Alcoholism: A Multi-Systemic Cellular Insult to Organs
Abstract
1. Introduction
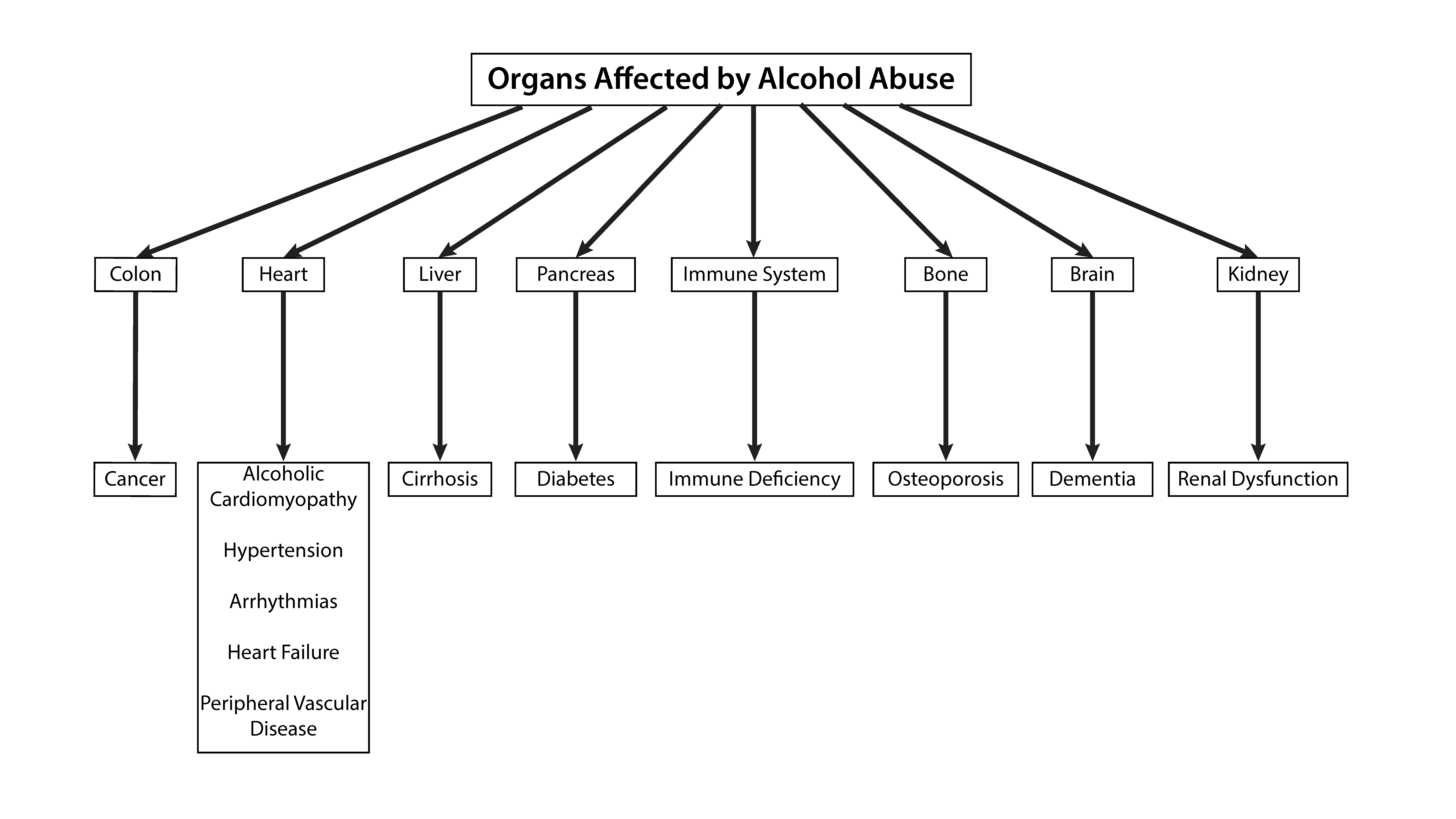
2. Systemic Effects of Alcohol
2.1. Cancer
2.2. Liver
2.3. Skeletal System
2.4. Kidney Disease
2.5. Pancreas
2.6. Immune System
2.7. Cardiovascular System
2.7.1. Clinical Studies and Public Health
2.7.2. Cellular Changes
2.8. Central Nervous System
2.9. Drugs: Effects of Alcohol
3. Epidemiology of Alcohol Abuse: Psychological and Societal Effects
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Peck, S.C.; Roeser, R.W.; Eccles, J.S. Exploring the roles of extracurricular activity quantity and quality in the educational resilience of vulnerable adolescents: Variable- and pattern-centered approaches. J. Soc. Issues 2008, 64, 20. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-V); American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Allen, N.A.L.; Benichou, J.; Beral, V.; Bloomfield, K.; Brooks, P.; Cai, L.; Crabb, D.; Eriiksson, P.; Gapstur, S.; Gmel, G.; et al. Alcohol consumption and ethyl carbamate. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2010; pp. 237–1280. [Google Scholar]
- Hamajima, N.; Hirose, K.; Tajima, K.; Rohan, T.; Calle, E.E.; Heath, C.W. Alcohol, tobacco and breast cancer—Collaborative reanalysis of individual data from 53 epidemiological studies, including 58 515 women with breast cancer and 95 067 women without the disease. Br. J. Cancer 2002, 87, 1234–1245. [Google Scholar] [PubMed]
- Na, H.K.; Lee, J.Y. Molecular Basis of Alcohol-Related Gastric and Colon Cancer. Int. J. Mol. Sci. 2017, 18, 1116. [Google Scholar] [CrossRef]
- Korsten, M.A.; Wilson, J.S.; Lieber, C.S. Interactive effects of dietary protein and ethanol on rat pancreas. Protein synthesis and enzyme secretion. Gastroenterology 1990, 99, 229–236. [Google Scholar] [CrossRef]
- Korsten, M.A.; Klapholz, M.B.; Leaf, M.A.; Lieber, C.S. Use of the triolein breath test in alcoholics with liver damage. J. Lab. Clin. Med. 1987, 109, 62–66. [Google Scholar] [PubMed]
- Babor, T.; Rehm, J.; Jernigan, D.; Vaeth, P.; Monteiro, M.; Lehman, H. Alcohol, diabetes, and public health in the Americas. Am. J. Public Health. 2012, 32, 151–155. [Google Scholar] [CrossRef]
- Abukhadir, S.S.; Mohamed, N.; Mohamed, N. Pathogenesis of Alcohol-Induced Osteoporosis and its Treatment: A Review. Curr. Drug Targets 2013, 14, 1601–1610. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.-S.; Liu, X.; Wang, L. Associations of alcohol consumption and mental health with the prevalence of arthritis among US adults: Data from the 2012 National Health Interview Survey. Rheumatol. Int. 2014, 34, 1241–1249. [Google Scholar] [CrossRef] [PubMed]
- Varga, Z.; Matyas, C.; Paloczi, J.; Pacher, P. Alcohol Misuse, and Kidney Injury: Epidemiological Evidence and Potential Mechanisms. Alcohol Res. Curr. Rev. 2017, 38, 283–288. [Google Scholar]
- Capurso, G.; Lahner, E. The interaction between smoking, alcohol and the gut microbiome. Best Pract. Res. Clin. Gastroenterol. 2017, 31, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Fagunwa, I.O.; Loughrey, M.B.; Coleman, H.G. Alcohol, smoking and the risk of premalignant and malignant colorectal neoplasms. Best Pract. Res. Clin. Gastroenterol. 2017, 31, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Nieminen, M.T.; Salaspuro, M. Local Acetaldehyde-An Essential Role in Alcohol-Related Upper Gastrointestinal Tract Carcinogenesis. Cancers 2018, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Curtis, B.J.; Zahs, A.; Kovacs, E.J. Epigenetic targets for reversing immune defects caused by alcohol exposure. Alcohol Res. 2013, 35, 97–113. [Google Scholar] [PubMed]
- Pan, X.Q.; Zhang, Y.H.; Liu, Y.Y.; Tong, W.J. Interaction between the C(-344)T polymorphism of CYP11B2 and alcohol consumption on the risk of essential hypertension in a Chinese Mongolian population. Eur. J. Epidemiol. 2010, 25, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Mukamal, K.J.; Krasnow, R.E.; Swan, G.E.; Reed, T. Higher usual alcohol consumption was associated with a lower 41-y mortality risk from coronary artery disease in men independent of genetic and common environmental factors: The prospective NHLBI Twin Study. Am. J. Clin. Nutr. 2015, 102, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Maisch, B. Alcoholic cardiomyopathy: The result of dosage and individual predisposition. Herz 2016, 41, 484–493. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Wallin, A.; Wolk, A. Alcohol consumption and risk of heart failure: Meta-analysis of 13 prospective studies. Clin. Nutr. 2017, in press. [Google Scholar] [CrossRef] [PubMed]
- Kuehn, D.; Aros, S.; Cassorla, F.; Avaria, M.; Unanue, N.; Henriquez, C.; Kleinsteuber, K.; Conca, B.; Avila, A.; Carter, T.C. A prospective cohort study of the prevalence of growth, facial, and central nervous system abnormalities in children with heavy prenatal alcohol exposure. Alcohol Clin. Exp. Res. 2012, 36, 1811–1819. [Google Scholar] [CrossRef] [PubMed]
- Neuberger, J.D. Treatment of liver disease. In Alcohol Abuse and Liver Disease; John Wiley and Sons Ltd.: Hoboken, NJ, USA, 2015. [Google Scholar]
- Jang, H.D.; Hong, J.Y.; Han, K.; Lee, J.C.; Shin, B.J.; Choi, S.W.; Suh, S.W.; Yang, J.H.; Park, S.Y.; Bang, C. Relationship between bone mineral density and alcohol intake: A nationwide health survey analysis of postmenopausal women. PLoS ONE 2017, 12, e0180132. [Google Scholar] [CrossRef] [PubMed]
- Baliunas, D.O.; Taylor, B.J.; Irving, H.; Roerecke, M.; Patra, J.; Mohapatra, S.; Rehm, J. Alcohol as a risk factor for type 2 diabetes: A systematic review and meta-analysis. Diabetes Care 2009, 32, 2123–2132. [Google Scholar] [CrossRef] [PubMed]
- Barr, T.; Helms, C.; Grant, K.; Messaoudi, I. Opposing effects of alcohol on the immune system. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 65, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Saha, B.; Momen-Heravi, F.; Furi, I.; Kodys, K.; Catalano, D.; Gangopadhyay, A.; Haraszti, R.; Satishchandran, A.; Adejumo, A.; Shaffer, S.A.; et al. Extracellular vesicles from mice with alcoholic liver disease carry a distinct protein cargo and induce macrophage activation through heat shock protein 90. Hepatology 2018, 67, 1986–2000. [Google Scholar] [CrossRef] [PubMed]
- Coleman, L.G.; Crews, F.T. Innate Immune Signaling and Alcohol Use Disorders. Handb. Exp. Pharmacol. 2018. [Google Scholar] [CrossRef]
- Romeo, J.; Warnberg, J.; Marcos, A. Drinking pattern and socio-cultural aspects on immune response: An overview. Proc. Nutr. Soc. 2010, 69, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Hillbom, M.; Juvela, S.; Numminen, H. Alcohol intake and the risk of stroke. J. Cardiovasc. Risk 1999, 6, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Tolstrup, J.S.; Wium-Andersen, M.K.; Ørsted, D.D.; Nordestgaard, B.G. Alcohol consumption and risk of atrial fibrillation: Observational and genetic estimates of association. Eur. J. Prev. Cardiol. 2016, 23, 1514–1523. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, C.; Hendriks, J.M.; Elliott, A.D.; Wong, C.X.; Rangnekar, G.; Middeldorp, M.E.; Mahajan, R.; Lau, D.H.; Sanders, P. Alcohol and incident atrial fibrillation—A systematic review and meta-analysis. Int. J. Cardiol. 2017, 246, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, D.P. Cardioprotective effects of light–moderate consumption of alcohol: A review of putative mechanisms. Alcohol Alcohol. 2002, 37, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Ronksley, P.E.; Brien, S.E.; Turner, B.J.; Mukamal, K.J.; Ghali, W.A. Association of alcohol consumption with selected cardiovascular disease outcomes: A systematic review and meta-analysis. Br. Med. J. 2011, 342, d671. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Franchini, M.; Favaloro, E.J.; Targher, G. Moderate red wine consumption and cardiovascular disease risk: Beyond the “French paradox”. Semin. Thromb. Hemost. 2010, 31, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Kalla, A.; Figueredo, V.M. Alcohol and cardiovascular disease in the geriatric population. Clin. Cardiol. 2017, 40, 444–449. [Google Scholar] [CrossRef] [PubMed]
- Briasoulis, A.; Agarwal, V.; Messerli, F.H. Alcohol consumption and the risk of hypertension in men and women: A systematic review and meta-analysis. J. Clin. Hypertens. 2012, 14, 792–798. [Google Scholar] [CrossRef] [PubMed]
- Umoh, N.A.; Walker, R.K.; Al-Rubaiee, M.; Jeffress, M.A.; Haddad, G.E. Acute alcohol modulates cardiac function as PI3K/Akt regulates oxidative stress. Alcohol. Clin. Exp. Res. 2014, 38, 1847–1864. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.K.; Cousins, V.M.; Umoh, N.A.; Jeffress, M.A.; Taghipour, D.; Al-Rubaiee, M.; Haddad, G.E. The good, the bad, and the ugly with alcohol use and abuse on the heart. Alcohol. Clin. Exp. Res. 2013, 37, 1253–1260. [Google Scholar] [CrossRef] [PubMed]
- Shiferaw, Y.; Aistrup, G.L.; Wasserstrom, J.A. Mechanism for Triggered Waves in Atrial Myocytes. Biophys. J. 2017, 113, 656–670. [Google Scholar] [CrossRef] [PubMed]
- Steiner, J.L.; Lang, C.H. Alcoholic Cardiomyopathy: Disrupted Protein Balance and Impaired Cardiomyocyte Contractility. Alcohol. Clin. Exp. Res. 2017, 41, 1392–1401. [Google Scholar] [CrossRef] [PubMed]
- Oscar-Berman, M. Alcoholism and asymmetries of brain function. Alcohol Health Res. World 1992, 16, 273. [Google Scholar]
- Nussbaum, R.L.; Ellis, C.E. Alzheimer’s disease and Parkinson’s disease. N. Eng. J. Med. 2003, 348, 1356–1364. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.J.; Zhang, X.; Chen, W.W. Association between alcohol and Alzheimer’s disease. Exp. Ther. Med. 2016, 12, 1247–1250. [Google Scholar] [CrossRef] [PubMed]
- Langballe, E.M.; Ask, H.; Holmen, J.; Stordal, E.; Saltvedt, I.; Selbæk, G.; Fikseaunet, A.; Bergh, S.; Nafstad, P.; Tambs, K. Alcohol consumption and risk of dementia up to 27 years later in a large, population-based sample: The HUNT study, Norway. Eur. J. Epidemiol. 2015, 30, 1049–1056. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.; Bateman, H.; Djonokusumo, E.; Hillmore, R.; Jackson, D.; Lakhani, I.; Lusznut, S.; McAdam, K.; Sargeant, H. Dictionary of Nursing; A&C Black Publishers Ltd.: Edinburgh, UK, 2007; pp. 346–348. [Google Scholar]
- Brokate, B.; Hildebrandt, H.; Eling, P.A.T. M.; Fichtner, H.; Runge, K.; Timm, C. Frontal lobe dysfunctions in Korsakoff’s syndrome and chronic alcoholism: Continuity or discontinuity? Neuropsychology 2003, 17, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-J.A.M.; Susan, E.; Parnell, S.E.; West, J.R. Alcohol and the Developing Brain: Neuroanatomical Studies. Alcohol Res. 2003, 27, 174–180. [Google Scholar]
- Mariën, P.P.P.F. A synthesis of the role of the cerebellum in cognition. Aphasiology 2005, 19, 16. [Google Scholar] [CrossRef]
- Letenneur, L. Risk of dementia and alcohol and wine consumption: A review of recent results. Biol. Res. 2004, 37, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.M.; Zheng, W.B.; Lian, G.P.; Zhang, H.D. Acute Effects of Alcohol on the Human Brain: Diffusion Tensor Imaging Study. Am. J. Neuroradiol. 2012, 33, 928–934. [Google Scholar] [CrossRef] [PubMed]
- Allan, A.M.; Harris, R.A. Gamma-aminobutyric acid and alcohol actions: Neurochemical studies of long sleep and short sleep mice. Life Sci. 1986, 39, 2005–2015. [Google Scholar] [CrossRef]
- Bjork, J.M.; Gilman, J.M. The effects of acute alcohol administration on the human brain: Insights from neuroimaging. Neuropharmacology 2014, 84, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Preidt, R. Opioids and Alcohol a Dangerous Cocktail: Drinking while Taking the Painkillers can Hamper Breathing, Especially among Seniors, Study Finds. Available online: https://www.webmd.com/mental-health/addiction/news/20170208/opioids-and-alcohol-a-dangerous-cocktail (accessed on 17 May 2018).
- Nathanson, V.; Jayesinghe, N.; Roycroft, G. Is it all right for women to drink small amounts of alcohol in pregnancy? No. BMJ 2007, 335, 857. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zheng, J.; Li, S.; Zhou, T.; Zhang, P.; Li, H.B. Alcoholic beverage consumption and chronic diseases. Int. J. Environ. Res. Public Health 2016, 13, 522. [Google Scholar] [CrossRef] [PubMed]
- Stockley, C.S. Is it merely a myth that alcoholic beverages such as red wine can be cardioprotective? J. Sci. Food Agric. 2012, 92, 1815–1821. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dguzeh, U.; Haddad, N.C.; Smith, K.T.S.; Johnson, J.O.; Doye, A.A.; Gwathmey, J.K.; Haddad, G.E. Alcoholism: A Multi-Systemic Cellular Insult to Organs. Int. J. Environ. Res. Public Health 2018, 15, 1083. https://doi.org/10.3390/ijerph15061083
Dguzeh U, Haddad NC, Smith KTS, Johnson JO, Doye AA, Gwathmey JK, Haddad GE. Alcoholism: A Multi-Systemic Cellular Insult to Organs. International Journal of Environmental Research and Public Health. 2018; 15(6):1083. https://doi.org/10.3390/ijerph15061083
Chicago/Turabian StyleDguzeh, Ucee, Natasha C. Haddad, Kathia T. S. Smith, John O. Johnson, Angelia A. Doye, Judith K. Gwathmey, and Georges E. Haddad. 2018. "Alcoholism: A Multi-Systemic Cellular Insult to Organs" International Journal of Environmental Research and Public Health 15, no. 6: 1083. https://doi.org/10.3390/ijerph15061083
APA StyleDguzeh, U., Haddad, N. C., Smith, K. T. S., Johnson, J. O., Doye, A. A., Gwathmey, J. K., & Haddad, G. E. (2018). Alcoholism: A Multi-Systemic Cellular Insult to Organs. International Journal of Environmental Research and Public Health, 15(6), 1083. https://doi.org/10.3390/ijerph15061083




