Detection of the Invasive Mosquito Species Aedes (Stegomyia) albopictus (Diptera: Culicidae) in Portugal
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mosquito Collection and Occurrence
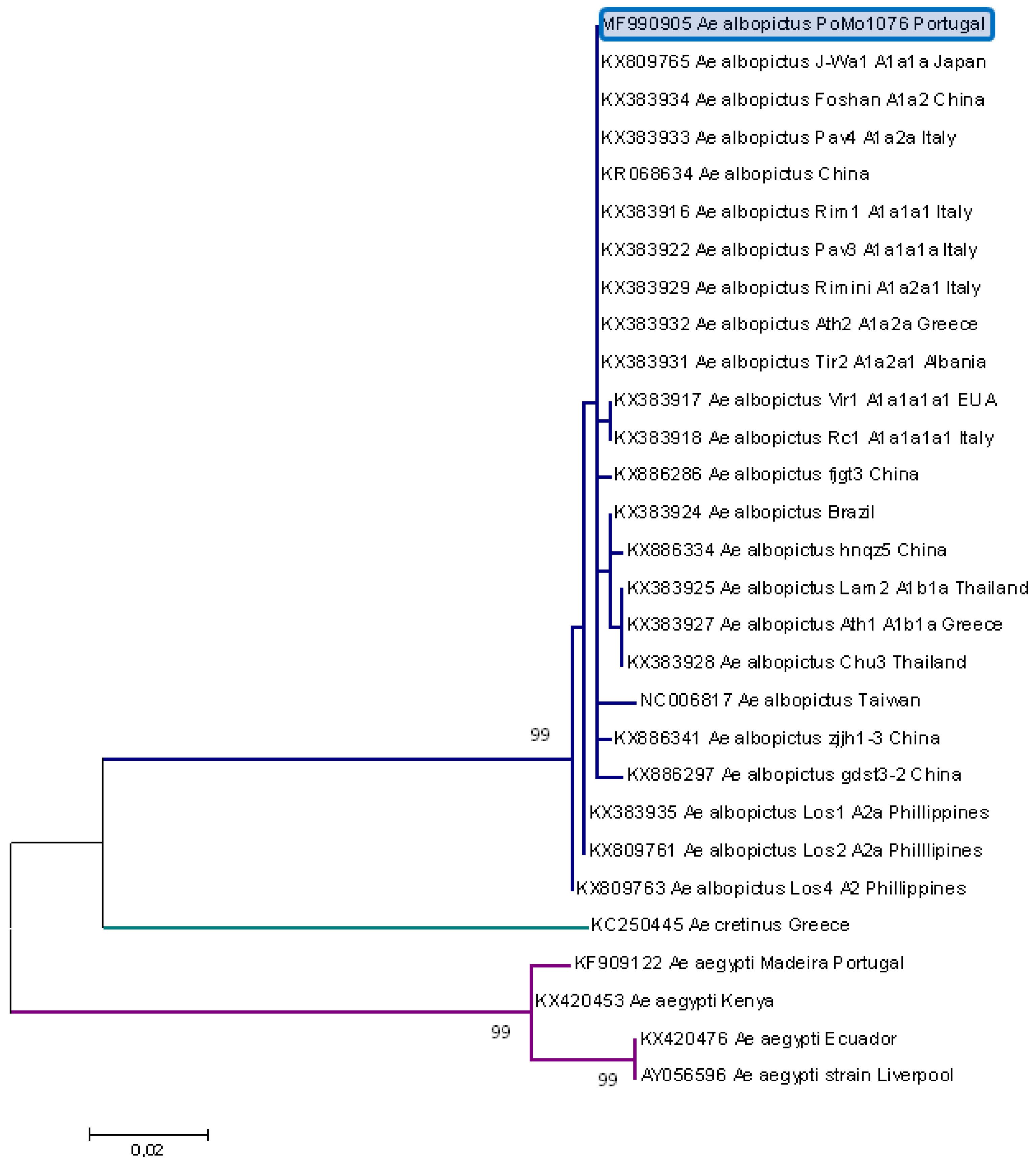
2.2. Molecular Typing
2.3. Arboviruses Screening
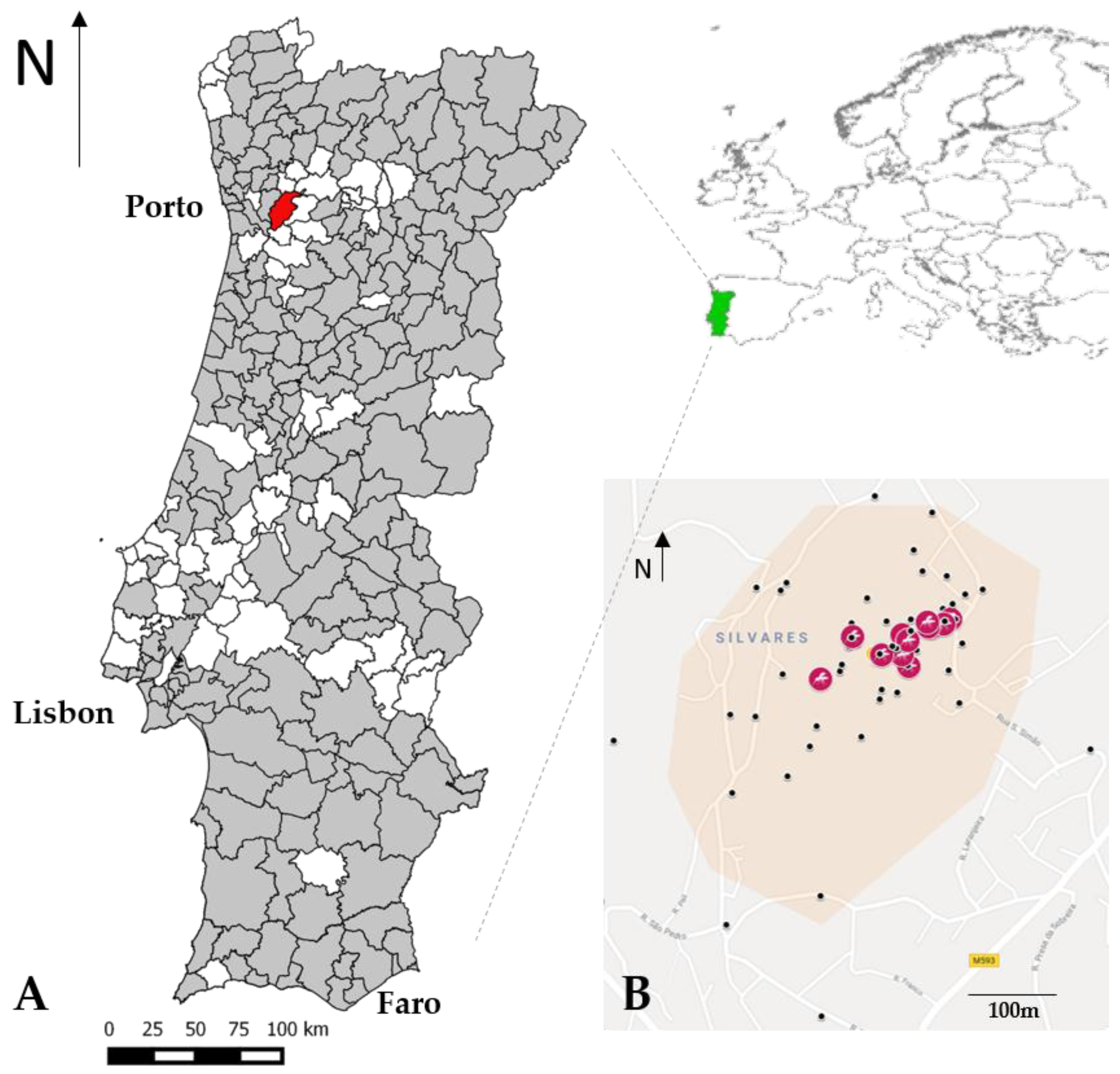
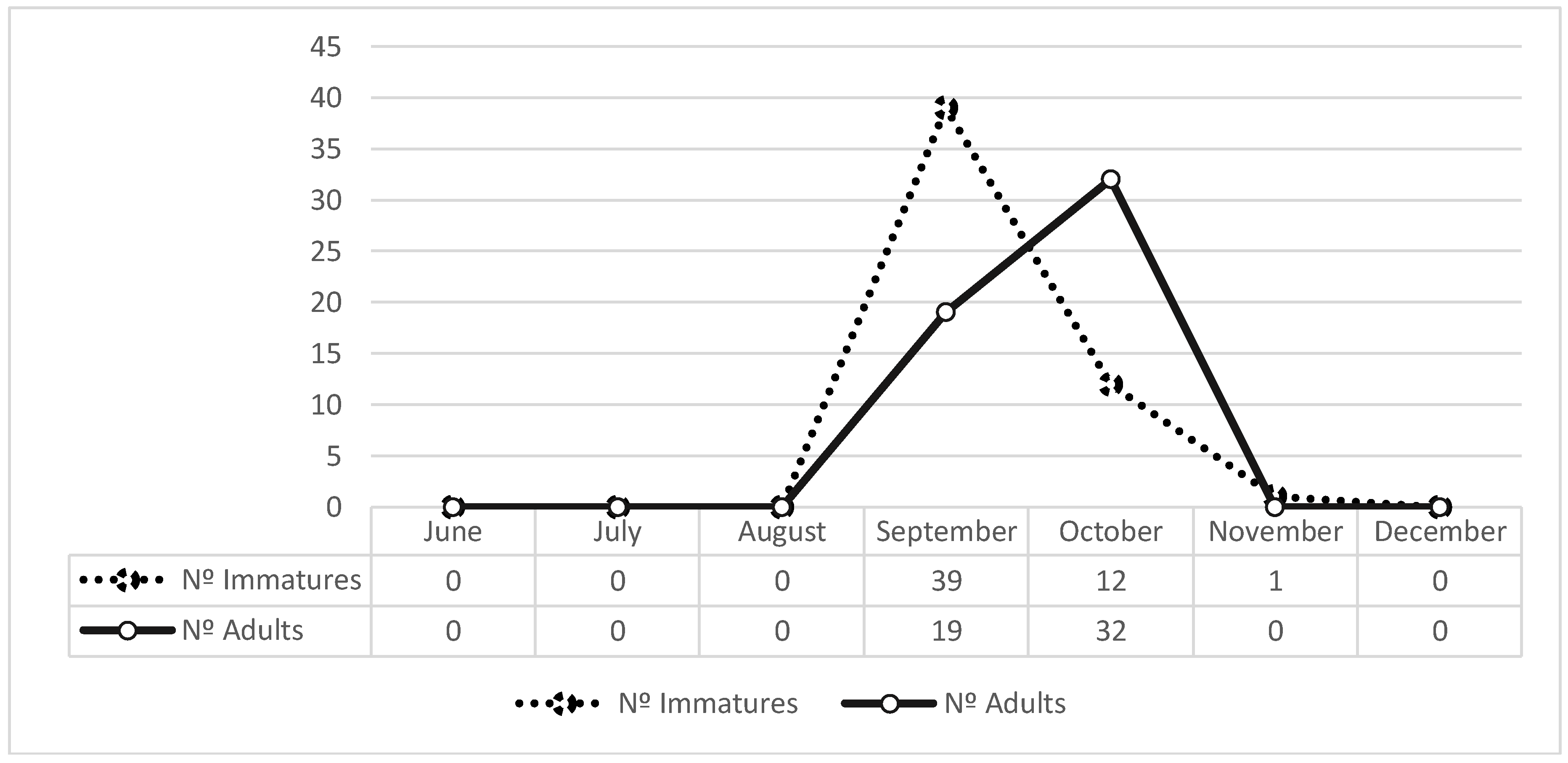
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Paupy, C.; Delatte, H.; Bagny, L.; Corbel, V.; Fontenille, D. Aedes. albopictus, an arbovirus vector: From the darkness to the light. Microbes Infect. 2009, 11, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Benedict, M.Q.; Levine, R.S.; Hawley, W.A.; Lounibos, L.P. Spread of the tiger: Global risk of invasion by the mosquito Aedes. Albopictus. Vector Borne Zoonotic Dis 2007, 7, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Adhami, J.; Reiter, P. Introduction and establishment of Aedes. (Stegomyia.) albopictus Skuse (Diptera: Culicidae) in Albania. J. Am. Mosq. Control. Assoc. 1998, 14, 340–343. [Google Scholar] [PubMed]
- Sabatini, A.; Raineri, V.; Trovato, G.; Coluzzi, M. Aedes. albopictus in Italy and possible diffusion of the species into the Mediterranean area. Parassitologia 1990, 32, 301–304. [Google Scholar] [PubMed]
- Romi, R.; Di Luca, M.; Majori, G. Current status of Aedes albopictus and Aedes atropalpus in Italy. J. Am. Mosq. Control. Assoc. 1999, 15, 425–427. [Google Scholar] [PubMed]
- Scholte, E.J.; Schaffner, F. Waiting for the tiger: Establishment and spread of the Aedes albopictus mosquito in Europe. In Emerging Pests and Vector-Borne Diseases in Europe; Takken, W., Knols, B.G.J., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2007; Volume 1, pp. 241–260. ISBN 978–90–8686–053–1. [Google Scholar]
- Collantes, F.; Delacour, S.; Alarcón-Elbal, P.M.; Ruiz-Arrondo, I.; Delgado, J.A.; Torrell-Sorio, A.; Bengoa, M.; Eritja, R.; Miranda, M.A.; Molina, R.; Lucientes, J. Review of ten-years presence of Aedes albopictus in Spain 2004–2014: Known distribution and public health concerns. Parasit Vectors 2015, 8, 655. [Google Scholar] [CrossRef] [PubMed]
- Collantes, F.; Delacour, S.; Delgado, J.A.; Bengoa, M.; Torrell-Sorio, A.; Guinea, H.; Ruiz, S.; Lucientes, J. Updating the known distribution of Aedes albopictus (Skuse, 1894) in Spain 2015. Acta Trop 2016, 164, 64–68. [Google Scholar] [CrossRef] [PubMed]
- ECDC—European Centre for Disease Control. Development of Aedes albopictus risk maps—Technical Report. ECDC: Stockholm, 2009. Available online: https://ecdc.europa.eu/sites/portal/files/media/en/publications/Publications/0905_TER_Development_of_Aedes_Albopictus_Risk_Maps.pdf (accessed on 20 December 2017).
- Medlock, J.; Hansford, K.; Versteirt, V.; Cull, B.; Kampen, H.; Fontenille, D.; Hendrickx, G.; Zeller, H.; Van Bortel, W.; Schaffner, F. An entomological review of invasive mosquitoes in Europe. Bull. Entomol. Res. 2015, 105, 637–663. [Google Scholar] [CrossRef] [PubMed]
- Angelini, R.; Finarelli, A.C.; Angelini, P.; Po, C.; Petropulacos, K.; Silvi, G.; Macini, P.; Fortuna, C.; Venturi, G.; Magurano, F.; et al. Chikungunya in north-eastern Italy: A summing up of the outbreak. Euro Surveill. 2007, 12, E071122.2. [Google Scholar] [CrossRef] [PubMed]
- Calba, C.; Guerbois-Galla, M.; Franke, F.; Jeannin, C.; Auzet-Caillaud, M.; Grard, G.; Pigaglio, L.; Decoppet, A.; Weicherding, J.; Savaill, M.C.; et al. Preliminary report of an autochthonous chikungunya outbreak in France, July to September 2017. Euro Surveill. 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Manica, M.; Guzzetta, G.; Poletti, P.; Filipponi, F.; Solimini, A.; Caputo, B.; Della Torre, A.; Rosà, R.; Merler, S. Transmission dynamics of the ongoing chikungunya outbreak in Central Italy: From coastal areas to the metropolitan city of Rome, summer 2017. Euro Surveill. 2017, 22. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, H.; Ramos, H.C. Identification keys of the mosquitoes of Continental Portugal, Açores and Madeira. Eur. Mosq. Bull. 1999, 3, 1–11. [Google Scholar]
- Osório, H.C.; Zé-Zé, L.; Amaro, F.; Alves, M.J. Mosquito surveillance for prevention and control of emerging mosquito-borne diseases in Portugal—2008—2014. Int. J. Environ. Res. Public Health 2014, 11, 11583–11596. [Google Scholar] [CrossRef] [PubMed]
- Margarita, Y.; Santos Grácio, A.J.; Lencastre, I.; Silva, A.C.; Novo, T.; Sousa, C. (First record of Aedes (Stegomyia) aegypti (Linnaeus, 1762) (Diptera, Culicidae) in Madeira Island—Portugal) (Portuguese, English abstract). Acta Parasitológica Portuguesa 2006, 13, 59–61. [Google Scholar]
- ECDC—European Centre for Disease Control. Guidelines for the Surveillance of Invasive Mosquitoes in Europe; ECDC: Stockholm, Sweden, 2012; Available online: https://ecdc.europa.eu/sites/portal/files/media/en/publications/Publications/TER-Mosquito-surveillance-guidelines.pdf (accessed on 14 March 2018).
- Schaffner, F.; Anges, G.; Geoffroy, B.; Hervy, J.P.; Rhaiem, A.; Brunhes, J. The Mosquitoes of Europe: An Identification and Training Programme; IRD: Montepellier, France, 2001. [Google Scholar]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar] [PubMed]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl. Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Battaglia, V.; Gabrieli, P.; Brandini, S.; Capodiferro, M.R.; Javier, P.A.; Chen, X.G.; Achilli, A.; Semino, O.; Gomulski, L.M.; Malacrida, A.R.; et al. The worldwide spread of the tiger mosquito as revealed by mitogenome haplogroup diversity. Front. Genet. 2016, 7, 208. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Briese, T.; Jia, X.Y.; Huang, C.; Grady, L.J.; Lipkin, W.I. Identification of a Kunjin/West Nile-like flavivirus in brains of patients with New York encephalitis. Lancet 1999, 354, 1261–1262. [Google Scholar] [CrossRef]
- Briese, T.; Rambaut, A.; Pathmajeyan, M.; Bishara, J.; Weinberger, M.; Pitlik, S.; Lipkin, W.I. Phylogenetic Analysis of a Human Isolate from the 2000 Israel West Nile virus Epidemic. Emerg. Infect. Dis. 2002, 8, 528–531. [Google Scholar] [CrossRef] [PubMed]
- WHO—World Health Organization. Chikungunya—Italy. World Health Organization. Available online: http://www.who.int/csr/don/15-september-2017-chikungunya-italy/en/ (accessed on 15 September 2017).
| Trap/Breeding Site (T/BS) | No. of T/BS (Positive T/BS) | No. of Immatures | No. of Adults | Total |
|---|---|---|---|---|
| BG sentinel | 7 (2) | 11 | 11 | |
| Flower pots | 1 (0) | |||
| Gutter | 8 (4) | 42 | 24 | 66 |
| Ovitrap | 48 (6) | 9 | 11 | 20 |
| Tyre | 1 (1) | 1 | 1 | |
| Water tank (>1000 L) | 2 (0) | |||
| Aspiration | 5 | 5 | ||
| Total | 52 | 51 | 103 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osório, H.C.; Zé-Zé, L.; Neto, M.; Silva, S.; Marques, F.; Silva, A.S.; Alves, M.J. Detection of the Invasive Mosquito Species Aedes (Stegomyia) albopictus (Diptera: Culicidae) in Portugal. Int. J. Environ. Res. Public Health 2018, 15, 820. https://doi.org/10.3390/ijerph15040820
Osório HC, Zé-Zé L, Neto M, Silva S, Marques F, Silva AS, Alves MJ. Detection of the Invasive Mosquito Species Aedes (Stegomyia) albopictus (Diptera: Culicidae) in Portugal. International Journal of Environmental Research and Public Health. 2018; 15(4):820. https://doi.org/10.3390/ijerph15040820
Chicago/Turabian StyleOsório, Hugo Costa, Líbia Zé-Zé, Maria Neto, Sílvia Silva, Fátima Marques, Ana Sofia Silva, and Maria João Alves. 2018. "Detection of the Invasive Mosquito Species Aedes (Stegomyia) albopictus (Diptera: Culicidae) in Portugal" International Journal of Environmental Research and Public Health 15, no. 4: 820. https://doi.org/10.3390/ijerph15040820
APA StyleOsório, H. C., Zé-Zé, L., Neto, M., Silva, S., Marques, F., Silva, A. S., & Alves, M. J. (2018). Detection of the Invasive Mosquito Species Aedes (Stegomyia) albopictus (Diptera: Culicidae) in Portugal. International Journal of Environmental Research and Public Health, 15(4), 820. https://doi.org/10.3390/ijerph15040820






