Exercise Early and Often: Effects of Physical Activity and Exercise on Women’s Bone Health
Abstract
1. Introduction
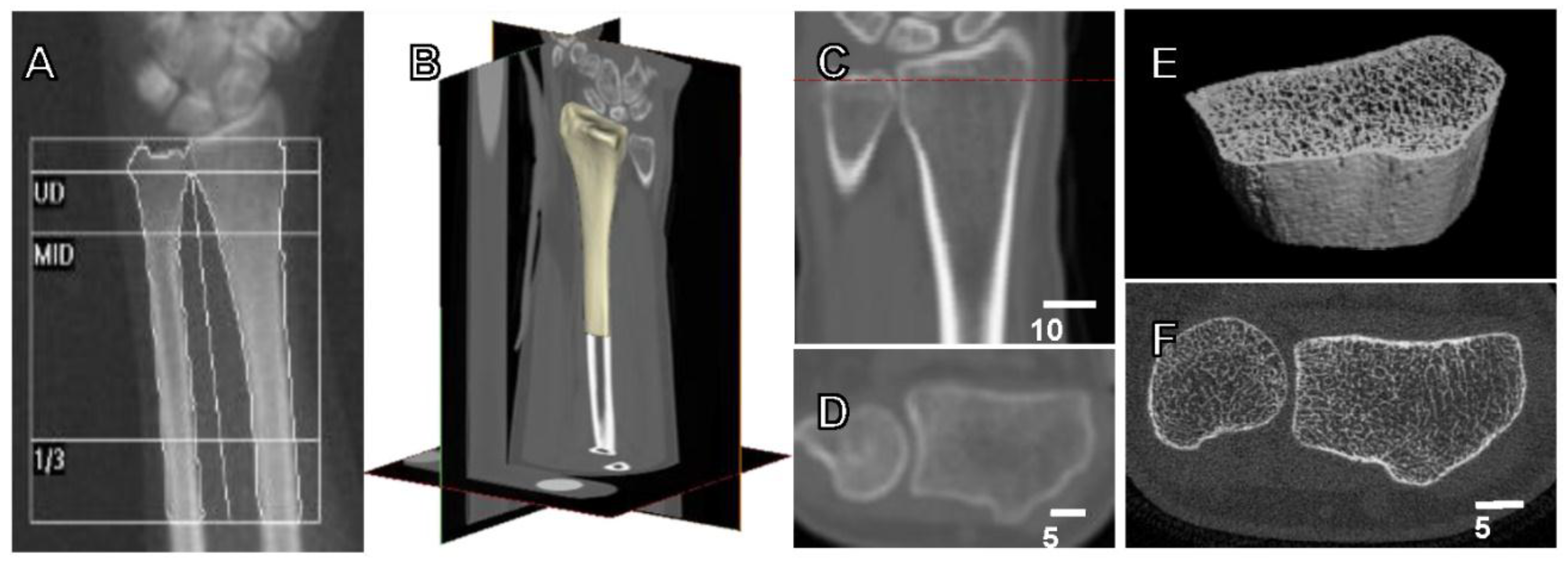
2. Measurement of Bone Strength and Fracture Risk
3. Bone Adaptation and the Biological Basis for Why Physical Activity and Exercise Matters
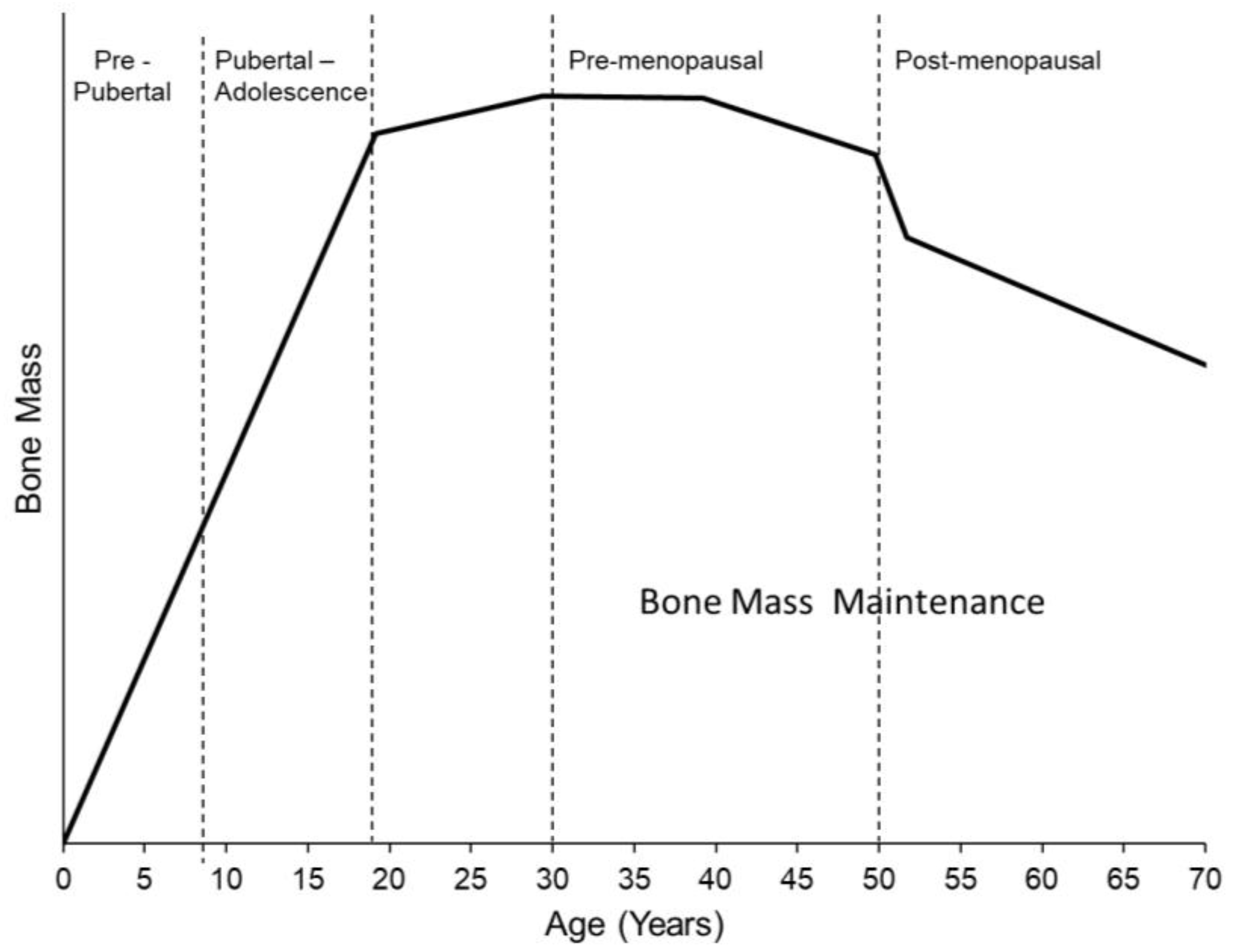
4. When in Life Does Physical Activity and Exercise Matter the Most?
5. Which Specific Types of Physical Activity Are Best for Bone?
6. Emerging Areas of Research about Exercise and Women’s Bone Health
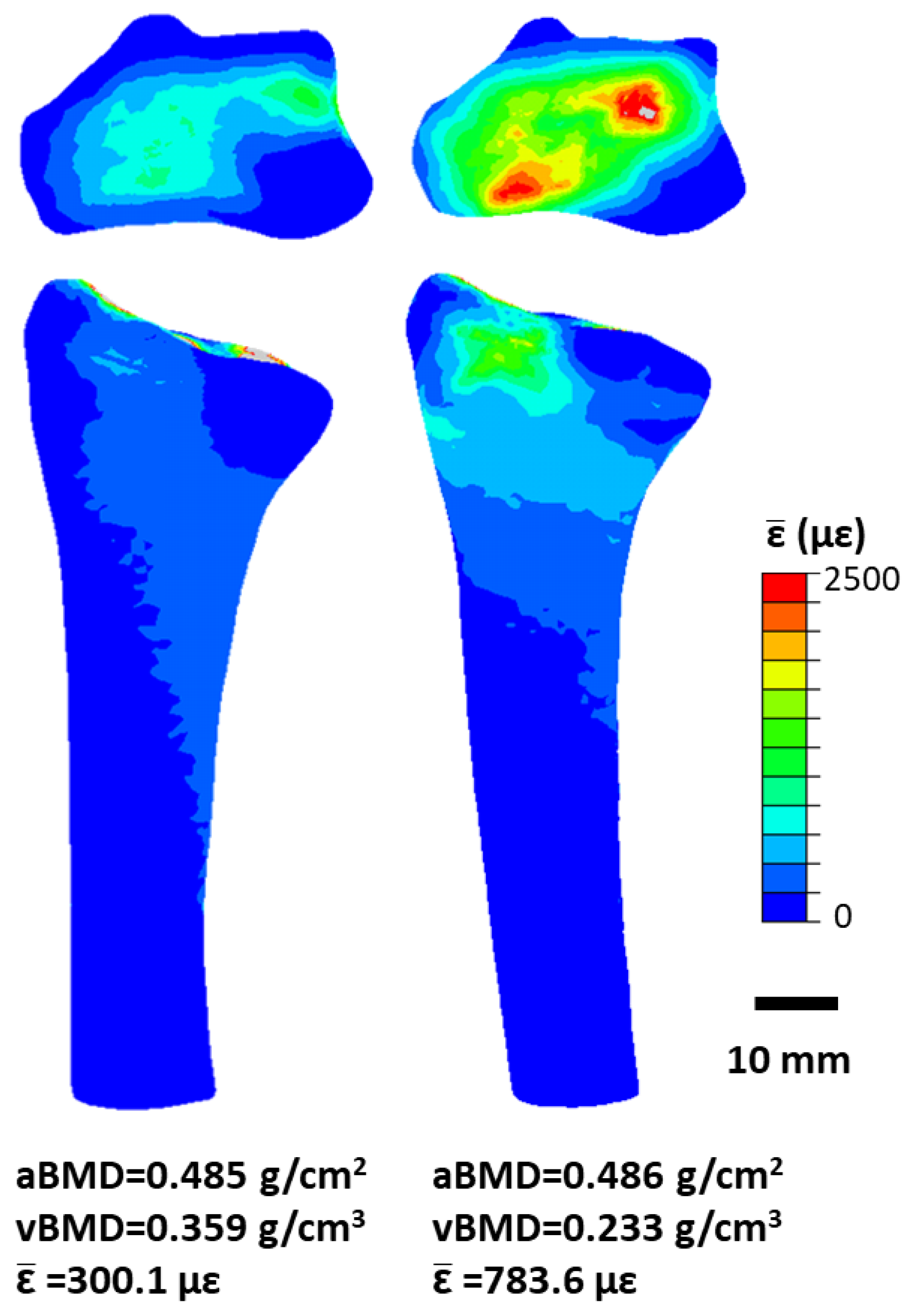
6.1. Measuring Bone Loading In Vivo
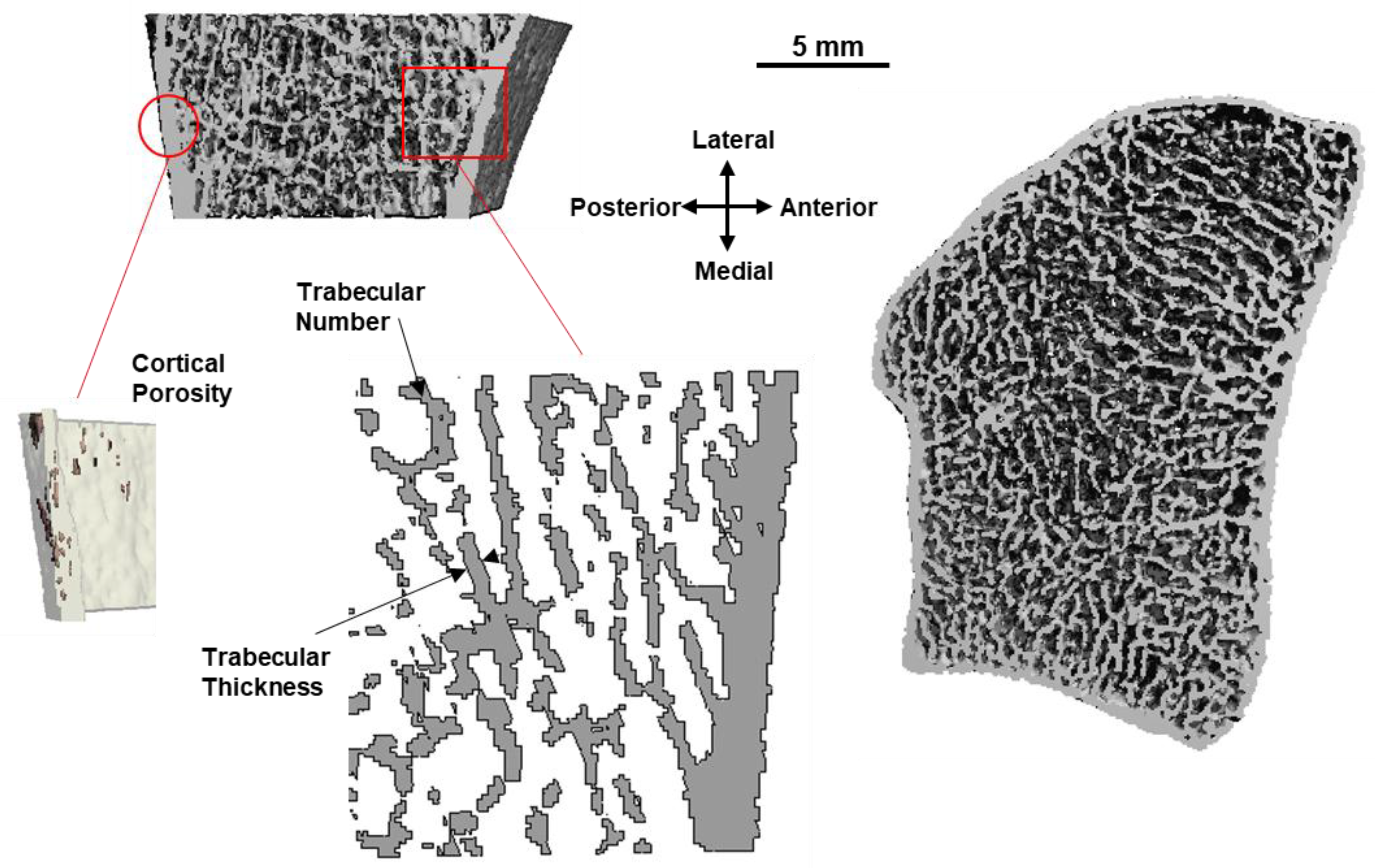
6.2. 3D and High Resolution Imaging of Bone
6.3. Detecting the Short-Term Response to Osteoporosis Treatment
6.4. Interactions between Drugs for Osteoporosis and Exercise
7. Conclusions
- Adolescent and prepubertal girls can derive the greatest benefit from bone-loading exercise. In this age group, exercise is an effective means of increasing peak bone mass, which provides lifelong fracture protection.
- High-impact exercises such as jumping or hopping, or resistance training combined with high- or odd-impact activities, are most consistently effective for bone.
- Two to four short (30 min/day or less) exercise sessions per week over a prolonged period are required to maintain or improve bone.
- For older women who have risk factors that prevent them from participating in high-impact activities, other weight-bearing activities such as resistance training, specific yoga postures, or walking, may maintain or improve bone.
- Other activities that preserve or improve mobility and strength are also beneficial, because they reduce fall risk, thereby reducing fracture risk.
Author Contributions
Acknowledgments
Conflicts of Interest
References
- United States Bone and Joint Initiative. The Burden of Musculoskeletal Diseases in the United States, 3rd ed.; Saag, K., Wright, N.C., Kilgore, M., Eds.; American Academy or Orthopaedic Surgeons: Rosemont, IL, USA, 2014; ISBN 9780996309103. [Google Scholar]
- Kanis, J.A.; Oden, A.; Johansson, H.; Borgström, F.; Ström, O.; McCloskey, E. FRAX®and its applications to clinical practice. Bone 2009, 44, 734–743. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, C.J.; Beaupré, G.S.; Carter, D.R. A theoretical analysis of the relative influences of peak BMD, age-related bone loss and menopause on the development of osteoporosis. Osteoporos. Int. 2003, 14, 843–847. [Google Scholar] [CrossRef] [PubMed]
- Cosman, F.; Lindsay, R.; LeBoff, M.S.; Jan de Beur, S.; Tanner, B. Clinician’s Guide to Prevention and Treatment of Osteoporosis. Natl. Osteoporos. Found. 2014, 1, 55. [Google Scholar] [CrossRef] [PubMed]
- National Osteoporosis Foundation. Osteoporosis Exercise for Strong Bones. Available online: https://www.nof.org/patients/fracturesfall-prevention/exercisesafe-movement/osteoporosis-exercise-for-strong-bones/ (accessed on 3 March 2018).
- Pfeifer, M.; Minne, H.; International Osteoporosis Foundation. Bone Loading Exercise Recommendations for Prevention and Treament of Osteoporosis. 2015. Available online: https://www.iofbonehealth.org/exercise-recommendations (accessed on 4 March 2018).
- NIAMS. Handout on Health: Osteoporosis. Available online: https://www.bones.nih.gov/health-info/bone/osteoporosis/osteoporosis-hoh (accessed on 4 March 2018).
- Martin, R.B.; Burr, D.B.; Sharkey, N.A.; Fyhrie, D.P. Skeletal Tissue Mechanics; Springer: New York, NY, USA, 2015; Volume 112, ISBN 0387984747. [Google Scholar]
- Morgan, E.F.; Bayraktar, H.H.; Keaveny, T.M. Trabecular bone modulus-density relationships depend on anatomic site. J. Biomech. 2003, 36, 897–904. [Google Scholar] [CrossRef]
- Keller, T.S. Predicting the compressive mechanical behavior of bone. J. Biomech. 1994, 27, 1159–1168. [Google Scholar] [CrossRef]
- Rho, J.Y.; Ashman, R.B.; Turner, C.H. Young’s modulus of trabecular and cortical bone material: Ultrasonic and microtensile measurements. J. Biomech. 1993, 26, 111–119. [Google Scholar] [CrossRef]
- Rodriguez-Florez, N.; Garcia-Tunon, E.; Mukadam, Q.; Saiz, E.; Oldknow, K.J.; Farquharson, C.; Millán, J.L.; Boyde, A.; Shefelbine, S.J. An investigation of the mineral in ductile and brittle cortical mouse bone. J. Bone Miner. Res. 2015, 30, 786–795. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Odgaard, A.; Linde, F.; Hvid, I. Age-related variations in the microstructure of human tibial cancellous bone. J. Orthop. Res. 2002, 20, 615–621. [Google Scholar] [CrossRef]
- Lewiecki, E.M.; Gordon, C.M.; Baim, S.; Leonard, M.B.; Bishop, N.J.; Bianchi, M.L.; Kalkwarf, H.J.; Langman, C.B.; Plotkin, H.; Rauch, F.; et al. The International Society for Clinical Densitometry. Official positions & pediatric official positions 2007. Bone 2008, 43, 1115–1121. [Google Scholar] [PubMed]
- Cody, D.D.; Gross, G.J.; Hou, F.J.; Spencer, H.J.; Goldstein, S.A.; Fyhire, D.P. Femoral strength is predicted better by finite element models than QCT and DXA. J. Biomech. 1999, 32, 1013–1020. [Google Scholar] [CrossRef]
- Johnell, O.; Kanis, J.A.; Oden, A.; Johansson, H.; De Laet, C.; Delmas, P.; Eisman, J.A.; Fujiwara, S.; Kroger, H.; Mellstrom, D.; et al. Predictive value of BMD for hip and other fractures. J. Bone Miner. Res. 2005, 20, 1185–1194. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J. WHO Scientific Group on the assessment of osteoporosis at the primary health care level. Available online: http://www.who.int/chp/topics/Osteoporosis.pdf (accessed on 27 April 2018).
- Edwards, W.B.; Schnitzer, T.J.; Troy, K.L. Torsional stiffness and strength of the proximal tibia are better predicted by finite element models than DXA or QCT. J. Biomech. 2013, 46, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Johannesdottir, F.; Thrall, E.; Muller, J.; Keaveny, T.M.; Kopperdahl, D.L.; Bouxsein, M.L. Comparison of non-invasive assessments of strength of the proximal femur. Bone 2017, 105, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Bareither, M.L.; Troy, K.L.; Grabiner, M.D. Bone mineral density of the proximal femur is not related to dynamic joint loading during locomotion in young women. Bone 2006, 38, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Dowthwaite, J.N.; Rosenbaum, P.F.; Scerpella, T.A. Mechanical loading during growth is associated with plane-specific differences in vertebral geometry: A cross-sectional analysis comparing artistic gymnasts vs. non-gymnasts. Bone 2011, 49, 1046–1054. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kontulainen, S.; Sievänen, H.; Kannus, P.; Pasanen, M.; Vuori, I. Effect of long-term impact-loading on mass, size, and estimated strength of humerus and radius of female racquet-sports players: A peripheral quantitative computed tomography study between young and old starters and controls. J. Bone Miner. Res. 2003, 18, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Frost, H.M. Bone’s mechanostat: A 2003 update. Anat. Rec. Part A 2003, 275A, 1081–1101. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, V.A.; Brent Edwards, W.; Johnson, J.E.; Troy, K.L. Short-Term Bone Formation is Greatest Within High Strain Regions of the Human Distal Radius: A Prospective Pilot Study. J. Biomech. Eng. 2015, 137. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.C.; Costa, A.G.; Cusano, N.E.; Kousteni, S.; Bilezikian, J.P. Catabolic and anabolic actions of parathyroid hormone on the skeleton. J. Endocrinol. Invest. 2011, 34, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Noble, B.S.; Peet, N.; Stevens, H.Y.; Brabbs, A.; Mosley, J.R.; Reilly, G.C.; Reeve, J.; Skerry, T.M.; Lanyon, L.E. Mechanical loading: Biphasic osteocyte survival and targeting of osteoclasts for bone destruction in rat cortical bone. AJP Cell Physiol. 2003, 284, C934–C943. [Google Scholar] [CrossRef] [PubMed]
- Klein-Nulend, J.; van Oers, R.F.M.; Bakker, A.D.; Bacabac, R.G. Bone cell mechanosensitivity, estrogen deficiency, and osteoporosis. J. Biomech. 2015, 48, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Mosley, J.R.; Lanyon, L.E. Strain rate as a controlling influence on adaptive modeling in response to dynamic loading of the ulna in growing male rats. Bone 1998, 23, 313–318. [Google Scholar] [CrossRef]
- Turner, C.H. Three rules for bone adaptation to mechanical stimuli. Bone 1998, 23, 399–407. [Google Scholar] [CrossRef]
- Shea, K.L.; Gavin, K.M.; Melanson, E.L.; Gibbons, E.; Stavros, A.; Wolfe, P.; Kittelson, J.M.; Vondracek, S.F.; Schwartz, R.S.; Wierman, M.E.; et al. Body composition and bone mineral density after ovarian hormone suppression with or without estradiol treatment. Menopause 2015, 22, 1045–1052. [Google Scholar] [CrossRef] [PubMed]
- Tan, V.P.; Macdonald, H.M.; Kim, S.; Nettlefold, L.; Gabel, L.; Ashe, M.C.; McKay, H.A. Influence of Physical Activity on Bone Strength in Children and Adolescents: A Systematic Review and Narrative Synthesis. J. Bone Miner. Res. 2014, 29, 2161–2181. [Google Scholar] [CrossRef] [PubMed]
- Henry, Y.M.; Fatayerji, D.; Eastell, R. Attainment of peak bone mass at the lumbar spine, femoral neck and radius in men and women: Relative contributions of bone size and volumetric bone mineral density. Osteoporos. Int. 2004, 15, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Baxter-Jones, A.D.G.; Faulkner, R.A.; Forwood, M.R.; Mirwald, R.L.; Bailey, D.A. Bone mineral accrual from 8 to 30 years of age: An estimation of peak bone mass. J. Bone Miner. Res. 2011, 26, 1729–1739. [Google Scholar] [CrossRef] [PubMed]
- Lorentzon, M.; Mellström, D.; Ohlsson, C. Age of attainment of peak bone mass is site specific in Swedish men–The GOOD study. J. Bone Miner. Res. 2005, 20, 1223–1227. [Google Scholar] [CrossRef] [PubMed]
- Specker, B.; Thiex, N.W.; Sudhagoni, R.G. Does Exercise Influence Pediatric Bone? A Systematic Review. Clin. Orthop. Relat. Res. 2015, 473, 3658–3672. [Google Scholar] [CrossRef] [PubMed]
- Weeks, B.K.; Beck, B.R. The BPAQ: A bone-specific physical activity assessment instrument. Osteoporos. Int. 2008, 19, 1567–1577. [Google Scholar] [CrossRef] [PubMed]
- Hind, K.; Burrows, M. Weight-bearing exercise and bone mineral accrual in children and adolescents: A review of controlled trials. Bone 2007, 40, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Nikander, R.; Sievänen, H.; Heinonen, A.; Daly, R.M.; Uusi-Rasi, K.; Kannus, P. Targeted exercise against osteoporosis: A systematic review and meta-analysis for optimising bone strength throughout life. BMC Med. 2010, 8, 47. [Google Scholar] [CrossRef] [PubMed]
- Morris, F.L.; Naughton, G.A.; Gibbs, J.L.; Carlson, J.S.; Wark, J.D. Prospective ten-month exercise intervention in premenarcheal girls: Positive effects on bone and lean mass. J. Bone Miner. Res. 1997, 12, 1453–1462. [Google Scholar] [CrossRef] [PubMed]
- Bradney, M.; Pearce, G.; Naughton, G.; Sullivan, C.; Bass, S.; Beck, T.; Carlson, J.; Seeman, E. Moderate exercise during growth in prepubertal boys: Changes in bone mass, size, volumetric density, and bone strength: A controlled prospective study. J. Bone Miner. Res. 1998, 13, 1814–1821. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, R.K.; Bauer, J.J.; Snow, C.M. Jumping improves hip and lumbar spine bone mass in prepubescent children: A randomized controlled trial. J. Bone Miner. Res. 2001, 16, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, A.; Sievänen, H.; Kannus, P.; Oja, P.; Pasanen, M.; Vuori, I. High-impact exercise and bones of growing girls: A 9-month controlled trial. Osteoporos. Int. 2000, 11, 1010–1017. [Google Scholar] [CrossRef] [PubMed]
- MacKelvie, K.J.; McKay, H.A.; Petit, M.A.; Moran, O.; Khan, K.M. Bone mineral response to a 7-month randomized controlled, school-based jumping intervention in 121 prepubertal boys: Associations with ethnicity and body mass index. J. Bone Miner. Res. 2002, 17, 834–844. [Google Scholar] [CrossRef] [PubMed]
- Gunter, K.; Baxter-Jones, A.D.G.; Mirwald, R.L.; Almstedt, H.; Fuchs, R.K.; Durski, S.; Snow, C. Impact exercise increases BMC during growth: An 8-year longitudinal study. J. Bone Miner. Res. 2008, 23, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Kontulainen, S.A.; Kannus, P.A.; Pasanen, M.E.; Sievänen, H.T.; Heinonen, A.O.; Oja, P.; Vuori, I. Does previous participation in high-impact training result in residual bone gain in growing girls? Int. J. Sports Med. 2002, 23, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, A.; Sievänen, H.; Kannus, P.; Oja, P.; Vuori, I. Site-specific skeletal response to long-term weight training seems to be attributable to principal loading modality: A pQCT study of female weightlifters. Calcif. Tissue Int. 2002, 70, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Ducher, G.; Bass, S.L.; Saxon, L.; Daly, R.M. Effects of repetitive loading on the growth-induced changes in bone mass and cortical bone geometry: A 12-month study in pre/peri- and postmenarcheal tennis players. J. Bone Miner. Res. 2011, 26, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Forwood, M.R.; Burr, D.B. Physical activity and bone mass: Exercises in futility? Bone Miner. 1993, 21, 89–112. [Google Scholar] [CrossRef]
- Haapasalo, H.; Kannus, P.; Sievänen, H.; Pasanen, M.; Uusi-Rasi, K.; Heinonen, A.; Oja, P.; Vuori, I. Effect of long-term unilateral activity on bone mineral density of female junior tennis players. J. Bone Miner. Res. 1998, 13, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Keaveny, T.M.; Kopperdahl, D.L.; Melton, L.J.; Hoffmann, P.F.; Amin, S.; Riggs, B.L.; Khosla, S. Age-dependence of femoral strength in white women and men. J. Bone Miner. Res. 2010, 25, 994–1001. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Cabello, A.; Ara, I.; González-Agüero, A.; Casajus, J.A.; Vicente-Rodriguez, G. Effects of training on bone mass in older adults: A systematic review. Sports Med. 2012, 42, 301–325. [Google Scholar] [CrossRef] [PubMed]
- Howe, T.E.; Shea, B.; Dawson, L.J.; Downie, F.; Murray, A.; Ross, C.; Harbour, R.T.; Caldwell, L.M.; Creed, G. Exercise for preventing and treating osteoporosis in postmenopausal women. Cochrane Database Syst. Rev. 2011, CD000333. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Xu, Z.; Zhao, M. Antiresorptive agents increase the effects of exercise on preventing postmenopausal bone loss in women: A meta-analysis. PLoS ONE 2015, 10, e0116729. [Google Scholar] [CrossRef] [PubMed]
- Martyn-St James, M.; Carroll, S. Meta-analysis of walking for preservation of bone mineral density in postmenopausal women. Bone 2008, 43, 521–531. [Google Scholar] [CrossRef] [PubMed]
- Wallace, B.A.; Cumming, R.G. Systematic review of randomized trials of the effect of exercise on bone mass in pre- and postmenopausal women. Calcif. Tissue Int. 2000, 67, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Zehnacker, C.H.; Bemis-Dougherty, A. Effect of weighted exercises on bone mineral density in post menopausal women. A systematic review. J. Geriatr. Phys. Ther. 2007, 30, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Martyn-St James, M.; Carroll, S. Progressive High-Intensity Resistance Training and Bone Mineral Density Changes Among Premenopausal Women. Sports Med. 2006, 36, 683–704. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, C.J.; Swan, V.J.D.; Jamal, S.A. The effects of exercise and physical activity participation on bone mass and geometry in postmenopausal women: A systematic review of pQCT studies. Osteoporos. Int. 2010, 21, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Polidoulis, I.; Beyene, J.; Cheung, M. The effect of exercise on pQCT parameters of bone structure and strength in postmenopausal women—A systematic review and meta-analysis of randomized controlled trials. Osteoporos. Int. 2012, 23, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Harding, A.; Beck, B. Exercise, Osteoporosis, and Bone Geometry. Sports 2017, 5, 29. [Google Scholar] [CrossRef]
- Watson, S.L.; Weeks, B.K.; Weis, L.J.; Harding, A.T.; Horan, S.A.; Beck, B.R. High-Intensity Resistance and Impact Training Improves Bone Mineral Density and Physical Function in Postmenopausal Women With Osteopenia and Osteoporosis: The LIFTMOR Randomized Controlled Trial. J. Bone Miner. Res. 2018, 33, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Zhang, M.; Zhang, Q. The Effectiveness of Combined Exercise Interventions for Preventing Postmenopausal Bone Loss: A Systematic Review and Meta-analysis. J. Orthop. Sports Phys. Ther. 2017, 47, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Sherk, V.D.; Barry, D.W.; Villalon, K.L.; Hansen, K.C.; Wolfe, P.; Kohrt, W.M. Bone loss over one year of training and competition in female cyclists. Clin. J. Sport Med. 2014, 24, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Bruton, A.; Gónzalez-Agüero, A.; Gómez-Cabello, A.; Casajús, J.A.; Vicente-Rodríguez, G. Is Bone Tissue Really Affected by Swimming? A Systematic Review. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Nikander, R.; Sievänen, H.; Uusi-Rasi, K.; Heinonen, A.; Kannus, P. Loading modalities and bone structures at nonweight-bearing upper extremity and weight-bearing lower extremity: A pQCT study of adult female athletes. Bone 2006, 39, 886–894. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.H.; Rosner, B.; Chang, G.; Fishman, L.M. Twelve-minute daily yoga regimen reverses osteoporotic bone loss. Top. Geriatr. Rehabil. 2016, 32, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Zhao, M.; Xu, Z. The effects of differing resistance training modes on the preservation of bone mineral density in postmenopausal women: A meta-analysis. Osteoporos. Int. 2015, 26, 1605–1618. [Google Scholar] [CrossRef] [PubMed]
- Guilak, F. Biomechanical factors in osteoarthritis. Best Pract. Res. Clin. Rheumatol. 2011, 25, 815–823. [Google Scholar] [CrossRef] [PubMed]
- Andriacchi, T.P.; Mündermann, A.; Smith, R.L.; Alexander, E.J.; Dyrby, C.O.; Koo, S. A framework for the in vivo pathomechanics of osteoarthritis at the knee. Ann. Biomed. Eng. 2004, 32, 447–457. [Google Scholar] [CrossRef] [PubMed]
- Alentorn-Geli, E.; Samuelsson, K.; Musahl, V.; Green, C.L.; Bhandari, M.; Karlsson, J. The Association of Recreational and Competitive Running With Hip and Knee Osteoarthritis: A Systematic Review and Meta-analysis. J. Orthop. Sports Phys. Ther. 2017, 47, 373–390. [Google Scholar] [CrossRef] [PubMed]
- De Kam, D.; Smulders, E.; Weerdesteyn, V.; Smits-Engelsman, B.C.M. Exercise interventions to reduce fall-related fractures and their risk factors in individuals with low bone density: A systematic review of randomized controlled trials. Osteoporos. Int. 2009, 20, 2111–2125. [Google Scholar] [CrossRef] [PubMed]
- Lanyon, L.E.; Hampson, W.G.J.; Goodship, A.E.; Shah, J.S. Bone Deformation Recorded in vivo from Strain Gauges Attached to the Human Tibial Shaft. Acta Orthop. Scand. 1975, 46, 256–268. [Google Scholar] [CrossRef] [PubMed]
- Burr, D.B.; Milgrom, C.; Fyhrie, D.; Forwood, M.; Nyska, M.; Finestone, A.; Hoshaw, S.; Saiag, E.; Simkin, A. In vivo measurement of human tibial strains during vigorous activity. Bone 1996, 18, 405–410. [Google Scholar] [CrossRef]
- Milgrom, C.; Finestone, A.; Simkin, A.; Ekenman, I.; Mendelson, S.; Millgram, M.; Nyska, M.; Larsson, E.; Burr, D. In-vivo strain measurements to evaluate the strengthening potential of exercises on the tibial bone. J. Bone Jt. Surg. Br. Vol. 2000, 82, 591–594. [Google Scholar] [CrossRef]
- Földhazy, Z.; Arndt, A.; Milgrom, C.; Finestone, A.; Ekenman, I. Exercise-induced strain and strain rate in the distal radius. Bone Jt. J. 2005, 87, 261–266. [Google Scholar] [CrossRef]
- Yang, P.F.; Sanno, M.; Ganse, B.; Koy, T.; Brüggemann, G.P.; Müller, L.P.; Rittweger, J. Torsion and antero-posterior bending in the in vivo human tibia loading regimes during walking and running. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.F.; Kriechbaumer, A.; Albracht, K.; Sanno, M.; Ganse, B.; Koy, T.; Shang, P.; Brüggemann, G.P.; Müller, L.P.; Rittweger, J. On the relationship between tibia torsional deformation and regional muscle contractions in habitual human exercises in vivo. J. Biomech. 2015, 48, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, V.A.; Edwards, W.B.; Troy, K.L. Predicting surface strains at the human distal radius during an in vivo loading task–Finite element model validation and application. J. Biomech. 2014, 47, 2759–2765. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Troy, K.L.; Edwards, W.B.; Bhatia, V.A.; Bareither, M. Lou In vivo loading model to examine bone adaptation in humans: A pilot study. J. Orthop. Res. 2013, 31, 1406–1413. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, M.E.; Johnson, J.E.; Ahmed, S.S.; Butler, T.A.; Troy, K.L. Distal Radius Microstructure and Finite Element Bone Strain Are Related to Site-Specific Mechanical Loading and Areal Bone Mineral Density in Premenopausal Women. Bone Rep. 2018. under review. [Google Scholar] [CrossRef]
- Engelke, K.; Lang, T.; Khosla, S.; Qin, L.; Zysset, P.; Leslie, W.D.; Shepherd, J.A.; Schousboe, J.T. Clinical Use of Quantitative Computed Tomography (QCT) of the Hip in the Management of Osteoporosis in Adults: The 2015 ISCD Official Positions-Part I. J. Clin. Densitom. 2015, 18, 338–358. [Google Scholar] [CrossRef] [PubMed]
- Keaveny, T.M.; Crittenden, D.B.; Bolognese, M.A.; Genant, H.K.; Engelke, K.; Oliveri, B.; Brown, J.P.; Langdahl, B.L.; Yan, C.; Grauer, A.; et al. Greater Gains in Spine and Hip Strength for Romosozumab Compared With Teriparatide in Postmenopausal Women With Low Bone Mass. J. Bone Miner. Res. 2017, 32, 1956–1962. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.C.; Varela, A.; Kostenuik, P.J.; Ominsky, M.S.; Keaveny, T.M. Finite Element Analysis of Denosumab Treatment Effects on Vertebral Strength in Ovariectomized Cynomolgus Monkeys. J. Bone Miner. Res. 2016, 31, 1586–1595. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.C.; Hoffmann, P.F.; Kopperdahl, D.L.; Keaveny, T.M. Phantomless calibration of CT scans for measurement of BMD and bone strength—Inter-operator reanalysis precision. Bone 2017, 103, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Laib, A.; Hauselmann, H.J.; Ruegsegger, P. In vivo high resolution 3D-QCT of the human forearm. Technol. Heal. Care 1998, 6, 329–337. [Google Scholar]
- Manske, S.L.; Zhu, Y.; Sandino, C.; Boyd, S.K. Human trabecular bone microarchitecture can be assessed independently of density with second generation HR-pQCT. Bone 2015, 79, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Kroker, A.; Zhu, Y.; Manske, S.L.; Barber, R.; Mohtadi, N.; Boyd, S.K. Quantitative in vivo assessment of bone microarchitecture in the human knee using HR-pQCT. Bone 2017, 97, 43–48. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, H.M.; Nishiyama, K.K.; Kang, J.; Hanley, D.A.; Boyd, S.K. Age-related patterns of trabecular and cortical bone loss differ between sexes and skeletal sites: A population-based HR-pQCT study. J. Bone Miner. Res. 2011, 26, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Kawalilak, C.E.; Johnston, J.D.; Olszynski, W.P.; Kontulainen, S.A. Characterizing microarchitectural changes at the distal radius and tibia in postmenopausal women using HR-pQCT. Osteoporos. Int. 2014, 25, 2057–2066. [Google Scholar] [CrossRef] [PubMed]
- Van Rietbergen, B.; Ito, K. A survey of micro-finite element analysis for clinical assessment of bone strength: The first decade. J. Biomech. 2015, 48, 832–841. [Google Scholar] [CrossRef] [PubMed]
- MacNeil, J.A.; Boyd, S.K. Bone strength at the distal radius can be estimated from high-resolution peripheral quantitative computed tomography and the finite element method. Bone 2008, 42, 1203–1213. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.E.; Troy, K.L. Simplified boundary conditions alter cortical-trabecular load sharing at the distal radius; A multiscale finite element analysis. J. Biomech. 2017. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.E.; Troy, K.L. Validation of a new multiscale finite element analysis approach at the distal radius. Med. Eng. Phys. 2017, 44, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Christen, D.; Melton, L.J.; Zwahlen, A.; Amin, S.; Khosla, S.; Müller, R. Improved fracture risk assessment based on nonlinear micro-finite element simulations from HRpQCT images at the distal radius. J. Bone Miner. Res. 2013, 28, 2601–2608. [Google Scholar] [CrossRef] [PubMed]
- Ural, A.; Bruno, P.; Zhou, B.; Shi, X.T.; Guo, X.E. A new fracture assessment approach coupling HR-pQCT imaging and fracture mechanics-based finite element modeling. J. Biomech. 2013, 46, 1305–1311. [Google Scholar] [CrossRef] [PubMed]
- Hambli, R. Micro-CT finite element model and experimental validation of trabecular bone damage and fracture. Bone 2013, 56, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Vasikaran, S.; Eastell, R.; Bruyère, O.; Foldes, A.J.; Garnero, P.; Griesmacher, A.; McClung, M.; Morris, H.A.; Silverman, S.; Trenti, T.; et al. Markers of bone turnover for the prediction of fracture risk and monitoring of osteoporosis treatment: A need for international reference standards. Osteoporos. Int. 2011, 22, 391–420. [Google Scholar] [CrossRef] [PubMed]
- Bauer, D.; Krege, J.; Lane, N.; Leary, E.; Libanati, C.; Miller, P.; Myers, G.; Silverman, S.; Vesper, H.W.; Lee, D.; et al. National Bone Health Alliance Bone Turnover Marker Project: Current practices and the need for US harmonization, standardization, and common reference ranges. Osteoporos. Int. 2012, 23, 2425–2433. [Google Scholar] [CrossRef] [PubMed]
- Kambas, A.; Leontsini, D.; Avloniti, A.; Chatzinikolaou, A.; Stampoulis, T.; Makris, K.; Draganidis, D.; Jamurtas, A.Z.; Tournis, S.; Fatouros, I.G. Physical activity may be a potent regulator of bone turnover biomarkers in healthy girls during preadolescence. J. Bone Miner. Metab. 2017, 35, 598–607. [Google Scholar] [CrossRef] [PubMed]
- Jürimäe, J.; Tillmann, V.; Cicchella, A.; Stefanelli, C.; Võsoberg, K.; Tamm, A.L.; Jürimäe, T. Increased sclerostin and preadipocyte factor-1 levels in prepubertal rhythmic gymnasts: Associations with bone mineral density, body composition, and adipocytokine values. Osteoporos. Int. 2016, 27, 1239–1243. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Bemben, M.G.; Knehans, A.W.; Bemben, D.A. Effects of an 8-month ashtanga-based yoga intervention on bone metabolism in middle-aged premenopausal women: A randomized controlled study. J. Sports Sci. Med. 2015, 14, 756–768. [Google Scholar] [PubMed]
- Morgan, AL.; Weiss, J.; Kelley, E.T. Bone Turnover Response to Acute Exercise with Varying Impact Levels: A preliminary investigation. Int. J. Exerc. Sci. 2015, 8, 6. [Google Scholar]
- Lutz, L.J.; Karl, J.P.; Rood, J.C.; Cable, S.J.; Williams, K.W.; Young, A.J.; Mcclung, J.P. Vitamin D status, dietary intake, and bone turnover in female Soldiers during military training. J. Int. Soc. Sports Nutr. 2012, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, K.; Lynch, R.P.; Reiger, J.; Yingling, V.R. Short-term jump activity on bone metabolism in female college-aged non-athletes. J. Sports Sci. Med. 2012, 11, 31–38. [Google Scholar] [PubMed]
- Lester, M.E.; Urso, M.L.; Evans, R.K.; Pierce, J.R.; Spiering, B.A.; Maresh, C.M.; Hatfield, D.L.; Kraemer, W.J.; Nindl, B.C. Influence of exercise mode and osteogenic index on bone biomarker responses during short-term physical training. Bone 2009, 45, 768–776. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, S.; Ichimura, S.; Iwamoto, J.; Takeda, T.; Toyama, Y. Effect of walking exercise on bone metabolism in postmenopausal women with osteopenia/osteoporosis. J. Bone Miner. Metab. 2004, 22, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Vincent, K.R.; Braith, R.W. Resistance exercise and bone turnover in elderly men and women. Med. Sci. Sports Exerc. 2002, 34, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Gombos, G.C.; Bajsz, V.; Pék, E.; Schmidt, B.; Sió, E.; Molics, B.; Betlehem, J. Direct effects of physical training on markers of bone metabolism and serum sclerostin concentrations in older adults with low bone mass. BMC Musculoskelet. Disord. 2016, 17, 254. [Google Scholar] [CrossRef] [PubMed]
- Wen, H.J.; Huang, T.H.; Li, T.L.; Chong, P.N.; Ang, B.S. Effects of short-term step aerobics exercise on bone metabolism and functional fitness in postmenopausal women with low bone mass. Osteoporos. Int. 2017, 28, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Robling, A.G.; Niziolek, P.J.; Baldridge, L.A.; Condon, K.W.; Allen, M.R.; Alam, I.; Mantila, S.M.; Gluhak-Heinrich, J.; Bellido, T.M.; Harris, S.E.; et al. Mechanical stimulation of bone in vivo reduces osteocyte expression of Sost/sclerostin. J. Biol. Chem. 2008, 283, 5866–5875. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, A.; Sugiyama, T.; Prasad, J.; Zaman, G.; Gross, T.S.; Lanyon, L.E.; Price, J.S. Mechanical loading-related changes in osteocyte sclerostin expression in mice are more closely associated with the subsequent osteogenic response than the peak strains engendered. Osteoporos. Int. 2012, 23, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Gao, R.; Cao, P.; Yuan, W. Additive effects of antiresorptive agents and exercise on lumbar spine bone mineral density in adults with low bone mass: A meta-analysis. Osteoporos. Int. 2014, 25, 1585–1594. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Zhao, M.; Zhang, L. Efficiency of Jumping Exercise in Improving Bone Mineral Density Among Premenopausal Women: A Meta-Analysis. Sports Med. 2014, 44, 1393–1402. [Google Scholar] [CrossRef] [PubMed]
- Bailey, C.A.; Brooke-Wavell, K. Exercise for optimising peak bone mass in women. Proc. Nutr. Soc. 2008, 67, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Martyn-St James, M.; Carroll, S. A meta-analysis of impact exercise on postmenopausal bone loss: The case for mixed loading exercise programmes. Br. J. Sports Med. 2009, 43, 898–908. [Google Scholar] [CrossRef] [PubMed]
- James, M.M.S.; Carroll, S. Effects of different impact exercise modalities on bone mineral density in premenopausal women: A meta-analysis. J. Bone Miner. Metab. 2010, 28, 251–267. [Google Scholar] [CrossRef] [PubMed]
- Marques, E.A.; Mota, J.; Carvalho, J. Exercise effects on bone mineral density in older adults: A meta-analysis of randomized controlled trials. Age 2012, 34, 1493–1515. [Google Scholar] [CrossRef] [PubMed]
- Langsetmo, L.; Hitchcock, C.; Kingwell, E.; Davison, K.; Berger, C.; Forsmo, S.; Zhou, W.; Kreiger, N.; Prior, J. Physical Activity, Body Mass Index and Bone Mineral Density—Associations in a Prospective Population-based Cohort of Women and Men: The Canadian Multicentre Osteoporosis Study (CaMos). Bone 2012, 50, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Beck, B.R.; Daly, R.M.; Singh, M.A.F.; Taaffe, D.R. Exercise and Sports Science Australia (ESSA) position statement on exercise prescription for the prevention and management of osteoporosis. J. Sci. Med. Sport 2017, 20, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Kemmler, W.; von Stengel, S.; Kohl, M. Exercise frequency and bone mineral density development in exercising postmenopausal osteopenic women. Is there a critical dose of exercise for affecting bone? Results of the Erlangen Fitness and Osteoporosis Prevention Study. Bone 2016, 89, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Babatunde, O.O.; Forsyth, J.J.; Gidlow, C.J. A meta-analysis of brief high-impact exercises for enhancing bone health in premenopausal women. Osteoporos. Int. 2012, 23, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Wu, L.; He, Z. Effects of walking on the preservation of bone mineral density in perimenopausal and postmenopausal women: A systematic review and meta-analysis. Menopause 2013, 20, 1216–1226. [Google Scholar] [CrossRef] [PubMed]
- Okubo, Y.; Osuka, Y.; Jung, S.; Rafael, F.; Tsujimoto, T.; Aiba, T.; Kim, T.; Tanaka, K. Walking can be more effective than balance training in fall prevention among community-dwelling older adults. Geriatr. Gerontol. Int. 2016, 16, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Kelley, G.A.; Kelley, K.S.; Kohrt, W.M. Effects of ground and joint reaction force exercise on lumbar spine and femoral neck bone mineral density in postmenopausal women: A meta-analysis of randomized controlled trials. BMC Musculoskelet. Disord. 2012, 13. [Google Scholar] [CrossRef] [PubMed]
- Kelley, G.A.; Kelley, K.S.; Kohrt, W.M. Exercise and bone mineral density in premenopausal women: A meta-analysis of randomized controlled trials. Int. J. Endocrinol. 2013, 2013. [Google Scholar] [CrossRef]
- Segev, D.; Hellerstein, D.; Dunsky, A. Physical activity-Does it really increase bone density in postmenopausal women? A Review of articles published between 2001–2016. Curr. Aging Sci. 2017. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Troy, K.L.; Mancuso, M.E.; Butler, T.A.; Johnson, J.E. Exercise Early and Often: Effects of Physical Activity and Exercise on Women’s Bone Health. Int. J. Environ. Res. Public Health 2018, 15, 878. https://doi.org/10.3390/ijerph15050878
Troy KL, Mancuso ME, Butler TA, Johnson JE. Exercise Early and Often: Effects of Physical Activity and Exercise on Women’s Bone Health. International Journal of Environmental Research and Public Health. 2018; 15(5):878. https://doi.org/10.3390/ijerph15050878
Chicago/Turabian StyleTroy, Karen L., Megan E. Mancuso, Tiffiny A. Butler, and Joshua E. Johnson. 2018. "Exercise Early and Often: Effects of Physical Activity and Exercise on Women’s Bone Health" International Journal of Environmental Research and Public Health 15, no. 5: 878. https://doi.org/10.3390/ijerph15050878
APA StyleTroy, K. L., Mancuso, M. E., Butler, T. A., & Johnson, J. E. (2018). Exercise Early and Often: Effects of Physical Activity and Exercise on Women’s Bone Health. International Journal of Environmental Research and Public Health, 15(5), 878. https://doi.org/10.3390/ijerph15050878



