A Physiologically-Based Pharmacokinetic Modeling Approach Using Biomonitoring Data in Order to Assess the Contribution of Drinking Water for the Achievement of an Optimal Fluoride Dose for Dental Health in Children
Abstract
1. Introduction
2. Materials and Methods
2.1. PBPK Model Development and Validation
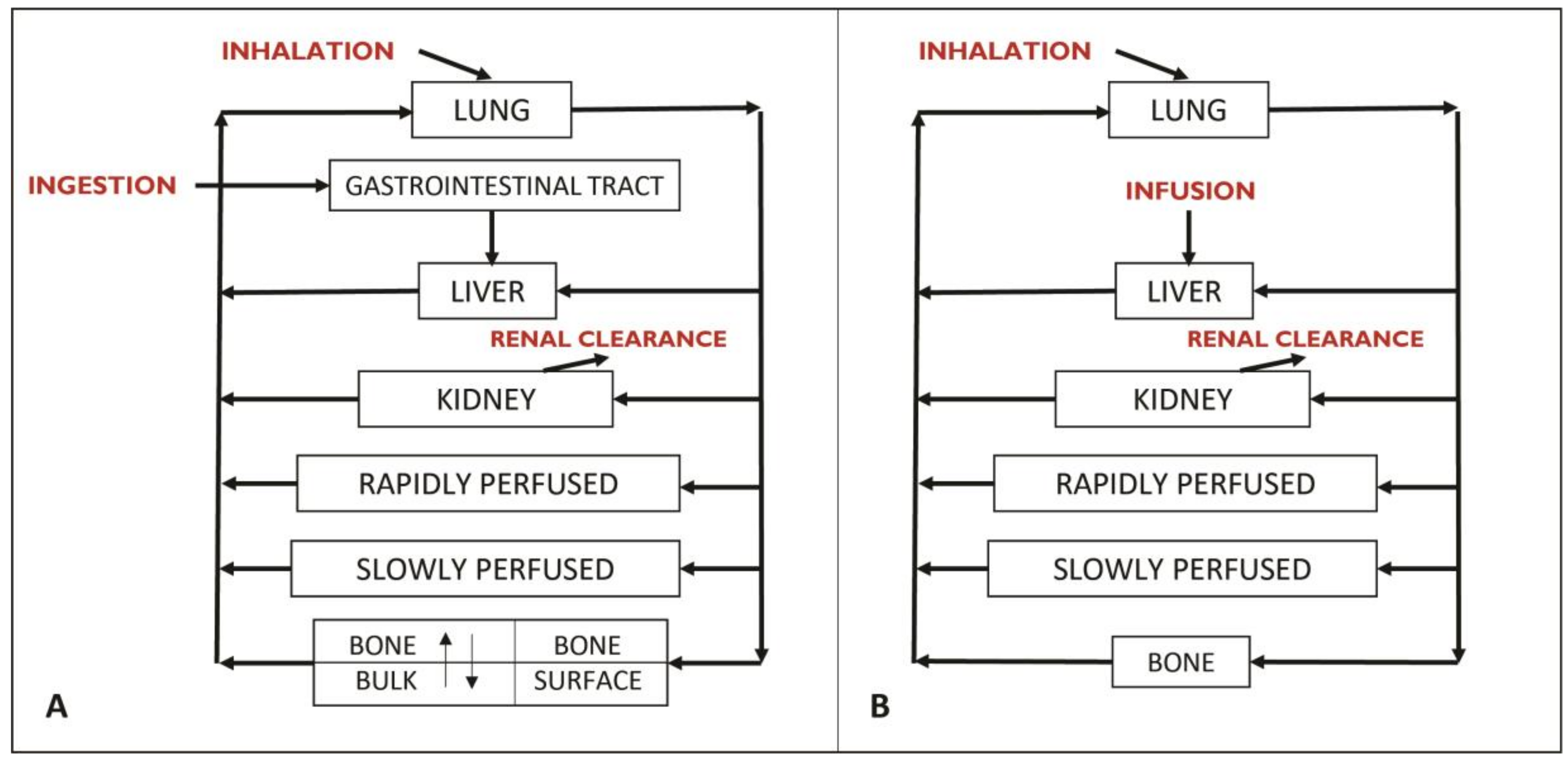
2.1.1. Model Structure
2.1.2. Model Parameters
2.1.3. Model Validation
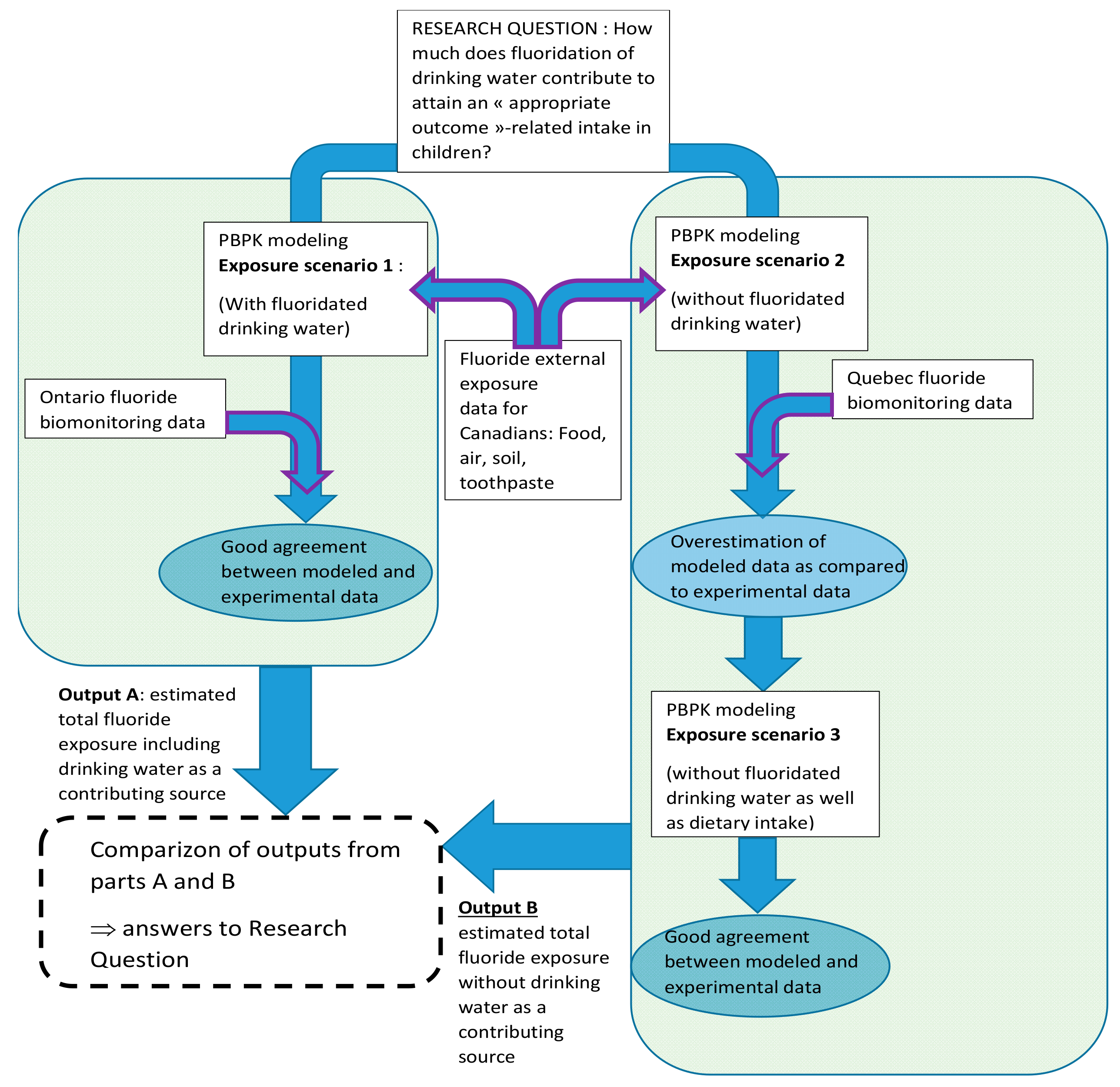
2.2. Exposure Scenarios
2.2.1. Subjects of Interest
2.2.2. Model Simulations of Internal Dose Metrics of Interest
2.2.3. Exposure Scenarios Simulated
2.3. Determination of Mean Absorbed Fluoride Dose in Average Children from Quebec and Ontario Using Biomonitoring Data
2.4. PBPK Model Sensitivity Analyses
3. Results
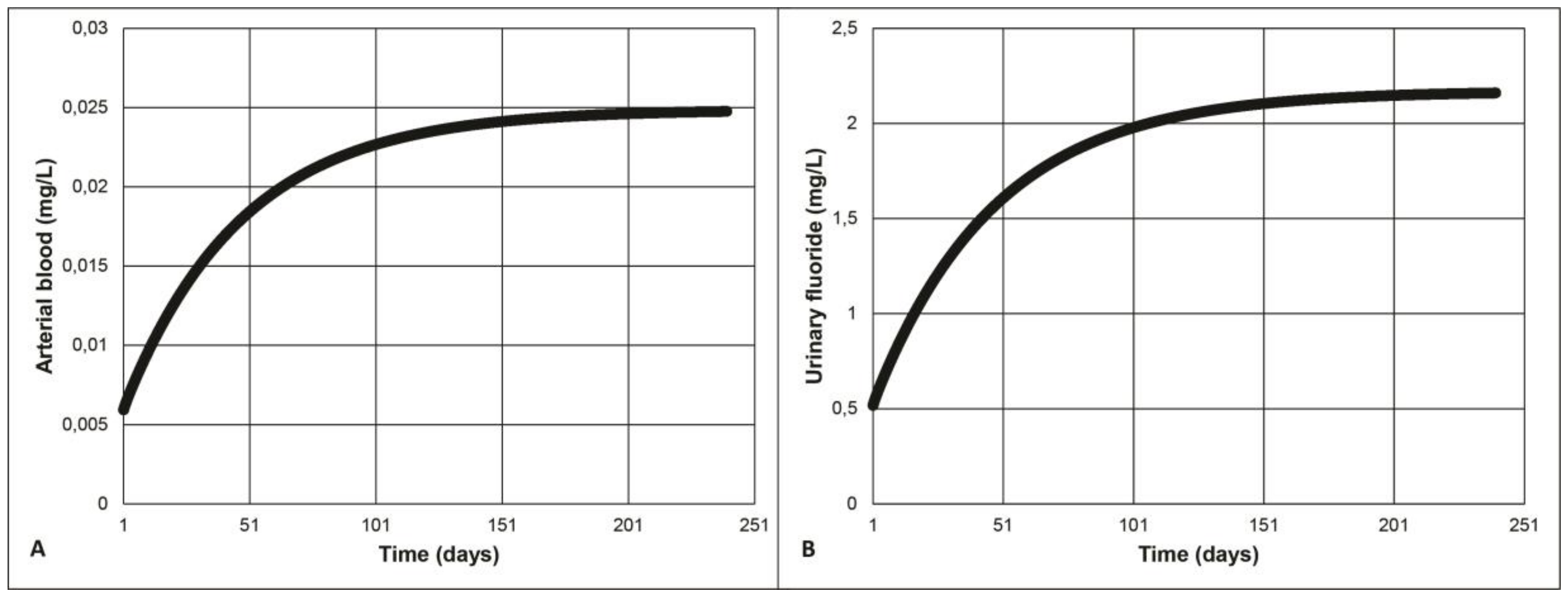
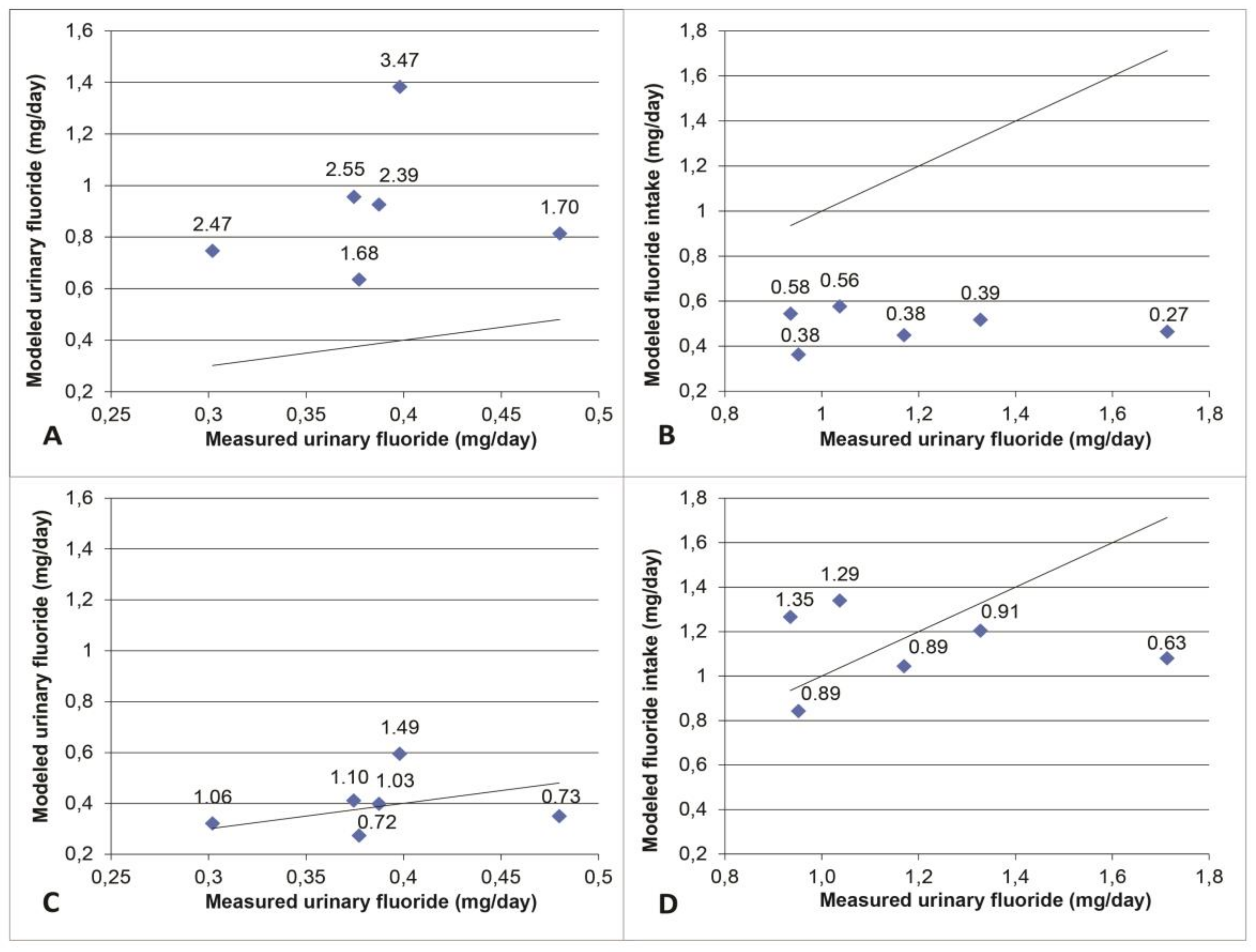
3.1. Model Validation
3.2. Modeled Exposure Scenarios
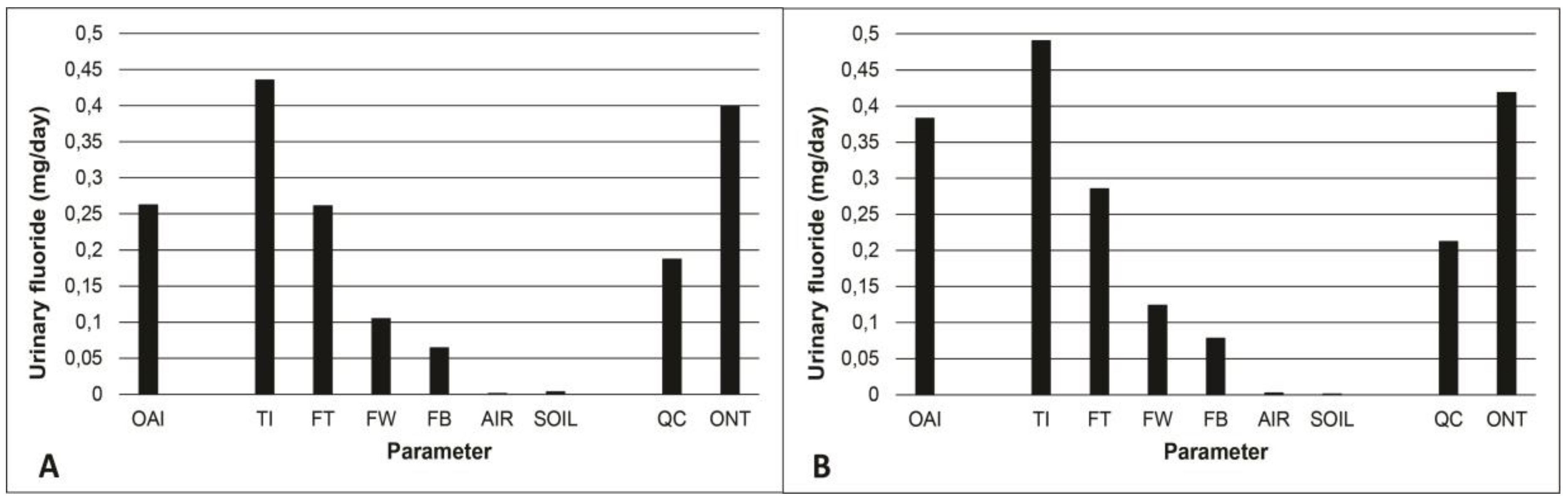
3.3. Contribution of Drinking Water to the Total Intake of Fluoride
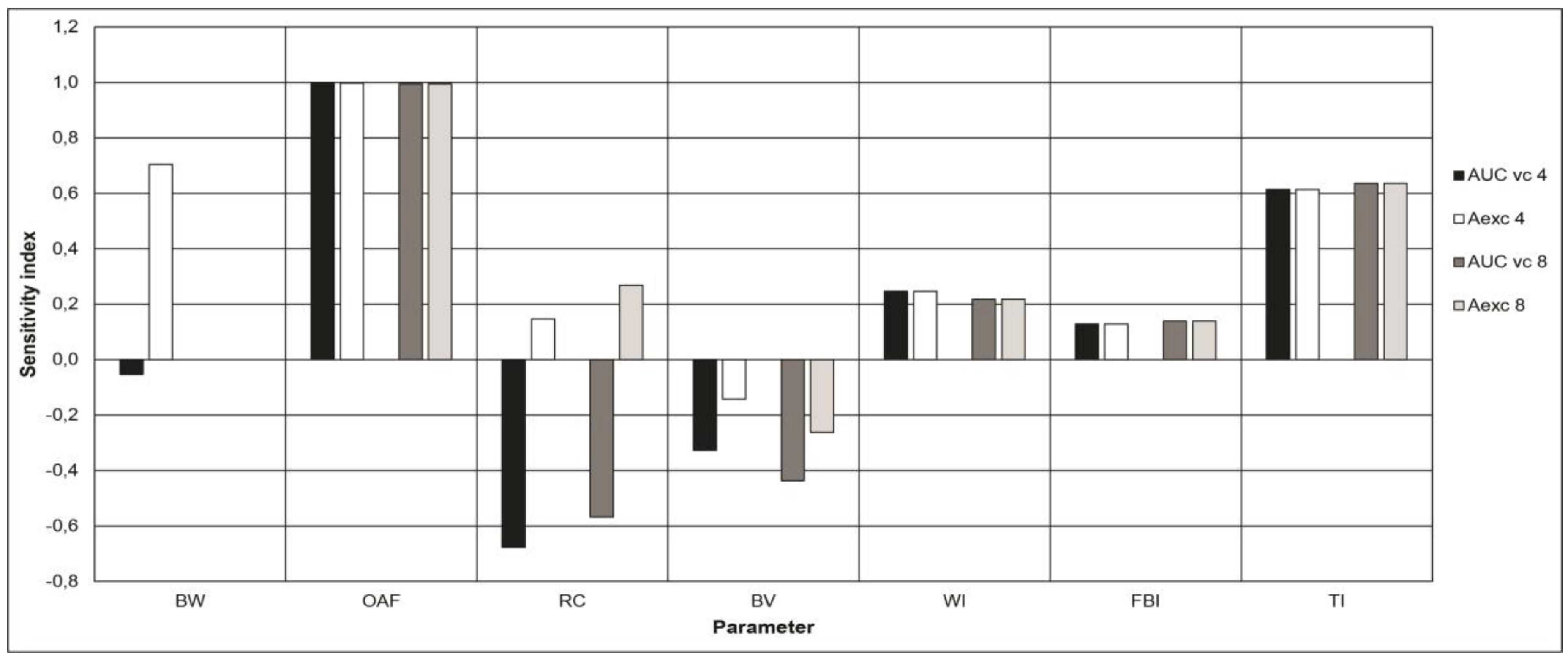
3.4. Sensitivity Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Age (year) | Weight (kg) | Water (mL/day) | Diet (mg/day) | Total (mg/day) | Intake (mg/kg/day) | Absorbed Dose (mg/kg/day) 1 |
|---|---|---|---|---|---|---|
| 1 to 3 | 8 à 16 | 0.39–0.56 | 0.027–0.265 | 0.417–0.825 | 0.026–0.103 (0.065) | 0.036–0.058 (0.047) |
| 4 to 6 | 13 à 24 | 0.52–0.745 | 0.036–0.360 | 0.556–1.105 | 0.023–0.085 (0.054) | 0.030–0.049 (0.040) |
| 7 to 9 | 16 à 35 | 0.65–0.93 | 0.045–0.450 | 0.695–1.380 | 0.020–0.086 (0.053) | 0.029–0.048 (0.039) |
| 10 to 12 | 25 à 54 | 0.81–1.66 | 0.056–0.560 | 0.866–1.725 | 0.016–0.069 (0.043) | 0.023–0.037 (0.031) |
References
- McClure, F.J. Ingestion of fluoride and dental caries: Quantitative relations based on food and water requirements of children one to twelve years old. Am. J. Dis. Child. 1943, 66, 362–369. [Google Scholar] [CrossRef]
- Galagan, D.J.; Vermillion, J.R. Determining optimum fluoride concentrations. Public Health Rep. 1957, 72, 491–493. [Google Scholar] [CrossRef] [PubMed]
- Health Canada. Fluoride and Oral Health. Available online: https://www.canada.ca/en/health-canada/services/healthy-living/your-health/environment/fluorides-human-health.html (accessed on 26 April 2018).
- Buzalaf, M.A.R.; Pessan, J.P.; Honório, H.M.; ten Cate, J.M. Mechanisms of Action of Fluoride for Caries Control. In Fluoride and the Oral Environment; Buzalaf, M.A.R., Ed.; Karger: Basel, Switzerland, 2011; Volume 22, pp. 20–36. [Google Scholar]
- Public Health Agency of Canada. The State of Community Water Fluoridation across Canada; Public Health Agency of Canada: Ottawa, ON, Canada, 2017; p. 13. Available online: https://www.canada.ca/content/dam/hc-sc/documents/services/publications/healthy-living/community-water-fluoridation-across-canada-2017/community-water-fluoridation-across-canada-2017-eng.pdf (accessed on 26 June 2018).
- Ontario Association of Public Health Dentistry. Mean dmft+DMFT and % Caries Free JK, SK and Grade 2 Children from Participating Ontario Health Units 2003 to 2014. In Wellington-Dufferin-Guelph Public Health, Oral Health Status Report; Ontario Association of Public Health Dentistry: Guelph, ON, Canada, 2015; p. 34. Available online: https://www.wdgpublichealth.ca/sites/default/files/file-attachments/report/hs_report_2015-oral-health-in-wdg-fullreport_access.pdf (accessed on 26 June 2018).
- Galarneau, C.; Arpin, S.; Boiteau, V.; Dube, M.A.; Hamel, D.; Wassef, N. Étude Clinique sur l’État de Santé Buccodentaire des Élèves Québécois du Primaire 2012–2013 (ÉCSBQ)—Rapport National, 2nd ed.; Institut National de Santé Publique du Québec: Montréal, QC, Canada, 2018; p. 181. Available online: https://www.inspq.qc.ca/sites/default/files/publications/2034_sante_buccodentaire_primaire.pdf (accessed on 6 June 2018).
- Institut National de Santé Publique du Qubec (INSPQ). Avis sur un Projet de Fluoration de l’Eau Potable; INSPQ: Montreal, QC, Canada, 2011; p. 13. Available online: https://www.inspq.qc.ca/publications/1278 (accessed on 13 February 2018).
- Health Canada; Canadian Health Measures Survey (CHMS). Oral Health Statistics 2007–2009. Available online: https://www.canada.ca/en/health-canada/services/healthy-living/reports-publications/oral-health/canadian-health-measures-survey.html (accessed on 26 April 2018).
- Canadian Institute of Health Information. National Health Expenditure Trends, 1975 to 2016; Canadian Institute for Health Information: Ottawa, ON, Canada, 2016; p. 44. Available online: https://secure.cihi.ca/free_products/NHEX-Trends-Narrative-Report_2016_EN.pdf (accessed on 13 February 2018).
- Health Canada. Guidelines for Canadian Drinking Water Quality: Guideline Technical Document—Fluoride; Minsitry of Health: Ottawa, ON, Canada, 2010; p. 104. Available online: https://www.canada.ca/content/dam/canada/health-canada/migration/healthy-canadians/publications/healthy-living-vie-saine/water-fluoride-fluorure-eau/alt/water-fluoride-fluorure-eau-eng.pdf (accessed on 13 February 2018).
- Cauley, J.A.; Murphy, P.A.; Riley, T.J.; Buhari, A.M. Effects of fluoridated drinking water on bone mass and fractures: The study of osteoporotic fractures. J. Bone Min. Res. 1995, 10, 1076–1086. [Google Scholar] [CrossRef] [PubMed]
- Karagas, M.R.; Baron, J.A.; Barrett, J.A.; Jacobsen, S.J. Patterns of fracture among the United States elderly: Geographic and fluoride effects. Ann. Epidemiol. 1996, 6, 209–216. [Google Scholar] [CrossRef]
- Leone, N.C.; Stevenson, C.A.; Hilbish, T.F.; Sosman, M.C. A roentgenologic study of a human population exposed to high-fluoride domestic water; a ten-year study. Am. J. Roentgenol. Radium Ther. Nucl. Med. 1955, 74, 874–885. [Google Scholar] [PubMed]
- Health Canada. Findings and Recommendations of the Fluoride Expert Panel (January 2007). https://www.canada.ca/en/health-canada/services/environmental-workplace-health/reports-publications/water-quality/findings-recommendations-fluoride-expert-panel-january-2007.html (accessed on 26 April 2018).
- Choi, A.L.; Sun, G.; Zhang, Y.; Grandjean, P. Developmental fluoride neurotoxicity: A systematic review and meta-analysis. Environ. Health Perspect. 2012, 120, 1362–1368. [Google Scholar] [CrossRef] [PubMed]
- Comber, H.; Deady, S.; Montgomery, E.; Gavin, A. Drinking water fluoridation and osteosarcoma incidence on the island of Ireland. Cancer Causes Control 2011, 22, 919–924. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.; Leclerc, B.S. Fluoride in drinking water and osteosarcoma incidence rates in the continental United States among children and adolescents. Cancer Epidemiol. 2012, 36, e83–e88. [Google Scholar] [CrossRef] [PubMed]
- Blakey, K.; Feltbower, R.G.; Parslow, R.C.; James, P.W.; Gómez Pozo, B.; Stiller, C.; Vincent, T.J.; Norman, P.; McKinney, P.A.; Murphy, M.F.; et al. Is fluoride a risk factor for bone cancer? Small area analysis of osteosarcoma and Ewing sarcoma diagnosed among 0–49-year-olds in Great Britain, 1980–2005. Int. J. Epidemiol. 2014, 43, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Rebhun, R.B.; Kass, P.H.; Kent, M.S.; Watson, K.D.; Withers, S.S.; Culp, W.T.N.; King, A.M. Evaluation of optimal water fluoridation on the incidence and skeletal distribution of naturally arising osteosarcoma in pet dogs. Vet. Comp. Oncol. 2017, 15, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Archer, N.P.; Napier, T.S.; Villanacci, J.F. Fluoride exposure in public drinking water and childhood and adolescent osteosarcoma in Texas. Cancer Causes Control 2016, 27, 863–868. [Google Scholar] [CrossRef] [PubMed]
- Barberio, A.M.; Hosein, F.S.; Quiñonez, C.; McLaren, L. Fluoride exposure and indicators of thyroid functioning in the Canadian population: Implications for community water fluoridation. J. Epidemiol. Community Health 2017, 71, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Rao, H.V.; Beliles, R.P.; Whitford, G.M.; Turner, C.H. A physiologically based pharmacokinetic model for fluoride uptake by bone. Regul. Toxicol. Pharmacol. 1995, 22, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Statistics Canada. Canadian Health Measures Survey (CHMS). Available online: http://www23.statcan.gc.ca/imdb/p2SV.pl?Function=getSurvey&Id=251160 (accessed on 26 April 2018).
- Institut National de Santé Publique du Québec (INSPQ). Extraction et Comparaison des Données Provinciales de Biosurveillance des Substances Chimiques de l’Environnement pour le Québec et l’Ontario Issues de l’Enquête Canadienne sur les Mesures de Santé; Unpublished Report; Institut National de Santé Publique du Québec: Montreal, QC, Canada, 2016; p. 86.
- Haddad, S.; Pelekis, M.; Krishnan, K. A methodology for solving physiologically based pharmacokinetic models without the use of simulation softwares. Toxicol. Lett. 1996, 85, 113–126. [Google Scholar] [CrossRef]
- Price, K.; Haddad, S.; Krishnan, K. Physiological modeling of age-specific changes in the pharmacokinetics of organic chemicals in children. J. Toxicol. Environ. Health A 2003, 66, 417–433. [Google Scholar] [CrossRef] [PubMed]
- Price, P.S.; Conolly, R.B.; Chaisson, C.F.; Gross, E.A.; Young, J.S.; Mathis, E.T.; Tedder, D.R. Modeling interindividual variation in physiological factors used in PBPK models of humans. Crit. Rev. Toxicol. 2003, 33, 469–503. [Google Scholar] [CrossRef] [PubMed]
- Valcke, M.; Krishnan, K. Evaluation of the impact of the exposure route on the human kinetic adjustment factor. Regul. Toxicol. Pharmacol. 2011, 59, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Haddad, S.; Tardif, G.C.; Tardif, R. Development of physiologically based toxicokinetic models for improving the human indoor exposure assessment to water contaminants: Trichloroethylene and trihalomethanes. J. Toxicol. Environ. Health A 2006, 69, 2095–2136. [Google Scholar] [CrossRef] [PubMed]
- Whitford, G.M. Fluoride metabolism and excretion in children. J. Public Health Dent. 1999, 59, 224–228. [Google Scholar] [CrossRef] [PubMed]
- Ekstrand, J.; Ziegler, E.E.; Nelson, S.E.; Fomon, S.J. Absorption and retention of dietary and supplemental fluoride by infants. Adv. Dent. Res. 1994, 8, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Trautner, K.; Siebert, G. An experimental study of bio-availability of fluoride from dietary sources in man. Arch. Oral Biol. 1986, 31, 223–228. [Google Scholar] [CrossRef]
- McIvor, M.E. Acute fluoride toxicity. Pathophysiology and management. Drug Saf. 1990, 5, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Ekstrand, J.; Ehrnebo, M. Absorption of fluoride from fluoride dentifrices. Caries Res. 1980, 14, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.K.; Kaushik, C.P.; Haritash, A.K.; Singh, B.; Raghuvanshi, S.P.; Kansal, A. Determination of exposure and probable ingestion of fluoride through tea, toothpaste, tobacco and pan masala. J. Hazard. Mater. 2007, 142, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Héctor, F.; Acevedo, A.; Margaret, P.; Anthony, V.; Rojas-Sanchez, F. Fluoride intake and urinary fluoride excretion in children attending a daycare center in Maracay, Aragua state, Venezuela. J. Dent. Oral Hyg. 2009, 1, 27–35. [Google Scholar]
- Villa, A.; Anabalon, M.; Cabezas, L. The fractional urinary fluoride excretion in young children under stable fluoride intake conditions. Community Dent. Oral Epidemiol. 2000, 28, 344–355. [Google Scholar] [CrossRef] [PubMed]
- Omid, N.; Maguire, A.; O’Hare, W.T.; Zohoori, F.V. Total daily fluoride intake and fractional urinary fluoride excretion in 4- to 6-year-old children living in a fluoridated area: Weekly variation? Community Dent. Oral Epidemiol. 2017, 45, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Haftenberger, M.; Viergutz, G.; Neumeister, V.; Hetzer, G. Total fluoride intake and urinary excretion in German children aged 3–6 years. Caries Res. 2001, 35, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Zohoori, F.V.; Walls, R.; Teasdale, L.; Landes, D.; Steen, I.N.; Moynihan, P.; Omid, N.; Maguire, A. Fractional urinary fluoride excretion of 6–7-year-old children attending schools in low-fluoride and naturally fluoridated areas in the UK. Br. J. Nutr. 2013, 109, 1903–1909. [Google Scholar] [CrossRef] [PubMed]
- Dietitians of Canada. WHO Growth Charts Set 2. Available online: https://www.dietitians.ca/Dietitians-Views/Prenatal-and-Infant/WHO-Growth-Charts/WHO-Growth-Charts-Set-2.aspx (accessed on 26 April 2018).
- Institut National de Santé Publique du Québec, Fluorures. Available online: https://www.inspq.qc.ca/eau-potable/fluorures (accessed on 26 April 2018).
- Ipsos MORI Social Research Institute. Tap Water Drinking Behaviour: A Study of Children Aged 0–15; DEFRA Drinking Water Inspectorate: London, UK, 2012; p. 84. Available online: http://dwi.defra.gov.uk/research/completed-research/reports/DWI70_2_251.pdf (accessed on 31 May 2018).
- Hays, S.M.; Becker, R.A.; Leung, H.W.; Aylward, L.L.; Pyatt, D.W. Biomonitoring equivalents: A screening approach for interpreting biomonitoring results from a public health risk perspective. Regul. Toxicol. Pharmacol. 2007, 47, 96–109. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Fourth National Report on Human Exposure to Environmental Chemicals; Centers for Disease Control and Prevention, Department of Health and Human Services: Atlanta, GA, USA, 2009; p. 530. Available online: https://www.cdc.gov/exposurereport/pdf/fourthreport.pdf (accessed on 16 April 2018).
- Umweltbundesamt (UBA). German Environmental Survey, GerES 2014–2017. Available online: https://www.umweltbundesamt.de/en/topics/health/assessing-environmentally-related-health-risks/german-environmental-surveys/german-environmental-survey-2014-2017-geres-v#textpart-1 (accessed on 31 May 2018).
- Becker, K.; Conrad, A.; Kirsch, N.; Kolossa-Gehring, M.; Schulz, C.; Seiwert, M.; Seifert, B. German Environmental Survey (GerES): Human biomonitoring as a tool to identify exposure pathways. Int. J. Hyg. Environ. Health 2007, 210, 267–269. [Google Scholar] [CrossRef] [PubMed]
- Health Canada. Fourth Report on Human Biomonitoring of Environmental Chemicals in Canada; Health Canada: Ottawa, ON, Canada, 2017; p. 247. Available online: https://www.canada.ca/content/dam/hc-sc/documents/services/environmental-workplace-health/reports-publications/environmental-contaminants/fourth-report-human-biomonitoring-environmental-chemicals-canada/fourth-report-human-biomonitoring-environmental-chemicals-canada-fra.pdf (accessed on 15 March 2018).
- Aylward, L.L.; Hays, S.M.; Vezina, A.; Deveau, M.; St-Amand, A.; Nong, A. Biomonitoring Equivalents for interpretation of urinary fluoride. Regul. Toxicol. Pharmacol. 2015, 72, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Aylward, L.L.; Kirman, C.R.; Blount, B.C.; Hays, S.M. Chemical-specific screening criteria for interpretation of biomonitoring data for volatile organic compounds (VOCs)—Application of steady-state PBPK model solutions. Regul. Toxicol. Pharmacol. 2010, 58, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.; Phillips, M.; Grulke, C.; Yoon, M.; Young, B.; McDougall, R.; Leonard, J.; Lu, J.; Lefew, W.; Tan, Y.M. Reconstructing exposures from biomarkers using exposure-pharmacokinetic modeling—A case study with carbaryl. Regul. Toxicol. Pharmacol. 2015, 73, 689–698. [Google Scholar] [CrossRef] [PubMed]
- Clewell, H.J.; Tan, Y.M.; Campbell, J.L.; Andersen, M.E. Quantitative interpretation of human biomonitoring data. Toxicol. Appl. Pharmacol. 2008, 231, 122–133. [Google Scholar] [CrossRef] [PubMed]
- Liao, K.H.; Tan, Y.M.; Clewell, H.J., 3rd. Development of a screening approach to interpret human biomonitoring data on volatile organic compounds: Reverse dosimetry on biomonitoring data for trichloroethylene. Risk Anal. 2007, 27, 1223–1236. [Google Scholar] [CrossRef] [PubMed]
- Lyons, M.A.; Yang, R.S.; Mayeno, A.N.; Reisfeld, B. Computational toxicology of chloroform: Reverse dosimetry using Bayesian inference, Markov chain Monte Carlo simulation, and human biomonitoring data. Environ. Health Perspect. 2008, 116, 1040–1046. [Google Scholar] [CrossRef] [PubMed]
- McNally, K.; Cotton, R.; Cocker, J.; Jones, K.; Bartels, M.; Rick, D.; Price, P.; Loizou, G. Reconstruction of Exposure to m-Xylene from Human Biomonitoring Data Using PBPK Modelling, Bayesian Inference, and Markov Chain Monte Carlo Simulation. J. Toxicol. 2012, 2012, 760281. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.M.; Liao, K.H.; Conolly, R.B.; Blount, B.C.; Mason, A.M.; Clewell, H.J. Use of a physiologically based pharmacokinetic model to identify exposures consistent with human biomonitoring data for chloroform. J. Toxicol. Environ. Health A 2006, 69, 1727–1756. [Google Scholar] [CrossRef] [PubMed]
- Timchalk, C.; Poet, T.S. Development of a physiologically based pharmacokinetic and pharmacodynamic model to determine dosimetry and cholinesterase inhibition for a binary mixture of chlorpyrifos and diazinon in the rat. Neurotoxicology 2008, 29, 428–443. [Google Scholar] [CrossRef] [PubMed]
- Buzalaf, M.A.R.; Whitford, G.M. Fluoride Metabolism. In Fluoride and the Oral Environment; Buzalaf, M.A.R., Ed.; Karger: Basel, Switzerland, 2011; Volume 22, pp. 20–36. [Google Scholar]
- Zohoori, F.V. Summary of General Discussion and Conclusions. Adv. Dent. Res. 2018, 29, 183–185. [Google Scholar] [CrossRef] [PubMed]
- Buzalaf, M.A.R. Review of Fluoride Intake and Appropriateness of Current Guidelines. Adv. Dent. Res. 2018, 29, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Health and Ecological Criteria Division Office of Water. Fluoride: Exposure and Relative Source Contribution Analysis; U.S. Environmental Protection Agency: Washington, DC, USA, 2010; p. 210.
- Abuhaloob, L.; Maguire, A.; Moynihan, P. Total daily fluoride intake and the relative contributions of foods, drinks and toothpaste by 3- to 4-year-old children in the Gaza Strip—Palestine. Int. J. Paediatr. Dent. 2015, 25, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Viswanathan, G.; Gopalakrishnan, S.; Siva Ilango, S. Assessment of water contribution on total fluoride intake of various age groups of people in fluoride endemic and non-endemic areas of Dindigul District, Tamil Nadu, South India. Water Res. 2010, 44, 6186–6200. [Google Scholar] [CrossRef] [PubMed]
- American Dental Association. Fluoridation Facts—2018 Edition; American Dental Association: Washington, DC, USA, 2018; p. 114. [Google Scholar]
- Institute of Medecine. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride; The National Academies Press: Washington, DC, USA, 1997; p. 432. [Google Scholar]
- Expert Working Group for Fluoride. Australian and New Zealand Nutrient Reference Values for Fluoride; Australian Government Department of Health; The New Zealand Ministry of Health: Thorndon, New Zealand, 2017; p. 97.
- Zohoori, F.V.; Maguire, A. Determining an Upper Reference Value for the Urinary Fluoride-Creatinine Ratio in Healthy Children Younger than 7 Years. Caries Res. 2017, 51, 283–289. [Google Scholar] [CrossRef] [PubMed]
| Age (years) | Children (n) | Intake (mg/day) | AuF-24 1 (mg/day) | Country | |||
|---|---|---|---|---|---|---|---|
| Diet | Toothpaste | Water | Supplement | ||||
| 4 | 31 | 0.560 | 0.706 | 0.042 | - | 0.3682 | Venezuela [37] |
| 4 | 20 | 0.533 | 0.254 | 0.231 | - | 0.358 | Chile [38] |
| 5 | 61 | 0.151 | 0.608 | 0.407 | - | 0.3705 | UK [39] |
| 5 | 11 | 0.092 | 0.274 | 0.111 | 0.455 2 | 0.476 | Germany [40] |
| 7 | 21 | 0.187 | 0.606 | 0.154 | - | 0.297 | UK [41] |
| 7 | 12 | 0.229 | 1.130 | 0.349 | - | 0.393 | UK [41] |
| Age (year) | Weight (kg) | Height (cm) | Sources of Fluorides | ||||
|---|---|---|---|---|---|---|---|
| Toothpaste (µg/kg/day) | Diet (µg/kg/day) | Soil (µg/kg/day) | Air (µg/kg/day) | Water (L/day) | |||
| 4 | 16 | 103 | 40 | 21 | 1.19 | 0.01 | 0.442 |
| 8 | 25 | 127 | 30 | 17.5 | 0.21 | 0.01 | 0.56 |
| Province | Scenario | Age-Specific Results | |||||
|---|---|---|---|---|---|---|---|
| 4 year-old Child | 8 year-old Child | ||||||
| Model | CHMS | Ratio 1 | Model | CHMS | Ratio 1 | ||
| Ontario | 1. (Fluoridation of drinking water at 0.7 mg/L) | 0.846 | 0.83 | 1.02 | 0.73 | 0.67 | 1.09 |
| Quebec | 2. (Fluoridation at 0.06 mg/L with dietary intake) | 0.708 | 0.39 | 1.81 | 0.605 | 0.34 | 1.78 |
| Quebec–modified | 3. (Fluoridation at 0.06 mg/L without dietary intake) | 0.574 | 1.47 | 0.48 | 1.41 | ||
| Age | Quebec, Geometric Mean (95th Percentile) | Ontario, Geometric Mean (95th Percentile) |
|---|---|---|
| 4 | 0.03 (0.13) | 0.06 (0.17) |
| 8 | 0.02 (0.05) | 0.05 (0.12) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jean, K.J.; Wassef, N.; Gagnon, F.; Valcke, M. A Physiologically-Based Pharmacokinetic Modeling Approach Using Biomonitoring Data in Order to Assess the Contribution of Drinking Water for the Achievement of an Optimal Fluoride Dose for Dental Health in Children. Int. J. Environ. Res. Public Health 2018, 15, 1358. https://doi.org/10.3390/ijerph15071358
Jean KJ, Wassef N, Gagnon F, Valcke M. A Physiologically-Based Pharmacokinetic Modeling Approach Using Biomonitoring Data in Order to Assess the Contribution of Drinking Water for the Achievement of an Optimal Fluoride Dose for Dental Health in Children. International Journal of Environmental Research and Public Health. 2018; 15(7):1358. https://doi.org/10.3390/ijerph15071358
Chicago/Turabian StyleJean, Keven J., Nancy Wassef, Fabien Gagnon, and Mathieu Valcke. 2018. "A Physiologically-Based Pharmacokinetic Modeling Approach Using Biomonitoring Data in Order to Assess the Contribution of Drinking Water for the Achievement of an Optimal Fluoride Dose for Dental Health in Children" International Journal of Environmental Research and Public Health 15, no. 7: 1358. https://doi.org/10.3390/ijerph15071358
APA StyleJean, K. J., Wassef, N., Gagnon, F., & Valcke, M. (2018). A Physiologically-Based Pharmacokinetic Modeling Approach Using Biomonitoring Data in Order to Assess the Contribution of Drinking Water for the Achievement of an Optimal Fluoride Dose for Dental Health in Children. International Journal of Environmental Research and Public Health, 15(7), 1358. https://doi.org/10.3390/ijerph15071358




