Lessons and Guidance from the Special Issue on Electronic Cigarette Use and Public Health
1. Introduction
- Degree of dependence, which is heavily influenced by nicotine receptor alleles [6], as well as age of smoking initiation [7]. Conventional smoking cessation aides such as Nicotine Replacement Therapy (NRT) and varenicline target these receptors. Some highly addicted people cannot imagine life without nicotine, even if they use relatively little, but they can use e-cigs instead of smoking.
- The rate of nicotine clearance, which is determined mostly by hepatic cytochrome P450 2A6 (CYP2A6) [8]. CYP2A6 phenotypes range from completely inactive (e.g., persons homozygous for deletion mutations) to hyperactive. The slowest metabolizers probably never smoke more than 7 cigarettes per day (CPD), may respond well to NRT [9], and might even feel intoxicated on high-dose NRT (e.g., 21 mg patches, clinical observation). Normal metabolizers are likely to smoke 10–20 CPD, while very rapid metabolizers smoke more, and may even wake from sleep specifically to smoke (clinical observation). There are no conventional drugs that target CYP2A6, although nicotyrine, found in cigarettes and aging or steeped e-liquids [10], is an irreversible inhibitor of the enzyme and might explain commonly observed changes in dependency measures when smokers switch to e-cigs [11].
- Benefits of nicotine use, which include mood modulation, mental focus, weight control, pain management, and social benefits [12,13,14,15,16,17]. For some of these purposes, chemical alternatives (or complements) to nicotine include caffeine, alcohol, cannabinoids, hallucinogens, benzodiazepines, cocaine, and opiates. Amongst these alternatives, nicotine and caffeine are by far the least damaging to cognition.
- Harms of nicotine use, which depend mostly on the delivery system. Nicotine is associated with minimal health harm for adults at the serum levels that people voluntarily maintain. Children and adolescents are probably vulnerable to central nervous system effects from nicotine (not just smoke) exposure [7], although most of the evidence comes from animal experimental studies or smoking rather than nicotine intake through a cleaner product [18]. Harms of delivery systems are highly variable. Tobacco cigarettes have long been recognized as one of the most dangerous product designs possible. Another nicotine-containing product, snus, is associated with substantially lower harm compared to smoking and this has long been established through clinical epidemiological evidence [19,20]. Although e-cigs are likely to be safer than smoking, there may be some variation in risk between e-liquids and amongst devices. Dual use of cigarettes and e-cigs is common. Although studies have shown similar levels of toxin exposure from dual use as from smoking [21], dual use is often a step toward smoking cessation [22,23], and long-term dual users who substantially decrease cigarette consumption probably reduce their health risks.
2. The Special Issue
2.1. Aerosol Chemistry
2.2. Adolescent Use
2.3. Vape Shops
2.4. Switching and Smoking Cessation
2.5. Consumer Issues
2.6. Policy Issues
3. Discussion
3.1. What We Learned
3.2. What We Want to Learn
Conflicts of Interest
References
- Farsalinos, K.E.; Polosa, R. Safety evaluation and risk assessment of electronic cigarettes as tobacco cigarette substitutes: A systematic review. Ther. Adv. Drug Saf. 2014, 5, 67–86. [Google Scholar] [CrossRef] [PubMed]
- Sumner, W. Permissive nicotine regulation as a complement to traditional tobacco control. BMC Public Health 2005, 5, 18. [Google Scholar] [CrossRef] [PubMed]
- Smoking Advisory Committee. Smoking and Health: Report of the Smoking Advisory Committee to the Surgeon General of the Public Health Service; Public Health Service: Washington, DC, USA, 1964.
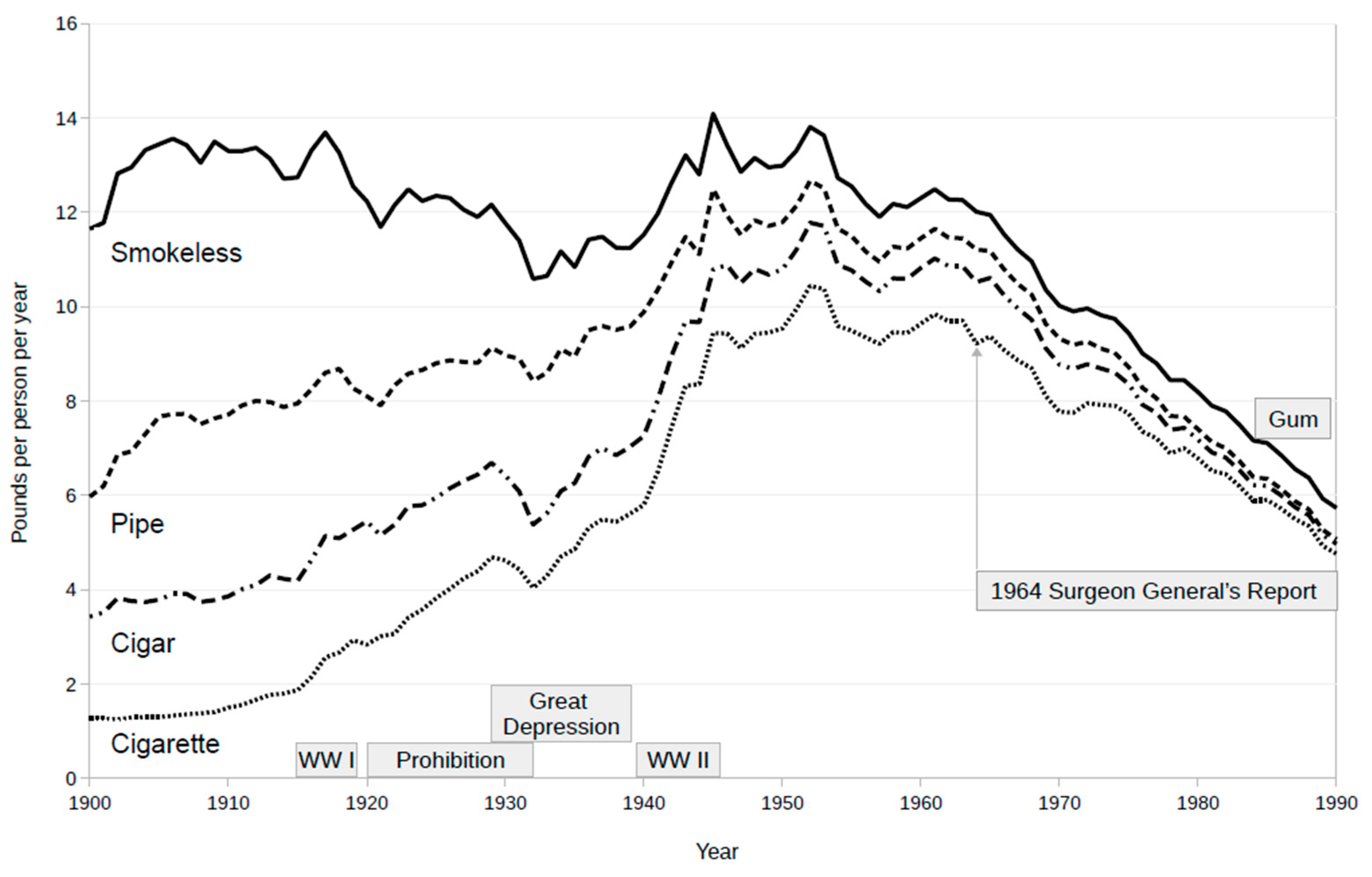
- Psoter, W.J.; Morse, D.E. Annual per capita apparent consumption of tobacco products in the United States: 1900–1990. Prev. Med. 2001, 32, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Sung, H.Y.; Keeler, T.E.; Marciniak, M. Cigarette consumption and sales of nicotine replacement products. Tob. Control 2000, 9, II60–II63. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Yang, Z.; Cui, W.; Li, M.D. Crucial roles of the CHRNB3-CHRNA6 gene cluster on chromosome 8 in nicotine dependence: Update and subjects for future research. Transl. Psychiatry 2016, 6, e843. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services. The Health Consequences of Smoking–50 Years of Progress; A Report of the Surgeon General; U.S. Government Printing Office: Rockvllle, MD, USA, 2014; pp. 121–123.
- Tyndale, R.F.; Sellers, E.M. Genetic variation in CYP2A6-mediated nicotine metabolism alters smoking behavior. Ther. Drug Monit. 2002, 24, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Schuit, E.; Panagiotou, O.A.; Munafo, M.R.; Bennett, D.A.; Bergen, A.W.; David, S.P. Pharmacotherapy for smoking cessation: Effects by subgroup defined by genetically informed biomarkers. Cochrane Database Syst. Rev. 2017, 9, CD011823. [Google Scholar] [CrossRef] [PubMed]
- Martinez, R.E.; Dhawan, S.; Sumner, W.; Williams, B.J. On-Line Chemical Composition Analysis of Refillable Electronic Cigarette Aerosol-Measurement of Nicotine and Nicotyrine. Nicotine Tob. Res. 2015, 17, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- Abramovitz, A.; McQueen, A.; Martinez, R.E.; Williams, B.J.; Sumner, W. Electronic cigarettes: The nicotyrine hypothesis. Med. Hypotheses 2015, 85, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Warburton, D.M. Nicotine and the smoker. Rev. Environ. Health 1985, 5, 343–390. [Google Scholar] [PubMed]
- Klesges, R.C.; Meyers, A.W.; Klesges, L.M.; La Vasque, M.E. Smoking, body weight, and their effects on smoking behavior: A comprehensive review of the literature. Psychol. Bull. 1989, 106, 204–230. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.G.; Covington, C.J. Predictors of cigarette smoking among adolescents. Psychol. Rep. 1997, 80, 481–482. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.R. Why does smoking so often produce dependence? A somewhat different view. Tob. Control 2001, 10, 62–64. [Google Scholar] [CrossRef] [PubMed]
- Kumari, V.; Postma, P. Nicotine use in schizophrenia: The self medication hypotheses. Neurosci. Biobehav. Rev. 2005, 29, 1021–1034. [Google Scholar] [CrossRef] [PubMed]
- LaRowe, L.R.; Langdon, K.J.; Zvolensky, M.J.; Zale, E.L.; Ditre, J.W. Pain-related anxiety as a predictor of early lapse and relapse to cigarette smoking. Exp. Clin. Psychopharmacol. 2017, 25, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Polosa, R.; Russell, C.; Nitzkin, J.; Farsalinos, K.E. A critique of the U.S. Surgeon General’s conclusions regarding e-cigarette use among youth and young adults in the United States of America. Harm. Reduct. J. 2017, 14, 61. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.N.; Hamling, J. Systematic review of the relation between smokeless tobacco and cancer in Europe and North America. BMC Med. 2009, 7, 36. [Google Scholar] [CrossRef] [PubMed]
- Hansson, J.; Galanti, M.R.; Hergens, M.P.; Fredlund, P.; Ahlbom, A.; Alfredsson, L.; Bellocco, R.; Eriksson, M.; Hallqvist, J.; Hedblad, B.; et al. Use of Snus and acute myocardial infarction: Pooled analysis of eight prospective observational studies. Eur. J. Epidemiol. 2012, 27, 771–779. [Google Scholar] [CrossRef] [PubMed]
- Shahab, L.; Goniewicz, M.L.; Blount, B.C.; Brown, J.; McNeill, A.; Alwis, K.U.; Feng, J.; Wang, L.; West, R. Nicotine, Carcinogen, and Toxin Exposure in Long-Term E-Cigarette and Nicotine Replacement Therapy Users: A Cross-sectional Study. Ann. Intern. Med. 2017, 166, 390–400. [Google Scholar] [CrossRef] [PubMed]
- Etter, J.F.; Bullen, C. A longitudinal study of electronic cigarette users. Addict. Behav. 2014, 39, 491–494. [Google Scholar] [CrossRef] [PubMed]
- Etter, J.F. Electronic cigarette: A longitudinal study of regular vapers. Nicotine Tob. Res. 2017. [Google Scholar] [CrossRef] [PubMed]
- Soulet, S.; Pairaud, C.; Lalo, H. A Novel Vaping Machine Dedicated to Fully Controlling the Generation of E-Cigarette Emissions. Int. J. Environ. Res. Public Health 2017, 14, 1225. [Google Scholar] [CrossRef] [PubMed]
- Eddingsaas, N.; Pagano, T.; Cummings, C.; Rahman, I.; Robinson, R.; Hensel, E. Qualitative Analysis of E-Liquid Emissions as a Function of Flavor Additives Using Two Aerosol Capture Methods. Int. J. Environ. Res. Public Health 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Kinnunen, J.M.; Ollila, H.; Minkkinen, J.; Lindfors, P.L.; Rimpela, A.H. A Longitudinal Study of Predictors for Adolescent Electronic Cigarette Experimentation and Comparison with Conventional Smoking. Int. J. Environ. Res. Public Health 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.S.; Hall, M.G.; Parada, H.; Peebles, K.; Brodar, K.E.; Brewer, N.T. Symptoms during Adolescents’ First Use of Cigarettes and E-Cigarettes: A Pilot Study. Int. J. Environ. Res. Public Health 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Mantey, D.S.; Harrell, M.B.; Case, K.; Crook, B.; Kelder, S.H.; Perry, C.L. Subjective experiences at first use of cigarette, e-cigarettes, hookah, and cigar products among Texas adolescents. Drug Alcohol Depend. 2017, 173, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Bauld, L.; MacKintosh, A.M.; Eastwood, B.; Ford, A.; Moore, G.; Dockrell, M.; Arnott, D.; Cheeseman, H.; McNeill, A. Young People’s Use of E-Cigarettes across the United Kingdom: Findings from Five Surveys 2015-2017. Int. J. Environ. Res. Public Health 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Kinouani, S.; Pereira, E.; Tzourio, C. Electronic Cigarette Use in Students and Its Relation with Tobacco-Smoking: A Cross-Sectional Analysis of the i-Share Study. Int. J. Environ. Res. Public Health 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Ward, E.; Cox, S.; Dawkins, L.; Jakes, S.; Holland, R.; Notley, C. A Qualitative Exploration of the Role of Vape Shop Environments in Supporting Smoking Abstinence. Int. J. Environ. Res. Public Health 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Escobedo, P.; Garcia, R.; Cruz, T.B.; Unger, J.B.; Baezconde-Garbanati, L.; Meza, L.; Sussman, S. A Descriptive Longitudinal Study of Changes in Vape Shop Characteristics and Store Policies in Anticipation of the 2016 FDA Regulations of Tobacco Products, Including E-Cigarettes. Int. J. Environ. Res. Public Health 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Truman, P.; Glover, M.; Fraser, T. An Online Survey of New Zealand Vapers. Int. J. Environ. Res. Public Health 2018, 15. [Google Scholar] [CrossRef] [PubMed]
- Adriaens, K.; Van Gucht, D.; Baeyens, F. Differences between Dual Users and Switchers Center around Vaping Behavior and Its Experiences Rather than Beliefs and Attitudes. Int. J. Environ. Res. Public Health 2017, 15. [Google Scholar] [CrossRef] [PubMed]
- Rodu, B.; Plurphanswat, N. Quit Methods Used by American Smokers, 2013–2014. Int. J. Environ. Res. Public Health 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Sears, C.G.; Hart, J.L.; Walker, K.L.; Robertson, R.M. Generally Recognized as Safe: Uncertainty Surrounding E-Cigarette Flavoring Safety. Int. J. Environ. Res. Public Health 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Gucht, D.V.; Adriaens, K.; Baeyens, F. Online Vape Shop Customers Who Use E-Cigarettes Report Abstinence from Smoking and Improved Quality of Life, But a Substantial Minority Still Have Vaping-Related Health Concerns. Int. J. Environ. Res. Public Health 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- McKeganey, N.; Dickson, T. Why Don’t More Smokers Switch to Using E-Cigarettes: The Views of Confirmed Smokers. Int. J. Environ. Res. Public Health 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Levy, D.T.; Yuan, Z.; Li, Y. The Prevalence and Characteristics of E-Cigarette Users in the U.S. Int. J. Environ. Res. Public Health 2017, 14. [Google Scholar] [CrossRef]
- McQueen, A.; Tower, S.; Sumner, W. Interviews with “vapers”: Implications for future research with electronic cigarettes. Nicotine Tob. Res. 2011, 13, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Sumner, H.M.; McQueen, A.; Scott, M.J.; Sumner, W. Analysis of Comments in a Petition Defending Electronic Cigarettes. Nicotine Tob. Res. 2014, 16, 1503–1511. [Google Scholar] [CrossRef] [PubMed]
- Foulds, J.; Veldheer, S.; Yingst, J.; Hrabovsky, S.; Wilson, S.J.; Nichols, T.T.; Eissenberg, T. Development of a Questionnaire for Assessing Dependence on Electronic Cigarettes Among a Large Sample of Ex-Smoking E-cigarette Users. Nicotine Tob. Res. 2015, 17, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Hartmann-Boyce, J.; McRobbie, H.; Bullen, C.; Begh, R.; Stead, L.F.; Hajek, P. Electronic cigarettes for smoking cessation. Cochrane Database Syst. Rev. 2016, 9, CD010216. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K. Electronic cigarettes: An aid in smoking cessation, or a new health hazard? Ther. Adv. Respir. Dis. 2018, 12, 1753465817744960. [Google Scholar] [CrossRef] [PubMed]
- Amato, M.S.; Boyle, R.G.; Levy, D. How to define e-cigarette prevalence? Finding clues in the use frequency distribution. Tob. Control 2016, 25, e24–e29. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sumner, W.; Farsalinos, K. Lessons and Guidance from the Special Issue on Electronic Cigarette Use and Public Health. Int. J. Environ. Res. Public Health 2018, 15, 1338. https://doi.org/10.3390/ijerph15071338
Sumner W, Farsalinos K. Lessons and Guidance from the Special Issue on Electronic Cigarette Use and Public Health. International Journal of Environmental Research and Public Health. 2018; 15(7):1338. https://doi.org/10.3390/ijerph15071338
Chicago/Turabian StyleSumner, Walton, and Konstantinos Farsalinos. 2018. "Lessons and Guidance from the Special Issue on Electronic Cigarette Use and Public Health" International Journal of Environmental Research and Public Health 15, no. 7: 1338. https://doi.org/10.3390/ijerph15071338
APA StyleSumner, W., & Farsalinos, K. (2018). Lessons and Guidance from the Special Issue on Electronic Cigarette Use and Public Health. International Journal of Environmental Research and Public Health, 15(7), 1338. https://doi.org/10.3390/ijerph15071338





