Influence of Latitude on the Prevalence of Kawasaki Disease: A Retrospective Cohort Study from the Taiwan National Health Insurance Database and Review of the Literature
Abstract
:1. Introduction
2. Methods
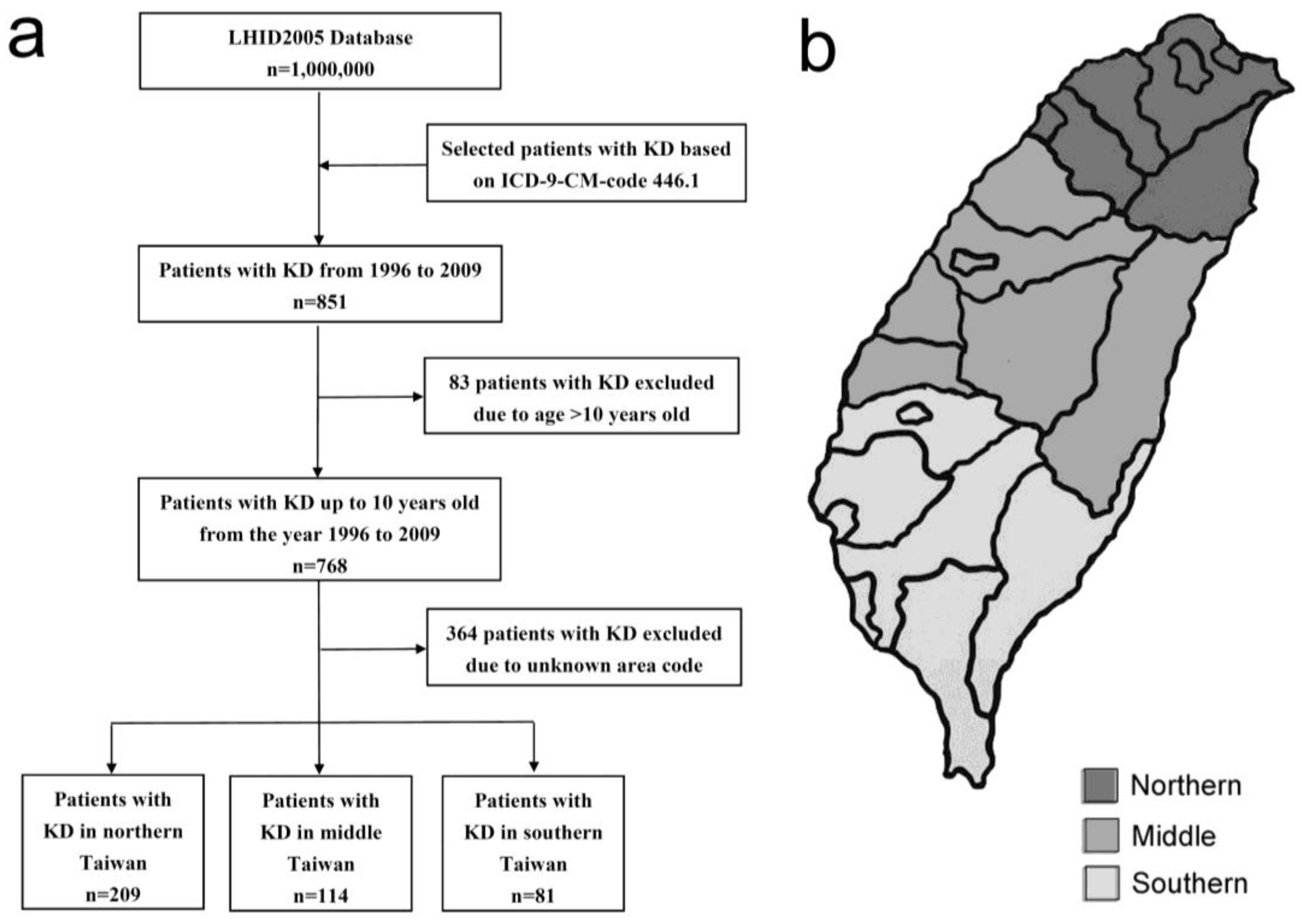
2.1. Database
2.2. Standard Protocol Approvals and Patient Consents
2.3. KD Period Prevalence
2.4. Climate Variables
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Newburger, J.W.; Takahashi, M.; Gerber, M.A.; Gewitz, M.H.; Tani, L.Y.; Burns, J.C.; Shulman, S.T.; Bolger, A.F.; Ferrieri, P.; Baltimore, R.S.; et al. Diagnosis, treatment, and long-term management of Kawasaki disease: A statement for health professionals from the Committee on Rheumatic Fever, Endocarditis and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation 2004, 110, 2747–2771. [Google Scholar] [CrossRef] [PubMed]
- Cohen, E.; Sundel, R. Kawasaki Disease at 50 Years. JAMA Pediatr. 2016, 170, 1093–1099. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Vignesh, P.; Burgner, D. The epidemiology of Kawasaki disease: A global update. Arch. Dis. Child. 2015, 100, 1084–1088. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T. Acute febrile mucocutaneous syndrome with lymphoid involvement with specific desquamation of the fingers and toes in children. Arerugi 1967, 16, 178–222. [Google Scholar] [PubMed]
- Rodo, X.; Ballester, J.; Cayan, D.; Melish, M.E.; Nakamura, Y.; Uehara, R.; Burns, J.C. Association of Kawasaki disease with tropospheric wind patterns. Sci. Rep. 2011, 1, 152. [Google Scholar] [CrossRef] [PubMed]
- Dedeoglu, F.; Sundel, R.P. Vasculitis in children. Pediatr. Clin. N. Am. 2005, 52, 547–575. [Google Scholar] [CrossRef] [PubMed]
- Makino, N.; Nakamura, Y.; Yashiro, M.; Ae, R.; Tsuboi, S.; Aoyama, Y.; Kojo, T.; Uehara, R.; Kotani, K.; Yanagawa, H. Descriptive epidemiology of Kawasaki disease in Japan, 2011–2012: From the results of the 22nd nationwide survey. J. Epidemiol. 2015, 25, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Uehara, R.; Belay, E.D. Epidemiology of Kawasaki disease in Asia, Europe, and the United States. J. Epidemiol. 2012, 22, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.B.; Park, S.; Eun, L.Y.; Kim, G.B.; Park, S.; Eun, L.Y.; Han, J.W.; Lee, S.Y.; Yoon, K.L.; Yu, J.J.; et al. Epidemiology and Clinical Features of Kawasaki Disease in South Korea, 2012–2014. Pediatr. Infect. Dis. J. 2017, 36, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Bhattad, S. Kawasaki disease incidence at Chandigarh, North India, during 2009–2014. Rheumatol Int. 2016, 36, 1391–1397. [Google Scholar] [CrossRef] [PubMed]
- Durongpisitkul, K.; Sangtawesin, C.; Khongphatthanayopthin, A.; Panamonta, M.; Sopontammarak, S.; Sittiwangkul, R.; Pongpanich, B. Epidemiologic study of Kawasaki disease and cases resistant to IVIG therapy in Thailand. Asian Pac. J. Allergy Immunol. 2006, 24, 27–32. [Google Scholar] [PubMed]
- Du, Z.D.; Zhao, D.; Du, J.; Zhang, Y.L.; Lin, Y.; Liu, C.; Zhang, T.; Beijing Kawasaki Research Group. Epidemiologic study on Kawasaki disease in Beijing from 2000 through 2004. Pediatr. Infect. Dis. J. 2007, 26, 449–451. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Ma, X.J.; Liu, F.; Yan, W.L.; Huang, M.R.; Huang, M.; Huang, G.Y.; Shanghai Kawasaki Disease Research Group. Epidemiologic Features of Kawasaki Disease in Shanghai from 2008 through 2012. Pediatr. Infect. Dis. J. 2016, 35, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Huang, L.M.; Chang, I.S.; Chang, L.Y.; Chiang, B.L.; Chen, P.J.; Wu, M.H.; Lue, H.C.; Lee, C.Y.; Kawasaki Disease Research Group. Epidemiologic features of Kawasaki disease in Taiwan, 2003–2006. Pediatrics 2009, 123, e401–e405. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.M.; Sung, R.Y.; So, L.Y.; Fong, N.C.; Ho, M.H.; Cheng, Y.W.; Lee, S.H.; Mak, W.C.; Wong, D.M.; Yam, M.C.; et al. Kawasaki disease in Hong Kong, 1994 to 2000. Hong Kong Med. J. 2005, 11, 331–335. [Google Scholar] [PubMed]
- Lin, Y.T.; Manlhiot, C.; Ching, J.C.; Han, R.K.; Nield, L.E.; Dillenburg, R.; Pepelassis, D.; Lai, L.S.; Smythe, J.F.; Chahal, N.; et al. Repeated systematic surveillance of Kawasaki disease in Ontario from 1995 to 2006. Pediatr. Int. 2010, 52, 699–706. [Google Scholar] [CrossRef] [PubMed]
- Holman, R.C.; Belay, E.D.; Christensen, K.Y.; Folkema, A.M.; Steiner, C.A.; Schonberger, L.B. Hospitalizations for Kawasaki syndrome among children in the United States, 1997–2007. Pediatr. Infect. Dis. J. 2010, 29, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Salo, E.; Griffiths, E.P.; Farstad, T.; Schiller, B.; Nakamura, Y.; Yashiro, M.; Uehara, R.; Best, B.M.; Burns, J.C. Incidence of Kawasaki disease in northern European countries. Pediatr. Int. 2012, 54, 770–772. [Google Scholar] [CrossRef] [PubMed]
- Lynch, M.; Holman, R.C.; Mulligan, A.; Belay, E.D.; Schonberger, L.B. Kawasaki syndrome hospitalizations in Ireland, 1996 through 2000. Pediatr. Infect. Dis. J. 2003, 22, 959–963. [Google Scholar] [CrossRef] [PubMed]
- Tacke, C.E.; Breunis, W.B.; Pereira, R.R.; Breur, J.M.; Kuipers, I.M.; Kuijpers, T.W. Five years of Kawasaki disease in the Netherlands: A national surveillance study. Pediatr. Infect. Dis. J. 2014, 33, 793–797. [Google Scholar] [CrossRef] [PubMed]
- Harnden, A.; Mayon-White, R.; Perera, R.; Yeates, D.; Goldacre, M.; Burgner, D. Kawasaki disease in England: Ethnicity, deprivation, and respiratory pathogens. Pediatr. Infect. Dis. J. 2009, 28, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Jakob, A.; Whelan, J.; Kordecki, M.; Berner, R.; Stiller, B.; Arnold, R.; von Kries, R.; Neumann, E.; Roubinis, N.; Robert, M.; et al. Kawasaki Disease in Germany: A Prospective, Population-based Study Adjusted for Underreporting. Pediatr. Infect. Dis. J. 2016, 35, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Heuclin, T.; Dubos, F.; Hue, V.; Godart, F.; Francart, C.; Vincent, P.; Hospital Network for Evaluating the Management of Common Childhood Diseases; Martinot, A. Increased detection rate of Kawasaki disease using new diagnostic algorithm, including early use of echocardiography. J Pediatr. 2009, 155, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Pinto, F.F.; Laranjo, S.; Mota Carmo, M.; Brito, M.J.; Cruz Ferreira, R. Twelve Years of Kawasaki Disease in Portugal: Epidemiology in Hospitalized Children. Pediatr. Infect. Dis. J. 2017, 36, 364–368. [Google Scholar] [CrossRef] [PubMed]
- Cimaz, R.; Fanti, E.; Mauro, A.; Voller, F.; Rusconi, F. Epidemiology of Kawasaki disease in Italy: Surveillance from national hospitalization records. Eur. J. Pediatr. 2017, 176, 1061–1065. [Google Scholar] [CrossRef] [PubMed]
- Rodo, X.; Curcoll, R.; Robinson, M.; Ballester, J.; Burns, J.C.; Cayan, D.R.; Lipkin, W.I.; Williams, B.L.; Couto-Rodriguez, M.; Nakamura, Y.; et al. Tropospheric winds from northeastern China carry the etiologic agent of Kawasaki disease from its source to Japan. Proc. Natl. Acad. Sci. USA 2014, 111, 7952–7957. [Google Scholar] [CrossRef] [PubMed]
- Round, J.L.; Mazmanian, S.K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 2009, 9, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Jorquera, H.; Borzutzky, A.; Hoyos-Bachiloglu, R.; Garcia, A. Association of Kawasaki disease with tropospheric winds in Central Chile: Is wind-borne desert dust a risk factor? Environ. Int. 2015, 78, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Zeft, A.S.; Burns, J.C.; Yeung, R.S.; McCrindle, B.W.; Newburger, J.W.; Dominguez, S.R.; Anderson, M.S.; Arrington, C.; Shulman, S.T.; Yoon, J.; et al. Kawasaki Disease and Exposure to Fine Particulate Air Pollution. J. Pediatr. 2016, 177, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.L.; Waller, P.L.; Mueller, B.A.; Dykewicz, C.A.; Schonberger, L.B. Kawasaki syndrome in Washington State. Race-specific incidence rates and residential proximity to water. Arch. Pediatr. Adolesc. Med. 1995, 149, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Oharaseki, T.; Kameoka, Y.; Kura, F.; Persad, A.S.; Suzuki, K.; Naoe, S. Susceptibility loci to coronary arteritis in animal model of Kawasaki disease induced with Candida albicans-derived substances. Microbiol. Immunol. 2005, 49, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.R.; Chen, W.T.; Lin, Y.T.; Hwang, B.F. Ambient Air Pollutant Exposures and Hospitalization for Kawasaki Disease in Taiwan: A Case-Crossover Study (2000–2010). Environ. Health Perspect. 2017, 125, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Meng, X.; Chen, R.; Huang, G.; Ma, X.; Chen, J.; Huang, M.; Huang, M.; Gui, Y.; Chu, C.; et al. Ambient air pollution, temperature and kawasaki disease in Shanghai, China. Chemosphere 2017, 186, 817–822. [Google Scholar] [CrossRef] [PubMed]
- Abrams, J.Y.; Blasé, J.L.; Belay, E.B.; Uehara, R.; Maddox, R.A.; Schonberger, L.B.; Nakamura, Y. Increased Kawasaki Disease Incidence Associated with Higher Precipitation and Lower Temperatures, Japan, 1991–2004. Pediatr. Infect. Dis. J. 2017. [Google Scholar] [CrossRef] [PubMed]
- Principi, N.; Rigante, D.; Esposito, S. The role of infection in Kawasaki syndrome. J Infect. 2013, 67, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Callinan, L.S.; Holman, R.C.; Vugia, D.J.; Schonberger, L.B.; Belay, E.D. Kawasaki disease hospitalization rate among children younger than 5 years in California, 2003–2010. Pediatr. Infect. Dis. J. 2014, 33, 781–783. [Google Scholar] [CrossRef] [PubMed]
- Holman, R.C.; Christensen, K.Y.; Belay, E.D.; Steiner, C.A.; Effler, P.V.; Miyamura, J.; Forbes, S.; Schonberger, L.B.; Melish, M. Racial/ethnic differences in the incidence of Kawasaki syndrome among children in Hawaii. Hawaii Med. J. 2010, 69, 194–197. [Google Scholar] [PubMed]
- Chang, L.Y.; Chang, I.S.; Lu, C.Y.; Chiang, B.L.; Lee, C.Y.; Chen, P.J.; Wang, J.T.; Ho, H.N.; Chen, D.S.; Huang, L.M. Epidemiologic features of Kawasaki disease in Taiwan, 1996–2002. Pediatrics 2004, 114, e678–e682. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Population ≤ 10 years | Kawasaki Disease | |||
|---|---|---|---|---|---|
| 61,830 | 404 | ||||
| Gender | Male | 32,064 | 51.9% | 237 | 58.7% |
| Female | 29,766 | 48.1% | 167 | 41.3% | |
| Age, year | <1 | 4087 | 6.6% | 35 | 8.7% |
| 1 | 4476 | 7.2% | 121 | 30.0% | |
| 2 | 4847 | 7.8% | 86 | 21.3% | |
| 3 | 5009 | 8.1% | 47 | 11.6% | |
| 4 | 5257 | 8.5% | 37 | 9.2% | |
| 5 | 6386 | 10.3% | 21 | 5.2% | |
| 6 | 5930 | 9.6% | 18 | 4.5% | |
| 7 | 5636 | 9.1% | 15 | 3.7% | |
| 8 | 6829 | 11.0% | 12 | 3.0% | |
| 9 | 6765 | 10.9% | 6 | 1.5% | |
| 10 | 6608 | 10.7% | 6 | 1.5% | |
| Region | Population ≤ 10 years | Kawasaki Disease | Prevalence | p-Value |
|---|---|---|---|---|
| Northern (N) | 26,933 | 209 (51.7%) | 77.6/10,000 | 0.0004 |
| Middle (M) | 17,486 | 114 (28.2%) | 65.2/10,000 | |
| Southern (S) | 17,411 | 81 (20.0%) | 46.5/10,000 | |
| N vs. M | 0.1327 | |||
| N vs. S | 0.0001 | |||
| M vs. S | 0.0193 | |||
| Total | 61,830 | 404 | 65.3/10,000 |
| Area | Mean Temperature (°C) | Mean Monthly Maximum Temperature (°C) | Maximun Temperature ≥ 30 °C (d/y) | Mean Temperature ≥ 25 °C (d/y) | Mean Temperature ≤ 10 °C (d/y) | Mean Monthly Minimum Temperature (°C) | Sunshine Duration (hr/y) | Precipitation (mm/y) | Mean Relative Humidity (%) |
|---|---|---|---|---|---|---|---|---|---|
| Northern (Taipei) | 23.0 | 26.6 | 134.0 | 153.7 | 7.4 | 20.4 | 1405.2 | 2405.1 | 76.6 |
| Middle (Taichung) | 23.3 | 28.1 | 162.2 | 171.3 | 10.7 | 19.8 | 2043.2 | 1773 | 75.6 |
| Southern (Kaohsiung) | 25.1 | 28.8 | 165.7 | 215.7 | 0.9 | 22.1 | 2212.2 | 1884.9 | 75.9 |
| Linear regression | Prevalence = b0 + b1 × Climate | ||||||||
| R2 | 0.9264 | 0.8990 | 0.7364 | 0.9837 | 0.5409 | 0.6228 | 0.8185 | 0.4787 | 0.3510 |
| t-Value | −3.548 | −2.984 | −1.671 | −7.761 | 1.085 | −1.285 | −2.123 | 0.958 | 0.735 |
| p-Value | 0.175 | 0.206 | 0.343 | 0.082 | 0.474 | 0.421 | 0.280 | 0.514 | 0.596 |
| Region | Latitude | Incidence a | Period | Citation |
|---|---|---|---|---|
| Asia | ||||
| Japan | 24–46 | 264.8 | 2012 | Makino et al. 2015 [7] |
| Korea | 33–39 | 194.7 | 2014 | Kim et al. 2017 [9] |
| Beijing | 39 | 55.1 | 2004 | Du et al. 2007 [12] |
| Shanghai | 31 | 55.5 | 2012 | Chen et al. 2016 [13] |
| Taiwan | 22–25 | 69.0 | 2003–2006 | Huang et al. 2009 [14] |
| Hong Kong | 22 | 39.0 | 1994–2000 | Ng et al. 2005 [15] |
| India | 8–37 | 7.0 | 2014 | Singh et al. 2016 [10] |
| Thailand | 5–20 | 2.6 | 2002 | Durongpisitkul et al. 2006 [11] |
| North America | ||||
| Canada | 41–83 | 26.2 | 1995–2006 | Lin et al. 2010 [16] |
| United States | 18–71 | 20.8 | 2006 | Holman et al. 2010 [17] |
| Europe | ||||
| Finland | 59–70 | 11.4 | 1998–2009 | Salo et al. 2012 [18] |
| Norway | 57–80 | 5.4 | 1998–2009 | Salo et al. 2012 [18] |
| Sweden | 55–69 | 7.4 | 1998–2009 | Salo et al. 2012 [18] |
| Ireland | 51–55 | 15.2 | 1996–2000 | Lynch et al. 2003 [19] |
| Netherlands | 50–53 | 5.8 | 2008–2012 | Tacke et al. 2014 [20] |
| England | 49–60 | 8.4 | 1998–2003 | Harnden et al. 2009 [21] |
| Germany | 47–54 | 7.2 | - | Jakob et al. 2016 [22] |
| France | 42–51 | 9.0 | 2005–2006 | Heuclin et al. 2009 [23] |
| Portugal | 37–42 | 6.5 | 2000–2011 | Pinto et al. 2017 [24] |
| Italy | 35–47 | 14.7 | 2008–2013 | Cimaz et al. 2017 [25] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chang, C.-L.; Wong, C.-S.; Yang, Y.-C.; Chiu, N.-C. Influence of Latitude on the Prevalence of Kawasaki Disease: A Retrospective Cohort Study from the Taiwan National Health Insurance Database and Review of the Literature. Int. J. Environ. Res. Public Health 2018, 15, 845. https://doi.org/10.3390/ijerph15050845
Chang C-L, Wong C-S, Yang Y-C, Chiu N-C. Influence of Latitude on the Prevalence of Kawasaki Disease: A Retrospective Cohort Study from the Taiwan National Health Insurance Database and Review of the Literature. International Journal of Environmental Research and Public Health. 2018; 15(5):845. https://doi.org/10.3390/ijerph15050845
Chicago/Turabian StyleChang, Chaw-Liang, Chih-Shung Wong, Yi-Chen Yang, and Nan-Chang Chiu. 2018. "Influence of Latitude on the Prevalence of Kawasaki Disease: A Retrospective Cohort Study from the Taiwan National Health Insurance Database and Review of the Literature" International Journal of Environmental Research and Public Health 15, no. 5: 845. https://doi.org/10.3390/ijerph15050845
APA StyleChang, C.-L., Wong, C.-S., Yang, Y.-C., & Chiu, N.-C. (2018). Influence of Latitude on the Prevalence of Kawasaki Disease: A Retrospective Cohort Study from the Taiwan National Health Insurance Database and Review of the Literature. International Journal of Environmental Research and Public Health, 15(5), 845. https://doi.org/10.3390/ijerph15050845





