Clinical Relevant Polymorphisms Affecting Clopidogrel Pharmacokinetics and Pharmacodynamics: Insights from the Puerto Rico Newborn Screening Program
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethics
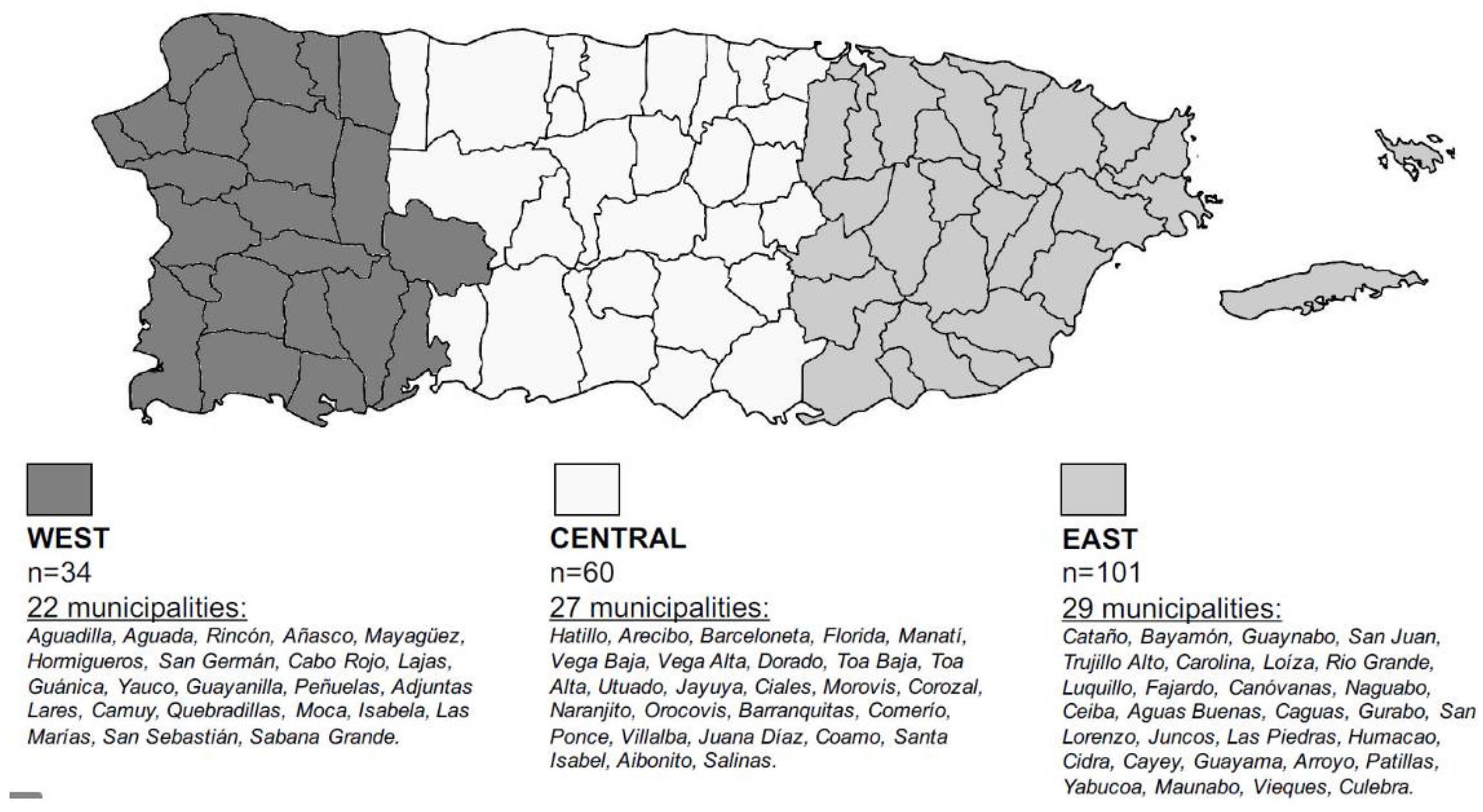
2.2. Sample and Data Collection
2.3. Genotyping Assay
2.4. Statistical Analyses
3. Results
3.1. ABCB1 Locus
3.2. PON1 Locus
3.3. P2RY12 Locus
3.4. CYP2C19 Locus
4. Discussion
5. Study Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Hernandez-Suarez, D.F.; Scott, S.A.; Tomey, M.I.; Melin, K.; Lopez-Candales, A.; Buckley, C.E.; Duconge, J. Clinical determinants of clopidogrel responsiveness in a heterogeneous cohort of Puerto Rican Hispanics. Ther. Adv. Cardiovasc. Dis. 2017, 11, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Gent, M.; Beaumont, D.; Blanchard, J.; Bousser, M.G.; Coffman, J.; Easton, J.D.; Hampton, J.R.; Harker, L.A.; Janzon, L.; Kusmierek, J.J.E.; et al. The CAPRIE Steering Committee. Randomized, blinded trial of clopidogrel versus aspirin in patients at risk of ischemic events (CAPRIE). Lancet 1996, 348, 1329–1339. [Google Scholar]
- Gladding, P.; Webster, M.; Zeng, I.; Farrell, H.; Stewart, J.; Ruygrok, P.; Ormiston, J.; El-Jack, S.; Armstrong, G.; Kay, P.; et al. The pharmacogenetics and pharmacodynamics of clopidogrel response: An analysis from the PRINC (Plavix Response in Coronary Intervention) trial. JACC Cardiovasc. Interv. 2008, 1, 620–627. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Suarez, D.F.; Núñez-Medina, H.; Scott, S.A.; Lopez-Candales, A.; Wiley, J.M.; Garcia, M.J.; Melin, K.; Nieves-Borrero, K.; Rodriguez-Ruiz, C.; Marshall, L.; et al. Effect of cilostazol on platelet reactivity among patients with peripheral artery disease on clopidogrel therapy. Drug Metab. Pers. Ther. 2018, 33, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Duconge, J.; Hernandez-Suarez, D.F. Potential Usefulness of Clopidogrel Pharmacogenetics in Cerebral Endovascular Procedures and Carotid Artery Stenting. Curr. Clin. Pharmacol. 2017, 12, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Savi, P.; Zachayus, J.L.; Delesque-Touchard, N.; Labouret, C.; Hervé, C.; Uzabiaga, M.F.; Pereillo, J.M.; Culouscou, J.M.; Bono, F.; Ferrara, P.; et al. The active metabolite of Clopidogrel disrupts P2Y12 receptor oligomers and partitions them out of lipid rafts. Proc. Natl. Acad. Sci. USA 2006, 103, 11069–11074. [Google Scholar] [CrossRef] [PubMed]
- Sabatine, M.S.; Cannon, C.P.; Gibson, C.M.; López-Sendón, J.L.; Montalescot, G.; Theroux, P.; Lewis, B.S.; Murphy, S.A.; McCabe, C.H.; Braunwald, E.; et al. Effect of clopidogrel pretreatment before percutaneous coronary intervention in patients with ST-elevation myocardial infarction treated with fibrinolytics: The PCI-CLARITY study. JAMA 2005, 294, 1224–1232. [Google Scholar] [CrossRef] [PubMed]
- Amsterdam, E.A.; Wenger, N.K.; Brindis, R.G.; Casey, D.E., Jr.; Ganiats, T.G.; Holmes, D.R., Jr.; Jaffe, A.S.; Jneid, H.; Kelly, R.F.; Kontos, M.C.; et al. 2014 AHA/ACC Guideline for the Management of Patients with Non-ST-Elevation Acute Coronary Syndromes: A report of the American College of Cardiology/American Heart Association Task Force on Practice. J. Am. Coll. Cardiol. 2014, 64, e139–e228. [Google Scholar] [CrossRef] [PubMed]
- Tantry, U.S.; Bliden, K.P.; Gurbel, P.A. Resistance to antiplatelet drugs: Current status and future research. Expert Opin. Pharmacother. 2005, 6, 2027–2045. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.K.; Lam, Y.Y.; Tan, V.P.; Yan, B.P. Variability in response to clopidogrel: How important are pharmacogenetics and drug interactions? Br. J. Clin. Pharmacol. 2011, 72, 697–706. [Google Scholar] [CrossRef] [PubMed]
- Krishna, V.; Diamond, G.A.; Kaul, S. Do platelet function testing and genotyping improve outcome in patients treated with antithrombotic agents?: The role of platelet reactivity and genotype testing in the prevention of atherothrombotic cardiovascular events remains unproven. Circulation 2012, 125, 1288–1303. [Google Scholar] [CrossRef] [PubMed]
- Spiliopoulos, S.; Pastromas, G. Current status of high on-treatment platelet reactivity in patients with coronary or peripheral arterial disease: Mechanisms, evaluation and clinical implications. World J. Cardiol. 2015, 7, 912–921. [Google Scholar] [CrossRef] [PubMed]
- Geisler, T.; Gawaz, M. Individualized antiplatelet therapy: What can a clinical score contribute? Hamostaseologie 2009, 29, 360–367. [Google Scholar] [PubMed]
- Cavallari, L.H.; Obeng, A.O. Genetic Determinants of P2Y12 Inhibitors and Clinical Implications. Interv. Cardiol. Clin. 2017, 6, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Viviani Anselmi, C.; Briguori, C.; Roncarati, R.; Papa, L.; Visconti, G.; Focaccio, A.; De Micco, F.; Latronico, M.V.; Pagnotta, P.; Condorelli, G. Routine assessment of on-clopidogrel platelet reactivity and gene polymorphisms in predicting clinical outcome following drug-eluting stent implantation in patients with stable coronary artery disease. JACC Cardiovasc. Interv. 2013, 6, 1166–1175. [Google Scholar] [CrossRef] [PubMed]
- Sanofi-Aventis. Bristol Myers Squibb. Plavix Label. 3-12-2010; Bristol-Myers Squibb/Sanofi Pharmaceuticals Partnership: Bridgewater, NJ, USA, 2010. [Google Scholar]
- Scott, S.A.; Sangkuhl, K.; Stein, C.M.; Hulot, J.S.; Mega, J.L.; Roden, D.M.; Klein, T.E.; Sabatine, M.S.; Johnson, J.A.; Shuldiner, A.R.; et al. Clinical Pharmacogenetics Implementation Consortium guidelines for CYP2C19 genotype and clopidogrel therapy: 2013 update. Clin. Pharmacol. Ther. 2013, 94, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Orengo-Mercado, C.; Nieves, B.; López, L.; Vallés-Ortiz, N.; Renta, J.Y.; Santiago-Borrero, P.J.; Cadilla, C.L.; Duconge, J. Frequencies of Functional Polymorphisms in Three Pharmacokinetic Genes of Clinical Interest within the Admixed Puerto Rican Population. J. Pharmacogenomics Pharmacoproteomics 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- ClinVar Aggregates Information about Genomic Variation and Its Relationship to Human Health (ClinVar). Available online: https://preview.ncbi.nlm.nih.gov/clinvar/ (accessed on 10 March 2018).
- Clinical Annotation for rs4244285 (CYP2C19), Clopidogrel, Acute Coronary Syndrome, Cardiovascular Diseases and Thrombosis (Level 1A Efficacy, Toxicity/ADR). Available online: https://www.pharmgkb.org/clinicalAnnotation/655384520 (accessed on 10 March 2018).
- Clinical Annotation for rs4986893 (CYP2C19), Clopidogrel, Acute Coronary Syndrome and Coronary Artery Disease (Level 1A Efficacy, Toxicity/ADR). Available online: https://www.pharmgkb.org/clinicalAnnotation/981201917 (accessed on 10 March 2018).
- Clinical Annotation for rs28399504 (CYP2C19), Clopidogrel, Acute Coronary Syndrome and Cardiovascular Diseases (level 1A Efficacy). Available online: https://www.pharmgkb.org/clinicalAnnotation/982028896 (accessed on 10 March 2018).
- Clinical Annotation for rs12248560 (CYP2C19), Clopidogrel, Acute Coronary Syndrome, Coronary Artery Disease and Myocardial Infarction (Level 1A Dosage, Efficacy, Toxicity/ADR). Available online: https://www.pharmgkb.org/clinicalAnnotation/655386913 (accessed on 10 March 2018).
- ClinVar Genomic Variation as It Relates to Human Health. Available online: https://preview.ncbi.nlm.nih.gov/clinvar/variation/225939/ (accessed on 10 March 2018).
- Clinical Annotation for P2RY12 H1P2RY12 H2 Related to Clopidogrel Efficacy. Available online: https://www.pharmgkb.org/gene/PA134971947/clinicalAnnotation/1043859330 (accessed on 10 March 2018).
- Clinical Annotation for rs662 Related to Clopidogrel Efficacy/Toxicity. Available online: https://www.pharmgkb.org/gene/PA33529/clinicalAnnotation/981202068 (accessed on 10 March 2018).
- Pharmacogene Variation Consortium (PharmVar). Available online: https://www.pharmvar.org/gene/CYP2C19 (accessed on 10 March 2018).
- Newcombe, R.G. Two-sided confidence intervals for the single proportion: Comparison of seven methods. Stat. Med. 1998, 17, 857–872. [Google Scholar] [CrossRef]
- IGSR: The International Genome Sample Resource. Providing Ongoing Support for the 1000 Genomes Project Data. Available online: http://www.internationalgenome.org/ (accessed on 10 March 2018).
- 1000 Genomes Browser Phase 3. Available online: https://www.ncbi.nlm.nih.gov/variation/tools/1000genomes/ (accessed on 10 March 2018).
- Amigo, J.; Salas, A.; Phillips, C.; Carracedo, A. SPSmart: Adapting population based SNP genotype databases for fast and comprehensive web access. BMC Bioinform. 2008, 9, 428. [Google Scholar] [CrossRef] [PubMed]
- 1000 Genomes Project Consortium; Auton, A.; Brooks, L.D.; Durbin, R.M.; Garrison, E.P.; Kang, H.M.; Korbel, J.O.; Marchini, J.L.; McCarthy, S.; McVean, G.A.; et al. A global reference for human genetic variation. Nature 2015, 526, 68–74. [Google Scholar]
- Via, M.; Gignoux, C.R.; Roth, L.A.; Fejerman, L.; Galanter, J.; Choudhry, S.; Toro-Labrador, G.; Viera-Vera, J.; Oleksyk, T.K.; Beckman, K.; et al. History shaped the geographic distribution of genomic admixture on the island of Puerto Rico. PLoS ONE 2011, 6, e16513. [Google Scholar] [CrossRef] [PubMed]
- Duconge, J.; Cadilla, C.; Renta, J.Y.; Silén-Rivera, P.; Piovanetti, P.; Garcia-Berdecia, R.; Castro-Rosario, L.M.; Monzón, S.; Vélez, L.; Rosas, G.; et al. Prevalence of CYP2C19 Gene Polymorphisms in the Puerto Rican Population: A preliminary report. PR Health Sci. J. 2008, 27, 357–358. [Google Scholar]
- Duconge, J.; Escalera, O.; Korchela, M.; Ruaño, G. Clinical Implications of Genetic Admixture in Hispanic Puerto Ricans: Impact on the Pharmacogenetics of CYP2C19 and PON1. In Clinical Applications of Pharmacogenetics, 1st ed.; Sanoudou, D., Ed.; IntechOpen: Rijeka, Croatia, 2012; pp. 151–164. ISBN 978-953-51-0389-9. [Google Scholar] [CrossRef]
- Ruaño, G.; Duconge, J.; Windemuth, A.; Santiago-Borrero, P.J. Physiogenomic analysis of the Puerto Rican population. Pharmacogenomics 2009, 10, 565–577. [Google Scholar] [CrossRef] [PubMed]
- Bouman, H.J.; Schömig, E.; van Werkum, J.W.; Velder, J.; Hackeng, C.M.; Hirschhäuser, C.; Waldmann, C.; Schmalz, H.G.; ten Berg, J.M.; Taubert, D. Paraoxonase-1 is a major determinant of clopidogrel efficacy. Nat. Med. 2011, 17, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Tselepis, A.D.; Tsoumani, M.E.; Kalantzi, K.I.; Dimitriou, A.A.; Tellis, C.C.; Goudevenos, I.A. Influence of high-density lipoprotein and paraoxonase-1 on platelet reactivity in patients with acute coronary syndromes receiving clopidogrel therapy. J. Thromb. Haemost. 2011, 9, 2371–2378. [Google Scholar] [CrossRef] [PubMed]
- Sibbing, D.; Koch, W.; Massberg, S.; Byrne, R.A.; Mehilli, J.; Schulz, S.; Mayer, K.; Bernlochner, I.; Schömig, A.; Kastrati, A. No association of paraoxonase-1 Q192R genotypes with platelet response to clopidogrel and risk of stent thrombosis after coronary stenting. Eur. Heart J. 2011, 32, 1605–1613. [Google Scholar] [CrossRef] [PubMed]
- Regieli, J.J.; Jukema, J.W.; Doevendans, P.A.; Zwinderman, A.H.; Kastelein, J.J.; Grobbee, D.E.; van der Graaf, Y. Paraoxonase variants relate to 10-year risk in coronary artery disease: Impact of a high-density lipoprotein-bound antioxidant in secondary prevention. J. Am. Coll. Cardiol. 2009, 54, 1238–1245. [Google Scholar] [CrossRef] [PubMed]
- Trenk, D.; Hochholzer, W. Genetics of platelet inhibitor treatment. Br. J. Clin. Pharmacol. 2014, 77, 642–653. [Google Scholar] [CrossRef] [PubMed]
- Carlquist, J.F.; Knight, S.; Horne, B.D.; Huntinghouse, J.A.; Rollo, J.S.; Muhlestein, J.B.; May, H.; Anderson, J.L. Cardiovascular risk among patients on clopidogrel anti-platelet therapy after placement of drug-eluting stents is modified by genetic variants in both the CYP2C19 and ABCB1 genes. Thromb. Haemost. 2013, 109, 744–754. [Google Scholar] [PubMed]
- Ameyaw, M.M.; Regateiro, F.; Li, T.; Liu, X.; Tariq, M.; Mobarek, A.; Thornton, N.; Folayan, G.O.; Githang’a, J.; Indalo, A.; et al. MDR1 pharmacogenetics: Frequency of the C3435T mutation in exon 26 is significantly influenced by ethnicity. Pharmacogenetics 2001, 11, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Calderón-Cruz, B.; Rodríguez-Galván, K.; Manzo-Francisco, L.A.; Vargas-Alarcón, G.; Fragoso, J.M.; Peña-Duque, M.A.; Reyes-Gómez, C.A.; Martínez-Ríos, M.A.; De la Peña-Díaz, A. C3435T polymorphism of the ABCB1 gene is associated with poor clopidogrel responsiveness in a Mexican population undergoing percutaneous coronary intervention. Thromb. Res. 2015, 136, 894–898. [Google Scholar] [CrossRef] [PubMed]
- Dorsam, R.T.; Kunapuli, S.P. Central role of the P2Y12 receptor in platelet activation. J. Clin. Investig. 2004, 113, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Angiolillo, D.J.; Fernandez-Ortiz, A.; Bernardo, E.; Ramírez, C.; Cavallari, U.; Trabetti, E.; Sabaté, M.; Jimenez-Quevedo, P.; Hernández, R.; Moreno, R.; et al. Lack of association between the P2Y12 receptor gene polymorphism and platelet response to clopidogrel in patients with coronary artery disease. Thromb. Res. 2005, 116, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Zee, R.Y.; Michaud, S.E.; Diehl, K.A.; Chasman, D.I.; Emmerich, J.; Gaussem, P.; Aiach, M.; Ridker, P.M. Purinergic receptor P2Y, G-protein coupled, 12 gene variants and risk of incident ischemic stroke, myocardial infarction, and venous thromboembolism. Atherosclerosis 2008, 197, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Oestreich, J.H.; Steinhubl, S.R.; Ferraris, S.P.; Loftin, C.D.; Akers, W.S. Effect of genetic variation in P2Y12 on TRAP-stimulated platelet response in healthy subjects. J. Thromb. Thrombolysis 2014, 38, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Staritz, P.; Kurz, K.; Stoll, M.; Giannitsis, E.; Katus, H.A.; Ivandic, B.T. Platelet reactivity and clopidogrel resistance are associated with the H2 haplotype of the P2Y12-ADP receptor gene. Int. J. Cardiol. 2009, 133, 341–345. [Google Scholar] [CrossRef] [PubMed]
| Status/Marker | ABCB1 (C > T) | PON1 (G > A) | CYP2C19*3 (G > A) | CYP2C19*4 (A > G) | CYP2C19*2 (G > A) | CYP2C19*17 (C > T) | P2RY12 H2 (C > T) * | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. | Freq. | No. | Freq. | No. | Freq. | No. | Freq. | No. | Freq. | No. | Freq. | No. | Freq. | |
| Genotypes | ||||||||||||||
| Homozygous wild-type (WT) | 69 | 0.350 | 59 | 0.299 | 196 | 1.000 | 195 | 0.995 | 145 | 0.739 | 144 | 0.738 | 152 | 0.786 |
| Heterozygous | 94 | 0.477 | 96 | 0.487 | 0 | 0 | 1 | 0.005 | 49 | 0.250 | 47 | 0.241 | 41 | 0.212 |
| Homozygous Variant | 34 | 0.173 | 42 | 0.213 | 0 | 0 | 0 | 0 | 2 | 0.010 | 4 | 0.021 | 0 | 0 |
| Totals (genotypes counts/freq.) | 197 | 1.000 | 197 | 1.000 | 196 | 1.000 | 196 | 1.000 | 196 | 1.000 | 195 | 1.000 | 193 | 1.000 |
| Alleles | ||||||||||||||
| Minor allele (variant) | 162 | 0.411 | 180 | 0.457 | 0 | 0 | 1 | 0.003 | 53 | 0.135 | 55 | 0.141 | 48 | 0.124 |
| Totals (allele counts/freq.) | 394 | 1.000 | 394 | 1.000 | 392 | 1.000 | 392 | 1.000 | 392 | 1.000 | 390 | 1.000 | 386 | 1.000 |
| Populations/Marker | ABCB1 N = 197; (MAFPR: 0.411) | PON1 N = 197; (MAFPR: 0.457) | CYP2C19*3 N = 196; (MAFPR: 0.000) | CYP2C19*4 N = 196; (MAFPR: 0.003) | CYP2C19*2 N = 196; (MAFPR: 0.135) | CYP2C19*17 N = 195; (MAFPR: 0.141) | P2RY12 H2 ** N = 193; (MAFPR: 0.124) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MAF | Sign. | MAF | Sign. | MAF | Sign. | MAF | Sign. | MAF | Sign. | MAF | Sign. | MAF | Sign. | |
| 1000 Genome Project Reference Populations | ||||||||||||||
| YRI (n = 88) | 0.119 | <0.01 * | 0.193 | <0.01 * | 0.000 | - | 0.000 | 0.630 | 0.165 | 0.510 | 0.256 | 0.019 * | 0.170 | 0.300 |
| CEU (n = 87) | 0.414 # | <0.01 * | 0.310 ¶ | <0.01 * | 0.000 | - | 0.000 | 0.630 | 0.138 | 0.950 | 0.224 | 0.083 | 0.195 | 0.120 |
| CHB (n = 97) | 0.397 | 0.820 | 0.397 | 0.330 | 0.046 | <0.01 * | 0.005 | 0.740 | 0.320 | <0.01 * | 0.260 | 0.013 * | 0.211 | 0.053 |
| AMR (n = 181) | 0.461 | 0.327 | 0.475 ¶ | 0.190 | 0.000 | - | 0.006 | 0.610 | 0.133 | 0.950 | 0.116 | 0.471 | 0.108 | 0.620 |
| Overall Population (n = 453) | 0.405 | 0.880 | 0.465 | 0.850 | 0.010 | 0.160 | 0.003 | 0.930 | 0.180 | 0.160 | 0.145 | 0.896 | 0.159 | 0.260 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernandez-Suarez, D.F.; Tomassini-Fernandini, J.C.; Cuevas, A.; Rosario-Berrios, A.N.; Nuñez-Medina, H.J.; Padilla-Arroyo, D.; Rivera, N.; Liriano, J.; Vega-Roman, R.K.; Renta, J.Y.; et al. Clinical Relevant Polymorphisms Affecting Clopidogrel Pharmacokinetics and Pharmacodynamics: Insights from the Puerto Rico Newborn Screening Program. Int. J. Environ. Res. Public Health 2018, 15, 1115. https://doi.org/10.3390/ijerph15061115
Hernandez-Suarez DF, Tomassini-Fernandini JC, Cuevas A, Rosario-Berrios AN, Nuñez-Medina HJ, Padilla-Arroyo D, Rivera N, Liriano J, Vega-Roman RK, Renta JY, et al. Clinical Relevant Polymorphisms Affecting Clopidogrel Pharmacokinetics and Pharmacodynamics: Insights from the Puerto Rico Newborn Screening Program. International Journal of Environmental Research and Public Health. 2018; 15(6):1115. https://doi.org/10.3390/ijerph15061115
Chicago/Turabian StyleHernandez-Suarez, Dagmar F., Jonnalie C. Tomassini-Fernandini, Angelica Cuevas, Anyelis N. Rosario-Berrios, Héctor J. Nuñez-Medina, Dariana Padilla-Arroyo, Nannette Rivera, Jennifer Liriano, Rocio K. Vega-Roman, Jessicca Y. Renta, and et al. 2018. "Clinical Relevant Polymorphisms Affecting Clopidogrel Pharmacokinetics and Pharmacodynamics: Insights from the Puerto Rico Newborn Screening Program" International Journal of Environmental Research and Public Health 15, no. 6: 1115. https://doi.org/10.3390/ijerph15061115
APA StyleHernandez-Suarez, D. F., Tomassini-Fernandini, J. C., Cuevas, A., Rosario-Berrios, A. N., Nuñez-Medina, H. J., Padilla-Arroyo, D., Rivera, N., Liriano, J., Vega-Roman, R. K., Renta, J. Y., Melin, K., & Duconge, J. (2018). Clinical Relevant Polymorphisms Affecting Clopidogrel Pharmacokinetics and Pharmacodynamics: Insights from the Puerto Rico Newborn Screening Program. International Journal of Environmental Research and Public Health, 15(6), 1115. https://doi.org/10.3390/ijerph15061115






