Surface Oxidation of Ethylenechlorotrifluoroethylene (ECTFE) Membrane for the Treatment of Real Produced Water by Membrane Distillation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Surface Modification
2.3. Characterization
2.4. Membrane Distillation
2.5. Regeneration
3. Results and Discussion
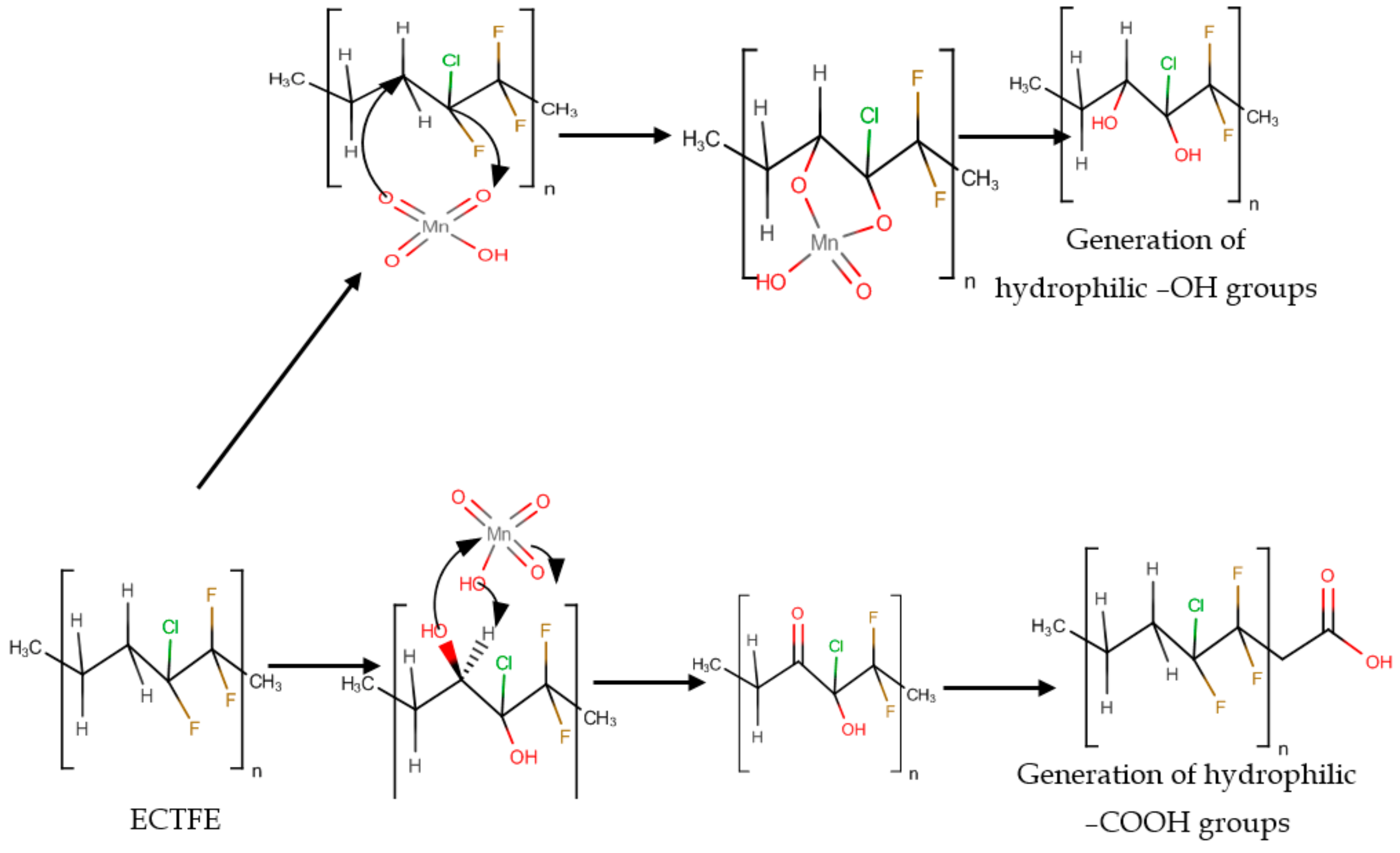
3.1. Surface Oxidation
3.2. Characterization of the Modified Surface
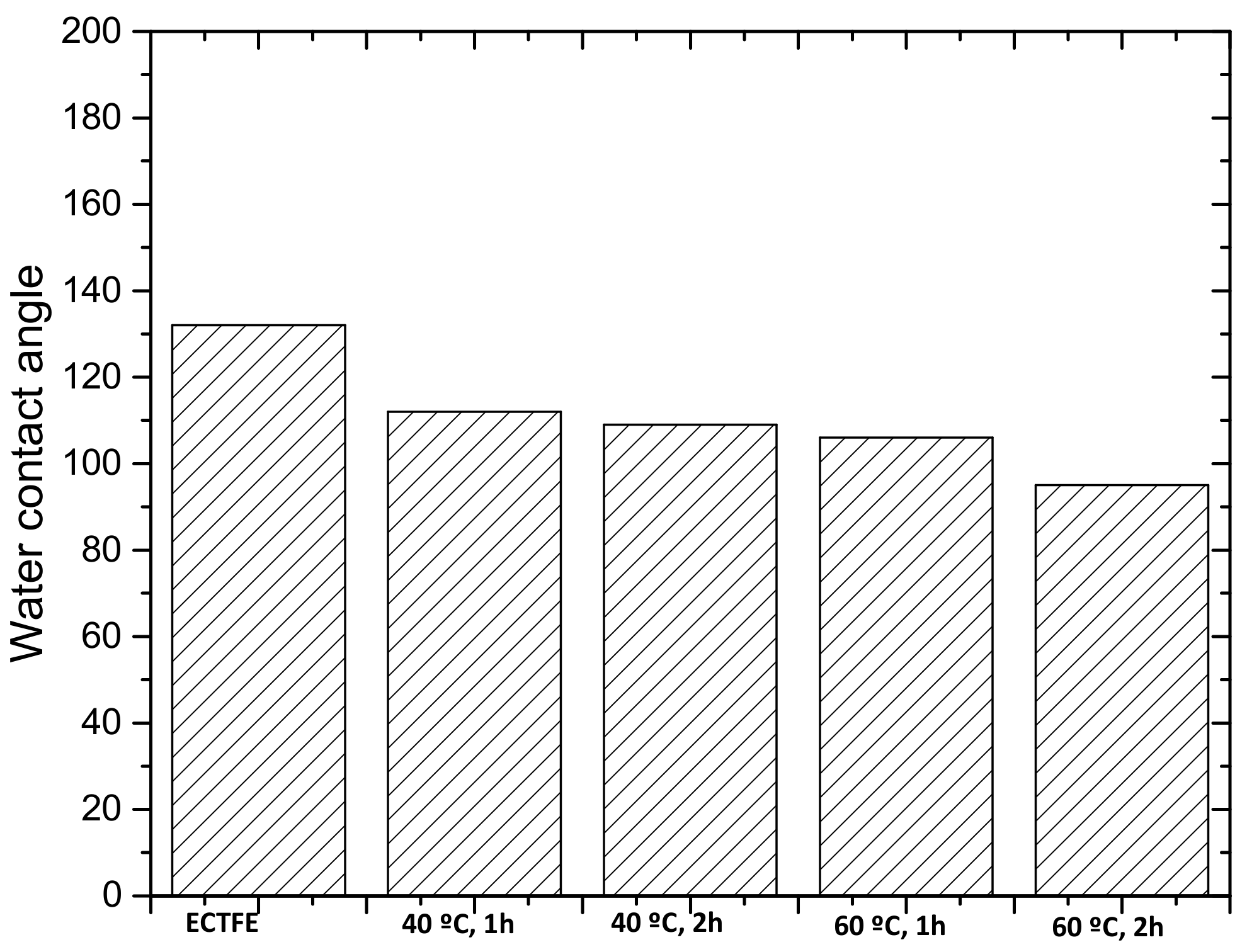
3.2.1. Water Contact Angle Measurement
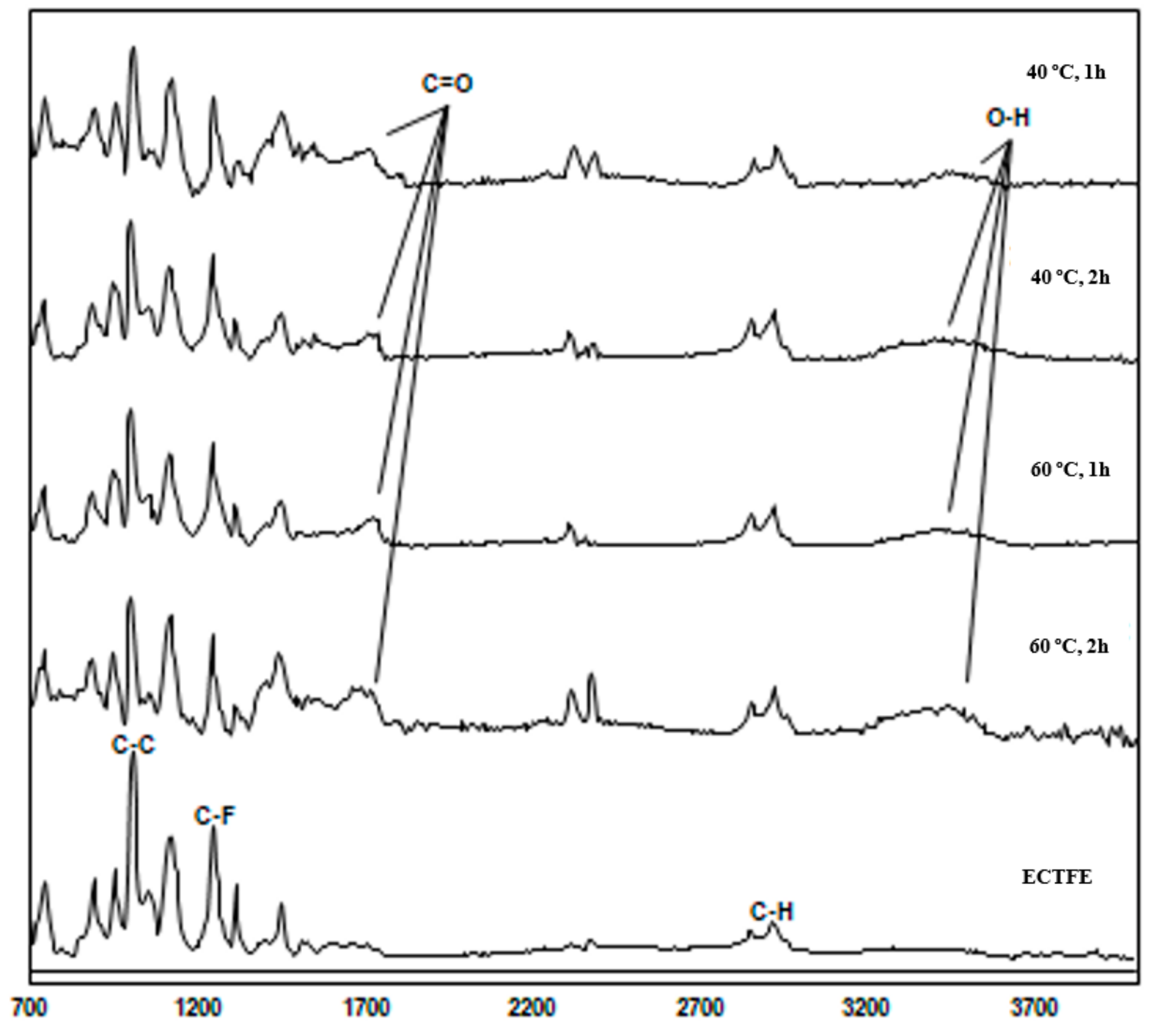
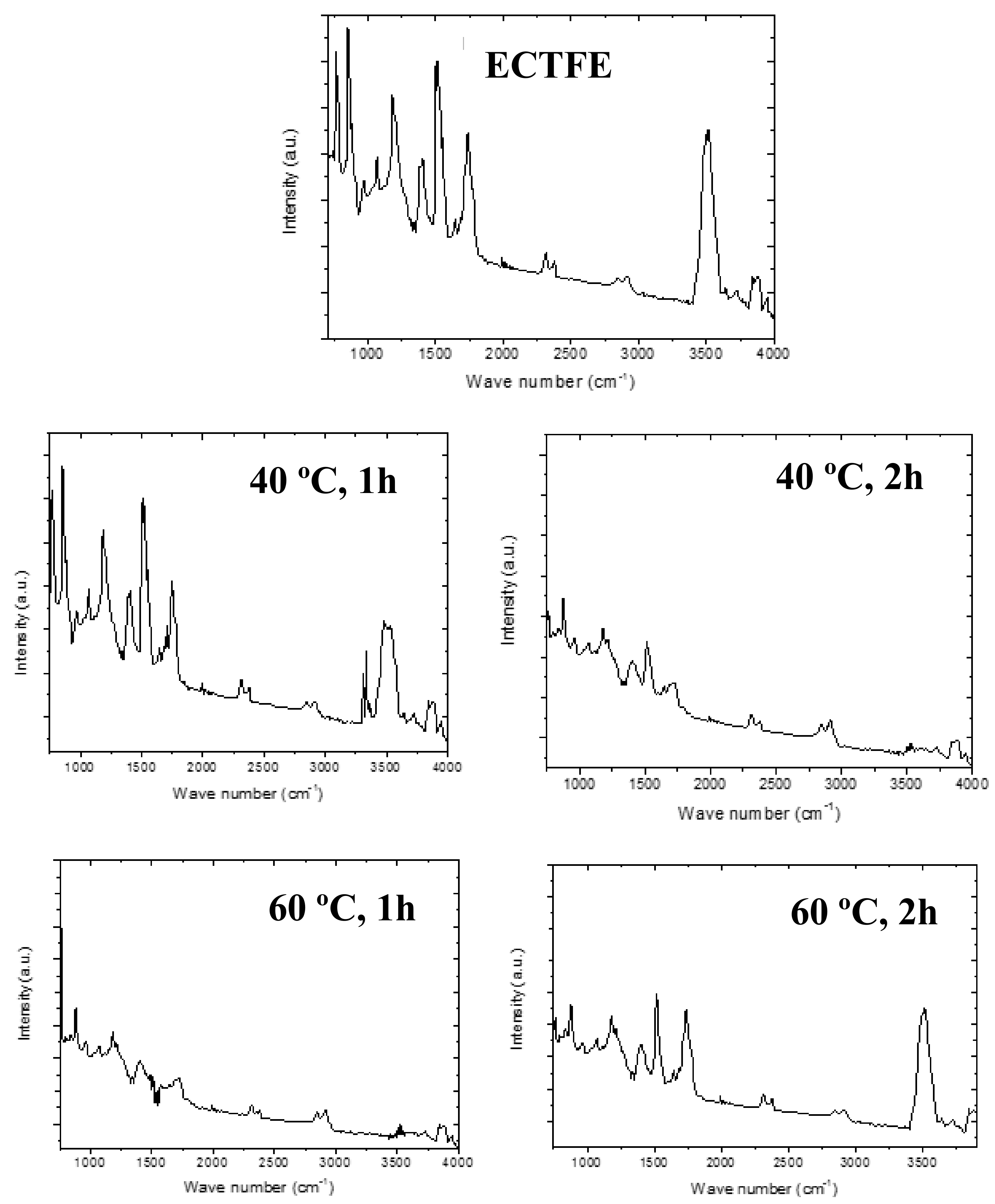
3.2.2. FTIR Characterization
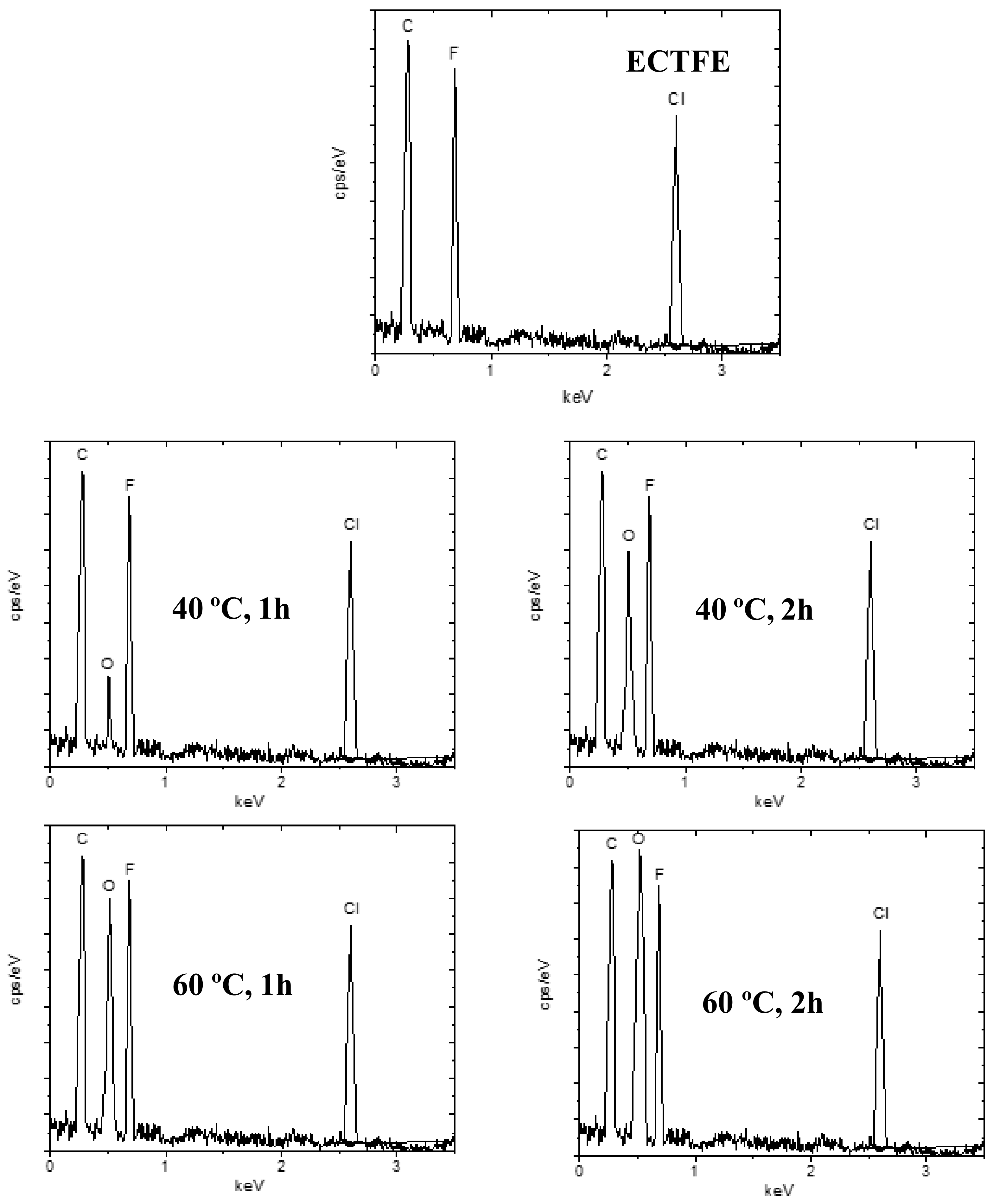
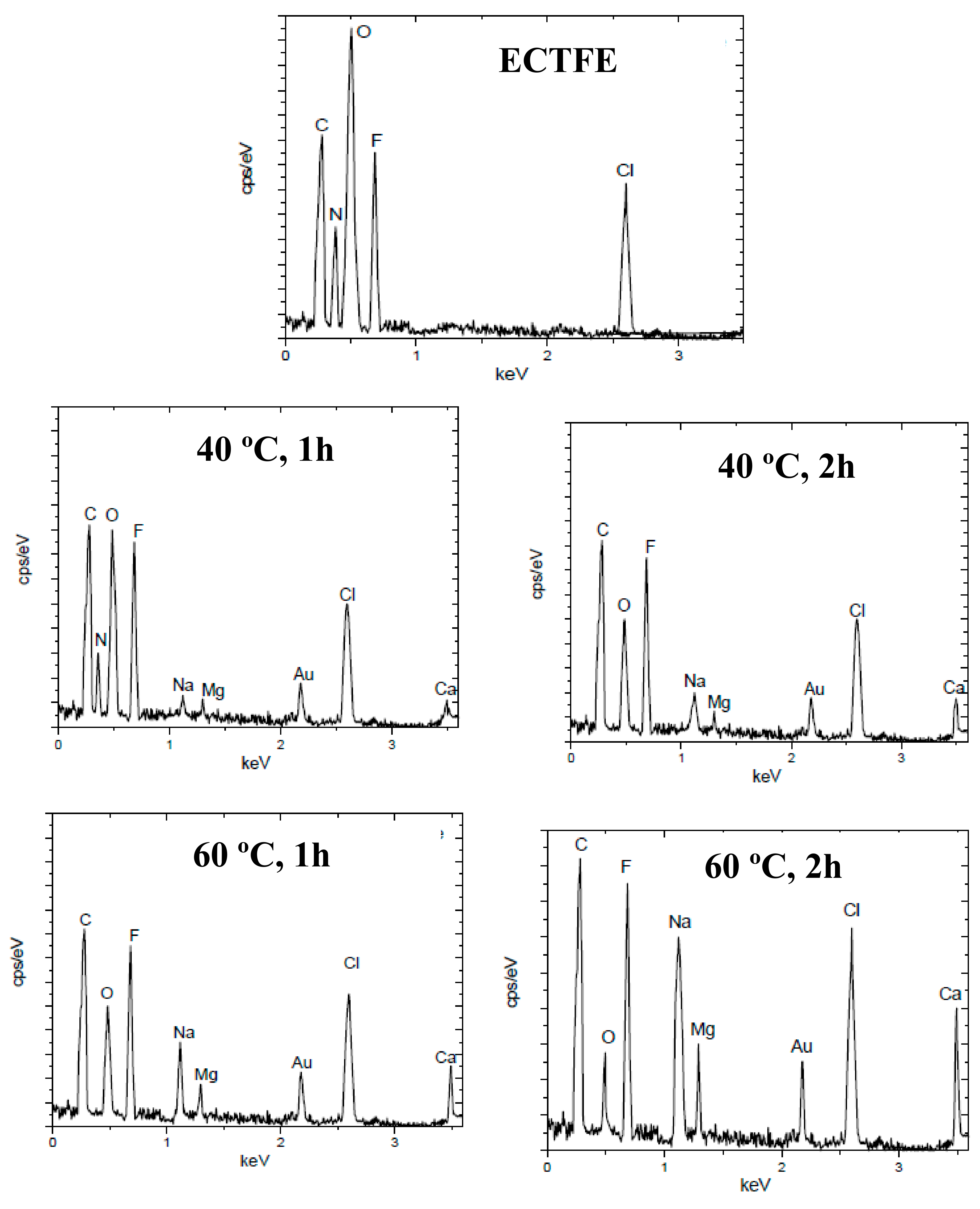
3.2.3. EDX Characterization of Modified Membranes
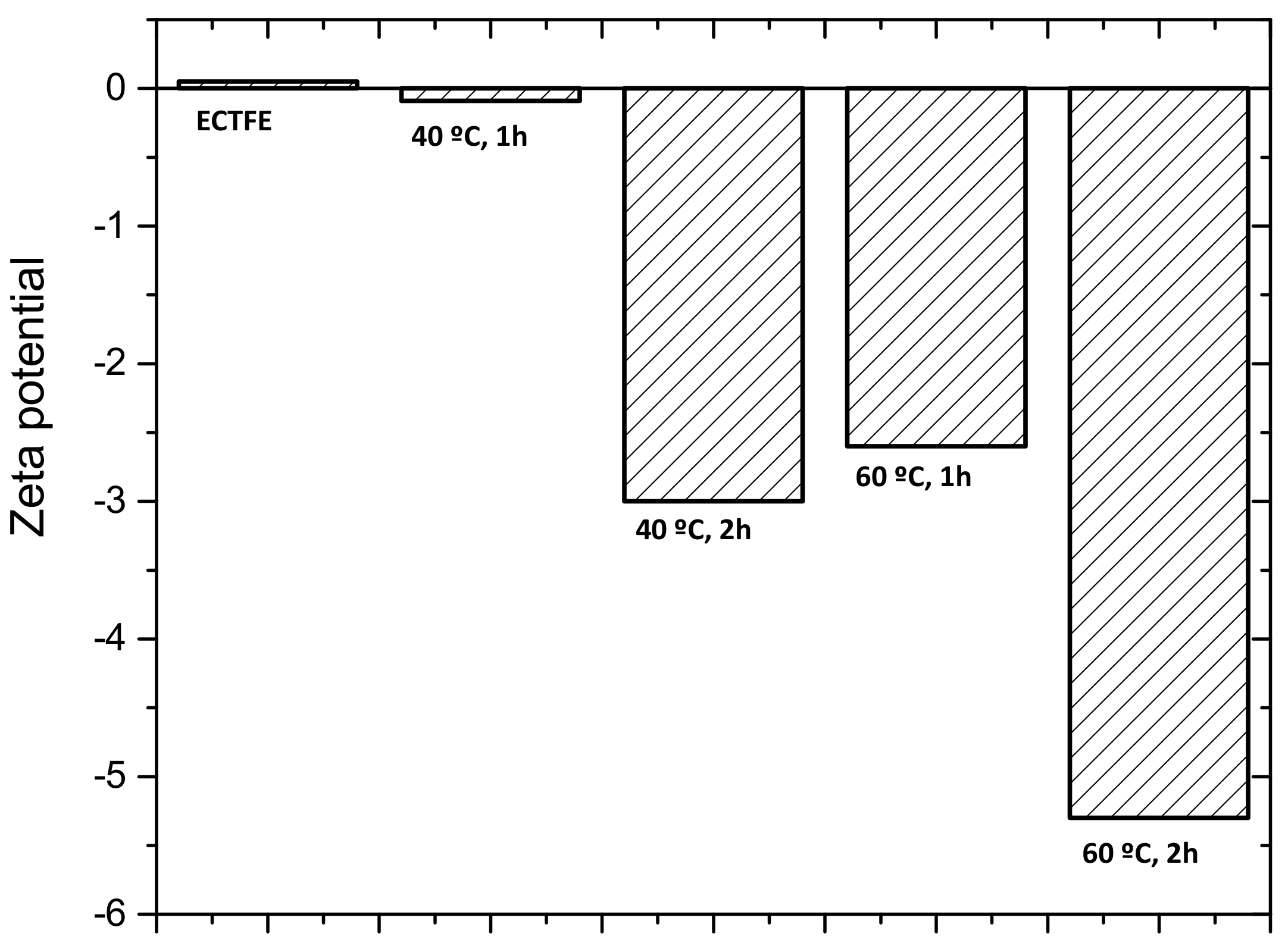
3.2.4. Zeta Potential Measurement
3.2.5. Relative Composition from XPS Analysis
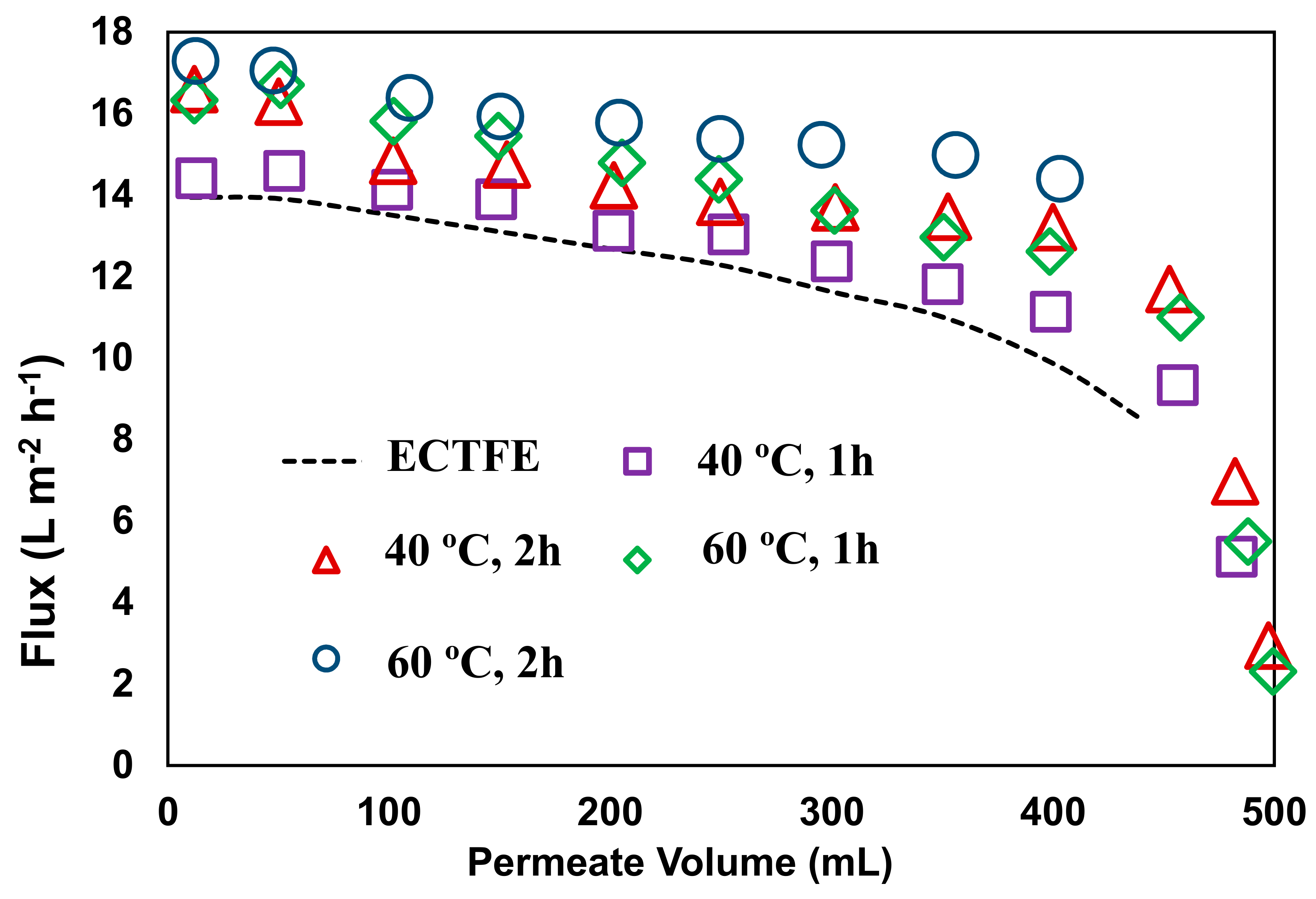
3.3. Membrane Performance
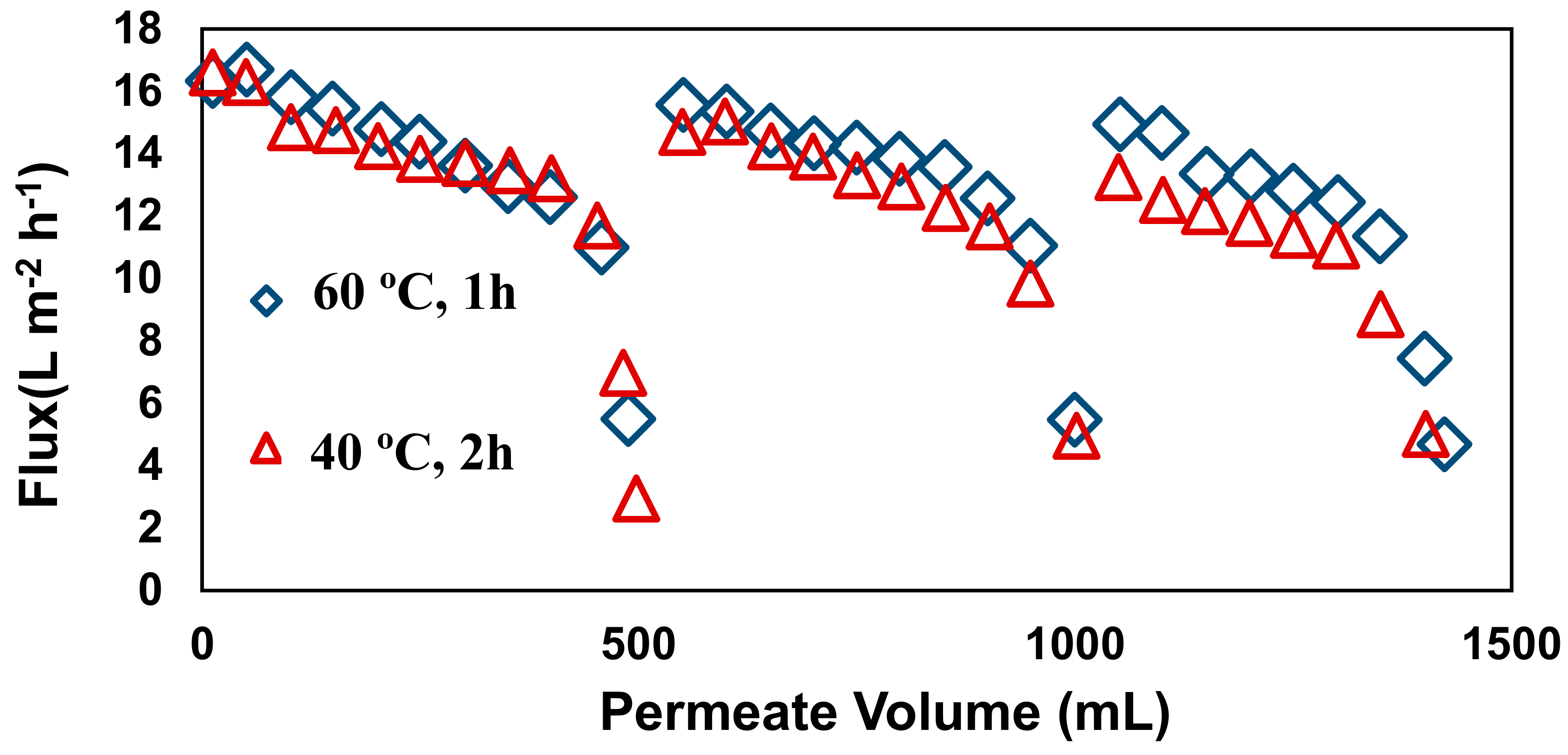
3.4. Reusability Test
3.5. Membrane Fouling
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ECTFE | Ethylenechlorotrifluoroethylene |
| PW | Produced Water |
| MD | Membrane Distillation |
| SEM | Scanning Electron Microscopy |
| bbl | Barrel |
| TDS | Total Dissolved Solid |
| PVDF | Polyvinylidene Fluoride |
| PP | Polypropylene |
| DI | De Ionized |
| FTIR | Fourier Transformed Infrared |
| EDX | Energy-Dispersive X-ray Spectroscopy |
| LMH | L m−2 h−1 |
References
- Himstedt, H.H.; Sengupta, A.; Qian, X.; Wickramasinghe, S.R. Magnetically responsive nano filtration membranes for treatment of coal bed methane produced water. J. Taiwan Inst. Chem. Eng. 2018. [Google Scholar] [CrossRef]
- Sardari, K.; Fyfe, P.; Lincicome, D.; Wickramasinghe, S.R. Aluminum electrocoagulation followed by forward osmosis for treating hydraulic fracturing produced waters. Desalination 2018, 428, 172–181. [Google Scholar] [CrossRef]
- Alessi, D.S.; Zolfaghari, A.; Kletke, S.; Gehman, J.; Allen, D.M.; Goss, G.G. Comparative analysis of hydraulic fracturing wastewater practices in unconventional shale development: Water sourcing, treatment and disposal practices. Can. Water Resour. J. 2017, 42, 105–121. [Google Scholar] [CrossRef]
- Lokare, O.R.; Tavakkoli, S.; Wadekar, S.; Khanna, V.; Vidic, R.D. Fouling in direct contact membrane distillation of produced water from unconventional gas extraction. J. Membr. Sci. 2017, 524, 493–501. [Google Scholar] [CrossRef]
- Kondash, A.J.; Albright, E.; Vengosh, A. Quantity of flowback and produced waters from unconventional oil and gas exploration. Sci. Total Environ. 2017, 574, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Gregory, K.B.; Vidic, R.D.; Dzombak, D.A. Water management challenges associated with the production of shale gas by hydraulic fracturing. Elements 2011, 7, 181–186. [Google Scholar] [CrossRef]
- Malmali, M.; Fyfe, P.; Lincicome, D.; Sardari, K.; Wickramasinghe, S.R. Selecting membranes for treating hydraulic fracturing produced waters by membrane distillation. Sep. Sci. Technol. 2017, 52, 266–275. [Google Scholar] [CrossRef]
- Lawson, K.W.; Lloyd, D.R. Membrane distillation. II. Direct contact MD. J. Membr. Sci. 1996, 120, 123–133. [Google Scholar] [CrossRef]
- Onsekizoglu, P. Membrane distillation: Principle, advances, limitations and future prospects in food industry. In Distillation-Advances from Modeling to Applications; InTech: London, UK, 2012. [Google Scholar] [CrossRef]
- Drioli, E.; Criscuoli, A.; Molero, L.P. Membrane Distillation. In Water Wastewater Treatment Technology, 2nd ed.; Vigneswaran, S., Ed.; EOLSS Publishers: Oxford, UK, 2009; Volume III, pp. 124–176. [Google Scholar]
- Alkhudhiri, A.; Darwish, N.; Hilal, N. Membrane distillation: A comprehensive review. Desalination 2012, 287, 2–18. [Google Scholar] [CrossRef]
- Pan, J.; Xiao, C.; Huang, Q.; Liu, H.; Hu, J. ECTFE porous membranes with conveniently controlled microstructures for vacuum membrane distillation. J. Mater. Chem. A 2015, 3, 23549–23559. [Google Scholar] [CrossRef]
- Zhou, B.; Li, Q.; Tang, Y.; Lin, Y.; Wang, X. Preparation of ECTFE membranes with bicontinuous structure via TIPS method by a binary diluent. Desalin. Water Treat. 2016, 57, 17646–17657. [Google Scholar] [CrossRef]
- Meng, J.Q.; Chen, C.L.; Huang, L.P.; Du, Q.Y.; Zhang, Y.F. Surface modification of PVDF membrane via AGET ATRP directly from the membrane surface. Appl. Surf. Sci. 2011, 257, 6282–6290. [Google Scholar] [CrossRef]
- Rahimpour, A.; Madaeni, S.S.; Zereshki, S.; Mansourpanah, Y. Preparation and characterization of modified nano-porous PVDF membrane with high antifouling property using UV photo-grafting. Appl. Surf. Sci. 2009, 255, 7455–7461. [Google Scholar] [CrossRef]
- Kang, G.D.; Cao, Y.M. Application and modification of poly(vinylidene fluoride) (PVDF) membranes—A review. J. Membr. Sci. 2014, 463, 145–165. [Google Scholar] [CrossRef]
- Wang, Z.; Jin, J.; Hou, D.; Lin, S. Tailoring surface charge and wetting property for robust oil-fouling mitigation in membrane distillation. J. Membr. Sci. 2016, 516, 113–122. [Google Scholar] [CrossRef]
- Hernández-Aguirre, O.A.; Núñez-Pineda, A.; Tapia-Tapia, M.; Espinosa, R.M.G. Surface Modification of Polypropylene Membrane Using Biopolymers with Potential Applications for Metal Ion Removal. J. Chem. 2016, 2016, 11. [Google Scholar] [CrossRef]
- Bae, B.; Chun, B.; Kim, D. Surface characterization of microporous polypropylene membranes modified by plasma treatment. Polymer 2001, 42, 7879–7885. [Google Scholar] [CrossRef]
- Meng, S.; Ye, Y.; Mansouri, J.; Chen, V. Fouling and crystallisation behaviour of superhydrophobic nano-composite PVDF membranes in direct contact membrane distillation. J. Membr. Sci. 2014, 463, 102–112. [Google Scholar] [CrossRef]
- Boo, C.; Lee, J.; Elimelech, M. Omniphobic polyvinylidene fluoride (PVDF) membrane for desalination of shale gas produced water by membrane distillation. Environ. Sci. Technol. 2016, 50, 12275–12282. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Lin, S. Membrane fouling and wetting in membrane distillation and their mitigation by novel membranes with special wettability. Water Res. 2017, 112, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Hou, D.; Lin, S. Composite membrane with underwater-oleophobic surface for anti-oil-fouling membrane distillation. Environ. Sci. Technol. 2016, 50, 3866–3874. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, A.; Boo, C.; Karanikola, V.; Lin, S.; Straub, A.P.; Tong, T.; Warsinger, D.M.; Elimelech, M. Membrane distillation at the water-energy nexus: Limits, opportunities, and challenges. Energy Environ. Sci. 2018, 11, 1177–1196. [Google Scholar] [CrossRef]
- Zhang, M.; Nguyen, Q.T.; Ping, Z. Hydrophilic modification of poly (vinylidene fluoride) microporous membrane. J. Membr. Sci. 2009, 327, 78–86. [Google Scholar] [CrossRef]
- Sarani, A.; de Geyter, N.; Nikiforov, A.Y.; Morent, R.; Leys, C.; Hubert, J.; Reniers, F. Surface modification of PTFE using an atmospheric pressure plasma jet in argon and argon + CO2. Surf. Coat. Technol. 2012, 206, 2226–2232. [Google Scholar] [CrossRef]
- Wang, S.; Li, J.; Suo, J.; Luo, T. Surface modification of porous poly(tetrafluoraethylene) film by a simple chemical oxidation treatment. Appl. Surf. Sci. 2010, 256, 2293–2298. [Google Scholar] [CrossRef]
- Shaabani, A.; Tavasoli-Rad, F.; Lee, D.G. Potassium permanganate oxidation of organic compounds. Synth. Commun. 2005, 35, 571–580. [Google Scholar] [CrossRef]
- Ethirajan, S.K.; Sengupta, A.; Jebur, M.; Kamaz, M.; Qian, X.; Wickramasinghe, R. Single-step synthesis of novel polyionic liquids having antibacterial activity and showing π-electron mediated selectivity in separation of aromatics. ChemistrySelect 2018, 3, 4959–4968. [Google Scholar] [CrossRef]
- Jebur, M.; Sengupta, A.; Chiao, Y.H.; Kamaz, M.; Qian, X.; Wickramasinghe, R. Pi electron cloud mediated separation of aromatics using supported ionic liquid (SIL) membrane having antibacterial activity. J. Membr. Sci. 2018, 556, 1–11. [Google Scholar] [CrossRef]
- Kamaz, M.; Rocha, P.; Sengupta, A.; Qian, X.; Wickramasinghe, R.S. Efficient removal of chemically toxic dyes using microorganism from activated sludge: Understanding sorption mechanism, kinetics, and associated thermodynamics. Sep. Sci. Technol. 2018, 53, 1–17. [Google Scholar] [CrossRef]
- Avram, A.M.; Morin, P.; Brownmiller, C.; Howard, L.R.; Sengupta, A.; Wickramasinghe, S.R. Concentrations of polyphenols from blueberry pomace extract using nanofiltration. Food Bioprod. Process. 2017, 106, 91–101. [Google Scholar] [CrossRef]
- Avram, A.; Sengupta, A.; Pfromm, P.H.; Zorn, H.; Lorenz, P.; Schwarz, T.; Nguyen, K.Q.; Czermak, P. Novel DyP from the basidiomycete Pleurotus sapidus: Substrate screening and kinetics. Biocatalysis 2018, 4, 1–13. [Google Scholar] [CrossRef]
- Njatawidjaja, E.; Kodama, M.; Matsuzaki, K.; Yasuda, K.; Matsuda, T.; Kogoma, M. Hydrophilic modification of expanded polytetrafluoroethylene (ePTFE) by atmospheric pressure glow discharge (APG) treatment. Surf. Coat. Technol. 2006, 201, 699–706. [Google Scholar] [CrossRef]
- Wang, C.; Chen, J.R.; Li, R. Studies on surface modification of poly(tetrafluoroethylene) film by remote and direct Ar plasma. Appl. Surf. Sci. 2008, 254, 2882–2888. [Google Scholar] [CrossRef]
- Lundstedt, A.; Papadakis, R.; Li, H.; Han, Y.; Jorner, K.; Bergman, J.; Leifer, K.; Grennberg, H.; Ottosson, H. White-light photoassisted covalent functionalization of graphene using 2-propanol. Small Methods 2017, 1, 1700214. [Google Scholar] [CrossRef]
- Saha, N.K.; Balakrishnan, M.; Ulbricht, M. Sugarcane juice ultrafiltration: FTIR and SEM analysis of polysaccharide fouling. J. Membr. Sci. 2007, 306, 287–297. [Google Scholar] [CrossRef]
- Yan, L.; Hong, S.; Li, M.L.; Li, Y.S. Application of the Al2O3-PVDF nanocomposite tubular ultrafiltration (UF) membrane for oily wastewater treatment and its antifouling research. Sep. Purif. Technol. 2009, 66, 347–352. [Google Scholar] [CrossRef]
- An, Y.; Wang, Z.; Wu, Z.; Yang, D.; Zhou, Q. Characterization of membrane foulants in an anaerobic non-woven fabric membrane bioreactor for municipal wastewater treatment. Chem. Eng. J. 2009, 155, 709–715. [Google Scholar] [CrossRef]
- Wang, Z.; Wu, Z.; Yin, X.; Tian, L. Membrane fouling in a submerged membrane bioreactor (MBR) under sub-critical flux operation: Membrane foulant and gel layer characterization. J. Membr. Sci. 2008, 325, 238–244. [Google Scholar] [CrossRef]


| Parameter | PW |
|---|---|
| Total dissolved solid (TDS) | 157,000 mgL−1 |
| Total suspended solid (TSS) | 1300 mg L−1 |
| Total organic carbon (TOC) | 12 mg L−1 |
| Turbidity | 273 NTU’s |
| Calcium | 12,000 mg L−1 |
| Chloride | 102,200 mg L−1 |
| Magnesium | 1250 mg L−1 |
| Sodium | 38,800 mg L−1 |
| Sulfate | 75.5 mg L−1 |
| Electroneutrality percent difference | <5% |
| Membrane | C (%) | F (%) | O (%) |
|---|---|---|---|
| ECTFE | 64 | 36 | - |
| 40 °C, 1 h | 61 | 32 | 7 |
| 40 °C, 2 h | 52 | 24 | 24 |
| 60 °C, 1 h | 56 | 21 | 23 |
| 60 °C, 2 h | 51 | 20 | 29 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anari, Z.; Sengupta, A.; Wickramasinghe, S.R. Surface Oxidation of Ethylenechlorotrifluoroethylene (ECTFE) Membrane for the Treatment of Real Produced Water by Membrane Distillation. Int. J. Environ. Res. Public Health 2018, 15, 1561. https://doi.org/10.3390/ijerph15081561
Anari Z, Sengupta A, Wickramasinghe SR. Surface Oxidation of Ethylenechlorotrifluoroethylene (ECTFE) Membrane for the Treatment of Real Produced Water by Membrane Distillation. International Journal of Environmental Research and Public Health. 2018; 15(8):1561. https://doi.org/10.3390/ijerph15081561
Chicago/Turabian StyleAnari, Zahra, Arijit Sengupta, and Sumith Ranil Wickramasinghe. 2018. "Surface Oxidation of Ethylenechlorotrifluoroethylene (ECTFE) Membrane for the Treatment of Real Produced Water by Membrane Distillation" International Journal of Environmental Research and Public Health 15, no. 8: 1561. https://doi.org/10.3390/ijerph15081561
APA StyleAnari, Z., Sengupta, A., & Wickramasinghe, S. R. (2018). Surface Oxidation of Ethylenechlorotrifluoroethylene (ECTFE) Membrane for the Treatment of Real Produced Water by Membrane Distillation. International Journal of Environmental Research and Public Health, 15(8), 1561. https://doi.org/10.3390/ijerph15081561





