Daily Intake Estimation for Young Children’s Ingestion of Residential Dust and Soils Contaminated with Chlorpyrifos and Cypermethrin in Taiwan
Abstract
1. Introduction
2. Materials and Methods
2.1. Analysis of Insecticides in Dust or Soils
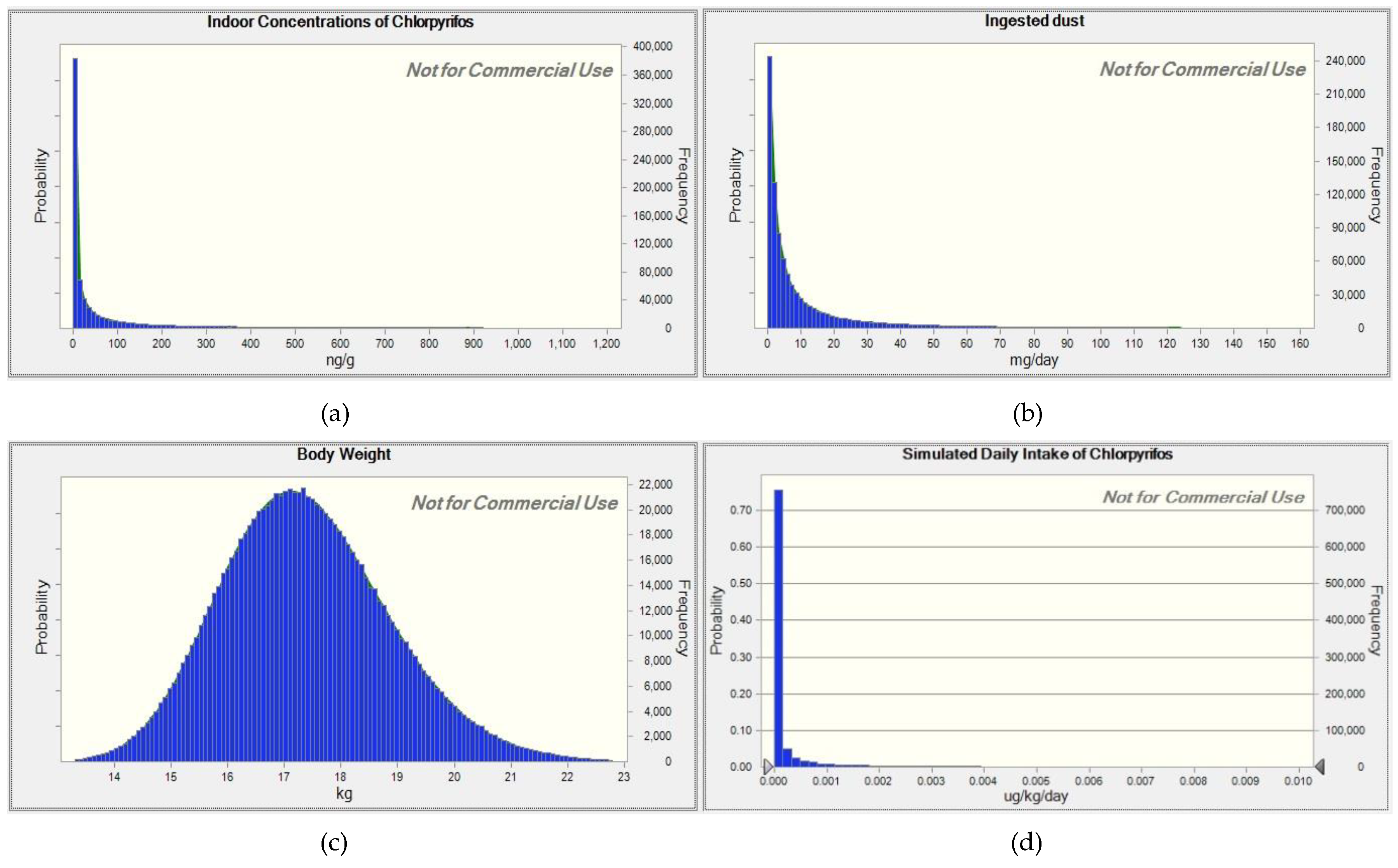
2.2. Estimation of Daily Intake
3. Results
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Environmental Sustainability Index, Benchmarking National Environmental Stewardship. 2005. Available online: http://archive.epi.yale.edu/files/2005_esi_summary_for_policymakers.pdf (accessed on 14 May 2018).
- Hung, C.C.; Huang, F.J.; Yang, Y.Q.; Hsieh, C.J.; Tseng, C.C.; Yiin, L.M. Pesticides in indoor and outdoor residential dust: A pilot study in a rural county of Taiwan. Environ. Sci. Pollut. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Gurunathan, S.; Robson, M.; Freeman, N.; Buckley, B.; Roy, A.; Meyer, R.; Bukowski, J.; Lioy, P.J. Accumulation of chlorpyrifos on residential surfaces and toys accessible to children. Environ. Health Perspect. 1998, 106, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Andersen, H.R.; Debes, F.; Wohlfahrt-Veje, C.; Murata, K.; Grandjean, P. Occupational pesticide exposure in early pregnancy associated with sex-specific neurobehavioral deficits in the children at school age. Neurotoxicol. Teratol. 2015, 47, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, M.F.; Chevrier, J.; Harley, K.G.; Kogut, K.; Vedar, M.; Calderon, N.; Trujillo, C.; Johnson, C.; Bradman, A.; Barr, D.B.; et al. Prenatal exposure to organophosphate pesticides and IQ in 7-year-old children. Environ. Health Perspect. 2011, 119, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; An, S.; Rauch, S.A.; Coker, E.S.; Maphula, A.; Obida, M.; Crause, M.; Kogut, K.R.; Bornman, R.; Chevrier, J. Prenatal exposure to DDT and pyrethroids for malaria control and child neurodevelopment: The VHEMBE cohort, South Africa. Environ. Health Perspect. 2018, 126, 047004. [Google Scholar] [CrossRef] [PubMed]
- Eskenazi, B.; Marks, A.R.; Bradman, A.; Harley, K.; Bart, D.B.; Johnson, C.; Morga, N.; Jewell, N.P. Organophosphate pesticide exposure and neurodevelopment in young Mexican-American children. Environ. Health Perspect. 2007, 115, 792–798. [Google Scholar] [CrossRef] [PubMed]
- Gunier, R.B.; Bradman, A.; Harley, K.G.; Kogut, K.; Eskenazi, B. Prenatal Residential Proximity to Agricultural Pesticide Use and IQ in 7-Year-Old Children. Environ. Health Perspect. 2017, 125, 057002. [Google Scholar] [CrossRef] [PubMed]
- Marks, A.R.; Harley, K.; Bradman, A.; Kogut, K.; Barr, D.B.; Johnson, C.; Calderon, N.; Eskenazi, B. Organophosphate pesticide exposure and attention in young Mexican-American children: The CHAMACOS study. Environ. Health Perspect. 2010, 118, 1768–1774. [Google Scholar] [CrossRef] [PubMed]
- Harari, R.; Julvez, J.; Murata, K.; Barr, D.; Debes, F.; Bellinger, D.C.; Grandjean, P. Neurobehavioral deficits and increased blood pressure in school-age children prenatally exposed to pesticides. Environ. Health Perspect. 2010, 118, 890–896. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.R.; Karr, C.J. Pesticide exposure in children. Pediatrics 2012, 130, e1765–e1788. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Quezada, M.T.; Lucero, B.A.; Barr, D.B.; Steenland, K.; Levy, K.; Ryan, P.B.; Iglesias, V.; Alvarado, S.; Concha, C.; Rojas, E. Neurodevelopmental effects in children associated with exposure to organophosphate pesticides: A systematic review. Neurotoxicology 2013, 39, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Fluegge, K.R.; Nishioka, M.; Wilkins, J.R., 3rd. Effects of simultaneous prenatal exposures to organophosphate and synthetic pyrethroid insecticides on infant neurodevelopment at three months of age. J. Environ. Toxiol. Public Health 2016, 1, 60–73. [Google Scholar]
- Furlong, M.A.; Barr, D.B.; Wolff, M.S.; Engel, S.M. Prenatal exposure to pyrethroid pesticides and childhood behavior and executive functioning. Neurotoxicology 2017, 62, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Viel, J.F.; Rouget, F.; Warembourg, C.; Monfort, C.; Limon, G.; Cordier, S.; Chevrier, C. Behavioural disorders in 6-year-old children and pyrethroid insecticide exposure: The PELAGIE mother-child cohort. Occup. Environ. Med. 2017, 74, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Viel, J.F.; Warembourg, C.; Le Maner-Idrissi, G.; Lacroix, A.; Limon, G.; Rouget, F.; Monfort, C.; Durand, G.; Cordier, S.; Chevrier, C. Pyrethroid insecticide exposure and cognitive developmental disabilities in children: The PELAGIE mother-child cohort. Environ. Int. 2015, 82, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Van Wendel de Joode, B.; Mora, A.M.; Lindh, C.H.; Hernandez-Bonilla, D.; Cordoba, L.; Wesseling, C.; Hoppin, J.A.; Mergler, D. Pesticide exposure and neurodevelopment in children aged 6–9 years from Talamanca, Costa Rica. Cortex 2016, 85, 137–150. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.J.; Du, J.C.; Chiou, H.C.; Chung, M.Y.; Yang, W.; Chen, Y.S.; Fuh, M.R.; Chien, L.C.; Hwang, B.; Chen, M.L. Increased risk of attention-deficit/hyperactivity disorder associated with exposure to organophosphate pesticide in Taiwanese children. Andrology 2016, 4, 695–705. [Google Scholar] [CrossRef] [PubMed]
- Zartarian, V.G.; Ozkaynak, H.; Burke, J.M.; Zufall, M.J.; Rigas, M.L.; Furtaw, E.J., Jr. A modeling framework for estimating children’s residential exposure and dose to chlorpyrifos via dermal residue contact and nondietary ingestion. Environ. Health Perspect. 2000, 108, 505–514. [Google Scholar] [CrossRef] [PubMed]
- USEPA. Stochastic Human Exposure and Dose Simulation (SHEDS) to Estimate Human Exposure to Chemicals. Available online: https://www.epa.gov/chemical-research/stochastic-human-exposure-and-dose-simulation-sheds-estimate-human-exposure (accessed on 23 April 2018).
- Özkaynak, H.; Xue, J.; Zartarian, V.G.; Glen, G.; Smith, L. Modeled estimates of soil and dust ingestion rates for children. Risk Anal. 2011, 31, 592–608. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Chang, M.-H. New growth charts for Taiwanese children and adolescents based on World Health Organization standards and health-related physical fitness. Pediatr. Neonatol. 2010, 51, 69–79. [Google Scholar] [CrossRef]
- Yuan, Y.; Chen, C.; Zheng, C.; Wang, X.; Yang, G.; Wang, Q.; Zhang, Z. Residue of chlorpyrifos and cypermethrin in vegetables and probabilistic exposure assessment for consumers in Zhejiang Province, China. Food Control 2014, 36, 63–68. [Google Scholar] [CrossRef]
- FAO/WHO. Pesticide Residues in Food 2006, Joint FAO/WHO Meeting on Pesticide Residues. Available online: http://www.fao.org/fileadmin/templates/agphome/documents/Pests_Pesticides/JMPR/JMPRrepor2006.pdf (accessed on 30 April 2018).
- Xue, J.; Zartarian, V.; Tornero-Velez, R.; Tulve, N.S. EPA’s SHEDS-multimedia model: Children’s cumulative pyrethroid exposure estimates and evaluation against NHANES biomarker data. Environ. Int. 2014, 73, 304–311. [Google Scholar] [CrossRef] [PubMed]
| Variable | N | Mean | SD | P5 | P25 | P50 | P75 | P95 | Maximum | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ingestion rate of dust (mg/day) a | 1000 | 26.65 | 36.54 | 0.66 | 4.06 | 10.80 | 28.72 | 100.97 | 901.96 | ||||
| Ingestion rate of soils (mg/day) a | 1000 | 40.96 | 78.29 | 0.15 | 5.26 | 15.34 | 44.85 | 175.60 | 1367.37 | ||||
| Indoor concentration of chlorpyrifos in dust (µg/g) b | 52 | 2.32 | 15.41 | <LOD | 0.08 | 0.11 | 0.30 | 0.91 | 112.34 | ||||
| Outdoor concentration of chlorpyrifos in soils (µg/g) b | 57 | 4.27 | 23.10 | <LOD | <LOD | <LOD | 0.11 | 16.19 | 134.60 | ||||
| Indoor concentration of cypermethrin in dust (µg/g) b | 52 | 18.33 | 55.92 | <LOD | 0.11 | 0.37 | 0.83 | 105.79 | 343.27 | ||||
| Outdoor concentration of cypermethrin in soils(µg/g) b | 57 | 4.29 | 22.32 | <LOD | <LOD | <LOD | 0.11 | 16.47 | 134.20 | ||||
| Variable | P3 | P15 | P25 | P50 | P75 | P85 | P97 | ||||||
| Body weight of 3–6 year old children at (kg) c | 13.5 | 15.1 | 15.8 | 17.3 | 19.0 | 20.1 | 22.9 | ||||||
| Percentile | Chlorpyrifos via Indoor Dust | Chlorpyrifos via Outdoor Soils | Cypermethrin via Indoor Dust | Cypermethrin via Outdoor Soils |
|---|---|---|---|---|
| P50 | <0.001% (<0.001%, <0.001%) | <0.001% (<0.001%, <0.001%) | <0.001% (<0.001%, <0.001%) | <0.001% (<0.001%, <0.001%) |
| P75 | 0.002% (0.002%, 0.002%) | 0.001% (0.001%, 0.001%) | 0.004% (0.004%, 0.004%) | <0.001% (<0.001%, <0.001%) |
| P90 | 0.019% (0.019%, 0.019%) | 0.007% (0.007%, 0.007%) | 0.052% (0.052%, 0.053%) | 0.005% (0.005%, 0.005%) |
| P95 | 0.079% (0.079%, 0.079%) | 0.029% (0.029%, 0.029%) | 0.208% (0.207%, 0.209%) | 0.023% (0.023%, 0.023%) |
| P97.5 | 0.254% (0.253%, 0.255%) | 0.102% (0.101%, 0.102%) | 0.629% (0.627%, 0.631%) | 0.080% (0.079%, 0.080%) |
| P99 | 0.884% (0.880%, 0.887%) | 0.412% (0.410%, 0.414%) | 2.042% (2.035%, 2.049%) | 0.310% (0.308%, 0.311%) |
| P99.9 | 8.313% (8.234%, 8.392%) | 4.819% (4.782%, 4.856%) | 16.89% (16.75%, 17.04%) | 3.155% (3.124%, 3.186%) |
| Percentile | Chlorpyrifos via Non-Dietary Ingestion | Chlorpyrifos via Dietary Ingestion a | Cypermethrin via Non-Dietary Ingestion | Cypermethrin via Dietary Ingestion a |
|---|---|---|---|---|
| P50 | <0.001% (<0.001%, <0.001%) | 1.39% (1.35%, 1.42%) | <0.001% (<0.001%, <0.001%) | 1.67% (1.64%, 1.70%) |
| P75 | 0.003% (0.003%, 0.003%) | NA | 0.004% (0.004%, 0.004%) | NA |
| P90 | 0.026% (0.026%, 0.026%) | 15.52% (15.35%, 15.70%) | 0.057% (0.057%, 0.058%) | 10.55% (10.44%, 10.67%) |
| P95 | 0.108% (0.108%, 0.108%) | NA | 0.231% (0.230%, 0.232%) | NA |
| P97.5 | 0.356% (0.354%, 0.357%) | 24.07% (23.69%, 24.47%) | 0.709% (0.706%, 0.711%) | 15.94% (15.68%, 16.19%) |
| P99 | 1.296% (1.290%, 1.301%) | 29.03% (28.43%, 29.66%) | 2.352% (2.343%, 2.360%) | 19.09% (18.70%, 19.52%) |
| P99.9 | 13.10% (13.02%, 13.25%) | 40.16% (38.39%, 42.41%) | 20.05% (19.87%, 20.23%) | 26.07% (24.87%, 27.42%) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.-Q.; Yiin, L.-M. Daily Intake Estimation for Young Children’s Ingestion of Residential Dust and Soils Contaminated with Chlorpyrifos and Cypermethrin in Taiwan. Int. J. Environ. Res. Public Health 2018, 15, 1327. https://doi.org/10.3390/ijerph15071327
Yang Y-Q, Yiin L-M. Daily Intake Estimation for Young Children’s Ingestion of Residential Dust and Soils Contaminated with Chlorpyrifos and Cypermethrin in Taiwan. International Journal of Environmental Research and Public Health. 2018; 15(7):1327. https://doi.org/10.3390/ijerph15071327
Chicago/Turabian StyleYang, Ya-Qing, and Lih-Ming Yiin. 2018. "Daily Intake Estimation for Young Children’s Ingestion of Residential Dust and Soils Contaminated with Chlorpyrifos and Cypermethrin in Taiwan" International Journal of Environmental Research and Public Health 15, no. 7: 1327. https://doi.org/10.3390/ijerph15071327
APA StyleYang, Y.-Q., & Yiin, L.-M. (2018). Daily Intake Estimation for Young Children’s Ingestion of Residential Dust and Soils Contaminated with Chlorpyrifos and Cypermethrin in Taiwan. International Journal of Environmental Research and Public Health, 15(7), 1327. https://doi.org/10.3390/ijerph15071327






