, C.; Duplan, H.; Bruel, S.; Perdu, E. Viable skin efficiently absorbs and metabolizes bisphenol A. Chemosphere 2011, 82, 424–430. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Hauser, R.; Marcus, M.; Olea, N.; Welshons, W.V. Human exposure to bisphenol A (BPA). Reprod. Toxicol. 2007, 24, 139–177. [Google Scholar] [CrossRef]
- Palacios-Arreola, M.I.; Nava-Castro, K.E.; Río-Araiza, V.H.D.; Pérez-Sánchez, N.Y.; Morales-Montor, J. A single neonatal administration of Bisphenol A induces higher tumour weight associated to changes in tumor microenvironment in the adulthood. Sci. Rep. 2017, 7, 10573. [Google Scholar] [CrossRef] [PubMed]
- Chalubinski, M.; Kowalski, M.L. Endocrine disrupters—Potential modulators of the immune system and allergic response. Allergy 2006, 61, 1326–1335. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Zaragoza, M.; Hernández, R.; Ostoa-Saloma, P. 2D immunoblots show differential response of mouse IgG and IgM antibodies to antigens of mammary carcinoma 4 T1 cells. Cancer Cell Int. 2014, 14, 9. [Google Scholar] [CrossRef] [PubMed]
- NORMA Oficial Mexicana NOM-062-ZOO-1999. Available online: https://www.biomedicas.unam.mx/wp-content/pdf/unidad-de-modelos-biologicos/nom-062-zoo-1999.pdf?x21431 (accessed on 11 April 2019).
- Pulaski, B.A.; Ostrand-Rosenberg, S. Mouse 4T1 breast tumor model. In Current Protocols in Immunology; Coligan, J., John, E., Eds.; Wiley & Sons Inc.: New York, NY, USA, 2001. [Google Scholar]
- Garín, M.I.; Chu, C.-C.; Dela, G.; Cernuda-Morollón, E.; Wait, R.; Lechler, R. Galectin-1: A key effector of regulation mediated by CD4 CD25 T cells. Blood 2007, 109, 2058–2065. [Google Scholar] [CrossRef]
- Dalotto-Moreno, T.; Croci, D.O.; Cerliani, J.P.; Martinez-Allo, V.C.; Dergan-Dylon, S.; Mendez-Huergo, S.P.; Stupirski, J.C.; Mazal, D.; Osinaga, E.; Toscano, M.A.; et al. Targeting Galectin-1 Overcomes Breast Cancer-Associated Immunosuppression and Prevents Metastatic Disease. Cancer Res. 2013, 73, 1107–1117. [Google Scholar] [CrossRef] [PubMed]
- MacLennan, I.C. Germinal centers. Annu. Rev. Immunol. 1994, 12, 117–139. [Google Scholar] [CrossRef] [PubMed]
- McHeyzer-Williams, M.; Okitsu, S.; Wang, N.; McHeyzer-Williams, L. Molecular programming of B cell memory. Nat. Rev. Immunol. 2011, 12, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Weill, J.C.; Le Gallou, S.; Hao, Y.; Reynaud, C.A. Multiple players in mouse B cell memory. Curr. Opin. Immunol. 2013, 25, 334–338. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Coutinho, A.; Kazatchkine, M.D.; Avrameas, S. Natural autoantibodies. Curr. Opin. Immunol. 1995, 7, 812–818. [Google Scholar] [CrossRef]
- Baumgarth, N.; Herman, O.C.; Jager, G.C.; Herzenberg, L.A.; Herzenberg, L.A. Innate and acquired humoral immunities to influenza virus are mediated by distinct arms of the immune system. Proc. Natl. Acad. Sci. USA 1999, 96, 2250–2255. [Google Scholar] [CrossRef]
- MacLennan, C.; Fieschi, C.; Lammas, D.A.; Picard, C.; Dorman, S.E.; Sanal, O.; MacLennan, J.M.; Holland, S.M.; Ottenhoff, T.H.; Casanova, J.L.; et al. Interleukin IL-12 and IL-23 are key cytokines for immunity against Salmonella in humans. J. Infect. Dis. 2004, 190, 1755–1757. [Google Scholar] [CrossRef]
- Jouanguy, E.; Döffinger, R.; Dupuis, S.; Pallier, A.; Altare, F.; Casanova, J.L. IL-12 and IFN-gamma in host defense against mycobacteria and salmonella in mice and men. Curr. Opin. Immunol. 1999, 11, 346–351. [Google Scholar] [CrossRef]
- Gostner, J.M.; Raggl, E.; Becker, K.; Überalla, F.; Schennach, H.; Pease, J.E.; Fuchs, D. Bisphenol A suppresses Th1-type immune response in human peripheral blood mononuclear cells in vitro. Immunol. Lett. 2015, 168, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Sawai, C.; Anderson, K.; Walser-Kuntz, D. Effect of Bisphenol A on Murine Immune Function: Modulation of Interferon-gamma, IgG2a and Disease Symptoms in NZB × NZW F1 Mice. Environ. Health Perspect. 2003, 111, 1883–1887. [Google Scholar] [CrossRef]
- Waite, K.J.; Floyd, Z.E.; Arbour-Reily, P.; Stephens, J.M. Interferon-gamma-induced regulation of peroxisome proliferator-activated receptor gamma and STATs in adipocytes. J. Biol. Chem. 2001, 276, 7062–7068. [Google Scholar] [CrossRef] [PubMed]
- Riu, A.; Grimaldi, M.; le Maire, A.; Bey, G.; Phillips, K.; Boulahtouf, A.; Perdu, E.; Zalko, D.; Bourguet, W.; Balaguer, P. Peroxisome proliferator-activated receptor gamma is a target for halogenated analogs of bisphenol A. Environ. Health Perspect. 2011, 119, 1227–1232. [Google Scholar] [CrossRef]
- Ramon, S.; Bancos, S.; Thatcher, T.H.; Murant, T.I.; Moshkani, S.; Sahler, J.M.; Bottaro, A.; Sime, P.J.; Phipps, R.P. Peroxisome proliferator-activated receptor gamma B cell-specific deficient mice have an impaired antibody response. J. Immunol. 2012, 189, 4740–4747. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández Avila, R.; Palacios-Arreola, M.I.; Nava-Castro, K.E.; Morales-Montor, J.; Ostoa-Saloma, P. Neonatal Bisphenol A Exposure Affects the IgM Humoral Immune Response to 4T1 Breast Carcinoma Cells in Mice. Int. J. Environ. Res. Public Health 2019, 16, 1784. https://doi.org/10.3390/ijerph16101784
Hernández Avila R, Palacios-Arreola MI, Nava-Castro KE, Morales-Montor J, Ostoa-Saloma P. Neonatal Bisphenol A Exposure Affects the IgM Humoral Immune Response to 4T1 Breast Carcinoma Cells in Mice. International Journal of Environmental Research and Public Health. 2019; 16(10):1784. https://doi.org/10.3390/ijerph16101784
Chicago/Turabian StyleHernández Avila, Ricardo, Margarita I. Palacios-Arreola, Karen E. Nava-Castro, Jorge Morales-Montor, and Pedro Ostoa-Saloma. 2019. "Neonatal Bisphenol A Exposure Affects the IgM Humoral Immune Response to 4T1 Breast Carcinoma Cells in Mice" International Journal of Environmental Research and Public Health 16, no. 10: 1784. https://doi.org/10.3390/ijerph16101784
APA StyleHernández Avila, R., Palacios-Arreola, M. I., Nava-Castro, K. E., Morales-Montor, J., & Ostoa-Saloma, P. (2019). Neonatal Bisphenol A Exposure Affects the IgM Humoral Immune Response to 4T1 Breast Carcinoma Cells in Mice. International Journal of Environmental Research and Public Health, 16(10), 1784. https://doi.org/10.3390/ijerph16101784




