Influence of Ammonium Ions, Organic Load and Flow Rate on the UV/Chlorine AOP Applied to Effluent of a Wastewater Treatment Plant at Pilot Scale
Abstract
:1. Introduction
2. Materials and Methods
2.1. Electrical Energy Consumption
2.2. Overview of Experiments
- Exp. A: Variation of NH4+-N concentration (0.5, 1.0, 1.5 mg/L) in spiked tap water (6.9 mg/L dosed free Cl2, 0.4 kW UV power, 1 m3/h flow rate)
- Exp. B: Variation of WWTE dilutions with spiked tap water (6.9 mg/L dosed free Cl2, 0.4 kW UV power, 1 m3/h flow rate)
- Exp. C: Variation of CC concentration (1–5 mg/L CC in UV chamber influent) on WWTE (0.0 and 0.4 kW UV power, 1 m3/h flow rate)
- Exp. D: Variation of flow rate (1, 2, 3 m3/h WWTE) at 3 mg/L FAC dosage in UV chamber influent and 0.4 kW UV power (0.13, 0.20, 0.40 kWh/m3 electrical energy consumption)
2.3. Chemicals and Reagents
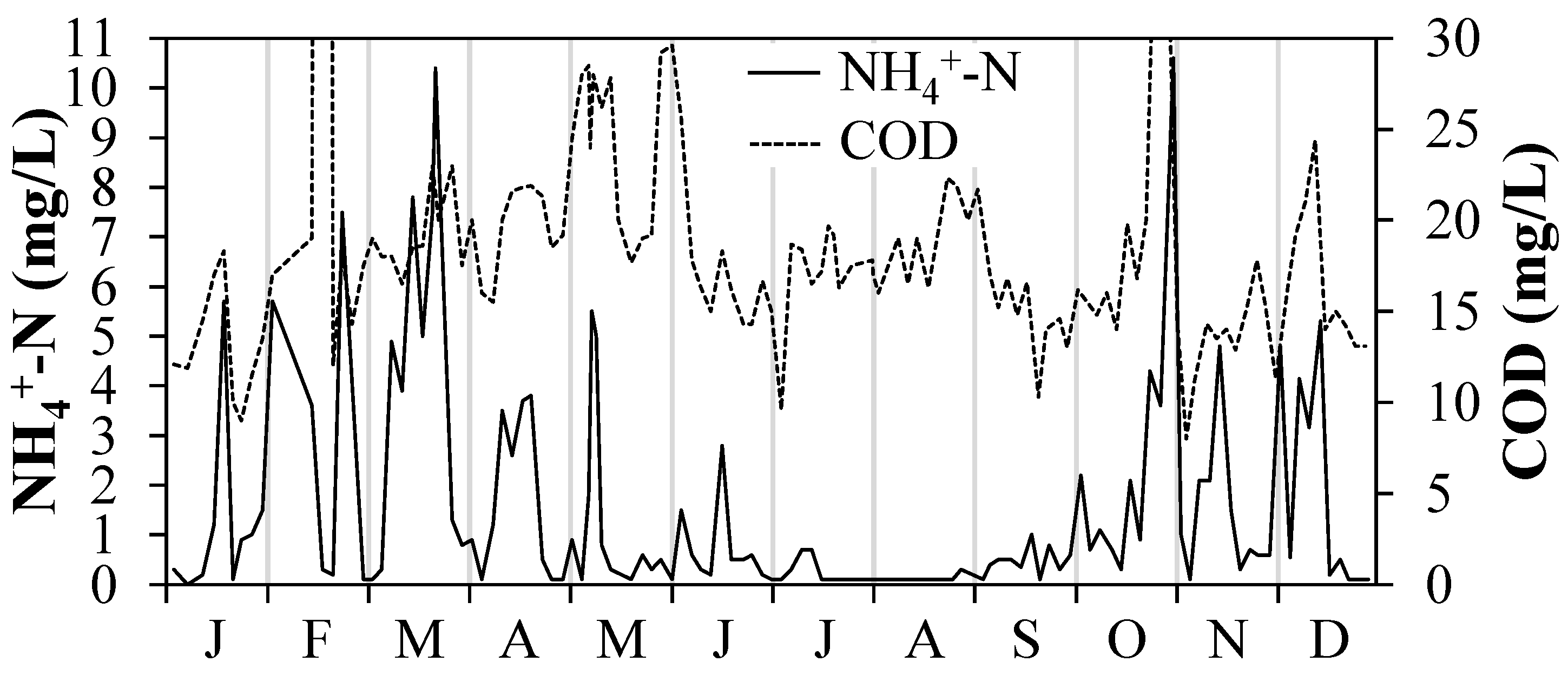
2.4. Tap Water (TW) and Wastewater Treatment Plant Effluent (WWTE)
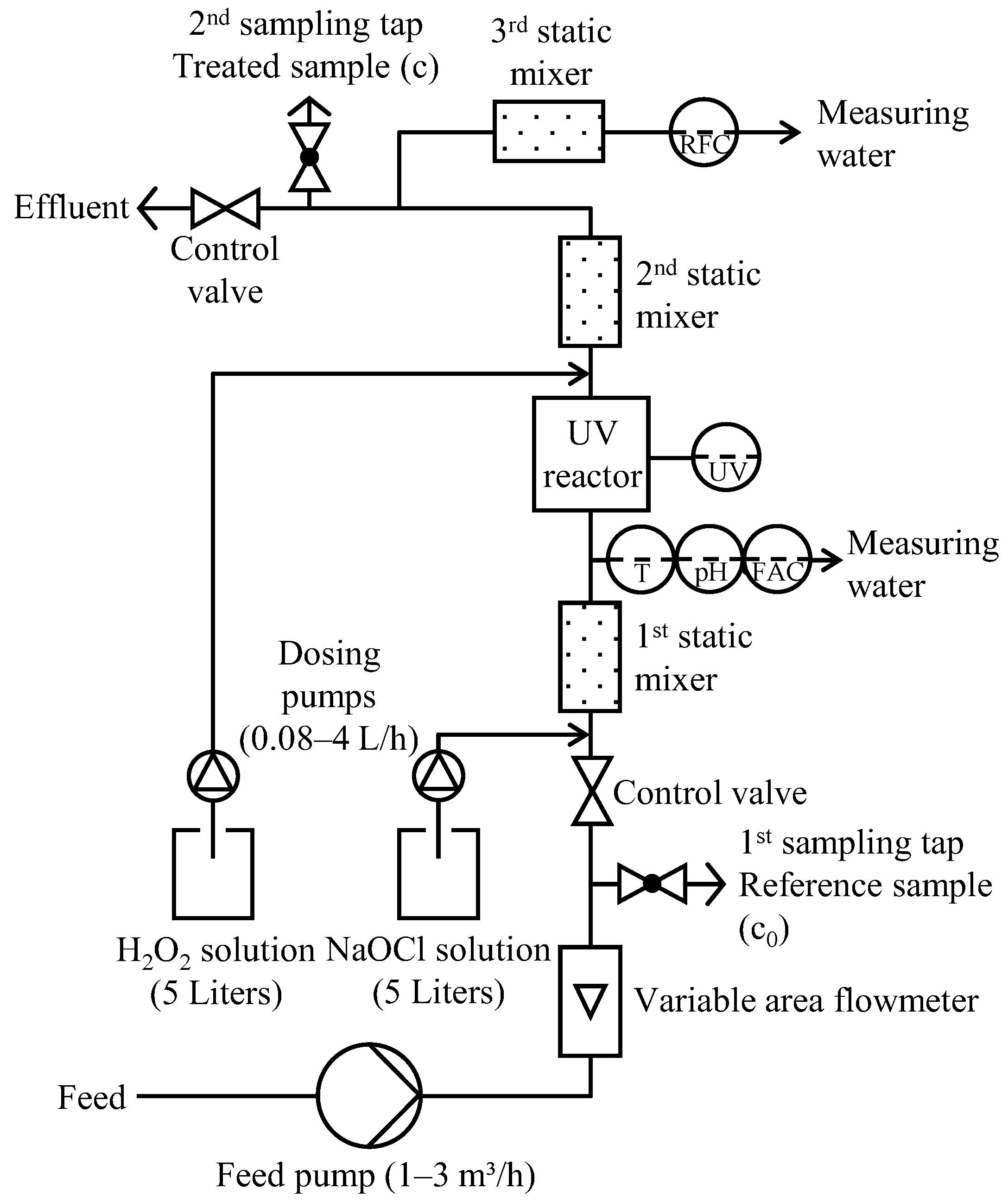
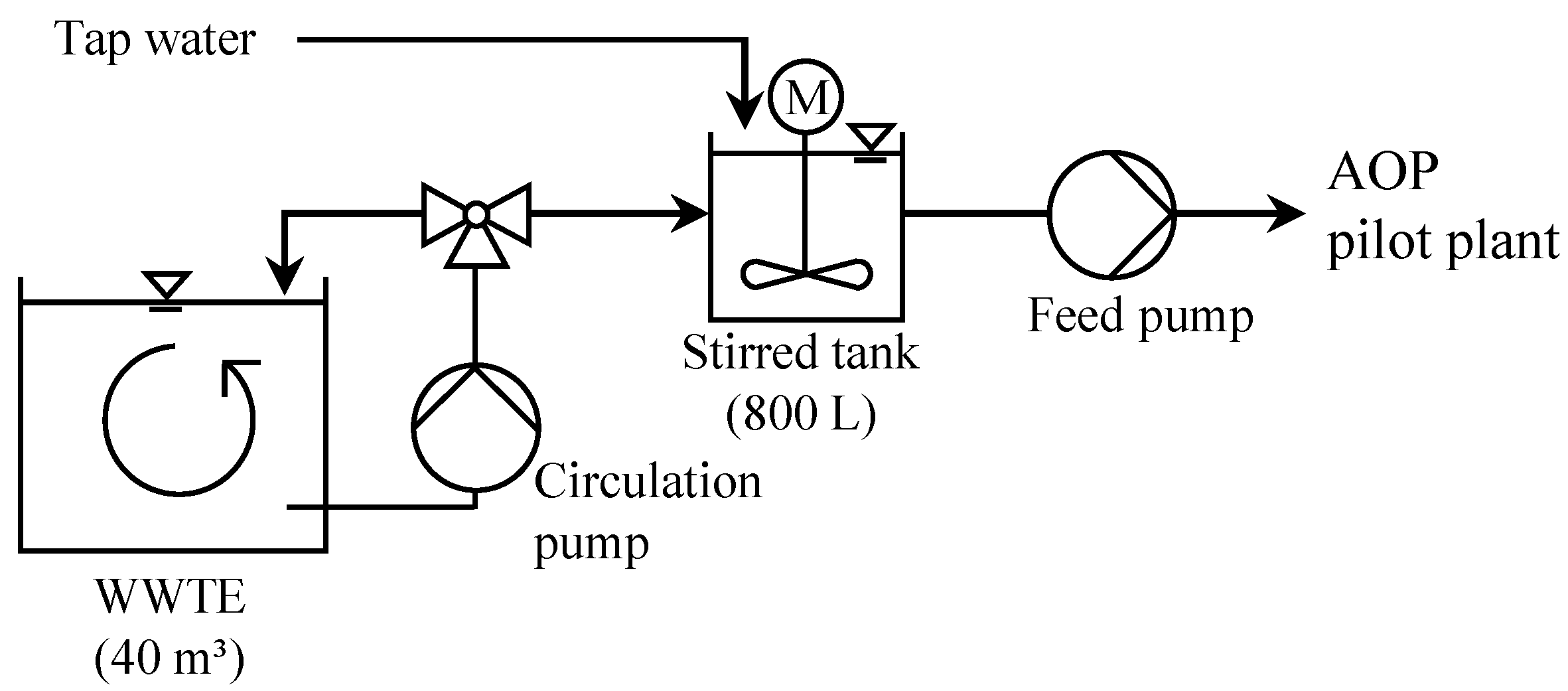
2.5. Pilot Plant
2.6. Experimental Procedure
2.6.1. Preparations
2.6.2. General Procedure
2.6.3. Exp. A: Variation of NH4+-N Concentration in Spiked Tap Water
2.6.4. Exp. B: Variation of WWTE Dilutions with Spiked Tap Water
2.6.5. Exp. C: Variation of CC Concentration on WWTE
2.6.6. Exp. D: Variation of Flow Rate
2.7. Analytical Methods
2.7.1. Free Cl2 (FAC, RFC), Combined Cl2 (CC), Total Cl2
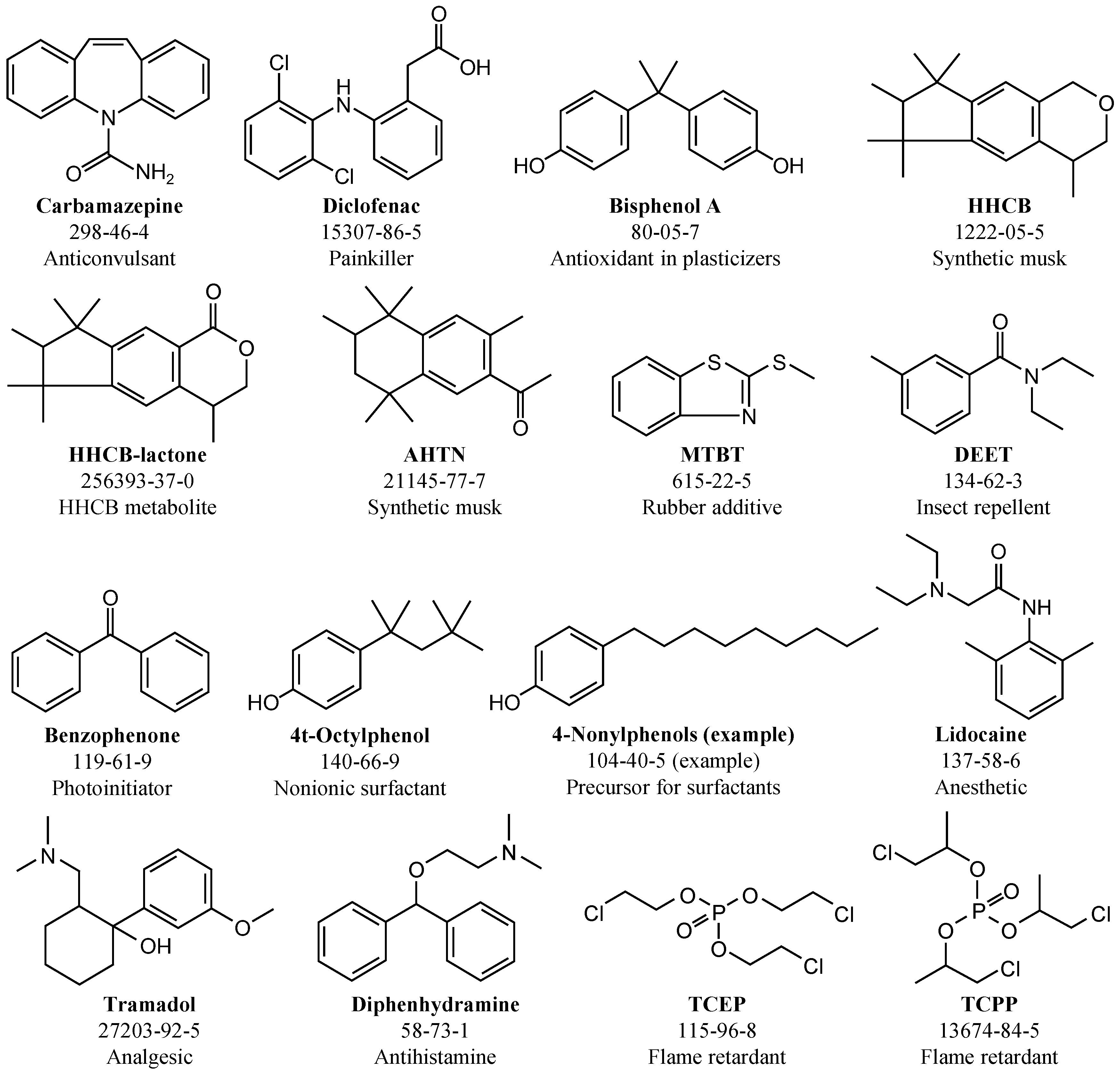
2.7.2. Emerging Contaminants
2.7.3. Other Parameters
2.7.4. Number of Measurements
3. Results and Discussion
3.1. Chlorine Species
3.1.1. Exp. A: Variation of NH4+-N Concentration in Spiked Tap Water
3.1.2. Exp. B: Variation of WWTE Dilutions with Spiked Tap Water
3.1.3. Exp. C: Variation of CC Concentration on WWTE
3.1.4. Exp. D: Variation of Flow Rate
3.2. Emerging Contaminants
3.2.1. Exp. A: Variation of NH4+-N Concentration and Exp. B: Variation of WWTE Dilutions
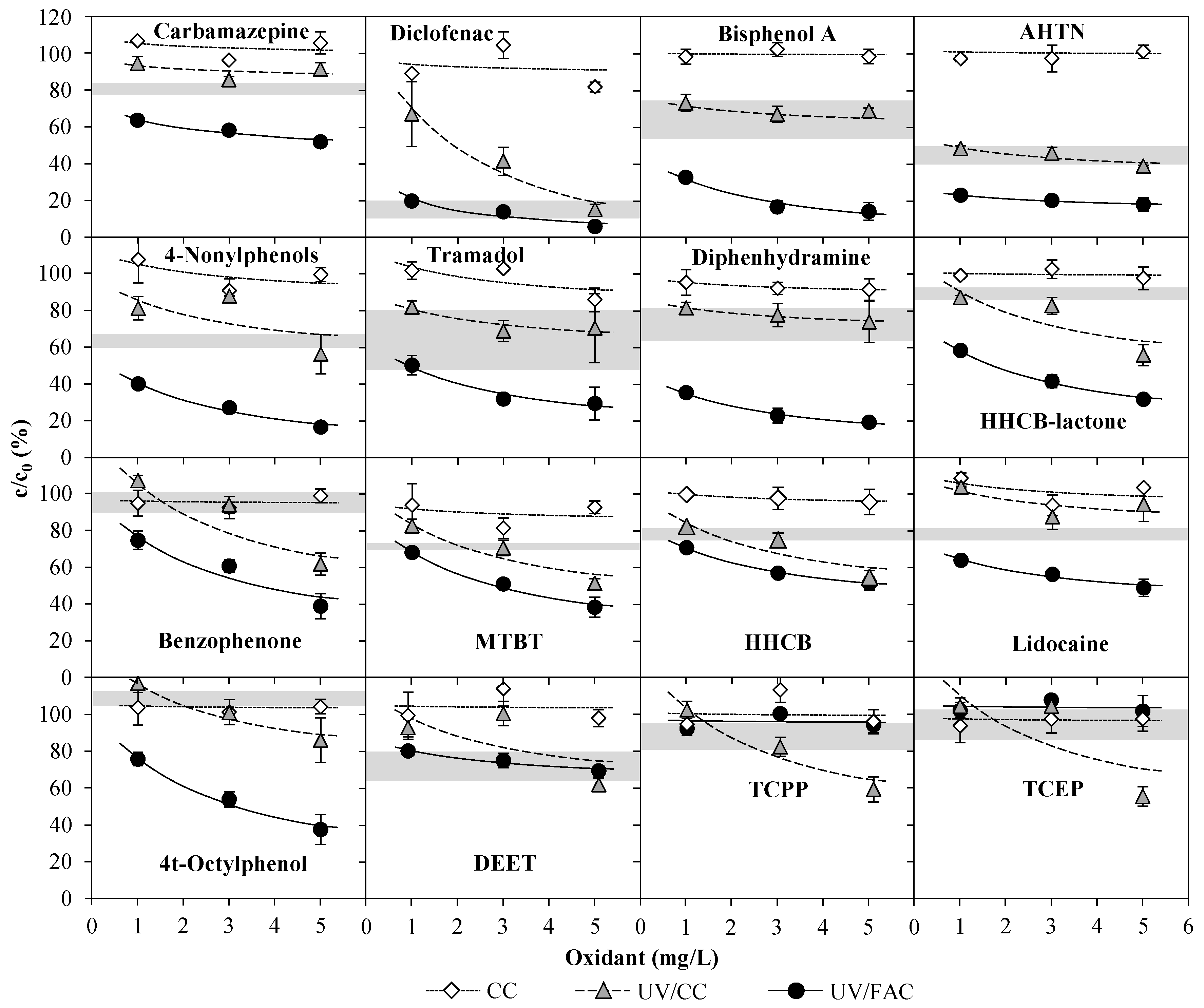
3.2.2. Exp. C: Variation of CC Concentration on WWTE
3.2.3. Exp. D: Variation of Flow Rate
4. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Auriol, M.; Filali-Meknassi, Y.; Tyagi, R.D.; Adams, C.D.; Surampalli, R.Y. Endocrine disrupting compounds removal from wastewater, a new challenge. Process Biochem. 2006, 41, 525–539. [Google Scholar] [CrossRef]
- Bolong, N.; Ismail, A.F.; Salim, M.R.; Matsuura, T. A review of the effects of emerging contaminants in wastewater and options for their removal. Desalination 2009, 239, 229–246. [Google Scholar] [CrossRef]
- Buxton, G.V.; Subhani, M.S. Radiation chemistry and photochemistry of oxychlorine ions. Part 2—Photodecomposition of aqueous solutions of hypochlorite ions. J. Chem. Soc. Faraday Trans. 1 1972, 68, 958–969. [Google Scholar] [CrossRef]
- Feng, Y.; Smith, D.W.; Bolton, J.R. Photolysis of aqueous free chlorine species (HOCl and OCl−) with 254 nm ultraviolet light. J. Environ. Eng. Sci. 2007, 6, 277–284. [Google Scholar] [CrossRef]
- Jin, J.; El-Din, M.G.; Bolton, J.R. Assessment of the UV/chlorine process as an advanced oxidation process. Water Res. 2011, 45, 1890–1896. [Google Scholar] [CrossRef] [PubMed]
- Watts, M.J.; Linden, K.G. Chlorine photolysis and subsequent OH radical production during UV treatment of chlorinated water. Water Res. 2007, 41, 2871–2878. [Google Scholar] [CrossRef] [PubMed]
- Rott, E.; Kuch, B.; Lange, C.; Richter, P.; Kugele, A.; Minke, R. Removal of Emerging Contaminants and Estrogenic Activity from Wastewater Treatment Plant Effluent with UV/Chlorine and UV/H2O2 Advanced Oxidation Treatment at Pilot Scale. Int. J. Environ. Res. Public Health 2018, 15, 935. [Google Scholar] [CrossRef] [PubMed]
- Shu, Z.; Bolton, J.R.; Belosevic, M.; El Din, M.G. Photodegradation of emerging micropollutants using the medium-pressure UV/H2O2 Advanced Oxidation Process. Water Res. 2013, 47, 2881–2889. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Sun, J.; Fu, W.; Shang, C.; Li, Y.; Chen, Y.; Gan, W.; Fang, J. PPCP degradation by UV/chlorine treatment and its impact on DBP formation potential in real waters. Water Res. 2016, 98, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Xia, Y.; Li, T.; Yao, T.; Shi, Z.; Zhu, S.; Gao, N. Degradation of carbamazepine by UV/chlorine advanced oxidation process and formation of disinfection by-products. Environ. Sci. Pollut. Res. Int. 2016, 23, 16448–16455. [Google Scholar] [CrossRef] [PubMed]
- Pressley, T.A.; Bishop, D.F.; Roan, S.G. Ammonia-nitrogen removal by breakpoint chlorination. Environ. Sci. Technol. 1972, 6, 622–628. [Google Scholar] [CrossRef]
- Wu, Q.-Y.; Hu, H.-Y.; Zhao, X.; Sun, Y.-X. Effect of Chlorination on the Estrogenic/Antiestrogenic Activities of Biologically Treated Wastewater. Environ. Sci. Technol. 2009, 43, 4940–4945. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Guo, K.; Fang, J.; Yang, X.; Xiao, H.; Hou, S.; Kong, X.; Shang, C.; Yang, X.; Meng, F.; et al. Factors affecting the roles of reactive species in the degradation of micropollutants by the UV/chlorine process. Water Res. 2017, 126, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Sichel, C.; Garcia, C.; Andre, K. Feasibility studies: UV/chlorine advanced oxidation treatment for the removal of emerging contaminants. Water Res. 2011, 45, 6371–6380. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-L.; Wu, Q.-Y.; Huang, N.; Wang, T.; Hu, H.-Y. Synergistic effect between UV and chlorine (UV/chlorine) on the degradation of carbamazepine: Influence factors and radical species. Water Res. 2016, 98, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Bolton, J.R.; Bircher, K.G.; Tumas, W.; Tolman, C.A. Figures-of-merit for the technical development and application of advanced oxidation technologies for both electric- and solar-driven systems. Pure Appl. Chem. 2001, 73, 627–637. [Google Scholar] [CrossRef]
- Zhang, Y.; Geissen, S.-U.; Gal, C. Carbamazepine and diclofenac: Removal in wastewater treatment plants and occurrence in water bodies. Chemosphere 2008, 73, 1151–1161. [Google Scholar] [CrossRef] [PubMed]
- American Chemical Society Database. Available online: https://scifinder.cas.org (accessed on 21 March 2018).
- Held, A.M.; Halko, D.J.; Hurst, J.K. Mechanisms of chlorine oxidation of hydrogen peroxide. J. Am. Chem. Soc. 1978, 100, 5732–5740. [Google Scholar] [CrossRef]
- Margerum, D.W.; Gray, E.T.; Huffman, R.P. Chlorination and the Formation of N-Chloro Compounds in Water Treatment. In Organometals and Organometalloids; Brinckman, F.E., Bellama, J.M., Eds.; American Chemical Society: Washington, DC, USA, 1979; pp. 278–291. [Google Scholar]
- Qiang, Z.; Adams, C.D. Determination of Monochloramine Formation Rate Constants with Stopped-Flow Spectrophotometry. Environ. Sci. Technol. 2004, 38, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Deborde, M.; von Gunten, U. Reactions of chlorine with inorganic and organic compounds during water treatment—Kinetics and mechanisms: A critical review. Water Res. 2008, 42, 13–51. [Google Scholar] [CrossRef] [PubMed]
- Chuang, Y.-H.; Chen, S.; Chinn, C.J.; Mitch, W.A. Comparing the UV/Monochloramine and UV/Free Chlorine Advanced Oxidation Processes (AOPs) to the UV/Hydrogen Peroxide AOP Under Scenarios Relevant to Potable Reuse. Environ. Sci. Technol. 2017, 51, 13859–13868. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Drinking Water and Health, Volume 7: Disinfectants and Disinfectant By-Products; National Academies Press: Washington, DC, USA, 1987.
- McKay, G.; Sjelin, B.; Chagnon, M.; Ishida, K.P.; Mezyk, S.P. Kinetic study of the reactions between chloramine disinfectants and hydrogen peroxide: Temperature dependence and reaction mechanism. Chemosphere 2013, 92, 1417–1422. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keen, O.S.; Thurman, E.M.; Ferrer, I.; Dotson, A.D.; Linden, K.G. Dimer formation during UV photolysis of diclofenac. Chemosphere 2013, 93, 1948–1956. [Google Scholar] [CrossRef] [PubMed]
- Soufan, M.; Deborde, M.; Delmont, A.; Legube, B. Aqueous chlorination of carbamazepine: Kinetic study and transformation product identification. Water Res. 2013, 47, 5076–5087. [Google Scholar] [CrossRef] [PubMed]
- Soufan, M.; Deborde, M.; Legube, B. Aqueous chlorination of diclofenac: Kinetic study and transformation products identification. Water Res. 2012, 46, 3377–3386. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, T. Different removal behaviours of multiple trace antibiotics in municipal wastewater chlorination. Water Res. 2013, 47, 2970–2982. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Xu, B.; Liungai, Z.; Hu, H.-Y.; Chen, C.; Qiao, J.; Lu, Y. The removal of estrogenic activity with UV/chlorine technology and identification of novel estrogenic disinfection by-products. J. Hazard. Mater. 2016, 307, 119–126. [Google Scholar] [CrossRef] [PubMed]
 ), CC (
), CC (  ) and OCRP (
) and OCRP (  ) in the UV chamber influent (left columns) and after UV treatment at 0.4 kW and subsequent quenching with H2O2 (right columns) by means of an AOP pilot plant. (A) Exp. A (1 m3/h TW, mainly <5 mg/L COD) and (B) Exp. B (1 m3/h, dilutions of WWTE, <0.1 mg/L NH4+-N).
) in the UV chamber influent (left columns) and after UV treatment at 0.4 kW and subsequent quenching with H2O2 (right columns) by means of an AOP pilot plant. (A) Exp. A (1 m3/h TW, mainly <5 mg/L COD) and (B) Exp. B (1 m3/h, dilutions of WWTE, <0.1 mg/L NH4+-N).
 ), CC (
), CC (  ) and OCRP (
) and OCRP (  ) in the UV chamber influent (left columns) and after UV treatment at 0.4 kW and subsequent quenching with H2O2 (right columns) by means of an AOP pilot plant. (A) Exp. A (1 m3/h TW, mainly <5 mg/L COD) and (B) Exp. B (1 m3/h, dilutions of WWTE, <0.1 mg/L NH4+-N).
) in the UV chamber influent (left columns) and after UV treatment at 0.4 kW and subsequent quenching with H2O2 (right columns) by means of an AOP pilot plant. (A) Exp. A (1 m3/h TW, mainly <5 mg/L COD) and (B) Exp. B (1 m3/h, dilutions of WWTE, <0.1 mg/L NH4+-N).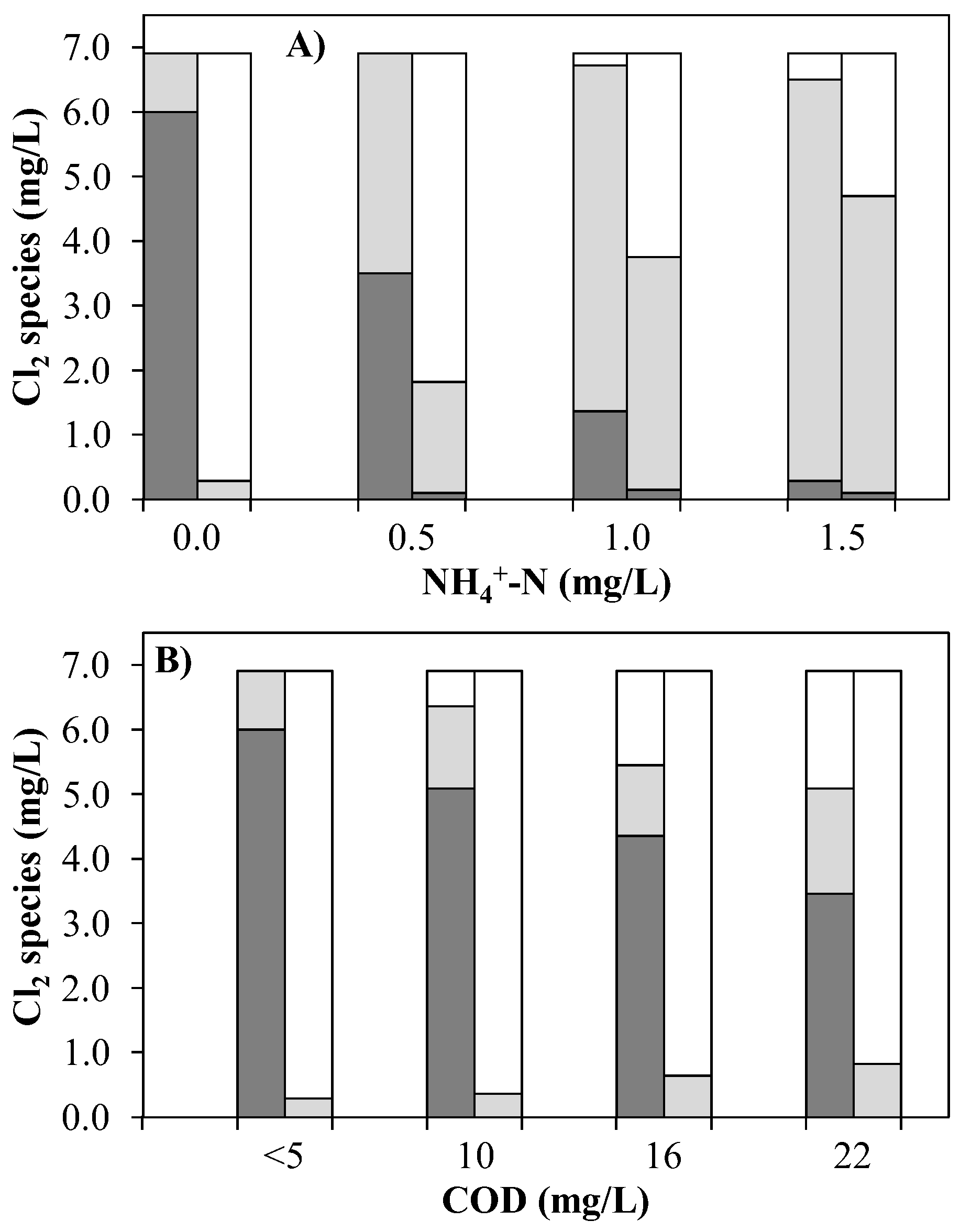
 ) and pilot plant effluent after treatment of 1 m3/h NH4+-loaded WWTE with CC (no UV) (
) and pilot plant effluent after treatment of 1 m3/h NH4+-loaded WWTE with CC (no UV) (  ), UV/CC (0.4 kW, no quenching) (
), UV/CC (0.4 kW, no quenching) (  ) and UV/CC (0.4 kW, 3.0–4.5 mg/L H2O2 quenching) (
) and UV/CC (0.4 kW, 3.0–4.5 mg/L H2O2 quenching) (  ) by means of an AOP pilot plant (Exp. C).
) by means of an AOP pilot plant (Exp. C).
 ) and pilot plant effluent after treatment of 1 m3/h NH4+-loaded WWTE with CC (no UV) (
) and pilot plant effluent after treatment of 1 m3/h NH4+-loaded WWTE with CC (no UV) (  ), UV/CC (0.4 kW, no quenching) (
), UV/CC (0.4 kW, no quenching) (  ) and UV/CC (0.4 kW, 3.0–4.5 mg/L H2O2 quenching) (
) and UV/CC (0.4 kW, 3.0–4.5 mg/L H2O2 quenching) (  ) by means of an AOP pilot plant (Exp. C).
) by means of an AOP pilot plant (Exp. C).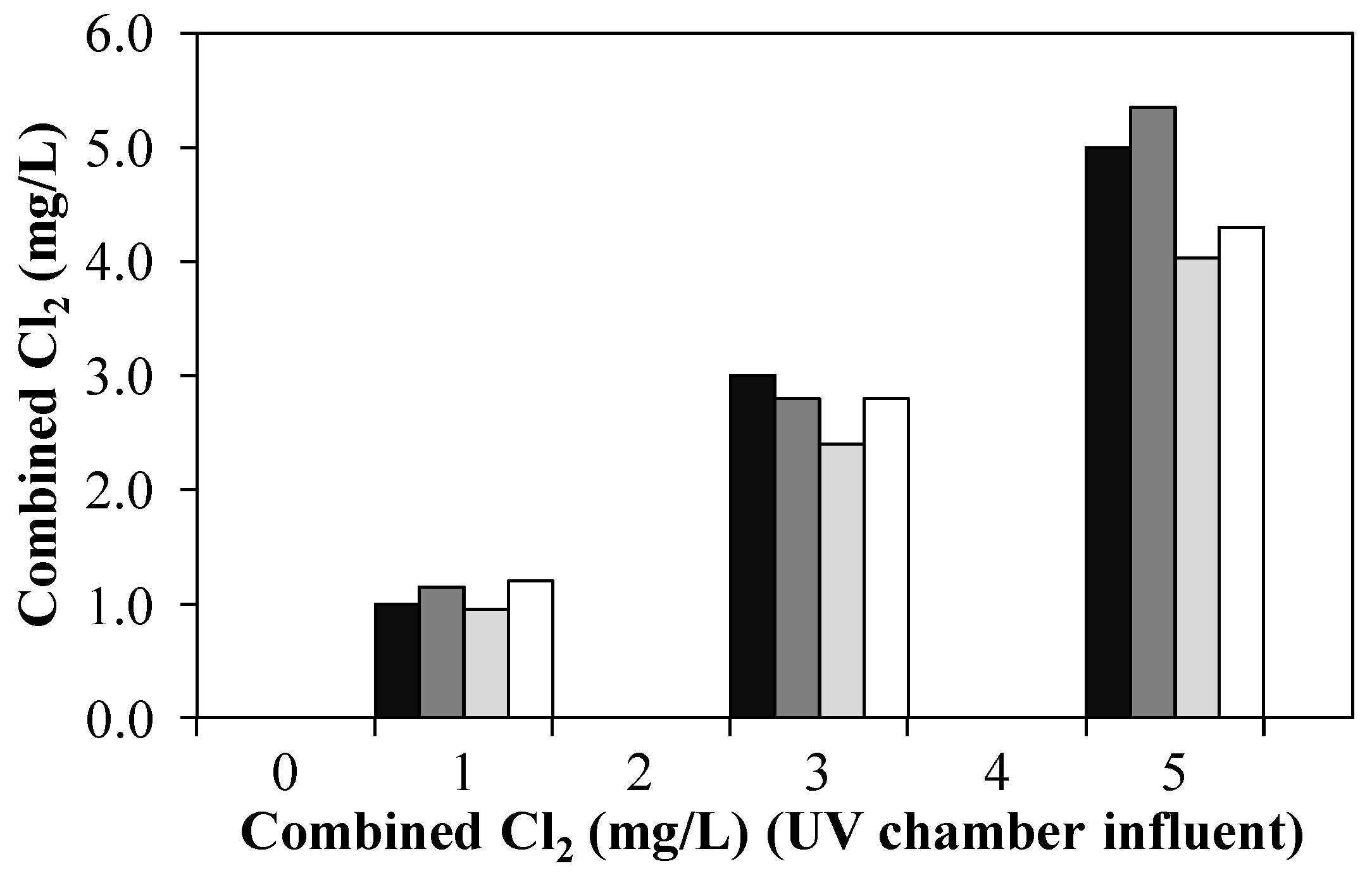
 ), CC (
), CC (  ) and OCRP (
) and OCRP (  ) in the UV chamber influent (left columns) and after UV treatment and subsequent quenching with H2O2 (right columns) by means of an AOP pilot plant at 0.4 kW (Exp. D: WWTE, 0.13–0.57 mg/L NH4+-N).
) in the UV chamber influent (left columns) and after UV treatment and subsequent quenching with H2O2 (right columns) by means of an AOP pilot plant at 0.4 kW (Exp. D: WWTE, 0.13–0.57 mg/L NH4+-N).
 ), CC (
), CC (  ) and OCRP (
) and OCRP (  ) in the UV chamber influent (left columns) and after UV treatment and subsequent quenching with H2O2 (right columns) by means of an AOP pilot plant at 0.4 kW (Exp. D: WWTE, 0.13–0.57 mg/L NH4+-N).
) in the UV chamber influent (left columns) and after UV treatment and subsequent quenching with H2O2 (right columns) by means of an AOP pilot plant at 0.4 kW (Exp. D: WWTE, 0.13–0.57 mg/L NH4+-N).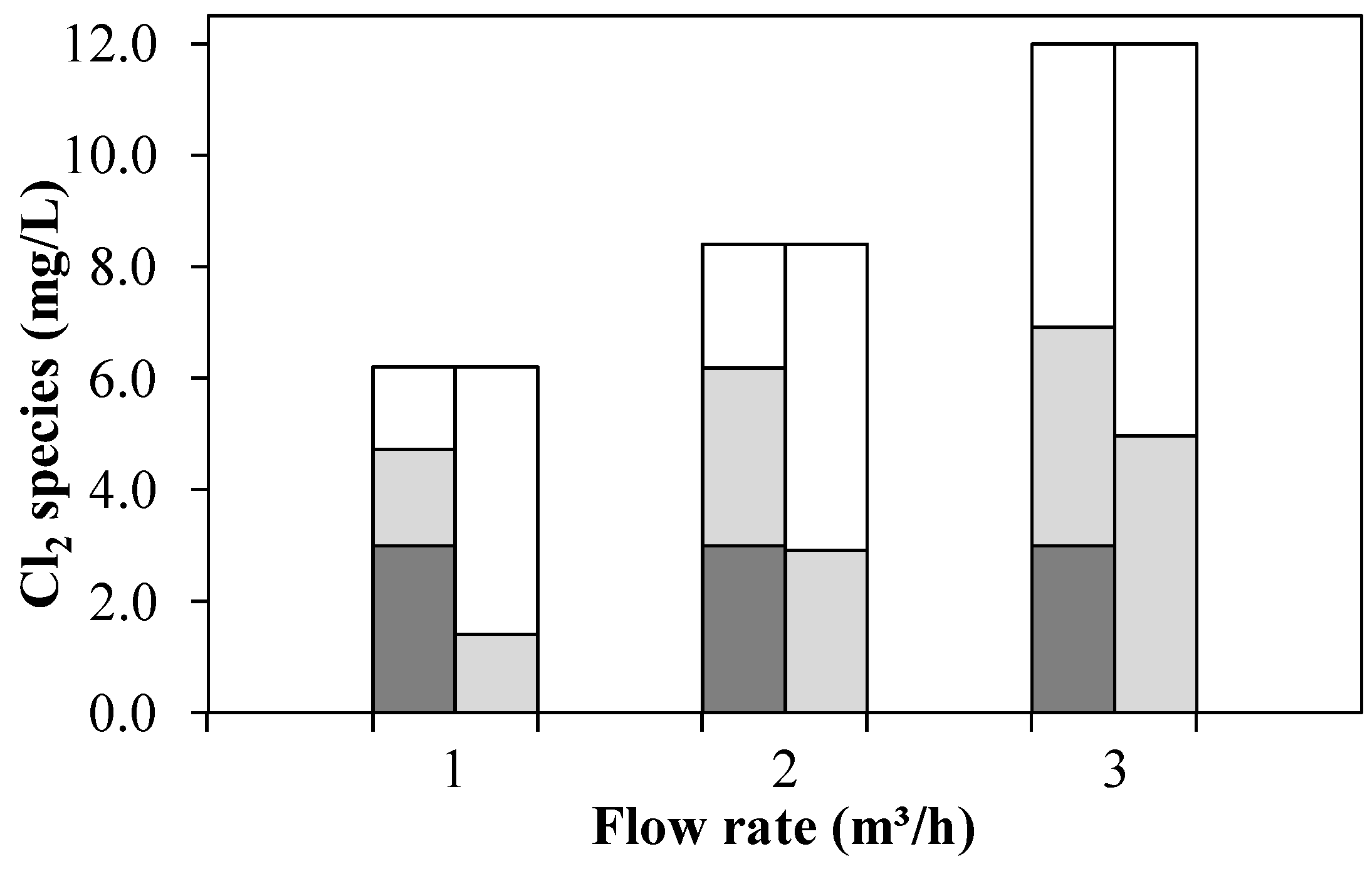
 ) and diclofenac (
) and diclofenac (  ) after treatment of tap water spiked with NH4Cl (Exp. A, <5 mg/L COD) and dilutions of WWTE with tap water (Exp. B, <0.1 mg/L NH4+-N) by means of an AOP pilot plant at 1 m3/h, 6.9 mg/L dosed Cl2, 0.4 kW UV lamp power (0.4 kWh/m3 electrical energy consumption) and subsequent quenching with around 3.2 mg/L H2O2 as functions of NH4+-N (A); COD (B) and FAC concentration as detected in the UV chamber influent (C: Exp. A; D: Exp. B). For EEO values, see Table S6.
) after treatment of tap water spiked with NH4Cl (Exp. A, <5 mg/L COD) and dilutions of WWTE with tap water (Exp. B, <0.1 mg/L NH4+-N) by means of an AOP pilot plant at 1 m3/h, 6.9 mg/L dosed Cl2, 0.4 kW UV lamp power (0.4 kWh/m3 electrical energy consumption) and subsequent quenching with around 3.2 mg/L H2O2 as functions of NH4+-N (A); COD (B) and FAC concentration as detected in the UV chamber influent (C: Exp. A; D: Exp. B). For EEO values, see Table S6.
 ) and diclofenac (
) and diclofenac (  ) after treatment of tap water spiked with NH4Cl (Exp. A, <5 mg/L COD) and dilutions of WWTE with tap water (Exp. B, <0.1 mg/L NH4+-N) by means of an AOP pilot plant at 1 m3/h, 6.9 mg/L dosed Cl2, 0.4 kW UV lamp power (0.4 kWh/m3 electrical energy consumption) and subsequent quenching with around 3.2 mg/L H2O2 as functions of NH4+-N (A); COD (B) and FAC concentration as detected in the UV chamber influent (C: Exp. A; D: Exp. B). For EEO values, see Table S6.
) after treatment of tap water spiked with NH4Cl (Exp. A, <5 mg/L COD) and dilutions of WWTE with tap water (Exp. B, <0.1 mg/L NH4+-N) by means of an AOP pilot plant at 1 m3/h, 6.9 mg/L dosed Cl2, 0.4 kW UV lamp power (0.4 kWh/m3 electrical energy consumption) and subsequent quenching with around 3.2 mg/L H2O2 as functions of NH4+-N (A); COD (B) and FAC concentration as detected in the UV chamber influent (C: Exp. A; D: Exp. B). For EEO values, see Table S6.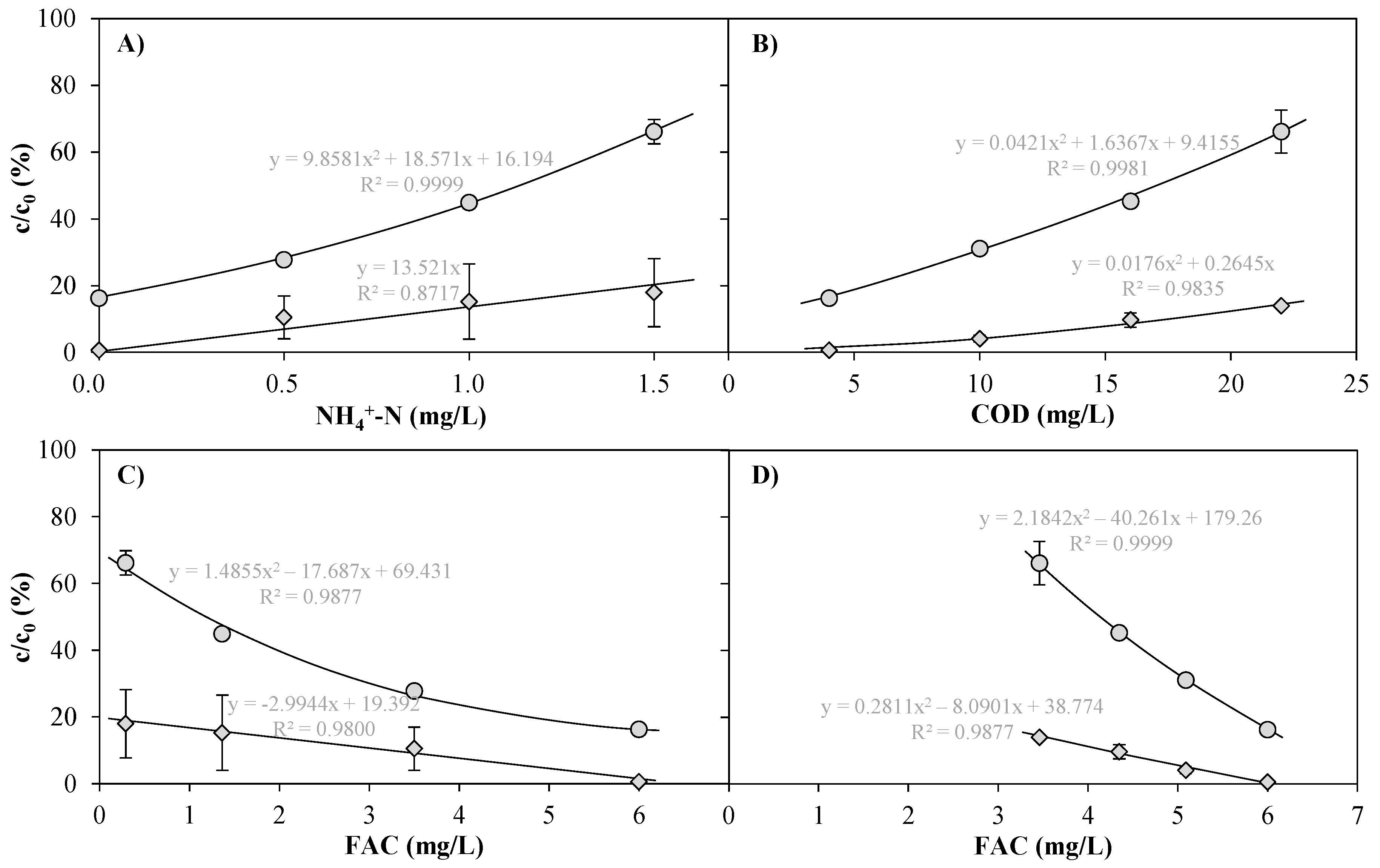

| Experiment | T | pH | COD | DOC | NH4+-N | Cond. | CBZ | DCF | |
|---|---|---|---|---|---|---|---|---|---|
| °C | - | mg/L | mg/L | mg/L | µS/cm | µg/L | µg/L | ||
| A | <0.1 mg/L NH4+-N | 16.3 | 7.4 | <5.0 | <1.5 | <0.1 | 334 ± 0 | 0.25 ± 0.00 | 1.70 ± 0.01 |
| 0.5 mg/L NH4+-N | 16.0 | 8.0 | <5.0 | <1.5 | 0.5 | 341 ± 0 | 0.94 ± 0.02 | 1.66 ± 0.18 | |
| 1.0 mg/L NH4+-N | 16.7 | 8.0 | <5.0 | <1.5 | 1.0 | 348 ± 3 | 0.59 ± 0.03 | 1.90 ± 0.03 | |
| 1.5 mg/L NH4+-N | 15.5 | 8.0 | <5.0 | <1.5 | 1.5 | 350 ± 0 | 1.07 ± 0.04 | 1.40 ± 0.25 | |
| B | <5 mg/L COD | 16.3 | 7.4 | <5.0 | <1.5 | <0.1 | 334 ± 0 | 0.25 ± 0.00 | 1.70 ± 0.01 |
| 10 mg/L COD | 17.7 | 8.2 | 12.1 ± 1.6 | 3.1 ± 0.2 | <0.1 | 520 ± 1 | 0.40 ± 0.00 | 3.08 ± 0.07 | |
| 16 mg/L COD | 17.1 | 8.2 | 16.2 ± 0.1 | 4.2 ± 0.4 | <0.1 | 717 ± 4 | 0.48 ± 0.07 | 3.01 ± 0.37 | |
| 22 mg/L COD | 18.2 | 8.2 | 21.0 ± 1.5 | 5.1 ± 0.1 | <0.1 | 909 ± 1 | 0.48 ± 0.01 | 2.08 ± 0.51 | |
| C | 1 mg/L CC | 13.9 | 6.9 | 31.2 ± 1.0 | 8.3 ± 0.9 | 1.58 ± 0.02 | 920 ± 0 | 0.68 ± 0.05 | 2.67 ± 0.18 |
| 3 mg/L CC | 14.8 | 7.0 | 23.0 ± 0.8 | 5.9 ± 0.3 | 6.26 ± 0.05 | 973 ± 0 | 0.57 ± 0.02 | 1.98 ± 0.25 | |
| 5 mg/L CC | 14.2 | 7.0 | 24.8 ± 1.5 | 6.5 ± 0.9 | 6.73 ± 0.85 | 1048 ± 9 | 0.57 ± 0.01 | 3.87 ± 0.45 | |
| D | 1 m3/h flow rate | 18.3 | 6.9 | 21.9 ± 0.1 | 4.5 ± 0.0 | 0.13 ± 0.00 | 823 ± 0 | 0.54 ± 0.02 | 1.31 ± 0.02 |
| 1 m3/h flow rate | 18.3 | 6.9 | 20.6 ± 2.3 | n.m. | 0.57 ± 0.00 | 822 ± 6 | 0.57 ± 0.02 | 1.42 ± 0.14 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rott, E.; Kuch, B.; Lange, C.; Richter, P.; Minke, R. Influence of Ammonium Ions, Organic Load and Flow Rate on the UV/Chlorine AOP Applied to Effluent of a Wastewater Treatment Plant at Pilot Scale. Int. J. Environ. Res. Public Health 2018, 15, 1276. https://doi.org/10.3390/ijerph15061276
Rott E, Kuch B, Lange C, Richter P, Minke R. Influence of Ammonium Ions, Organic Load and Flow Rate on the UV/Chlorine AOP Applied to Effluent of a Wastewater Treatment Plant at Pilot Scale. International Journal of Environmental Research and Public Health. 2018; 15(6):1276. https://doi.org/10.3390/ijerph15061276
Chicago/Turabian StyleRott, Eduard, Bertram Kuch, Claudia Lange, Philipp Richter, and Ralf Minke. 2018. "Influence of Ammonium Ions, Organic Load and Flow Rate on the UV/Chlorine AOP Applied to Effluent of a Wastewater Treatment Plant at Pilot Scale" International Journal of Environmental Research and Public Health 15, no. 6: 1276. https://doi.org/10.3390/ijerph15061276
APA StyleRott, E., Kuch, B., Lange, C., Richter, P., & Minke, R. (2018). Influence of Ammonium Ions, Organic Load and Flow Rate on the UV/Chlorine AOP Applied to Effluent of a Wastewater Treatment Plant at Pilot Scale. International Journal of Environmental Research and Public Health, 15(6), 1276. https://doi.org/10.3390/ijerph15061276





