The Social and Spatial Ecology of Dengue Presence and Burden during an Outbreak in Guayaquil, Ecuador, 2012
Abstract
1. Introduction
2. Materials and Methods
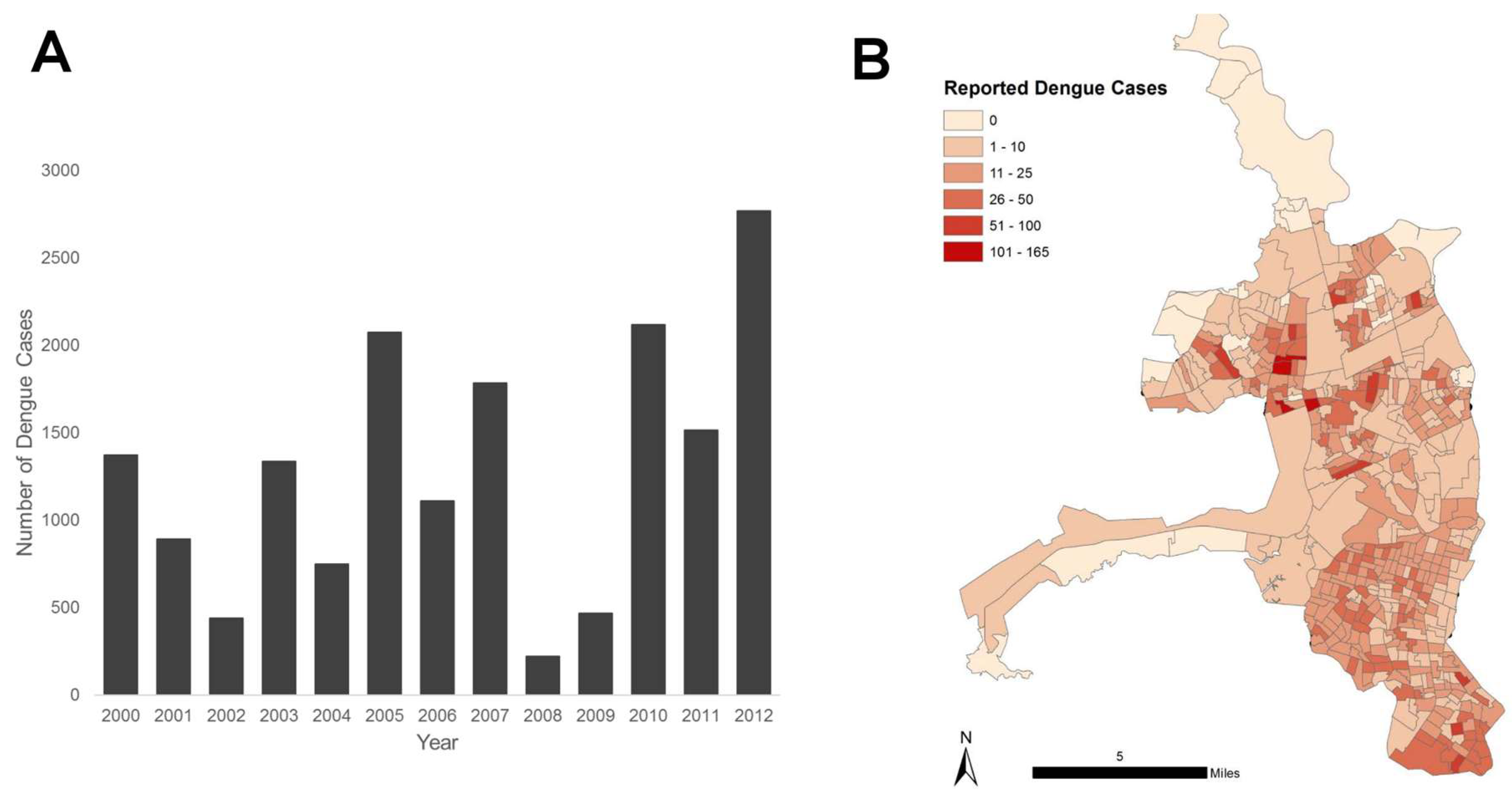
3. Results
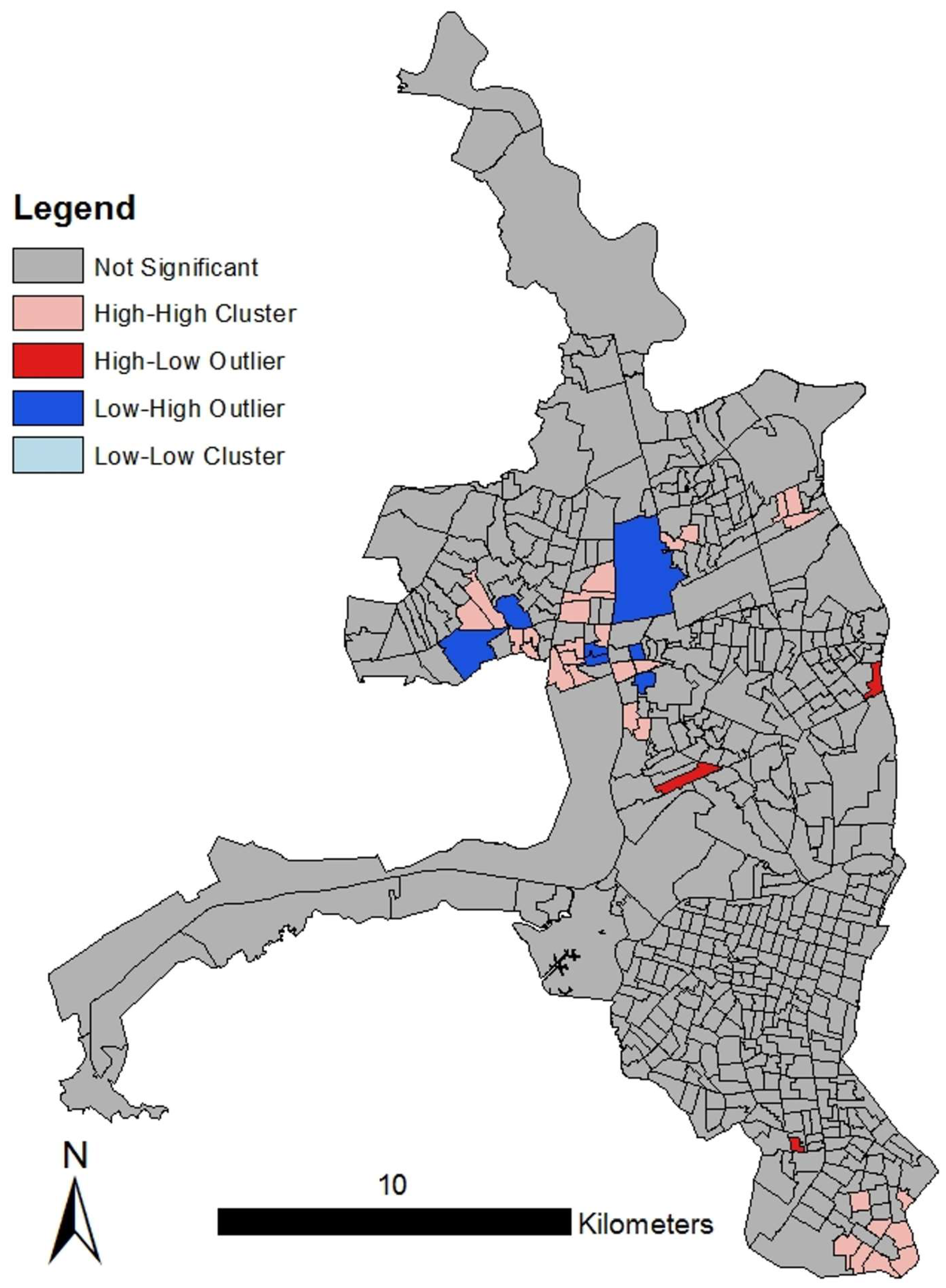
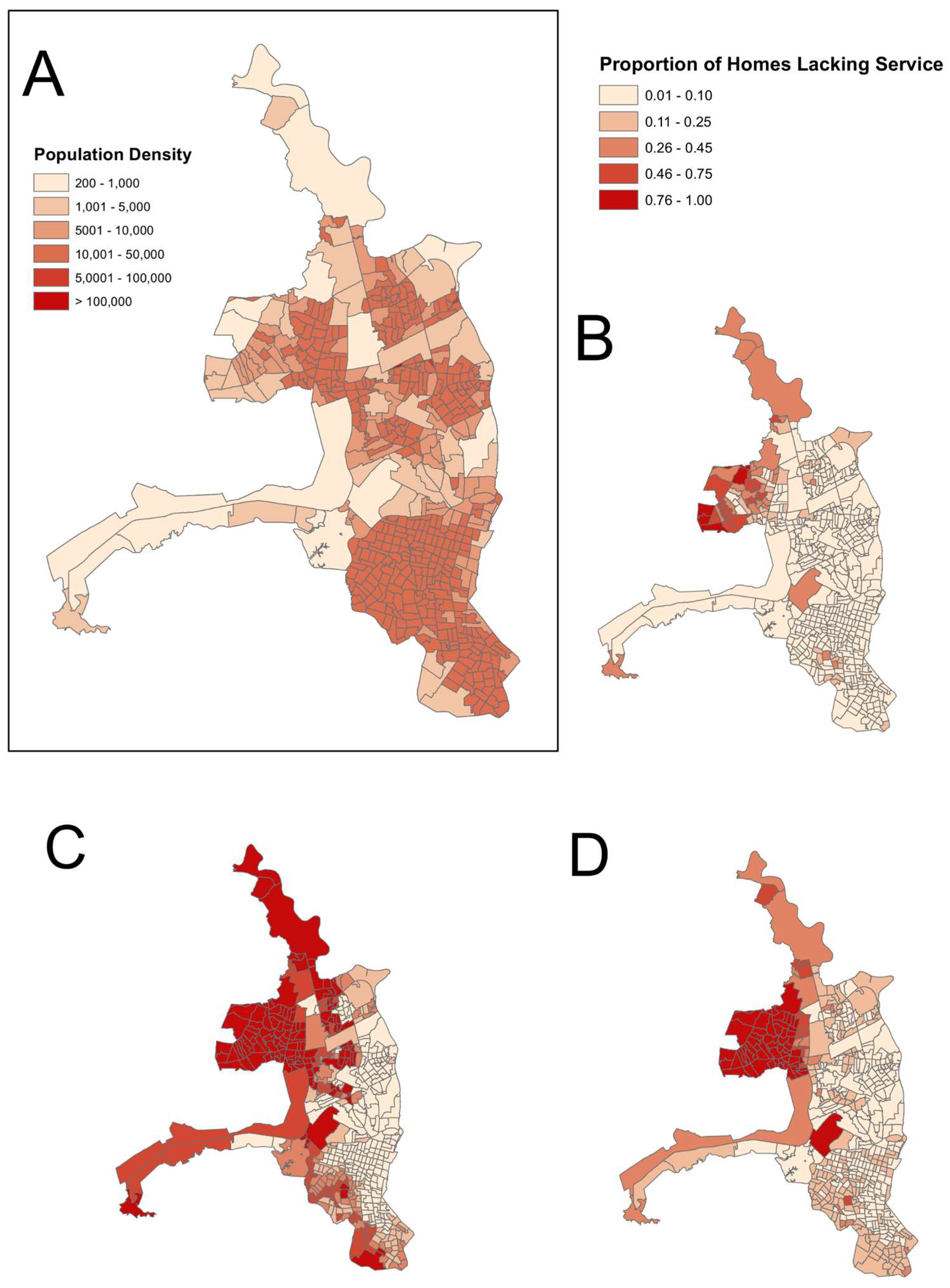
3.1. Spatial Analyses
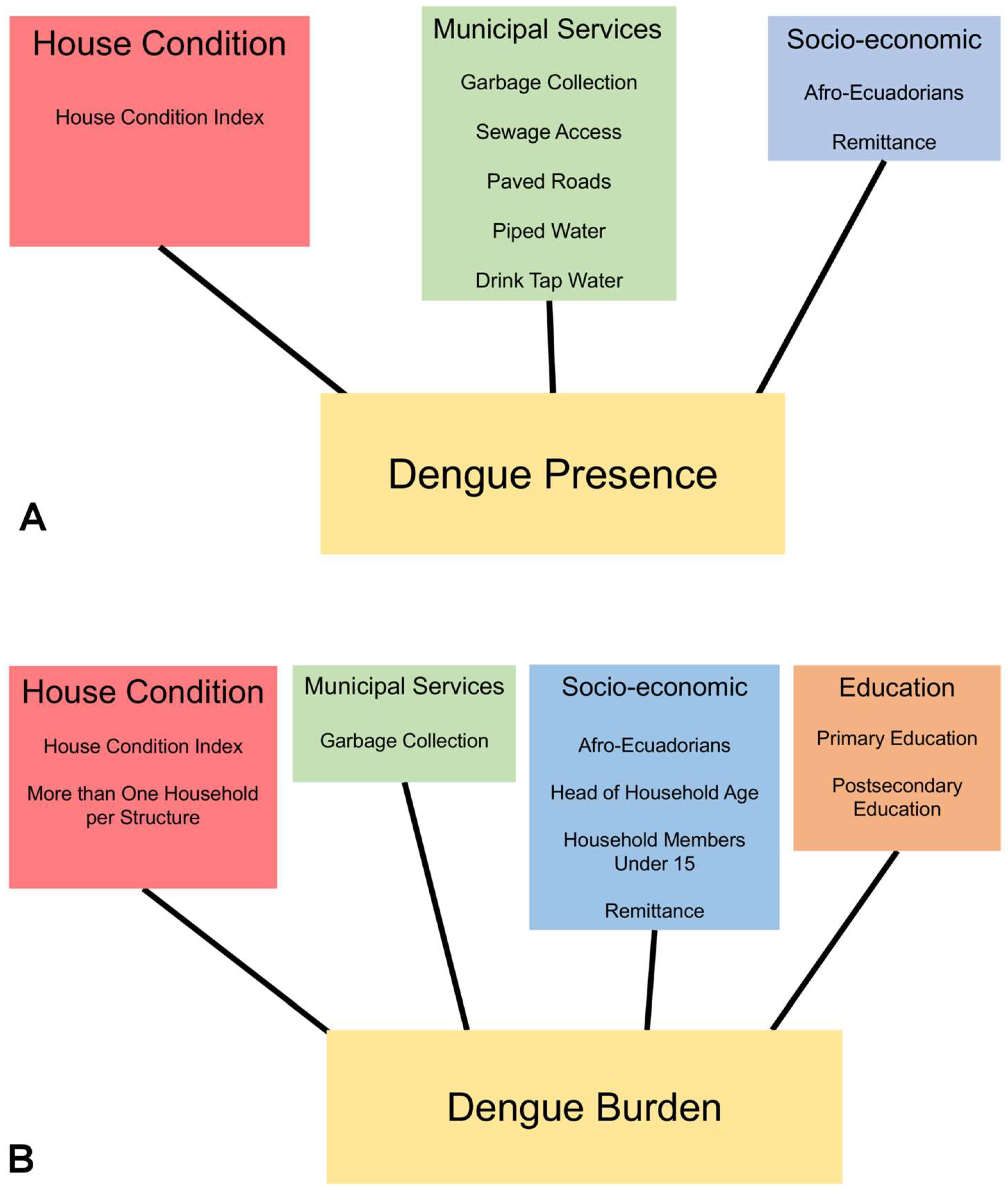
3.2. Social-Ecological Risk Factors
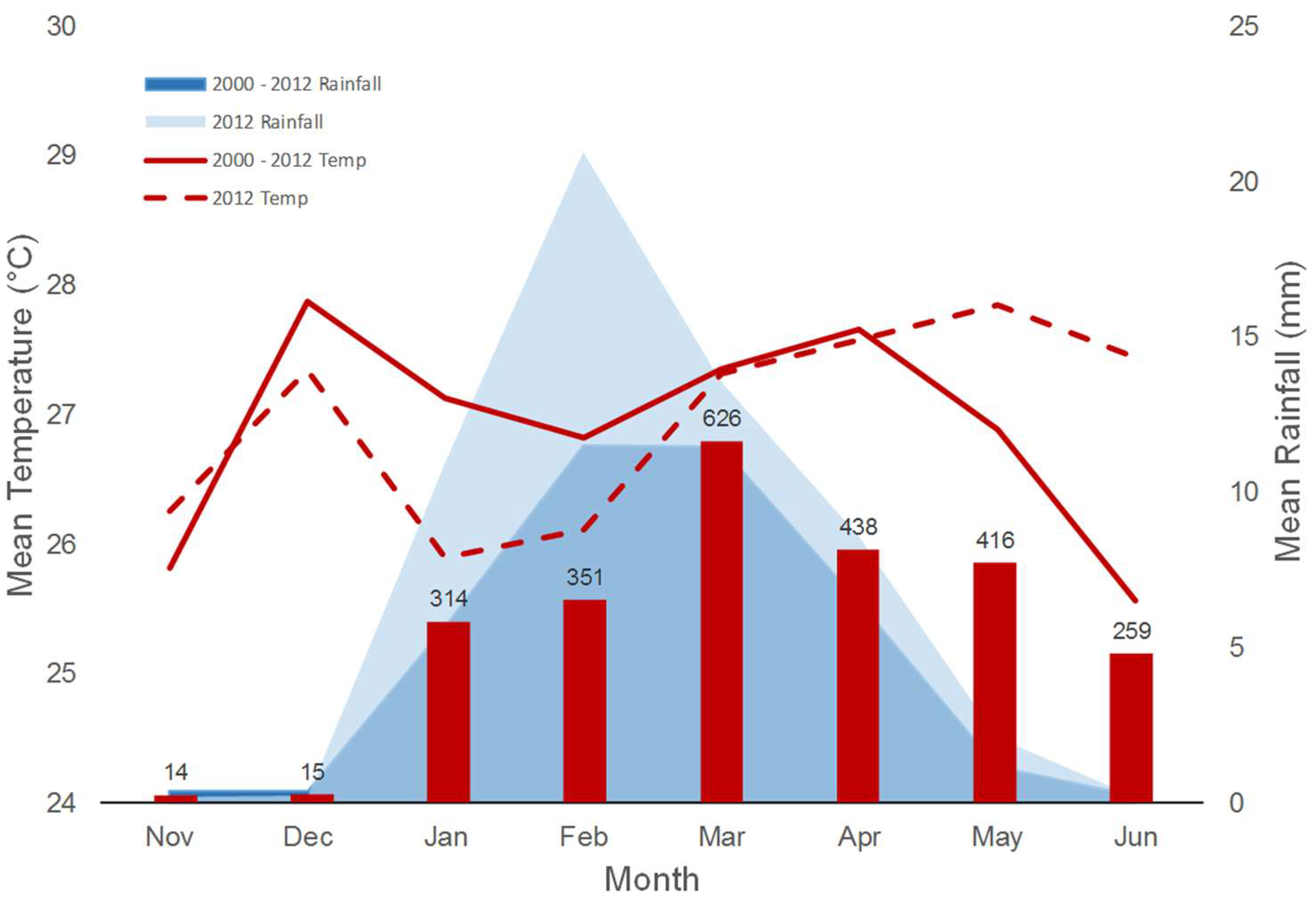
3.3. Climate Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zambrano, H.; Waggoner, J.J.; Almeida, C.; Rivera, L.; Benjamin, J.Q.; Pinsky, B.A. Zika Virus and Chikungunya Virus CoInfections: A Series of Three Cases from a Single Center in Ecuador. Am. J. Trop. Med. Hyg. 2016, 95, 894–896. Available online: http://www.ajtmh.org/cgi/doi/10.4269/ajtmh.16-0323 (accessed on 16 September 2016). [CrossRef] [PubMed]
- Dick, O.B.; Martín, J.L.S.; Montoya, R.H.; del Diego, J.; Zambrano, B.; Dayan, G.H. The History of Dengue Outbreaks in the Americas. Am. J. Trop. Med. Hyg. 2012, 87, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Pan American Health Organization, World Health Organization. Zika Suspected and Confirmed Cases Reported by Countries and Territories in the Americas. Cumulative Cases, 2015–2016; PAN/WHO: Washington, DC, USA, 2016; Available online: http://www.paho.org/hq/index.php?option=com_content&view=article&id=12390&Itemid=42090&lang=en (accessed on 17 April 2018).
- Muñoz, Á.G.; Thomson, M.C.; Goddard, L.; Aldighieri, S. Analyzing climate variations at multiple timescales can guide Zika virus response measures. GigaScience 2016, 5, 41. Available online: http://gigascience.biomedcentral.com/articles/10.1186/s13742-016-0146-1 (accessed on 17 October 2016).
- Ibarra, A.M.S.; Luzadis, V.A.; Cordova, M.J.B.; Silva, M.; Ordoñez, T.; Ayala, E.B.; Ryan, S.J. A social-ecological analysis of community perceptions of dengue fever and Aedes aegypti in Machala, Ecuador. BMC Public Health 2014, 14, 1135. Available online: http://bmcpublichealth.biomedcentral.com/articles/10.1186/1471-2458-14-1135 (accessed on 16 August 2016). [CrossRef] [PubMed]
- Castillo, K.C.; Körbl, B.; Stewart, A.; Gonzalez, J.F.; Ponce, F. Application of spatial analysis to the examination of dengue fever in Guayaquil, Ecuador. Procedia Environ. Sci. 2011, 7, 188–193. [Google Scholar] [CrossRef]
- Castillo, K.C. Zeit-Raumanalyse des Einflusses von Umwelt und sozialen Faktoreauf den Ausbruch von Denguefieber im Zeitraum 2005–2009 in Guayaquil-Ecuador. Ph.D. Thesis, Heinrich-Heine-Universität Düsseldorf, Düsseldorf, Germany, 2011. [Google Scholar]
- Kuhn, K.; Campbell-Lendrum, D.; Haines, A.; Cox, J. Using Climate to Predict Infectious Disease Epidemics. World Health Organization. 2005. Available online: http://www.who.int/globalchange/publications/infectdiseases.pdf?ua=1 (accessed on 16 April 2018).
- Thomson, M.C.; García Herrera, R.; Beniston, M. Seasonal Forecasts, Climatic Change and Human Health: Health and Climate Dordrecht; Springer: London, UK, 2008; Available online: http://site.ebrary.com/id/11029199 (accessed on 2 August 2016).
- WHO|Global Strategy for Dengue Prevention and Control, 2012–2020 WHO. Available online: http://www.who.int/denguecontrol/9789241504034/en/ (accessed on 5 May 2014).
- Banaitiene, N. (Ed.) Risk Management—Current Issues and Challenges; InTech: London, UK, 2012; Available online: http://www.intechopen.com/books/risk-management-current-issues-and-challenges (accessed on 17 October 2016).
- Almeida, A.S.D.; Medronho, R.D.A.; Valencia, L.I.O. Spatial analysis of dengue and the socioeconomic context of the city of Rio de Janeiro (Southeastern Brazil). Rev. Saúde Pública 2009, 43, 666–673. [Google Scholar] [CrossRef] [PubMed]
- De Mattos Almeida, M.C.; Caiaffa, W.T.; Assunção, R.M.; Proietti, F.A. Spatial Vulnerability to Dengue in a Brazilian Urban Area During a 7-Year Surveillance. J. Urban Health 2007, 84, 334–345. [Google Scholar] [CrossRef] [PubMed]
- Mondini, A.; de Moraes Bronzoni, R.V.; Nunes, S.H.P.; Chiaravalloti Neto, F.; Massad, E.; Alonso, W.J.; Lázzaro, E.S.; Ferraz, A.A.; de Andrade Zanotto, P.M.; Nogueira, M.L. Spatio-Temporal Tracking and Phylodynamics of an Urban Dengue 3 Outbreak in São Paulo, Brazil. PLoS Negl. Trop. Dis. 2009, 3, e448. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, M.D.G.; Barreto, M.L.; Costa, M.D.C.N.; Ferreira, L.D.A.; Vasconcelos, P.F.C.; Cairncross, S. Dynamics of dengue virus circulation: A silent epidemic in a complex urban area. Trop. Med. Int. Health 2002, 7, 757–762. [Google Scholar] [CrossRef]
- Stewart Ibarra, A.M.; Ryan, S.J.; Beltrán, E.; Mejía, R.; Silva, M.; Muñoz, Á. Dengue Vector Dynamics (Aedes aegypti) Influenced by Climate and Social Factors in Ecuador: Implications for Targeted Control. PLoS ONE 2013, 8, e78263. [Google Scholar] [CrossRef] [PubMed]
- Stewart-Ibarra, A.M.; Muñoz, Á.G.; Ryan, S.J.; Ayala, E.B.; Borbor-Cordova, M.J.; Finkelstein, J.L.; Mejía, R.; Ordoñez, T.; Recalde-Coronel, G.C.; Rivero, K. Spatiotemporal clustering, climate periodicity, and social-ecological risk factors for dengue during an outbreak in Machala, Ecuador, in 2010. BMC Infect. Dis. 2014, 14, 610. Available online: http://bmcinfectdis.biomedcentral.com/articles/10.1186/s12879-014-0610-4 (accessed on 3 August 2016). [CrossRef] [PubMed]
- Alava, A.; Mosquera, C.; Vargas, W.; Real, J. Dengue en el Ecuador 1989–2002. Rev. Ecuat. Hig. Med. Trop. 2005, 42, 11–34. [Google Scholar]
- INEC. Censo de Población y Vivienda; Instituto Nacional de Estadística y Censos: Quito, Ecuador, 2010. [Google Scholar]
- Muñoz, Á.G.; Stewart-Ibarra, A.M.; Ruiz-Carrascal, D. Desarrollo de Modelos de Pronóstico Experimental: Análisis Socio Ecológico de Riesgo a Dengue y Análisis Estadístico de Patrones Climáticos, Entomológicos y Epidemiológicos en Modelos de Dengue; Technical Report, CLIDEN Project; INAMHI-SENESCYT: Quito, Ecuador, 2013. [Google Scholar]
- Real, J.; Mosquera, C. Detección del Virus Dengue en el Ecuador. Una Vision Epidemiologica. Período 1988–2012; Instituto Nacional de Higiene y Medicina Tropical: Guayaquil, Ecuador, 2012. [Google Scholar]
- Borbor-Cordova, M.J.; Boyer, E.W.; McDowell, W.H.; Hall, C.A. Nitrogen and phosphorus budgets for a tropical watershed impacted by agricultural land use: Guayas, Ecuador. Biogeochemistry 2006, 79, 135–161. [Google Scholar] [CrossRef]
- Hartter, J.; Ryan, S.J.; MacKenzie, C.A.; Parker, J.N.; Strasser, C.A. Spatially Explicit Data: Stewardship and Ethical Challenges in Science. PLoS Biol. 2013, 11, e1001634. [Google Scholar] [CrossRef] [PubMed]
- Environmental Systems Research Institute (ESRI). ArcGIS 10.3.1.; ESRI: Redlands, CA, USA, 2016. [Google Scholar]
- Global Administrative Areas. GADM Database Glob. Adm. Areas Version 28. 2015. Available online: www.gadm.org (accessed on 16 April 2018).
- US Military, Department of Defense (all branches). Digital Chart of the World (DCW); MIL-D-89009 (specifications); US Military: Washington, DC, USA, 1992. [Google Scholar]
- Ministerio de Salud Pública. Boletín Epidemiológico de la Situación del Dengue en el Ecuador. 2013. Available online: http://www.salud.gob.ec/boletin-epidemiologico-de-la-situacion-del-dengue-en-el-ecuador-no-46-07-de-enero-de-2013/ (accessed on 3 August 2016).
- Huang, B.; Banzon, V.F.; Freeman, E.; Lawrimore, J.; Liu, W.; Peterson, T.C.; Smith, T.M.; Thorne, P.W.; Woodruff, S.D.; Zhang, H.M. Extended Reconstructed Sea Surface Temperature Version 4 (ERSST.v4). Part I: Upgrades and Intercomparisons. J. Clim. 2015, 28, 911–930. [Google Scholar] [CrossRef]
- Kanamitsu, M.; Ebisuzaki, W.; Woollen, J.; Yang, S.-K.; Hnilo, J.J.; Fiorino, M.; Potter, G.L. NCEP–DOE AMIP-II Reanalysis (R-2). Bull. Am. Meteorol. Soc. 2002, 83, 1631–1643. [Google Scholar] [CrossRef]
- Anselin, L. Local Indicators of Spatial Association-LISA. Geogr. Anal. 2010, 27, 93–115. [Google Scholar] [CrossRef]
- Calcagno, V.; de Mazancourt, C. Glmulti: An R Package for Easy Automated Model Selection with (Generalized) Linear Models. J. Stat. Softw. 2010, 34, 1–29. Available online: http://www.jstatsoft.org/v34/i12/ (accessed on 3 August 2016). [CrossRef]
- Burnham, K.P.; Anderson, D.R.; Burnham, K.P. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Oliveira, M.A.D.; Ribeiro, H.; Castillo-Salgado, C. Geospatial analysis applied to epidemiological studies of dengue: A systematic review. Rev. Bras. Epidemiol. 2013, 16, 907–917. [Google Scholar] [CrossRef] [PubMed][Green Version]
- San Martín, J.L.; Brathwaite, O.; Zambrano, B.; Solórzano, J.O.; Bouckenooghe, A.; Dayan, G.H.; Guzmán, M.G. The Epidemiology of Dengue in the Americas over the Last Three Decades: A Worrisome Reality. Am. J. Trop. Med. Hyg. 2010, 82, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Reiner, R.C.; Stoddard, S.T.; Forshey, B.M.; King, A.A.; Ellis, A.M.; Lloyd, A.L.; Long, K.C.; Rocha, C.; Vilcarromero, S.; Astete, H.; et al. Time-varying, serotype-specific force of infection of dengue virus. Proc. Natl. Acad. Sci. USA 2014, 111, E2694–E2702. [Google Scholar] [CrossRef] [PubMed]
- Galli, B.; Chiaravalloti Neto, F. Temporal-spatial risk model to identify areas at high-risk for occurrence of dengue fever. Rev. Saúde Pública 2008, 42, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Prokopec, G.M.; Kitron, U.; Montgomery, B.; Horne, P.; Ritchie, S.A. Quantifying the Spatial Dimension of Dengue Virus Epidemic Spread within a Tropical Urban Environment. PLoS Negl. Trop. Dis. 2010, 4, e920. [Google Scholar] [CrossRef] [PubMed]
- Cazelles, B.; Chavez, M.; McMichael, A.J.; Hales, S. Nonstationary Influence of El Niño on the Synchronous Dengue Epidemics in Thailand. PLoS Med. 2005, 2, e106. [Google Scholar] [CrossRef] [PubMed]
- Cummings, D.A.T.; Irizarry, R.A.; Huang, N.E.; Endy, T.P.; Nisalak, A.; Ungchusak, K.; Burke, D.S. Travelling waves in the occurrence of dengue haemorrhagic fever in Thailand. Nature 2004, 427, 344–347. [Google Scholar] [CrossRef] [PubMed]
- Endy, T.P.; Nisalak, A.; Chunsuttiwat, S.; Libraty, D.H.; Green, S.; Rothman, A.L.; Vaughn, D.W.; Ennis, F.A. Spatial and Temporal Circulation of Dengue Virus Serotypes: A Prospective Study of Primary School Children in Kamphaeng Phet, Thailand. Am. J. Epidemiol. 2002, 156, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Yoon, I.-K.; Getis, A.; Aldstadt, J.; Rothman, A.L.; Tannitisupawong, D.; Koenraadt, C.J.M.; Fansiri, T.; Jones, J.W.; Morrison, A.C.; Jarman, R.G.; et al. Fine Scale Spatiotemporal Clustering of Dengue Virus Transmission in Children and Aedes aegypti in Rural Thai Villages. PLoS Negl. Trop. Dis. 2012, 6, e1730. [Google Scholar] [CrossRef] [PubMed]
- Stoddard, S.T.; Morrison, A.C.; Vazquez-Prokopec, G.M.; Paz Soldan, V.; Kochel, T.J.; Kitron, U.; Elder, J.P.; Scott, T.W. The Role of Human Movement in the Transmission of Vector-Borne Pathogens. PLoS Negl. Trop. Dis. 2009, 3, e481. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Prokopec, G.M.; Stoddard, S.T.; Paz-Soldan, V.; Morrison, A.C.; Elder, J.P.; Kochel, T.J.; Scott, T.W.; Kitron, U. Usefulness of commercially available GPS data-loggers for tracking human movement and exposure to dengue virus. Int. J. Health Geogr. 2009, 8, 68. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, K.; Dixon, J.; Joanna Sinn, C.-L.; Elliott, S.J. Is dengue a disease of poverty? A systematic review. Pathog. Glob. Health 2015, 109, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Van Benthem, B.H.B.; Vanwambeke, S.O.; Khantikul, N.; Burghoorn-Maas, C.; Panart, K.; Oskam, L.; Lambin, E.F.; Somboon, P. Spatial patterns of and risk factors for seropositivity for dengue infection. Am. J. Trop. Med. Hyg. 2005, 72, 201–208. [Google Scholar] [PubMed]
- Bradley, J.; Rehman, A.M.; Schwabe, C.; Vargas, D.; Monti, F.; Ela, C.; Riloha, M.; Kleinschmidt, I. Reduced Prevalence of Malaria Infection in Children Living in Houses with Window Screening or Closed Eaves on Bioko Island, Equatorial Guinea. PLoS ONE 2013, 8, e80626. [Google Scholar] [CrossRef] [PubMed]
- Hiscox, A.; Khammanithong, P.; Kaul, S.; Sananikhom, P.; Luthi, R.; Hill, N.; Brey, P.T.; Lindsay, S.W. Risk Factors for Mosquito House Entry in the Lao PDR. PLoS ONE 2013, 8, e62769. [Google Scholar] [CrossRef] [PubMed]
- Jokisch, B.; Pribilsky, J. The Panic to Leave: Economic Crisis and the “New Emigration” from Ecuador. Int. Migr. 2002, 40, 75–102. [Google Scholar] [CrossRef]
- Bertoli, S.; Fernandez-Huertas Moraga, J.; Ortega, F. Immigration Policies and the Ecuadorian Exodus. World Bank Econ. Rev. 2011, 25, 57–76. [Google Scholar] [CrossRef]
- Bertoli, S.; Marchetta, F. Migration, Remittances and Poverty in Ecuador. J. Dev. Stud. 2014, 50, 1067–1089. [Google Scholar] [CrossRef]
- Schmidt, W.-P.; Suzuki, M.; Dinh Thiem, V.; White, R.G.; Tsuzuki, A.; Yoshida, L.-M.; Yanai, H.; Haque, U.; Anh, D.D.; Ariyoshi, K. Population Density, Water Supply, and the Risk of Dengue Fever in Vietnam: Cohort Study and Spatial Analysis. PLoS Med. 2011, 8, e1001082. [Google Scholar] [CrossRef] [PubMed]
- Padmanabha, H.; Durham, D.; Correa, F.; Diuk-Wasser, M.; Galvani, A. The Interactive Roles of Aedes aegypti Super-Production and Human Density in Dengue Transmission. PLoS Negl. Trop. Dis. 2012, 6, e1799. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.; Marques, G.; Serpa, L.; Arduino, M.; Voltolini, J.; Barbosa, G.; Andrade, V.R.; de Lima, V.L. Density of Aedes aegypti and Aedes albopictus and its association with number of residents and meteorological variables in the home environment of dengue endemic area, São Paulo, Brazil. Parasit. Vectors 2015, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Van Benthem, B.H.B.; Khantikul, N.; Panart, K.; Kessels, P.J.; Somboon, P.; Oskam, L. Knowledge and use of prevention measures related to dengue in northern Thailand. Trop. Med. Int. Health 2002, 7, 993–1000. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Mean | SD |
|---|---|---|
| Housing conditions | ||
| House condition index (HCI), 0 to 1, where 1 is poor condition | 0.27 | 0.12 |
| More than four people per bedroom | 16.78% | 0.08 |
| People per household | 3.88 | 0.34 |
| Municipal garbage collection | 93.06% | 0.15 |
| People in household drink tap water | 76.85% | 0.09 |
| Piped water inside the home | 77.03% | 0.31 |
| Municipal sewage | 62.52% | 0.39 |
| Access to paved roads | 80.06% | 0.25 |
| More than one household per structure | 1.90% | 0.01 |
| Unoccupied households | 16.08% | 0.56 |
| Rental homes | 1.55% | 0.17 |
| Demographics | ||
| Receive remittances | 8.85% | 0.04 |
| People emigrate for work | 1.88% | 0.01 |
| Mean age of the head of the household (years) | 45.69 | 4.54 |
| Mean household age (years) | 29.36 | 4.29 |
| Proportion of household under 15 years of age | 28.34% | 0.06 |
| Proportion of household under 5 years of age | 9.31% | 0.03 |
| Head of the household has primary education or less | 30.94% | 0.15 |
| Head of household has secondary education | 31.73% | 0.07 |
| Head of household has post-secondary education | 25.77% | 0.21 |
| Afro-Ecuadorian | 10.13% | 0.07 |
| Head of the household is unemployed | 26.84% | 0.06 |
| Head of the household is a woman | 33.29% | 0.04 |
| Model | Estimate | 95% CI | SE | AICc | p-Value |
|---|---|---|---|---|---|
| Intercept | 3.84 | 0.54–7.25 | 1.71 | 369.85 | 0.03 |
| House condition | 24.55 | 17.62–32.11 | 3.69 | <0.001 | |
| Proportion of Afro-Ecuadorians | −9.69 | −15.72–−3.76 | 3.04 | 0.001 | |
| Municipal garbage collection | 4.70 | 2.27–7.37 | 1.29 | <0.001 | |
| Piped water | 3.50 | 1.38–5.72 | 1.10 | 0.002 | |
| Municipal sewage | 2.04 | 0.44–3.62 | 0.81 | 0.012 | |
| Access by paved roads | −3.36 | −6.36–−0.54 | 1.48 | 0.023 | |
| Drink tap water | −10.74 | −16.53–−5.28 | 2.86 | <0.001 | |
| Remittance | 23.20 | 10.83–36.15 | 6.44 | <0.001 |
| Model | Estimate | 95% CI | SE | AICc | p-Value |
|---|---|---|---|---|---|
| Intercept | 1.04 | −4.09–6.25 | 2.54 | 2920.67 | 0.682 |
| House condition | 10.95 | 6.77–15.13 | 2.09 | <0.001 | |
| Postsecondary education | −2.53 | −4.72–−0.34 | 1.07 | 0.018 | |
| Primary education | −5.11 | −8.52–−1.71 | 1.62 | 0.002 | |
| Proportion of Afro-Ecuadorians | −4.23 | −6.43–−1.94 | 1.25 | <0.001 | |
| Proportion of household members under 15 | −9.02 | −15.58–−2.50 | 3.46 | 0.009 | |
| Head of household age | −0.12 | −0.19–−0.05 | 0.03 | <0.001 | |
| Municipal garbage collection | 2.82 | 1.77–3.87 | 0.61 | <0.001 | |
| More than 1 household per structure | 7.57 | −4.66–20.03 | 6.26 | 0.227 | |
| Remittance | 4.76 | −1.27–10.87 | 3.07 | 0.121 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lippi, C.A.; Stewart-Ibarra, A.M.; Muñoz, Á.G.; Borbor-Cordova, M.J.; Mejía, R.; Rivero, K.; Castillo, K.; Cárdenas, W.B.; Ryan, S.J. The Social and Spatial Ecology of Dengue Presence and Burden during an Outbreak in Guayaquil, Ecuador, 2012. Int. J. Environ. Res. Public Health 2018, 15, 827. https://doi.org/10.3390/ijerph15040827
Lippi CA, Stewart-Ibarra AM, Muñoz ÁG, Borbor-Cordova MJ, Mejía R, Rivero K, Castillo K, Cárdenas WB, Ryan SJ. The Social and Spatial Ecology of Dengue Presence and Burden during an Outbreak in Guayaquil, Ecuador, 2012. International Journal of Environmental Research and Public Health. 2018; 15(4):827. https://doi.org/10.3390/ijerph15040827
Chicago/Turabian StyleLippi, Catherine A., Anna M. Stewart-Ibarra, Ángel G. Muñoz, Mercy J. Borbor-Cordova, Raúl Mejía, Keytia Rivero, Katty Castillo, Washington B. Cárdenas, and Sadie J. Ryan. 2018. "The Social and Spatial Ecology of Dengue Presence and Burden during an Outbreak in Guayaquil, Ecuador, 2012" International Journal of Environmental Research and Public Health 15, no. 4: 827. https://doi.org/10.3390/ijerph15040827
APA StyleLippi, C. A., Stewart-Ibarra, A. M., Muñoz, Á. G., Borbor-Cordova, M. J., Mejía, R., Rivero, K., Castillo, K., Cárdenas, W. B., & Ryan, S. J. (2018). The Social and Spatial Ecology of Dengue Presence and Burden during an Outbreak in Guayaquil, Ecuador, 2012. International Journal of Environmental Research and Public Health, 15(4), 827. https://doi.org/10.3390/ijerph15040827






