A Nationwide Registry-Based Study on Mortality Due to Rare Congenital Anomalies
Abstract
1. Introduction
2. Materials and Methods
Research Ethics
3. Results
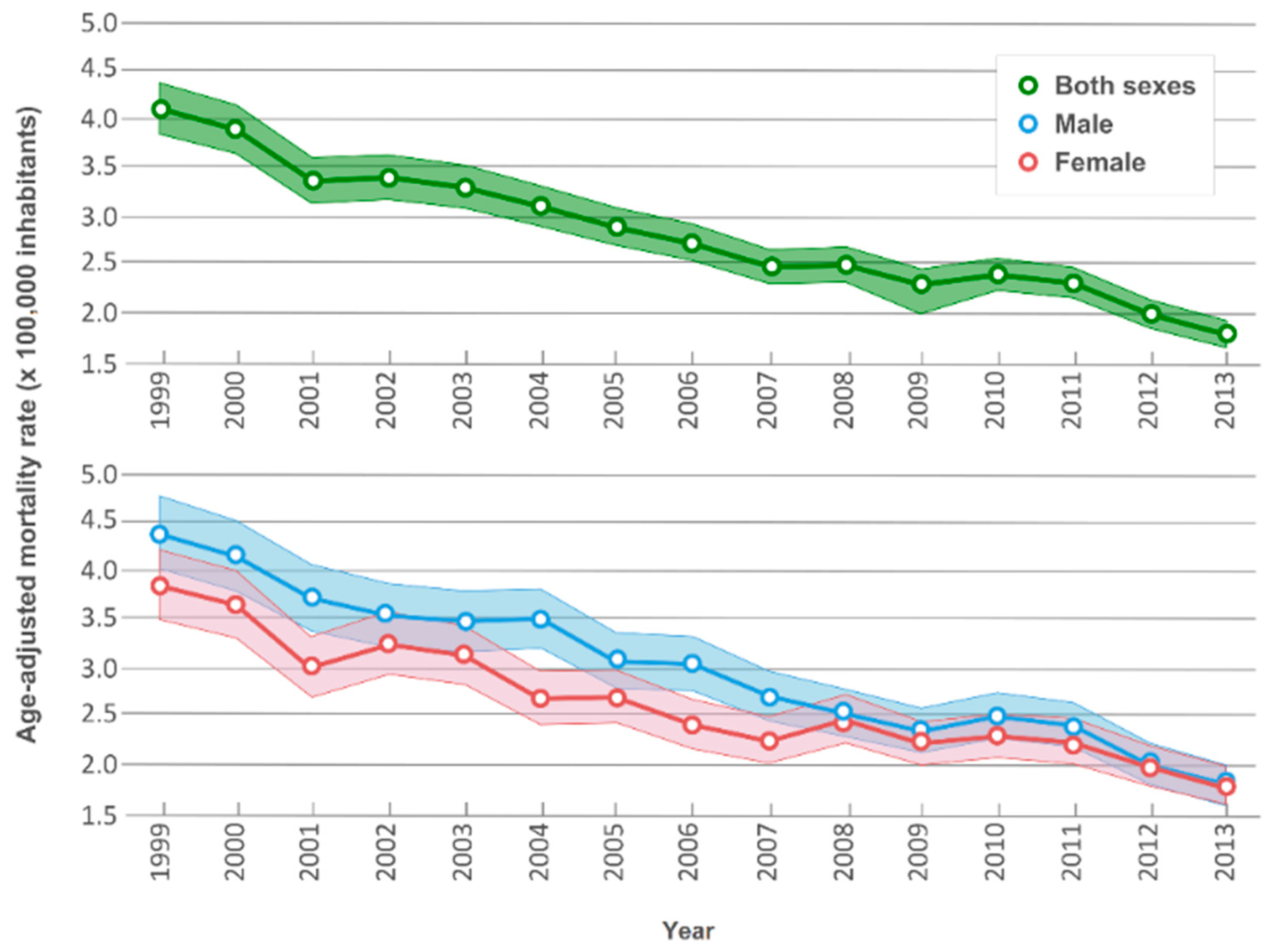
3.1. Time Trends
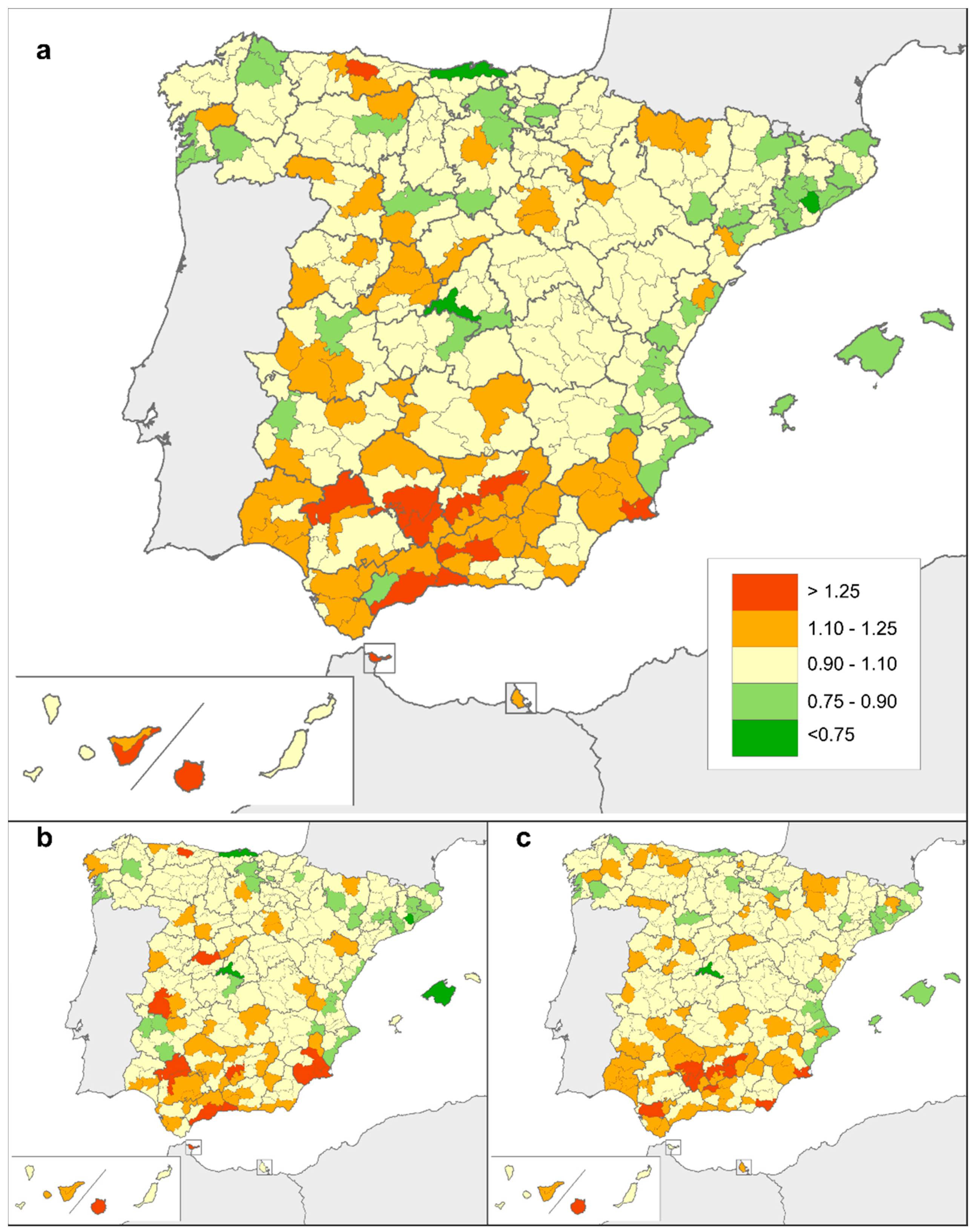
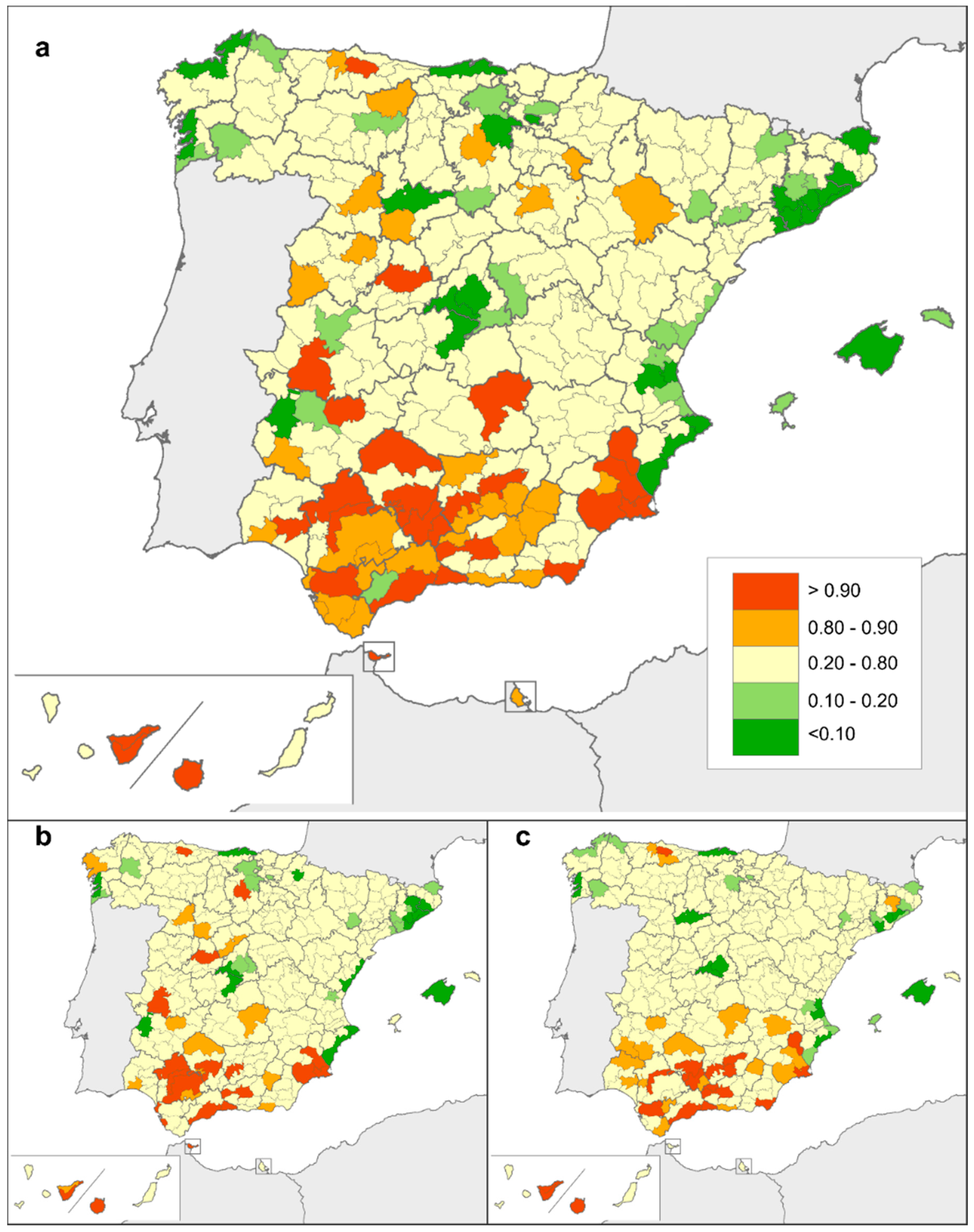
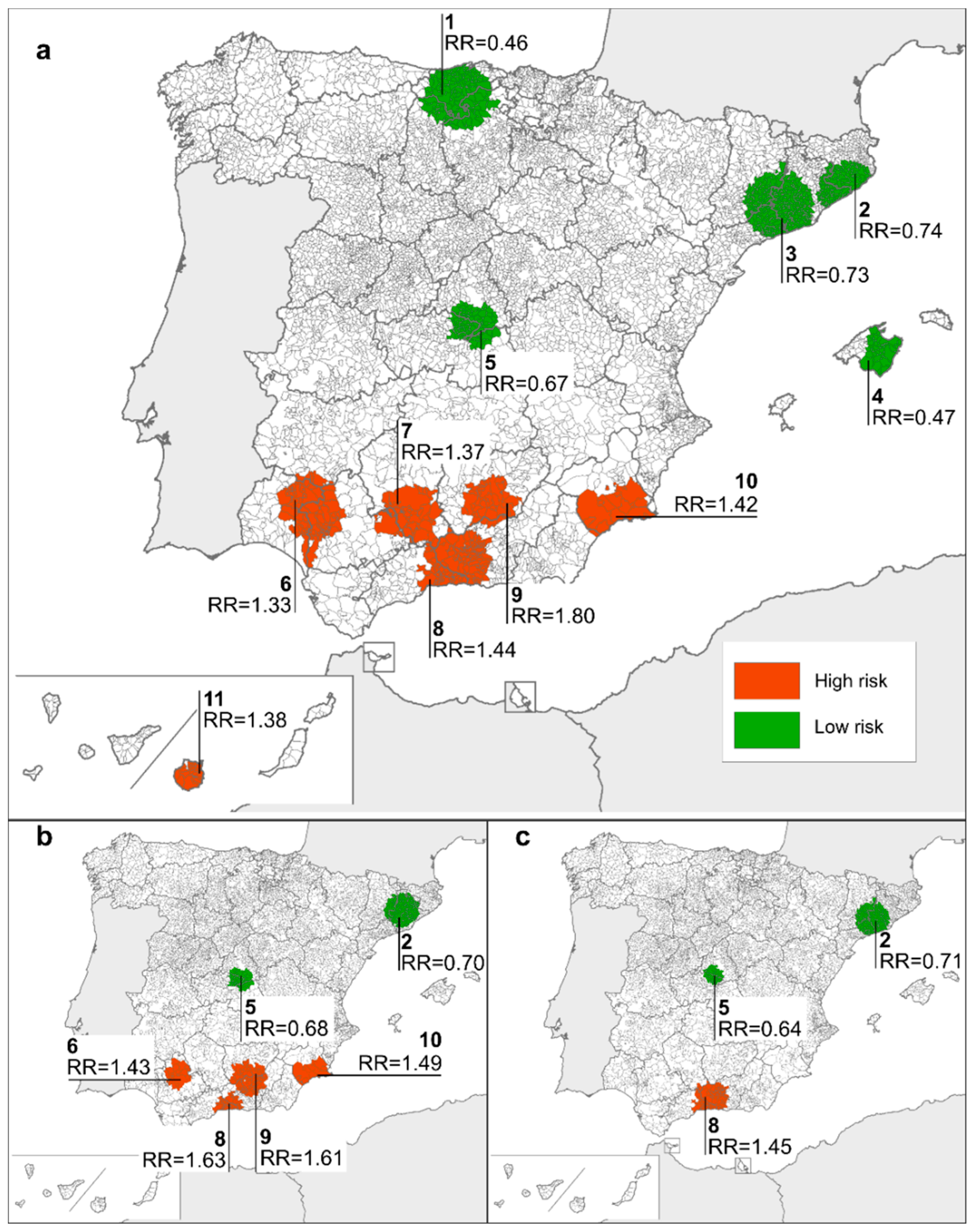
3.2. Geographical Distribution
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- GBD 2015 Mortality and Causes of Death Collaborators. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1459–1544. [Google Scholar] [CrossRef]
- Liu, L.; Oza, S.; Hogan, D.; Chu, Y.; Perin, J.; Zhu, J.; Lawn, J.E.; Cousens, S.; Mathers, C.; Black, R.E. Global, regional, and national causes of under-5 mortality in 2000–2015: An updated systematic analysis with implications for the Sustainable Development Goals. Lancet 2016, 388, 3027–3035. [Google Scholar] [CrossRef]
- Oza, S.; Lawn, J.E.; Hogan, D.R.; Mathers, C.; Cousens, S.N. Neonatal cause-of-death estimates for the early and late neonatal periods for 194 countries: 2000–2013. Bull. World Health Organ. 2015, 93, 19–28. [Google Scholar] [CrossRef] [PubMed]
- European Surveillance of Congenital Anomalies, EUROCAT. Available online: http://www.eurocat-network.eu/ (accessed on 25 June 2018).
- International Clearinghouse for Birth Defects Surveillance and Research, ICBDSR. Available online: http://www.icbdsr.org/ (accessed on 25 June 2018).
- Kinsner-Ovaskainen, A.; Lanzoni, M.; Garne, E.; Loane, M.; Morris, J.; Neville, A.; Nicholl, C.; Rankin, J.; Rissmann, A.; Tucker, D.; et al. A Sustainable Solution for the Activities of the European Network for Surveillance of Congenital Anomalies: EUROCAT as Part of the EU Platform on Rare Diseases Registration. Eur. J. Med. Genet. 2018. [CrossRef] [PubMed]
- Coi, A.; Santoro, M.; Pierini, A.; Marrucci, S.; Pieroni, F.; Bianchi, F. Prevalence Estimates of Rare Congenital Anomalies by Integrating Two Population-Based Registries in Tuscany, Italy. Public Health Genom. 2017, 20, 229–234. [Google Scholar] [CrossRef] [PubMed]
- De la Paz, M.P.; Villaverde-Hueso, A.; Alonso, V.; János, S.; Zurriaga, O.; Pollán, M.; Abaitua-Borda, I. Rare diseases epidemiology research. Adv. Exp. Med. Biol. 2010, 686, 17–39. [Google Scholar] [CrossRef] [PubMed]
- Arias Merino, G.; Sánchez Díaz, G.; Villaverde-Hueso, A.; Posada de la Paz, M.; Alonso Ferreira, V. Mortality Statistics and their Contribution to Improving the Knowledge of Rare Diseases Epidemiology: The Example of Hereditary Ataxia in Europe. Adv. Exp. Med. Biol. 2017, 1031, 521–533. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.F.; Martuzzi, M.; Elliott, P. Cluster analysis and disease mapping—Why, when, and how? A step-by-step guide. BMJ 1996, 313, 863–866. [Google Scholar] [CrossRef] [PubMed]
- Bermejo-Sánchez, E.; Posada de la Paz, M. Congenital Anomalies: Cluster Detection and Investigation. Adv. Exp. Med. Biol. 2017, 1031, 535–557. [Google Scholar] [CrossRef] [PubMed]
- Yazdy, M.M.; Werler, M.M.; Feldkamp, M.L.; Shaw, G.M.; Mosley, B.S.; Vieira, V.M.; National Birth Defects Prevention Study. Spatial analysis of gastroschisis in the National Birth Defects Prevention Study. Birth Defects Res. A Clin. Mol. Teratol. 2015, 103, 544–553. [Google Scholar] [CrossRef] [PubMed]
- Castilla, E.E.; Mastroiacovo, P.; López-Camelo, J.S.; Saldarriaga, W.; Isaza, C.; Orioli, I.M. Sirenomelia and cyclopia cluster in Cali, Colombia. Am. J. Med. Genet. A 2008, 146A, 2626–2636. [Google Scholar] [CrossRef] [PubMed]
- Bronberg, R.; Gili, J.; Chaves, E.; Dipierri, J. Infant mortality due to congenital malformation in Argentina (1998–2009): Temporal and spatial analysis of neonatal and postneonatal components. J. Community Genet. 2013, 4, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Fazzo, L.; Belli, S.; Minichilli, F.; Mitis, F.; Santoro, M.; Martina, L.; Pizzuti, R.; Comba, P.; Martuzzi, M.; Bianchi, F.; the Working Group. Cluster analysis of mortality and malformations in the provinces of Naples and Caserta (Campania Region). Ann. Ist. Super. Sanita 2008, 44, 99–111. [Google Scholar] [PubMed]
- Armstrong, B.G.; Dolk, H.; Pattenden, S.; Vrijheid, M.; Loane, M.; Rankin, J.; Dunn, C.E.; Grundy, C.; Abramsky, L.; Boyd, P.A.; et al. Geographic variation and localised clustering of congenital anomalies in Great Britain. Emerg. Themes Epidemiol. 2007, 4, 14. [Google Scholar] [CrossRef] [PubMed]
- Boyle, B.; Addor, M.C.; Arriola, L.; Barisic, I.; Bianchi, F.; Csáky-Szunyogh, M.; de Walle, H.E.K.; Dias, C.M.; Draper, E.; Gatt, M.; et al. Estimating Global Burden of Disease due to congenital anomaly: An analysis of European data. Arch. Dis. Child. Fetal. Neonatal 2018, 103, F22–F28. [Google Scholar] [CrossRef] [PubMed]
- The Spanish Rare Diseases Registries Research Network, SpainRDR. Available online: https://spainrdr.isciii.es/ (accessed on 25 June 2018).
- Arias López, M. La Comarca Como Entidad Natural, Histórica Y Administrativa; Dykinson, S.L.: Madrid, Spain, 2013; pp. 1–518. [Google Scholar]
- Sánchez-Díaz, G.; Arias-Merino, G.; Villaverde-Hueso, A.; Morales-Piga, A.; Abaitua-Borda, I.; Hens, M.; Bermejo-Sánchez, E.; Posada de la Paz, M.; Alonso-Ferreira, V. Monitoring Huntington’s Disease Mortality across a 30-Year Period: Geographic and Temporal Patterns. Neuroepidemiology 2016, 47, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Besag, J.E.; York, J.; Molliè, A. A Bayesian image restoration with two applications in spatial statistics. Ann. Inst. Stat. Math. 1991, 43, 1–59. [Google Scholar] [CrossRef]
- Kulldorf, M.; Mostashari, F.; Duczmal, L.; Katherine, Y.W.; Kleinman, K.; Platt, R. Multivariate scan statistics for disease surveillance. Stat. Med. 2007, 26, 1824–1833. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization, WHO. Mortality. Available online: http://apps.who.int/healthinfo/statistics/mortality/whodpms/ (accessed on 25 June 2018).
- Gupta, N.; Leven, L.; Stewart, M.; Cheung, M.; Patel, N. Transport of infants with congenital heart disease: Benefits of antenatal diagnosis. Eur. J. Pediatr. 2014, 173, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.K.; Springett, A.L.; Greenlees, R.; Loane, M.; Addor, M.C.; Arriola, L.; Barisic, I.; Bergman, J.E.H.; Csaky-Szunyogh, M.; Dias, C.; et al. Trends in congenital anomalies in Europe from 1980 to 2012. PLoS ONE 2018, 13, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Lara, D.A.; Fixler, D.E.; Ethen, M.K.; Canfield, M.A.; Nembhard, W.N.; Morris, S.A. Prenatal diagnosis, hospital characteristics, and mortality in transposition of the great arteries. Birth Defects Res. A Clin. Mol. Teratol. 2016, 106, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Arth, A.C.; Tinker, S.C.; Simeone, R.M.; Ailes, E.C.; Cragan, J.D.; Grosse, S.D. Inpatient Hospitalization Costs Associated with Birth Defects Among Persons of All Ages—United States, 2013. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 41–46. [Google Scholar] [CrossRef] [PubMed]
- GBD 2016. Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1211–1259. [Google Scholar] [CrossRef]
- Modell, B.; Berry, R.J.; Boyle, C.A.; Christianson, A.; Darlison, M.; Dolk, H.; Howson, C.P.; Mastroiacovo, P.; Mossey, P.; Rankin, J. Global regional and national causes of child mortality. Lancet 2012, 380, 1556. [Google Scholar] [CrossRef]
- Johansson, L.; Westerling, R. Comparing hospital discharge records with death certificates: Can the differences be explained? J. Epidemiol. Community Health 2002, 56, 301–308. [Google Scholar] [CrossRef] [PubMed]
| Rare CAs as Cause of Death | Number of Deaths | |||
|---|---|---|---|---|
| Total | Men | Women | ||
| Rare CAs of the Nervous System | ||||
| Nervous system | Q00, Q01, Q02, Q03, Q04, Q05, Q06, Q07 | 1251 | 645 | 606 |
| Neural Tube Defects | Q00, Q01, Q05 | 272 | 140 | 132 |
| Anencephalus and similar | Q00 | 97 | 49 | 48 |
| Encephalocele | Q01 | 15 | 10 | 5 |
| Myelomeningocele/Spina Bifida | Q05 | 160 | 81 | 79 |
| Hydrocephalus | Q03 | 199 | 119 | 80 |
| Severe microcephaly | Q02 | 54 | 24 | 30 |
| Arhinencephaly/holoprosencephaly | Q04.1, Q04.2 | 59 | 24 | 35 |
| Rare CAs of the Eye, Ear, Face and Neck | ||||
| Eye | Q10–Q15 | 1 | 0 | 1 |
| Anophthalmos/microphthalmos | Q11.0, Q11.1, Q11.2 | 1 | 0 | 1 |
| Anophthalmos | Q11.0, Q11.1 | 0 | 0 | 0 |
| Congenital cataract | Q12.0 | 0 | 0 | 0 |
| Congenital glaucoma | Q15.0 | 0 | 0 | 0 |
| Ear, face and neck | Q16, Q17, Q18 | 0 | 0 | 0 |
| Anotia | Q16.0 | 0 | 0 | 0 |
| Rare CAs of the Circulatory System | ||||
| Congenital Heart Defects (CHD) | Q20–Q26 | 4927 | 2780 | 2147 |
| Severe CHD | Q20.0, Q20.1, Q20.3, Q20.4, Q21.2, Q21.3, Q22.0, Q22.4, Q22.5, Q22.6, Q23.0, Q23.2, Q23.3, Q23.4, Q25.1, Q25.2, Q26.2 | 1475 | 841 | 634 |
| Common arterial truncus | Q20.0 | 44 | 20 | 24 |
| Double outlet right ventricle | Q20.1 | 19 | 12 | 7 |
| Transposition of great vessels | Q20.3 | 235 | 151 | 84 |
| Single ventricle | Q20.4 | 75 | 40 | 35 |
| AVSD | Q21.2 | 126 | 52 | 74 |
| Tetralogy of Fallot | Q21.3 | 360 | 210 | 150 |
| Triscuspid atresia and stenosis | Q22.4 | 34 | 15 | 19 |
| Ebstein’s anomaly | Q22.5 | 75 | 32 | 43 |
| Pulmonary valve stenosis | Q22.1 | 6 | 4 | 2 |
| Pulmonary valve atresia | Q22.0 | 5 | 2 | 3 |
| Aortic valve atresia/stenosis | Q23.0 | 57 | 37 | 20 |
| Mitral valve anomalies | Q23.2, Q23.3 | 27 | 15 | 12 |
| Hypoplastic left heart | Q23.4 | 256 | 155 | 101 |
| Hypoplastic right heart | Q22.6 | 5 | 3 | 2 |
| Coarctation of aorta | Q25.1 | 150 | 93 | 57 |
| Total anomalous pulmonary venous return | Q26.2 | 7 | 4 | 3 |
| PDA as only CHD in term infants (GA + 37 weeks) | Q25.0 | 290 | 155 | 135 |
| Rare CAs of the Respiratory System | ||||
| Respiratory | Q30.0, Q32–Q34 | 347 | 203 | 144 |
| Choanal atresia | Q30.0 | 8 | 5 | 3 |
| Cleft lip and Cleft Palate | ||||
| Oro-facial clefts | Q35–Q37 | 4 | 1 | 3 |
| Cleft lip with or without cleft palate | Q36, Q37 | 0 | 0 | 0 |
| Cleft palate | Q35 | 4 | 1 | 3 |
| Rare CAs of the Digestive System | ||||
| Digestive system | Q38–Q45 | 1092 | 593 | 499 |
| Oesophageal atresia with or without tracheaoesophageal fistula | Q39.0–Q39.1 | 107 | 55 | 52 |
| Duodenal atresia or stenosis | Q41.0 | 11 | 7 | 4 |
| Atresia or stenosis of other parts of small intestine | Q41.1–Q41.8 | 17 | 9 | 8 |
| Ano-rectal atresia and stenosis | Q42.0–Q42.3 | 6 | 4 | 2 |
| Hirschsprung’s disease | Q43.1 | 47 | 27 | 20 |
| Atresia of bile ducts | Q44.2 | 55 | 30 | 25 |
| Annular pancreas | Q45.1 | 0 | 0 | 0 |
| Rare CAs of the Urinary System | ||||
| Urinary | Q60–Q64, Q79.4 | 756 | 393 | 363 |
| Bilateral renal agenesis including Potter syndrome | Q60.1, Q60.6 | 84 | 53 | 31 |
| Renal dysplasia | Q61.4 | 28 | 20 | 8 |
| Bladder exstrophy and/or epispadia | Q64.0, Q64.1 | 2 | 2 | 0 |
| Posterior urethral valve and/or prune belly | Q64.2, Q64.3, Q79.4 | 7 | 6 | 1 |
| Rare CAs of Genital Organs | ||||
| Genital | Q50–Q52, Q54–Q56 | 1 | 1 | 0 |
| Hypospadias | Q54 | 1 | 1 | 0 |
| Indeterminate sex | Q56 | 0 | 0 | 0 |
| Rare CAs of the Musculoskeletal System | ||||
| Craniosynostosis | Q75.0 | 6 | 3 | 3 |
| Diaphragmatic hernia | Q79.0 | 339 | 191 | 148 |
| Limb | Q65–Q74 | 56 | 24 | 32 |
| Limb reduction defects | Q71–Q73 | 0 | 0 | 0 |
| Polydactyly | Q69 | 0 | 0 | 0 |
| Syndactyly | Q70 | 0 | 0 | 0 |
| Abdominal wall defects | Q79.2, Q79.3, Q79.5 | 66 | 42 | 24 |
| Gastroschisis | Q79.3 | 44 | 30 | 14 |
| Omphalocele | Q79.2 | 22 | 12 | 10 |
| Other Rare Congenital Malformations | ||||
| Other anomalies/syndromes | ||||
| Congenital skin disorders | Q80–Q82 | 93 | 62 | 31 |
| Fetal alcohol syndrome | Q86.0 | 2 | 2 | 0 |
| Situs inversus | Q89.3 | 31 | 16 | 15 |
| Conjoined twins | Q89.4 | 11 | 2 | 9 |
| Rare Chromosomal Abnormalities, not elsewhere classified | ||||
| Chromosomal | Q90–Q92, Q93, Q96–Q99 | 2312 | 1068 | 1244 |
| Patau syndrome/trisomy 13 | Q91.4–Q91.7 | 143 | 62 | 81 |
| Edwards syndrome / trisomy 18 | Q91.0–Q91.3 | 288 | 81 | 207 |
| Turner syndrome | Q96 | 23 | 1 | 22 |
| Klinefelter syndrome | Q98.0–Q98.4 | 3 | 3 | 0 |
| Year | Rare CAs of the Nervous System | Neural Tube Defects | ||||||||||
| Both Sexes | Men | Women | Both Sexes | Men | Women | |||||||
| n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | |
| 1999 | 93 | 0.37 (0.29–0.45) | 47 | 0.35 (0.25–0.47) | 46 | 0.39 (0.28–0.53) | 23 | 0.10 (0.06–0.15) | 11 | 0.08 (0.04–0.15) | 12 | 0.12 (0.06–0.20) |
| 2000 | 95 | 0.36 (0.29–0.45) | 51 | 0.37 (0.27–0.50) | 44 | 0.35 (0.25–0.48) | 25 | 0.10 (0.06–0.15) | 12 | 0.09 (0.05–0.16) | 13 | 0.10 (0.05–0.18) |
| 2001 | 87 | 0.34 (0.27–0.43) | 54 | 0.42 (0.32–0.56) | 33 | 0.26 (0.17–0.37) | 24 | 0.09 (0.06–0.14) | 15 | 0.11 (0.06–0.19) | 9 | 0.07 (0.03–0.14) |
| 2002 | 79 | 0.29 (0.23–0.37) | 34 | 0.25 (0.17–0.35) | 45 | 0.35 (0.25–0.47) | 15 | 0.07 (0.04–0.11) | 9 | 0.08 (0.04–0.15) | 6 | 0.06 (0.02–0.12) |
| 2003 | 93 | 0.34 (0.27–0.42) | 47 | 0.34 (0.25–0.46) | 46 | 0.34 (0.25–0.46) | 20 | 0.07 (0.04–0.11) | 14 | 0.10 (0.05–0.17) | 6 | 0.05 (0.02–0.10) |
| 2004 | 95 | 0.31 (0.32–0.39) | 56 | 0.38 (0.28–0.50) | 39 | 0.26 (0.18–0.36) | 18 | 0.06 (0.03–0.10) | 9 | 0.05 (0.02–0.11) | 9 | 0.06 (0.03–0.13) |
| 2005 | 82 | 0.26 (0.21–0.33) | 45 | 0.29 (0.21–0.40) | 37 | 0.24 (0.16–0.33) | 16 | 0.05 (0.03–0.09) | 10 | 0.07 (0.03–0.13) | 6 | 0.03 (0.01–0.08) |
| 2006 | 80 | 0.25 (0.21–0.33) | 46 | 0.28 (0.20–0.37) | 34 | 0.23 (0.16–0.32) | 21 | 0.06 (0.04–0.10) | 12 | 0.06 (0.03–0.12) | 9 | 0.06 (0.03–0.11) |
| 2007 | 80 | 0.25 (0.20–0.31) | 41 | 0.25 (0.18–0.34) | 39 | 0.25 (0.18–0.35) | 16 | 0.05 (0.03–0.08) | 6 | 0.04 (0.01–0.09) | 10 | 0.06 (0.03–0.11) |
| 2008 | 73 | 0.22 (0.18–0.28) | 35 | 0.21 (0.14–0.29) | 38 | 0.25 (0.17–0.34) | 16 | 0.05 (0.03–0.08) | 11 | 0.07 (0.03–0.12) | 5 | 0.03 (0.01–0.08) |
| 2009 | 74 | 0.21 (0.17–0.27) | 33 | 0.19 (0.13–0.26) | 41 | 0.24 (0.17–0.34) | 20 | 0.06 (0.03–0.09) | 8 | 0.05 (0.02–0.10) | 12 | 0.07 (0.03–0.12) |
| 2010 | 93 | 0.26 (0.21–0.32) | 42 | 0.24 (0.17–0.32) | 51 | 0.28 (0.21–0.38) | 12 | 0.03 (0.01–0.05) | 4 | 0.02 (0.01–0.06) | 8 | 0.04 (0.01–0.08) |
| 2011 | 82 | 0.22 (0.17–0.27) | 44 | 0.23 (0.16–0.31) | 38 | 0.20 (0.14–0.29) | 18 | 0.04 (0.02–0.07) | 6 | 0.03 (0.01–0.06) | 12 | 0.06 (0.03–0.10) |
| 2012 | 77 | 0.21 (0.16–0.26) | 37 | 0.21 (0.14–0.29) | 40 | 0.20 (0.14–0.28) | 14 | 0.03 (0.02–0.05) | 6 | 0.03 (0.01–0.06) | 8 | 0.04 (0.01–0.08) |
| 2013 | 68 | 0.20 (0.15–0.25) | 33 | 0.18 (0.12–0.26) | 35 | 0.22 (0.15–0.31) | 14 | 0.04 (0.02–0.06) | 7 | 0.04 (0.01–0.08) | 7 | 0.04 (0.01–0.08) |
| APC | −4.49 (p < 0.001) | −5.19 (p < 0.001) | −3.87 (p < 0.001) | −6.60 (p < 0.001) | −2.40 (p = 0.30) | −8.24 (p < 0.001) | ||||||
| Year | Hydrocephalus | Rare CAs of the Circulatory System | ||||||||||
| Both Sexes | Men | Women | Both Sexes | Men | Women | |||||||
| n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | |
| 1999 | 20 | 0.08 (0.05–0.13) | 14 | 0.11 (0.06–0.19) | 6 | 0.05 (0.02–0.11) | 486 | 1.90 (1.73–2.09) | 274 | 2.11 (1.86–2.38) | 212 | 1.69 (1.46–1.95) |
| 2000 | 22 | 0.09 (0.05–0.14) | 13 | 0.10 (0.05–0.18) | 9 | 0.07 (0.03–0.14) | 480 | 1.85 (1.68–2.03) | 268 | 2.06 (1.82–2.34) | 212 | 1.63 (1.40–1.88) |
| 2001 | 12 | 0.05 (0.02–0.08) | 6 | 0.04 (0.02–0.10) | 6 | 0.05 (0.02–0.11) | 387 | 1.51 (1.36–1.67) | 224 | 1.69 (1.46–1.93) | 163 | 1.33 (1.12–1.56) |
| 2002 | 22 | 0.08 (0.05–0.12) | 10 | 0.07 (0.03–0.13) | 12 | 0.09 (0.05–0.17) | 421 | 1.60 (1.44–1.76) | 241 | 1.79 (1.57–2.04) | 180 | 1.39 (1.19–1.62) |
| 2003 | 15 | 0.05 (0.03–0.09) | 7 | 0.05 (0.02–0.10) | 8 | 0.06 (0.02–0.12) | 391 | 1.38 (1.24–1.53) | 211 | 1.48 (1.28–1.70) | 180 | 1.28 (1.09–1.49) |
| 2004 | 16 | 0.05 (0.03–0.09) | 12 | 0.08 (0.04–0.14) | 4 | 0.03 (0.01–0.07) | 404 | 1.39 (1.25–1.53) | 254 | 1.71 (1.50–1.94) | 150 | 1.05 (0.88–1.24) |
| 2005 | 12 | 0.04 (0.02–0.07) | 10 | 0.06 (0.03–0.11) | 2 | 0.02 (0.00–0.06) | 341 | 1.16 (1.03–1.29) | 205 | 1.37 (1.19–1.58) | 136 | 0.93 (0.78–1.11) |
| 2006 | 9 | 0.03 (0.01–0.06) | 7 | 0.05 (0.02–0.10) | 2 | 0.01 (0.00–0.05) | 380 | 1.21 (1.09–1.34) | 220 | 1.38 (1.20–1.58) | 160 | 1.04 (0.88–1.22) |
| 2007 | 11 | 0.03 (0.02–0.06) | 7 | 0.04 (0.02–0.09) | 4 | 0.03 (0.01–0.08) | 318 | 0.99 (0.88–1.11) | 170 | 1.09 (0.93–1.26) | 140 | 0.9 (0.75–1.07) |
| 2008 | 14 | 0.04 (0.02–0.07) | 7 | 0.04 (0.01–0.08) | 7 | 0.05 (0.02–0.10) | 350 | 1.05 (0.94–1.17) | 190 | 1.14 (0.98–1.32) | 160 | 0.95 (0.81–1.12) |
| 2009 | 9 | 0.02 (0.01–0.05) | 4 | 0.02 (0.01–0.06) | 5 | 0.03 (0.01–0.07) | 341 | 0.97 (0.86–1.08) | 190 | 1.06 (0.91–1.23) | 151 | 0.87 (0.73–1.02) |
| 2010 | 13 | 0.04 (0.02–0.06) | 8 | 0.05 (0.02–0.09) | 5 | 0.02 (0.01–0.06) | 349 | 0.97 (0.87–1.08) | 190 | 1.06 (0.91–1.23) | 159 | 0.88 (0.74–1.03) |
| 2011 | 10 | 0.03 (0.01–0.05) | 9 | 0.04 (0.02–0.09) | 1 | 0.01 (0.00–0.04) | 321 | 0.9 (0.80–1.01) | 184 | 1.01 (0.87–1.17) | 137 | 0.8 (0.66–0.95) |
| 2012 | 6 | 0.02 (0.01–0.03) | 1 | 0.01 (0.00–0.03) | 5 | 0.02 (0.01–0.06) | 295 | 0.81 (0.71–0.91) | 153 | 0.84 (0.71–0.99) | 142 | 0.77 (0.64–0.92) |
| 2013 | 8 | 0.02 (0.01–0.05) | 4 | 0.02 (0.01–0.06) | 4 | 0.02 (0.01–0.06) | 246 | 0.67 (0.59–0.77) | 134 | 0.71 (0.59–0.85) | 112 | 0.64 (0.52–0.78) |
| APC | −9.65 (p < 0.001) | −9.11 (p < 0.001) | −9.08 (p < 0.001) | −6.44 (p < 0.001) | −6.70 (p < 0.001) | −6.08 (p < 0.001) | ||||||
| Year | Severe CHD | Transposition of Great Vessels | ||||||||||
| Both Sexes | Men | Women | Both Sexes | Men | Women | |||||||
| n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | |
| 1999 | 145 | 0.59 (0.50–0.70) | 89 | 0.72 (0.57–0.89) | 56 | 0.47 (0.35–0.62) | 28 | 0.12 (0.08–0.18) | 18 | 0.16 (0.09–0.25) | 10 | 0.09 (0.04–0.16) |
| 2000 | 148 | 0.59 (0.49–0.69) | 75 | 0.59 (0.46–0.74) | 73 | 0.58 (0.45–0.74) | 30 | 0.13 (0.08–0.18) | 18 | 0.15 (0.09–0.24) | 12 | 0.10 (0.05–0.18) |
| 2001 | 100 | 0.39 (0.32–0.48) | 56 | 0.42 (0.31–0.55) | 44 | 0.36 (0.26–0.49) | 15 | 0.06 (0.03–0.10) | 9 | 0.07 (0.03–0.14) | 6 | 0.05 (0.02–0.11) |
| 2002 | 119 | 0.46 (0.38–0.56) | 66 | 0.49 (0.38–0.63) | 53 | 0.43 (0.32–0.57) | 22 | 0.09 (0.06–0.14) | 15 | 0.11 (0.06–0.19) | 7 | 0.06 (0.03–0.13) |
| 2003 | 102 | 0.38 (0.31–0.47) | 52 | 0.39 (0.29–0.51) | 50 | 0.38 (0.28–0.50) | 13 | 0.05 (0.03–0.09) | 7 | 0.06 (0.02–0.12) | 6 | 0.05 (0.02–0.11) |
| 2004 | 117 | 0.40 (0.33–0.49) | 79 | 0.53 (0.42–0.67) | 38 | 0.27 (0.19–0.37) | 22 | 0.08 (0.05–0.12) | 13 | 0.09 (0.05–0.16) | 9 | 0.07 (0.03–0.13) |
| 2005 | 101 | 0.33 (0.27–0.41) | 58 | 0.37 (0.28–0.49) | 43 | 0.29 (0.21–0.39) | 17 | 0.06 (0.03–0.09) | 13 | 0.08 (0.04–0.15) | 4 | 0.03 (0.01–0.07) |
| 2006 | 109 | 0.35 (0.29–0.43) | 60 | 0.38 (0.29–0.49) | 49 | 0.33 (0.24–0.44) | 15 | 0.05 (0.03–0.08) | 9 | 0.06 (0.02–0.11) | 6 | 0.04 (0.01–0.09) |
| 2007 | 101 | 0.31 (0.25–0.38) | 63 | 0.39 (0.29–0.50) | 38 | 0.23 (0.16–0.32) | 12 | 0.04 (0.02–0.07) | 9 | 0.06 (0.03–0.11) | 3 | 0.02 (0.00–0.06) |
| 2008 | 83 | 0.26 (0.20–0.32) | 48 | 0.29 (0.21–0.38) | 35 | 0.22 (0.15–0.31) | 14 | 0.05 (0.02–0.08) | 7 | 0.05 (0.02–0.09) | 7 | 0.05 (0.02–0.10) |
| 2009 | 80 | 0.23 (0.18–0.29) | 51 | 0.29 (0.21–0.38) | 29 | 0.17 (0.11–0.25) | 12 | 0.04 (0.02–0.07) | 9 | 0.06 (0.03–0.11) | 3 | 0.02 (0.00–0.06) |
| 2010 | 71 | 0.19 (0.15–0.25) | 38 | 0.21 (0.14–0.28) | 33 | 0.18 (0.13–0.26) | 9 | 0.03 (0.01–0.05) | 6 | 0.03 (0.01–0.07) | 3 | 0.02 (0.00–0.06) |
| 2011 | 65 | 0.18 (0.14–0.23) | 35 | 0.19 (0.13–0.26) | 30 | 0.17 (0.12–0.25) | 11 | 0.03 (0.01–0.05) | 9 | 0.05 (0.02–0.09) | 2 | 0.01 (0.00–0.04) |
| 2012 | 68 | 0.19 (0.14–0.24) | 36 | 0.20 (0.14–0.27) | 32 | 0.18 (0.12–0.25) | 9 | 0.03 (0.01–0.05) | 6 | 0.03 (0.01–0.08) | 3 | 0.02 (0.00–0.05) |
| 2013 | 66 | 0.18 (0.14–0.24) | 35 | 0.19 (0.13–0.26) | 31 | 0.18 (0.12–0.26) | 6 | 0.02 (0.01–0.04) | 3 | 0.02 (0.00–0.05) | 3 | 0.02 (0.00–0.05) |
| APC | −8.55 (p < 0.001) | −8.59 (p < 0.001) | −8.53 (p < 0.001) | −11.80 (p < 0.001) | −11.03 (p < 0.001) | −12.43 (p < 0.001) | ||||||
| Year | Hypoplastic Left Heart | Rare CAs of the Respiratory System | ||||||||||
| Both Sexes | Men | Women | Both Sexes | Men | Women | |||||||
| n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | |
| 1999 | 23 | 0.11 (0.07–0.16) | 13 | 0.12 (0.06–0.21) | 10 | 0.10 (0.05–0.18) | 25 | 0.11 (0.07–0.17) | 11 | 0.10 (0.05–0.18) | 14 | 0.13 (0.07–0.21) |
| 2000 | 19 | 0.09 (0.05–0.14) | 9 | 0.08 (0.04–0.16) | 10 | 0.10 (0.05–0.18) | 23 | 0.10 (0.06–0.15) | 14 | 0.12 (0.07–0.21) | 9 | 0.07 (0.03–0.14) |
| 2001 | 22 | 0.10 (0.06–0.15) | 11 | 0.10 (0.05–0.18) | 11 | 0.11 (0.05–0.19) | 24 | 0.11 (0.07–0.16) | 16 | 0.14 (0.08–0.22) | 8 | 0.08 (0.03–0.15) |
| 2002 | 25 | 0.11 (0.07–0.16) | 14 | 0.12 (0.07–0.20) | 11 | 0.10 (0.05–0.18) | 22 | 0.09 (0.05–0.14) | 14 | 0.11 (0.06–0.19) | 8 | 0.07 (0.03–0.14) |
| 2003 | 21 | 0.09 (0.05–0.14) | 13 | 0.11 (0.06–0.18) | 8 | 0.07 (0.03–0.14) | 35 | 0.14 (0.10–0.19) | 24 | 0.18 (0.12–0.28) | 11 | 0.09 (0.04–0.16) |
| 2004 | 21 | 0.08 (0.05–0.13) | 15 | 0.12 (0.07–0.19) | 6 | 0.05 (0.02–0.11) | 31 | 0.12 (0.08–0.17) | 17 | 0.12 (0.07–0.20) | 14 | 0.12 (0.06–0.19) |
| 2005 | 17 | 0.06 (0.04–0.10) | 9 | 0.07 (0.03–0.13) | 8 | 0.06 (0.03–0.12) | 25 | 0.09 (0.06–0.14) | 15 | 0.11 (0.06–0.18) | 10 | 0.08 (0.04–0.14) |
| 2006 | 21 | 0.08 (0.05–0.12) | 15 | 0.10 (0.06–0.17) | 6 | 0.05 (0.02–0.10) | 20 | 0.07 (0.04–0.11) | 12 | 0.08 (0.04–0.14) | 8 | 0.06 (0.02–0.11) |
| 2007 | 17 | 0.06 (0.04–0.10) | 13 | 0.09 (0.05–0.15) | 4 | 0.03 (0.01–0.08) | 28 | 0.10 (0.06–0.14) | 17 | 0.11 (0.06–0.18) | 11 | 0.08 (0.04–0.15) |
| 2008 | 16 | 0.05 (0.03–0.09) | 11 | 0.07 (0.04–0.13) | 5 | 0.04 (0.01–0.08) | 20 | 0.06 (0.04–0.10) | 10 | 0.07 (0.03–0.12) | 10 | 0.06 (0.03–0.11) |
| 2009 | 11 | 0.04 (0.02–0.06) | 7 | 0.04 (0.02–0.09) | 4 | 0.03 (0.01–0.07) | 22 | 0.06 (0.04–0.10) | 14 | 0.08 (0.04–0.14) | 8 | 0.04 (0.02–0.09) |
| 2010 | 12 | 0.04 (0.02–0.07) | 8 | 0.05 (0.02–0.10) | 4 | 0.03 (0.01–0.07) | 22 | 0.07 (0.04–0.10) | 15 | 0.09 (0.05–0.14) | 7 | 0.04 (0.02–0.09) |
| 2011 | 12 | 0.04 (0.02–0.07) | 6 | 0.04 (0.01–0.08) | 6 | 0.04 (0.01–0.09) | 32 | 0.10 (0.07–0.14) | 16 | 0.10 (0.05–0.16) | 16 | 0.1 (0.06–0.17) |
| 2012 | 10 | 0.03 (0.02–0.06) | 4 | 0.02 (0.01–0.06) | 6 | 0.04 (0.01–0.09) | 23 | 0.07 (0.05–0.11) | 14 | 0.08 (0.05–0.14) | 9 | 0.06 (0.03–0.11) |
| 2013 | 9 | 0.03 (0.01–0.06) | 7 | 0.04 (0.02–0.09) | 2 | 0.01 (0.00–0.05) | 16 | 0.05 (0.03–0.08) | 9 | 0.06 (0.03–0.11) | 7 | 0.05 (0.02–0.09) |
| APC | −9.00 (p < 0.001) | −7.47 (p < 0.001) | −10.53 (p < 0.001) | −4.07 (p < 0.001) | −4.46 (p < 0.001) | −3.72 (p = 0.87) | ||||||
| Year | Rare CAs of the Digestive System | Rare CAs of the Urinary System | ||||||||||
| Both Sexes | Men | Women | Both Sexes | Men | Women | |||||||
| n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | |
| 1999 | 42 | 0.15 (0.10–0.21) | 28 | 0.2 (0.13–0.29) | 14 | 0.11 (0.06–0.19) | 60 | 0.16 (0.12–0.22) | 39 | 0.24 (0.16–0.34) | 21 | 0.10 (0.05–0.17) |
| 2000 | 37 | 0.14 (0.09–0.19) | 21 | 0.15 (0.09–0.24) | 16 | 0.12 (0.07–0.21) | 70 | 0.17 (0.13–0.22) | 37 | 0.20 (0.13–0.29) | 33 | 0.14 (0.09–0.21) |
| 2001 | 37 | 0.12 (0.08–0.17) | 22 | 0.16 (0.10–0.25) | 15 | 0.08 (0.04–0.15) | 52 | 0.13 (0.09–0.18) | 24 | 0.14 (0.09–0.22) | 28 | 0.11 (0.07–0.18) |
| 2002 | 42 | 0.15 (0.11–0.21) | 18 | 0.15 (0.09–0.23) | 24 | 0.15 (0.09–0.24) | 50 | 0.12 (0.09–0.17) | 27 | 0.16 (0.10–0.24) | 23 | 0.09 (0.05–0.15) |
| 2003 | 48 | 0.16 (0.11–0.21) | 34 | 0.22 (0.15–0.31) | 14 | 0.1 (0.05–0.18) | 46 | 0.11 (0.08–0.16) | 27 | 0.15 (0.10–0.23) | 19 | 0.07 (0.04–0.13) |
| 2004 | 56 | 0.18 (0.13–0.23) | 29 | 0.17 (0.11–0.26) | 27 | 0.18 (0.12–0.27) | 35 | 0.08 (0.05–0.11) | 19 | 0.09 (0.05–0.15) | 16 | 0.07 (0.03–0.12) |
| 2005 | 64 | 0.19 (0.15–0.25) | 32 | 0.19 (0.13–0.28) | 32 | 0.19 (0.13–0.28) | 49 | 0.11 (0.08–0.15) | 23 | 0.11 (0.07–0.17) | 26 | 0.11 (0.07–0.17) |
| 2006 | 39 | 0.13 (0.09–0.18) | 17 | 0.12 (0.07–0.19) | 22 | 0.14 (0.08–0.22) | 51 | 0.12 (0.08–0.16) | 32 | 0.17 (0.11–0.24) | 19 | 0.07 (0.04–0.13) |
| 2007 | 61 | 0.16 (0.12–0.21) | 35 | 0.19 (0.13–0.27) | 26 | 0.13 (0.08–0.20) | 39 | 0.09 (0.06–0.13) | 22 | 0.12 (0.07–0.18) | 17 | 0.06 (0.03–0.11) |
| 2008 | 58 | 0.15 (0.11–0.20) | 33 | 0.17 (0.11–0.24) | 25 | 0.13 (0.08–0.20) | 44 | 0.11 (0.08–0.15) | 16 | 0.08 (0.05–0.14) | 28 | 0.13 (0.08–0.20) |
| 2009 | 46 | 0.12 (0.09–0.16) | 19 | 0.11 (0.06–0.17) | 27 | 0.13 (0.08–0.20) | 39 | 0.07 (0.05–0.10) | 16 | 0.06 (0.03–0.11) | 23 | 0.08 (0.05–0.13) |
| 2010 | 53 | 0.13 (0.10–0.18) | 24 | 0.12 (0.07–0.18) | 29 | 0.15 (0.10–0.23) | 61 | 0.13 (0.09–0.17) | 31 | 0.15 (0.10–0.22) | 30 | 0.10 (0.06–0.16) |
| 2011 | 68 | 0.15 (0.12–0.20) | 36 | 0.18 (0.12–0.25) | 32 | 0.12 (0.08–0.19) | 44 | 0.08 (0.06–0.12) | 22 | 0.09 (0.05–0.14) | 22 | 0.08 (0.04–0.13) |
| 2012 | 56 | 0.12 (0.09–0.16) | 29 | 0.13 (0.08–0.19) | 27 | 0.12 (0.07–0.18) | 50 | 0.07 (0.05–0.10) | 25 | 0.09 (0.05–0.14) | 25 | 0.06 (0.04–0.10) |
| 2013 | 46 | 0.09 (0.06–0.13) | 25 | 0.1 (0.06–0.15) | 21 | 0.09 (0.05–0.15) | 63 | 0.11 (0.08–0.14) | 31 | 0.13 (0.08–0.19) | 32 | 0.09 (0.05–0.14) |
| APC | −1.49 (p = 0.20) | −2.61 (p = 0.10) | −0.16 (p = 0.90) | −3.96 (p < 0.001) | −5.10 (p < 0.001) | −2.08 (p = 0.20) | ||||||
| Year | Rare CAs of the Musculoskeletal System | Diaphragmatic Hernia | ||||||||||
| Both Sexes | Men | Women | Both Sexes | Men | Women | |||||||
| n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | |
| 1999 | 61 | 0.26 (0.20–0.34) | 34 | 0.29 (0.20–0.41) | 27 | 0.23 (0.15–0.34) | 32 | 0.15 (0.10–0.21) | 19 | 0.18 (0.11–0.27) | 13 | 0.13 (0.07–0.22) |
| 2000 | 59 | 0.25 (0.19–0.32) | 27 | 0.23 (0.15–0.34) | 32 | 0.26 (0.18–0.38) | 26 | 0.12 (0.08–0.18) | 16 | 0.15 (0.08–0.24) | 10 | 0.10 (0.05–0.18) |
| 2001 | 49 | 0.21 (0.15–0.28) | 31 | 0.26 (0.17–0.37) | 18 | 0.15 (0.09–0.25) | 29 | 0.13 (0.09–0.19) | 19 | 0.17 (0.10–0.27) | 10 | 0.10 (0.05–0.18) |
| 2002 | 51 | 0.21 (0.15–0.27) | 25 | 0.20 (0.13–0.30) | 26 | 0.21 (0.14–0.32) | 22 | 0.10 (0.06–0.15) | 14 | 0.12 (0.07–0.20) | 8 | 0.07 (0.03–0.14) |
| 2003 | 67 | 0.23 (0.18–0.30) | 35 | 0.24 (0.17–0.34) | 32 | 0.22 (0.15–0.32) | 30 | 0.13 (0.09–0.18) | 17 | 0.14 (0.08–0.22) | 13 | 0.11 (0.06–0.19) |
| 2004 | 40 | 0.13 (0.09–0.19) | 22 | 0.16 (0.10–0.25) | 18 | 0.11 (0.06–0.18) | 13 | 0.05 (0.03–0.09) | 9 | 0.07 (0.03–0.13) | 4 | 0.03 (0.01–0.09) |
| 2005 | 46 | 0.15 (0.11–0.21) | 19 | 0.12 (0.07–0.19) | 27 | 0.19 (0.12–0.28) | 23 | 0.09 (0.05–0.13) | 12 | 0.08 (0.04–0.15) | 11 | 0.09 (0.04–0.16) |
| 2006 | 50 | 0.17 (0.12–0.22) | 35 | 0.23 (0.16–0.33) | 15 | 0.10 (0.06–0.17) | 25 | 0.09 (0.06–0.14) | 17 | 0.12 (0.07–0.19) | 8 | 0.06 (0.03–0.12) |
| 2007 | 43 | 0.14 (0.10–0.19) | 25 | 0.16 (0.10–0.24) | 18 | 0.12 (0.07–0.20) | 18 | 0.06 (0.04–0.10) | 9 | 0.06 (0.03–0.12) | 9 | 0.07 (0.03–0.13) |
| 2008 | 56 | 0.18 (0.13–0.23) | 28 | 0.17 (0.11–0.25) | 28 | 0.18 (0.12–0.27) | 25 | 0.09 (0.06–0.13) | 11 | 0.07 (0.04–0.13) | 14 | 0.10 (0.05–0.17) |
| 2009 | 40 | 0.12 (0.08–0.16) | 21 | 0.12 (0.07–0.19) | 19 | 0.11 (0.06–0.17) | 16 | 0.05 (0.03–0.08) | 8 | 0.05 (0.02–0.10) | 8 | 0.05 (0.02–0.10) |
| 2010 | 47 | 0.14 (0.10–0.18) | 28 | 0.17 (0.11–0.24) | 19 | 0.10 (0.06–0.17) | 26 | 0.08 (0.06–0.12) | 17 | 0.11 (0.06–0.17) | 9 | 0.06 (0.03–0.11) |
| 2011 | 62 | 0.18 (0.14–0.23) | 28 | 0.16 (0.11–0.23) | 34 | 0.20 (0.14–0.28) | 25 | 0.08 (0.05–0.12) | 12 | 0.07 (0.04–0.13) | 13 | 0.09 (0.05–0.15) |
| 2012 | 28 | 0.08 (0.06–0.12) | 10 | 0.06 (0.03–0.11) | 18 | 0.11 (0.06–0.18) | 11 | 0.04 (0.02–0.06) | 3 | 0.02 (0.00–0.05) | 8 | 0.05 (0.02–0.11) |
| 2013 | 29 | 0.08 (0.06–0.12) | 11 | 0.07 (0.03–0.12) | 18 | 0.10 (0.06–0.17) | 18 | 0.06 (0.04–0.09) | 8 | 0.05 (0.02–0.10) | 10 | 0.07 (0.03–0.13) |
| APC | −5.75 (p < 0.001) | −6.48 (p < 0.001) | −4.76 (p < 0.001) | −6.45 (p < 0.001) | −8.09 (p < 0.001) | −3.85 (p < 0.001) | ||||||
| Year | Other Rare Congenital Malformations | Rare Chromosomal Abnormalities, Not Elsewhere Classified | ||||||||||
| Both Sexes | Men | Women | Both Sexes | Men | Women | |||||||
| n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | n | AR (CI) | |
| 1999 | 158 | 0.63 (0.53–0.74) | 87 | 0.68 (0.54–0.85) | 71 | 0.58 (0.45–0.74) | 157 | 0.55 (0.46–0.55) | 64 | 0.55 (0.46–0.65) | 93 | 0.67 (0.53–0.83) |
| 2000 | 168 | 0.64 (0.55–0.75) | 87 | 0.67 (0.53–0.83) | 81 | 0.62 (0.48–0.78) | 119 | 0.42 (0.35–0.51) | 55 | 0.42 (0.35–0.51) | 64 | 0.47 (0.36–0.61) |
| 2001 | 154 | 0.61 (0.51–0.71) | 89 | 0.68 (0.54–0.85) | 65 | 0.53 (0.40–0.68) | 116 | 0.38 (0.31–0.46) | 47 | 0.38 (0.31–0.46) | 69 | 0.47 (0.36–0.61) |
| 2002 | 158 | 0.57 (0.48–0.68) | 84 | 0.62 (0.49–0.78) | 74 | 0.52 (0.40–0.66) | 123 | 0.4 (0.33–0.49) | 51 | 0.4 (0.33–0.49) | 72 | 0.48 (0.37–0.62) |
| 2003 | 136 | 0.47 (0.39–0.56) | 84 | 0.57 (0.45–0.71) | 52 | 0.36 (0.26–0.48) | 163 | 0.51 (0.43–0.60) | 63 | 0.51 (0.43–0.60) | 100 | 0.67 (0.54–0.82) |
| 2004 | 123 | 0.41 (0.34–0.50) | 67 | 0.44 (0.34–0.57) | 56 | 0.38 (0.28–0.50) | 171 | 0.5 (0.43–0.59) | 80 | 0.5 (0.43–0.59) | 91 | 0.55 (0.44–0.68) |
| 2005 | 143 | 0.44 (0.37–0.53) | 77 | 0.46 (0.36–0.58) | 66 | 0.43 (0.33–0.55) | 176 | 0.5 (0.43–0.59) | 78 | 0.5 (0.43–0.59) | 98 | 0.57 (0.54–0.70) |
| 2006 | 130 | 0.42 (0.34–0.50) | 73 | 0.46 (0.36–0.58) | 57 | 0.37 (0.28–0.49) | 148 | 0.39 (0.33–0.46) | 68 | 0.39 (0.33–0.46) | 80 | 0.45 (0.35–0.56) |
| 2007 | 122 | 0.36 (0.30–0.43) | 70 | 0.41 (0.32–0.53) | 52 | 0.3 (0.22–0.40) | 161 | 0.41 (0.35–0.48) | 85 | 0.41 (0.35–0.48) | 76 | 0.41 (0.32–0.52) |
| 2008 | 136 | 0.38 (0.32–0.46) | 70 | 0.39 (0.31–0.50) | 66 | 0.37 (0.28–0.48) | 146 | 0.36 (0.30–0.43) | 69 | 0.33 (0.26–0.42) | 77 | 0.39 (0.31–0.50) |
| 2009 | 126 | 0.35 (0.29–0.42) | 65 | 0.34 (0.26–0.44) | 61 | 0.35 (0.27–0.46) | 175 | 0.42 (0.36–0.50) | 93 | 0.44 (0.36–0.55) | 82 | 0.41 (0.32–0.51) |
| 2010 | 121 | 0.31 (0.26–0.38) | 70 | 0.35 (0.27–0.45) | 51 | 0.28 (0.21–0.37) | 172 | 0.42 (0.36–0.49) | 79 | 0.37 (0.29–0.47) | 93 | 0.46 (0.37–0.57) |
| 2011 | 117 | 0.32 (0.26–0.38) | 59 | 0.32 (0.24–0.41) | 58 | 0.32 (0.24–0.42) | 164 | 0.39 (0.34–0.46) | 78 | 0.36 (0.29–0.46) | 86 | 0.43 (0.34–0.54) |
| 2012 | 99 | 0.25 (0.21–0.31) | 51 | 0.26 (0.19–0.35) | 48 | 0.25 (0.18–0.34) | 159 | 0.38 (0.32–0.44) | 77 | 0.36 (0.28–0.45) | 82 | 0.4 (0.32–0.51) |
| 2013 | 87 | 0.23 (0.18–0.29) | 42 | 0.22 (0.16–0.30) | 45 | 0.24 (0.18–0.29) | 163 | 0.37 (0.31–0.43) | 81 | 0.36 (0.28–0.45) | 82 | 0.38 (0.30–0.47) |
| APC | −6.66 (p < 0.001) | −7.19 (p < 0.001) | −6.00 (p < 0.001) | −1.77 (p < 0.001) | −0.16 (p = 0.90) | −3.05 (p < 0.001) | ||||||
| Year | Edwards Syndrome/Trisomy 18 | |||||||||||
| Both Sexes | Men | Women | ||||||||||
| n | AR (CI) | n | AR (CI) | n | AR (CI) | |||||||
| 1999 | 26 | 0.12 (0.08–0.18) | 4 | 0.04 (0.01–0.09) | 22 | 0.21 (0.13–0.32) | ||||||
| 2000 | 20 | 0.09 (0.06–0.14) | 7 | 0.06 (0.03–0.13) | 13 | 0.12 (0.07–0.21) | ||||||
| 2001 | 21 | 0.10 (0.06–0.15) | 4 | 0.04 (0.01–0.09) | 17 | 0.16 (0.09–0.26) | ||||||
| 2002 | 14 | 0.06 (0.03–0.10) | 5 | 0.04 (0.01–0.10) | 9 | 0.08 (0.04–0.15) | ||||||
| 2003 | 26 | 0.11 (0.07–0.16) | 3 | 0.02 (0.01–0.07) | 23 | 0.20 (0.13–0.30) | ||||||
| 2004 | 24 | 0.10 (0.06–0.14) | 7 | 0.05 (0.02–0.11) | 17 | 0.14 (0.08–0.23) | ||||||
| 2005 | 26 | 0.10 (0.06–0.15) | 9 | 0.07 (0.03–0.13) | 17 | 0.13 (0.08–0.21) | ||||||
| 2006 | 17 | 0.06 (0.04–0.10) | 3 | 0.02 (0.00–0.06) | 14 | 0.11 (0.06–0.18) | ||||||
| 2007 | 19 | 0.07 (0.04–0.10) | 4 | 0.03 (0.01–0.07) | 15 | 0.11 (0.06–0.18) | ||||||
| 2008 | 21 | 0.07 (0.04–0.11) | 9 | 0.06 (0.03–0.11) | 12 | 0.08 (0.04–0.15) | ||||||
| 2009 | 17 | 0.05 (0.03–0.09) | 6 | 0.04 (0.01–0.08) | 11 | 0.07 (0.04–0.13) | ||||||
| 2010 | 17 | 0.05 (0.03–0.09) | 3 | 0.02 (0.00–0.06) | 14 | 0.09 (0.05–0.16) | ||||||
| 2011 | 20 | 0.06 (0.04–0.10) | 8 | 0.05 (0.02–0.10) | 12 | 0.08 (0.04–0.14) | ||||||
| 2012 | 9 | 0.03 (0.01–0.06) | 4 | 0.03 (0.01–0.06) | 5 | 0.03 (0.01–0.08) | ||||||
| 2013 | 11 | 0.04 (0.02–0.06) | 5 | 0.03 (0.01–0.07) | 6 | 0.04 (0.01–0.09) | ||||||
| APC | −6.60 (p < 0.001) | −2.44 (p = 0.30) | −8.24 (p < 0.001) | |||||||||
| Standardised Mortality Ratio for Rare Congenital Anomalies, 1999–2013 (95% CI) | |||||
|---|---|---|---|---|---|
| District | Province | Location | Both Sexes | Men | Women |
| Significantly Lower than the Expected for the Country as Reference | |||||
| Marquesado | Alicante | E | 0.69 (0.48–0.97) | – | – |
| Central | Alicante | E | 0.76 (0.64–0.90) | 0.78 (0.62–0.98) | 0.74 (0.57–0.94) |
| Meridional | Alicante | E | 0.84 (0.71–0.98) | 0.80 (0.63–0.99) | – |
| Badajoz | Badajoz | W | 0.68 (0.47–0.94) | 0.58 (0.33–0.95) | – |
| Ibiza | Baleares | E * | – | – | 0.42 (0.17–0.86) |
| Mallorca | Baleares | E * | 0.71 (0.61–0.83) | 0.66 (0.53–0.82) | 0.77 (0.62–0.95) |
| Osona | Barcelona | NE | – | 0.57 (0.32–0.94) | – |
| Penedès | Barcelona | NE | 0.76 (0.58–0.98) | – | 0.62 (0.39–0.94) |
| Vallès Oriental | Barcelona | NE | 0.76 (0.61–0.94) | – | 0.69 (0.48–0.95) |
| Vallès Occidental | Barcelona | NE | 0.71 (0.62–0.82) | 0.68 (0.56–0.83) | 0.75 (0.61–0.92) |
| Litoral del Norte | Castellón | E | – | 0.42 (0.13–0.97) | – |
| Cerdanya | Girona | NE | 0.00 (0.00–0.87) | – | – |
| La Selva | Girona | NE | 0.63 (0.43–0.90) | 0.58 (0.32–0.96) | – |
| Bajo Cinca | Huesca | N | – | 0.00 (0.00–0.96) | – |
| Costa | Lugo | NW | – | – | 0.22 (0.02–0.79) |
| Sur Occidental | Madrid | C | 0.61 (0.52–0.72) | 0.62 (0.49–0.77) | 0.61 (0.47–0.78) |
| Litoral | Pontevedra | NW | 0.76 (0.65–0.89) | 0.76 (0.60–0.94) | 0.78 (0.61–0.97) |
| Costera | Cantabria | N | 0.62 (0.50–0.76) | 0.61 (0.45–0.81) | 0.63 (0.46–0.85) |
| Sagra–Toledo | Toledo | C | 0.71 (0.53–0.92) | 0.60 (0.39–0.88) | – |
| Riberas del Júcar | Valencia | E | – | – | 0.67 (0.44–0.99) |
| Significantly Higher than the Expected for the Country as Reference | |||||
| Campo de Níjar y Bajo Andarax | Almería | SE | 1.25 (1.02–1.52) | – | 1.40 (1.04–1.83) |
| Ávila | Avila | C | – | 1.85 (1.05–3.00) | – |
| Don Benito | Badajoz | W | 1.43 (1.02–1.94) | – | – |
| Osona | Barcelona | NE | – | – | 1.48 (1.02–2.07) |
| Arlanzón | Burgos | N | – | 1.40 (1.01–1.88) | – |
| Caceres | Caceres | W | 1.42 (1.02–1.86) | 1.82 (1.28–2.52) | 1.44 (1.14–1.80) |
| Campiña de Cádiz | Cadiz | SW | 1.20 (1.01–1.42) | – | – |
| Los Pedroches | Cordoba | S | 1.47 (1.01–2.07) | – | – |
| Campiña Baja | Cordoba | S | 1.29 (1.11–1.50) | – | 1.35 (1.07–1.67) |
| Las Colonias | Cordoba | S | 1.95 (1.05–3.00) | 2.13 (1.02–3.92) | – |
| Campiña Alta | Cordoba | S | 1.33 (1.04–1.67) | – | 1.73 (1.26–2.31) |
| Serranía Baja | Cuenca | C | – | 3.63 (1.17–8.47) | – |
| La Vega | Granada | S | 1.32 (1.15–1.51) | 1.29 (1.06–1.56) | 1.36 (1.11–1.65) |
| Guadix | Granada | S | 1.77 (1.11–2.67) | – | – |
| Montefrío | Granada | S | – | – | 3.56 (1.77–6.36) |
| Sierra | Huelva | SW | – | – | 2.16 (1.08–3.87) |
| La Loma | Jaén | S | 1.44 (1.03–1.96) | – | 1.85 (1.19–2.16) |
| Campiña del Sur | Jaén | S | 1.66 (1.36–2.02) | 1.88 (1.44–2.42) | 1.41 (1.00–1.92) |
| Mágina | Jaén | S | – | – | 2.21 (1.06–4.06) |
| Montaña de Riaño | León | N | 2.37 (1.08–4.49) | – | – |
| Guadalhorce | Malaga | S | 1.31 (1.19–1.43) | 1.34 (1.18–1.52) | 1.27 (1.11–1.45) |
| Vélez–Málaga | Malaga | S | 1.40 (1.10–1.75) | 1.51 (1.10–2.02) | – |
| Río Segura | Murcia | SE | 1.22 (1.07–1.37) | 1.31 (1.11–1.53) | – |
| Suroeste y Valle del Guadalentín | Murcia | SE | 1.31 (1.05–1.60) | 1.41 (1.06–1.84) | – |
| Campo de Cartagena | Murcia | SE | 1.59 (1.36–1.84) | 1.68 (1.37–2.04) | 1.48 (1.16–1.85) |
| Grado | Asturias | N | – | – | 2.33 (1.16–4.17) |
| Oviedo | Asturias | N | 1.40 (1.17–1.66) | 1.55 (1.22–1.94) | – |
| Gran Canaria | Las Palmas | SW * | 1.34 (1.20–1.49) | 1.33 (1.14–1.54) | 1.36 (1.15–1.59) |
| Sur de Tenerife | Tenerife | SW * | 1.30 (1.11–1.51) | 1.32 (1.06–1.62) | 1.28 (1.01–1.60) |
| Sierra Norte | Seville | SW | 1.77 (1.22–2.49) | 2.40 (1.54–3.57) | – |
| La Vega | Seville | SW | 1.27 (1.15–1.39) | 1.29 (1.14–1.47) | 1.24 (1.07–1.42) |
| Ceuta | Ceuta | S | 1.61 (1.18–2.14) | 1.97 (1.34–2.80) | – |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alonso-Ferreira, V.; Sánchez-Díaz, G.; Villaverde-Hueso, A.; Posada de la Paz, M.; Bermejo-Sánchez, E. A Nationwide Registry-Based Study on Mortality Due to Rare Congenital Anomalies. Int. J. Environ. Res. Public Health 2018, 15, 1715. https://doi.org/10.3390/ijerph15081715
Alonso-Ferreira V, Sánchez-Díaz G, Villaverde-Hueso A, Posada de la Paz M, Bermejo-Sánchez E. A Nationwide Registry-Based Study on Mortality Due to Rare Congenital Anomalies. International Journal of Environmental Research and Public Health. 2018; 15(8):1715. https://doi.org/10.3390/ijerph15081715
Chicago/Turabian StyleAlonso-Ferreira, Verónica, Germán Sánchez-Díaz, Ana Villaverde-Hueso, Manuel Posada de la Paz, and Eva Bermejo-Sánchez. 2018. "A Nationwide Registry-Based Study on Mortality Due to Rare Congenital Anomalies" International Journal of Environmental Research and Public Health 15, no. 8: 1715. https://doi.org/10.3390/ijerph15081715
APA StyleAlonso-Ferreira, V., Sánchez-Díaz, G., Villaverde-Hueso, A., Posada de la Paz, M., & Bermejo-Sánchez, E. (2018). A Nationwide Registry-Based Study on Mortality Due to Rare Congenital Anomalies. International Journal of Environmental Research and Public Health, 15(8), 1715. https://doi.org/10.3390/ijerph15081715





