The Relationship between Persistent Organic Pollutants Exposure and Type 2 Diabetes among First Nations in Ontario and Manitoba, Canada: A Difference in Difference Analysis
Abstract
1. Introduction
2. Methods
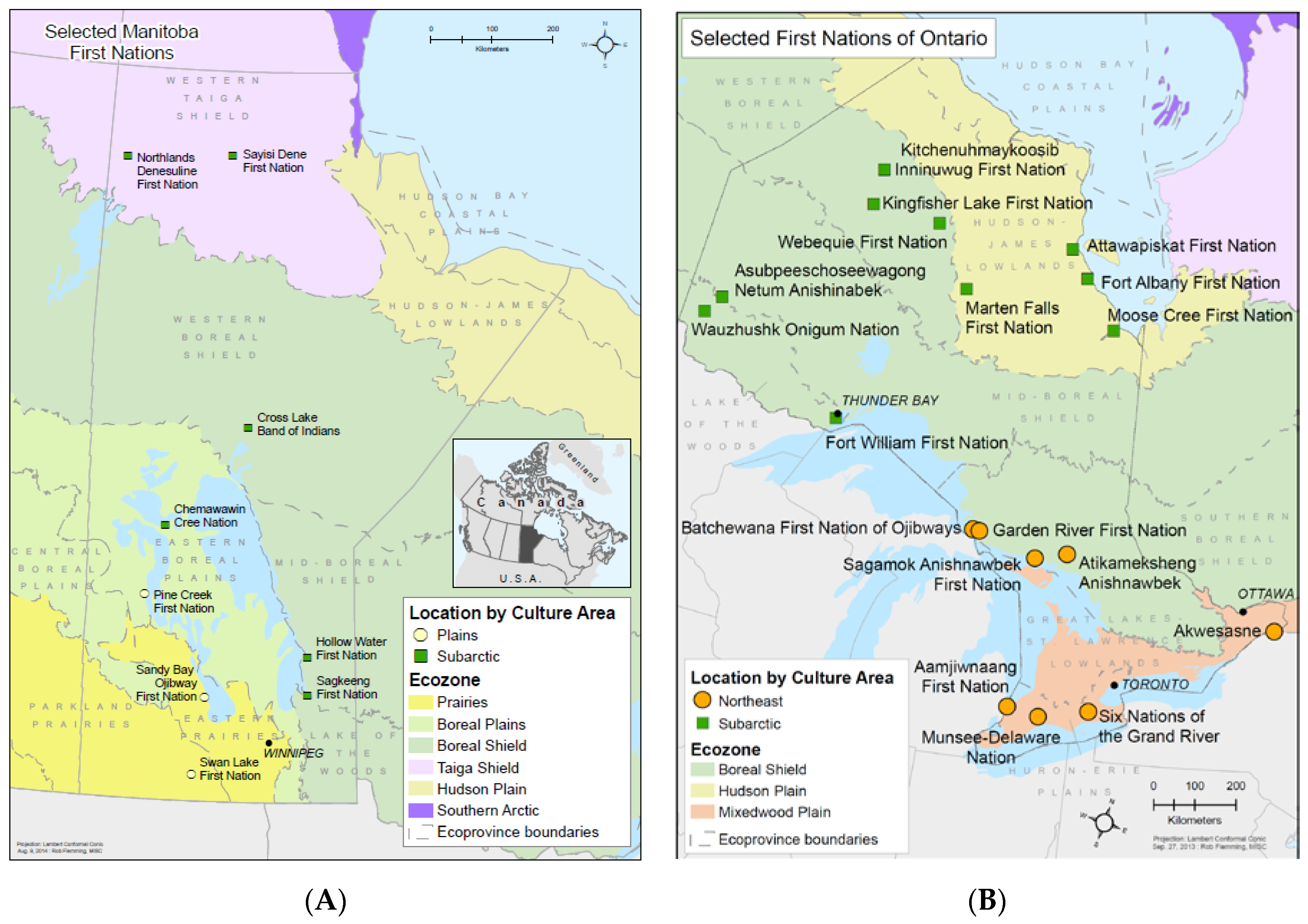
2.1. Manitoba and Ontario First Nations
2.2. Data Collection
2.3. Fish Sampling and Contaminants Analysis
2.4. Estimation of Fish, Dietary POPs (DDE, PCBs), and Long-Chain Omega-3 FA Intake
2.5. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| BMI | body mass index |
| CI | confident interval |
| DDE | dichlorodiphenyldichloroethylene |
| DID | difference in difference method |
| DHA | docosahexaenoic acid |
| EPA | eicosapentaenoic acid |
| FA | fatty acids |
| FFQ | food frequency questionnaire |
| FNFNES | First Nations Food Nutrition and Environment Study |
| OR | odds ratio |
| PCBs | polychlorinated biphenyls |
| POPs | persistent organic pollutants |
| SHL | Socio-health-lifestyle questionnaire |
| T2D | type 2 diabetes |
References
- Acton, K.J.; Ríos Burrows, N.; Moore, K.; Querec, L.; Geiss, L.S.; Engelgau, M.M. Trends in diabetes prevalence among American Indian and Alaska Native children, adolescents, and young adults. Am. J. Public Health 2002, 92, 1485–1490. [Google Scholar] [CrossRef] [PubMed]
- O’dea, K. The price of progress. Diabetes Soc. 2005, 50, 28–30. [Google Scholar]
- Young, T.K.; Reading, J.; Elias, B. Type 2 diabetes mellitus in Canada ‚s First Nations: Status of an epidemic in progress. Can. Med. Assoc. 2000, 163, 561–566. [Google Scholar]
- Pelletier, C.; Dai, S.; Roberts, K.C.; Bienek, A.; Onysko, J.; Pelletier, L. Diabetes in Canada: Facts and Figures from a Public Health Perspective. 2012. Available online: http://www.phac-aspc.gc.ca/cd-mc/diabetes-diabete/index-eng.php (accessed on 4 August 2017).
- First Nations Information Governance Centre. First Nations Regional Health Survey (RHS) 2008/10: National Report on Adults, Youth and Children Living in First Nations Communities; FNIGC: Ottawa, ON, Canada, 2012.
- Taylor, K.W.; Novak, R.F.; Anderson, H.A.; Birnbaum, L.S.; Blystone, C.; De Vito, M.; Jacobs, D.; Köhrle, J.; Lee, D.H.; Rylander, L.; et al. Evaluation of the association between persistent organic pollutants (POPs) and diabetes in epidemiological studies: A national toxicology program workshop review. Environ. Health Perspect. 2013, 121, 774–783. [Google Scholar] [CrossRef] [PubMed]
- Silverstone, A.E.; Rosenbaum, P.F.; Weinstock, R.S.; Bartell, S.M.; Foushee, H.R.; Shelton, C.; Pavuk, M. Polychlorinated biphenyl (PCB) exposure and diabetes: Results from the Anniston community health survey. Environ. Health Perspect. 2012, 120, 727–732. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-H.H.; Steffes, M.W.; Sjödin, A.; Jones, R.S.; Needham, L.L.; Jacobs, D.R. Low dose organochlorine pesticides and polychlorinated biphenyls predict obesity, dyslipidemia, and insulin resistance among people free of diabetes. PLoS ONE 2011, 6, e15977. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Bertrand, K.A.; Choi, A.L.; Hu, F.B.; Laden, F.; Grandjean, P.; Sun, Q. Persistent organic pollutants and type 2 diabetes: A prospective analysis in the nurses’ health study and meta-analysis. Environ. Health Perspect. 2013, 121, 153–161. [Google Scholar] [PubMed]
- Everett, C.J.; Thompson, O.M. Associations of dioxins, furans and dioxin-like PCBs with diabetes and pre-diabetes: Is the toxic equivalency approach useful? Environ. Res. 2012, 118, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Turyk, M.; Anderson, H.; Knobeloch, L.; Imm, P.; Persky, V. Organochlorine exposure and incidence of diabetes in a cohort of great lakes sport fish consumers. Environ. Health Perspect. 2009, 117, 1076–1082. [Google Scholar] [CrossRef] [PubMed]
- Turyk, M.; Anderson, H.A.; Knobeloch, L.; Imm, P.; Persky, V.W. Prevalence of diabetes and body burdens of polychlorinated biphenyls, polybrominated diphenyl ethers, and p,p′-diphenyldichloroethene in Great Lakes sport fish consumers. Chemosphere 2009, 75, 674–679. [Google Scholar] [CrossRef] [PubMed]
- Philibert, A.; Schwartz, H.; Mergler, D. An exploratory study of diabetes in a first nation community with respect to serum concentrations of p,p′-DDE and PCBs and fish consumption. Int. J. Environ. Res. Public Health 2009, 6, 3179–3189. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, M.E.; Borch-Johnsen, K.; Bjerregaard, P. A cross-sectional study of the association between persistent organic pollutants and glucose intolerance among Greenland Inuit. Diabetologia 2008, 51, 1416–1422. [Google Scholar] [CrossRef] [PubMed]
- Aminov, Z.; Haase, R.; Rej, R.; Schymura, M.J.; Santiago-Rivera, A.; Morse, G.; DeCaprio, A.; Carpenter, D.O.; Akwesasne Task Force on the Environment. Diabetes prevalence in relation to serum concentrations of polychlorinated biphenyl (PCB) congener groups and three chlorinated pesticides in a Native American population. Environ. Health Perspect. 2016, 124, 1376. [Google Scholar] [CrossRef] [PubMed]
- Aminov, Z.; Haase, R.; Carpenter, D.O. Diabetes in Native Americans: Elevated risk as a result of exposure to polychlorinated biphenyls (PCBs). Rev. Environ. Health 2016, 31, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Chan, H.M. Persistent organic pollutants and diabetes among Inuit in the Canadian Arctic. Environ. Int. 2017, 101, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Grice, B.A.; Nelson, R.G.; Williams, D.E.; Knowler, W.C.; Mason, C.; Hanson, R.L.; Bullard, K.M.; Pavkov, M.E. Associations between persistent organic pollutants, type 2 diabetes, diabetic nephropathy and mortality. Occup. Environ. Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Seabert, T.A.; Pal, S.; Pinet, B.M.; Haman, F.; Robidoux, M.A.; Imbeault, P.; Krümmel, E.M.; Kimpe, L.E.; Blais, J.M. Elevated contaminants contrasted with potential benefits of ω-3 fatty acids in wild food consumers of two remote first nations communities in Northern Ontario, Canada. PLoS ONE 2014, 9, e90315. [Google Scholar] [CrossRef] [PubMed]
- Kuhnlein, H.V.; Receveur, O. Local cultural animal food contributes high levels of nutrients for Arctic Canadian Indigenous adults and children. J. Nutr. 2007, 137, 1110–1114. [Google Scholar] [CrossRef] [PubMed]
- Sheehy, T.; Kolahdooz, F.; Schaefer, S.E.; Douglas, D.N.; Corriveau, A.; Sharma, S. Traditional food patterns are associated with better diet quality and improved dietary adequacy in Aboriginal peoples in the Northwest Territories, Canada. J. Hum. Nutr. Diet. 2015, 28, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Stevens, S.; Gorman, D.; Pan, A.; Warnakula, S.; Chowdhury, S.; Ward, H.; Johnson, L.; Crowe, F.; Hu, F.B.; et al. Association between fish consumption, long chain omega 3 fatty acids, and risk of cerebrovascular disease: Systematic review and meta-analysis. BMJ 2012, 345, e6698. [Google Scholar] [CrossRef] [PubMed]
- He, K. Fish, Long-Chain Omega-3 Polyunsaturated Fatty Acids and Prevention of Cardiovascular Disease-Eat Fish or Take Fish Oil Supplement? Prog. Cardiovasc. Dis. 2009, 52, 95–114. [Google Scholar] [CrossRef] [PubMed]
- Leung Yinko, S.S.L.; Stark, K.D.; Thanassoulis, G.; Pilote, L. Fish consumption and acute coronary syndrome: A meta-analysis. Am. J. Med. 2014, 127, 848–857. [Google Scholar] [CrossRef] [PubMed]
- Fedor, D.; Kelley, D.S. Prevention of insulin resistance by n-3 polyunsaturated fatty acids. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Yu, X.; Shao, S. Effects of Omega-3 Fatty Acid Supplementation on Glucose Control and Lipid Levels in Type 2 Diabetes: A Meta-Analysis. PLoS ONE 2015, 10, e0139565. [Google Scholar] [CrossRef] [PubMed]
- Nanri, A.; Mizoue, T.; Noda, M.; Takahashi, Y.; Matsushita, Y.; Poudel-tandukar, K. Fish intake and type 2 diabetes in Japanese men and women : The Japan Public Health Center—Based Prospective Study 1–3. Am. J. Clin. Nutr. 2011, 94, 884–891. [Google Scholar] [CrossRef] [PubMed]
- Nkondjock, A.; Receveur, O. Fish-seafood consumption, obesity, and risk of type 2 diabetes: An ecological study. Diabetes Metab. 2003, 29, 635–642. [Google Scholar] [CrossRef]
- Rylander, C.; Sandanger, T.M.; Engeset, D.; Lund, E. Consumption of lean fish reduces the risk of type 2 diabetes mellitus: A prospective population based cohort study of Norwegian women. PLoS ONE 2014, 9, e89845. [Google Scholar] [CrossRef] [PubMed]
- Kuijsten, A.N.; Sijbrands, E.J.; Geleijnse, J.M.; Hofman, A.L.; Witteman, J.C.; Feskens, E.J. Eating Fish and Risk of Type 2 Diabetes: A population-based, prospective follow-up study. Diabetes Care 2009, 32, 2021–2026. [Google Scholar] [CrossRef]
- Djoussé, L.; Gaziano, J.M.; Buring, J.E.; I-Min, L. Dietary omega-3 fatty acids and fish consumption and risk of type 2 diabetes. Am. J. Clin. Nutr. 2011, 93, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, M.; Mozaffarian, D. Long-chain omega-3 fatty acids, fish intake, and the risk of type 2 diabetes mellitus. Am. J. Clin. Nutr. 2009, 90, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-H.; Jacobs, D.R. Inconsistent epidemiological findings on fish consumption may be indirect evidence of harmful contaminants in fish. J. Epidemiol. Community Health 2010, 64, 190–192. [Google Scholar] [CrossRef] [PubMed]
- Wallin, A.; Di Giuseppe, D.; Orsini, N.; Åkesson, A.; Forouhi, N.G.; Wolk, A. Fish consumption and frying of fish in relation to type 2 diabetes incidence: A prospective cohort study of Swedish men. Eur. J. Nutr. 2015. [Google Scholar] [CrossRef] [PubMed]
- Turyk, M.; Fantuzzi, G.; Persky, V.; Freels, S.; Lambertino, A.; Pini, M.; Rhodes, D.H.; Anderson, H.A. Persistent organic pollutants and biomarkers of diabetes risk in a cohort of Great Lakes sport caught fish consumers. Environ. Res. 2015, 140, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Marushka, L.; Batal, M.; Sharp, D.; Schwartz, H.; Ing, A.; Fediuk, K.; Black, A.; Tikhonov, C.; Chan, H.M. Fish consumption is inversely associated with type 2 diabetes in Manitoba First Nations communities. FACETS 2017. [Google Scholar] [CrossRef]
- Marushka, L.; Batal, M.; David, W.; Schwartz, H.; Ing, A.; Fediuk, K.; Sharp, D.; Black, A.; Tikhonov, C.; Chan, H.M. Association between fish consumption, dietary omega-3 fatty acids and persistent organic pollutants intake, and type 2 diabetes in 18 First Nations in Ontario, Canada. Environ. Res. 2017, 156, 725–737. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.; Receveur, O.; Sharp, D.; Schwartz, H.; Tikhonov, C. First Nations Food, Nutrition and Environment Study (FNFNES): Results from Manitoba (2010); University of Northern British Columbia: Prince George, BC, Canada, 2012. [Google Scholar]
- Chan, L.; Receveur, O.; Batal, M.; David, W.; Schwartz, H.; Ing, A.; Fediuk, K.; Black, A.; Tikhonov, C. First Nations Food, Nutrition and Environment Study (FNFNES): Results from Ontario (2011/2012); University of Ottawa: Ottawa, ON, Canada, 2014. [Google Scholar]
- Dimick, J.B.; Ryan, A.M. Methods for evaluating changes in health care policy: The difference-in-differences approach. JAMA 2014, 312, 2401–2402. [Google Scholar] [CrossRef] [PubMed]
- Benmarhnia, T.; Bailey, Z.; Kaiser, D.; Auger, N.; King, N.; Kaufman, J.S. A difference-in-differences approach to assess the effect of a heat action plan on heat-related mortality, and differences in effectiveness according to sex, age, and socioeconomic status (Montreal, Quebec). Environ. Health Perspect. 2016, 124, 1694–1699. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.J.; Kang, C. Identification for difference in differences with cross-section and panel data. Econ. Lett. 2006, 92, 270–276. [Google Scholar] [CrossRef]
- Soares, R. Life Expectancy and Welfare in Latin America and the Caribbean. Health Econ. 2009, 18, 37–54. [Google Scholar] [CrossRef] [PubMed]
- Statistics Canada. Aboriginal Peoples Survey. 94F0043Xie. 2001. Available online: http://www.statcan.gc.ca/bsolc/olc-cel/olc-cel?catno=94F0043XIE&lang=eng (accessed on 5 August 2017).
- Lee, D.H.; Lee, I.K.; Song, K.; Steffes, M.; Toscano, W.; Baker, B.A.; Jacobs, D.R. A Strong Dose-Response Relation Between Serum Concentrations of Persistent Organic Pollutants and Diabetes: Results from the National Health and Examination Survey 1999–2002. Diabetes Care 2006, 29, 1638–1644. [Google Scholar] [CrossRef] [PubMed]
- Codru, N.; Schymura, M.J.; Negoita, S.; Rej, R.; Carpenter, D.O. Diabetes in relation to serum levels of polychlorinated biphenyls and chlorinated pesticides in adult native Americans. Environ. Health Perspect. 2007, 115, 1442–1447. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-H.; Steffes, M.W.; Sjödin, A.; Jones, R.S.; Needham, L.L.; Jacobs, D.R. Low dose of some persistent organic pollutants predicts type 2 diabetes: A nested case-control study. Environ. Health Perspect. 2010, 118, 1235–1242. [Google Scholar] [CrossRef] [PubMed]
- Vasiliu, O.; Cameron, L.; Gardiner, J.; DeGuire, P.; Karmaus, W. Polybrominated Biphenyls, Polychlorinated Biphenyls, Body Weight, and Incidence of Adult-Onset Diabetes Mellitus. Epidemiology 2006, 17, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, E.F.; Deres, D.A.; Hwang, S.A.; Bush, B.; Yang, B.Z.; Tarbell, A.; Jacobs, A. Local fish consumption and serum PCB concentrations among Mohawk men at Akwesasne. Environ Res. 1999. [Google Scholar] [CrossRef] [PubMed]
- Duarte-Davidson, R.; Jones, K.C. Polychlorinated biphenyls (PCBs) in the UK population: Estimated intake, exposure and body burden. Sci. Total Environ. 1994, 151, 131–152. [Google Scholar] [CrossRef]
- Gray, S.L.; Shaw, A.C.; Gagne, A.X.; Chan, H.M. Chronic exposure to PCBs (Aroclor 1254) exacerbates obesity-induced insulin resistance and hyperinsulinemia in mice. J. Toxicol. Environ. Health A 2013, 76, 701–715. [Google Scholar] [CrossRef] [PubMed]
- Ruzzin, J.; Petersen, R.; Meugnier, E.; Madsen, L.; Lock, E.J.; Lillefosse, H.; Ma, T.; Pesenti, S.; Sonne, S.B.; Marstrand, T.T.; et al. Persistent organic pollutant exposure leads to insulin resistance syndrome. Environ. Health Perspect. 2010, 118, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.M.; Fjære, E.; Lock, E.J.; Naville, D.; Amlund, H.; Meugnier, E.; Battistoni, B.L.M.; Frøyland, L.; Madsen, L.; Jessen, N. Chronic consumption of farmed salmon containing persistent organic pollutants causes insulin resistance and obesity in mice. PLoS ONE 2011, 6, e25170. [Google Scholar] [CrossRef] [PubMed]
- Neel, B.A.; Sargis, R.M. The paradox of progress: Environmental disruption of metabolism and the diabetes epidemic. Diabetes 2011, 60, 1838–1848. [Google Scholar] [CrossRef] [PubMed]
- Newbold, R.R.; Padilla-Banks, E.; Jefferson, W.N.; Heindel, J.J. Effects of endocrine disruptors on obesity. Int. J. Androl. 2008, 31, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, M.K.; Turner, N. Mitochondrial dysfunction and insulin resistance: An update. Endocr. Connect. 2015, 4, R1–R15. [Google Scholar] [CrossRef] [PubMed]
- De Tata, V. Association of dioxin and other persistent organic pollutants (POPs) with diabetes: Epidemiological evidence and new mechanisms of beta cell dysfunction. Int. J. Mol. Sci. 2014, 15, 7787–7811. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.S.; Sharp, S.J.; Luben, R.N.; Khaw, K.T.; Bingham, S.A.; Wareham, N.J.; Forouhi, N.G. Association Between Type of Dietary Fish and Seafood Intake and the Risk of Incident Type 2 Diabetes. Diabetes Care 2009, 32, 1857–1863. [Google Scholar] [CrossRef] [PubMed]
- Villegas, R.; Xiang, Y.B.; Elasy, T.; Li, H.L.; Yang, G.; Cai, H.; Ye, F.; Gao, Y.T.; Shyr, Y.; Zheng, W.; et al. Fish, shellfish, and long-chain n23 fatty acid consumption and risk of incident type 2 diabetes in middle-aged Chinese men and women 1–3. Am. J. Clin. Nutr. 2011, 94, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Paquet, C.; Propsting, S.; Daniel, M. Total n-3 fatty acid and SFA intakes in relation to insulin resistance in a Canadian First Nation at risk for the development of type 2 diabetes. Public Health Nutr. 2014, 17, 1337–1341. [Google Scholar] [CrossRef] [PubMed]
- Ebbesson, S.O.E.; Risica, P.M.; Ebbesson, L.O.E.E.A. Omega-3 Fatty Acids Improve Glucose Tolerance and Components of the Metabolic Syndrome in Alaskan Eskimos. Int. J. Circumpolar. Health 2005, 64, 396–408. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.-S.; Huang, T.; Yang, J.; Fu, Y.-Q.; Li, D. Marine N-3 Polyunsaturated Fatty Acids Are Inversely Associated with Risk of Type 2 Diabetes in Asians: A Systematic Review and Meta-Analysis. PLoS ONE 2012, 7, e44525. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, A.; Borrell, A.; Reijnders, P.J.H. Patterns in geographical and temporal variation of organochlorine pollutant concentrations in marine mammals. Mar. Environ. Res. 2002, 53, 425–452. [Google Scholar] [CrossRef]
- Chevalier, N.; Fénichel, P. Endocrine disruptors: New players in the pathophysiology of type 2 diabetes? Diabetes Metab. 2015, 41, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Welshons, W.V.; Thayer, K.A.; Judy, B.M.; Taylor, J.A.; Curran, E.M.; vom Saal, F.S. Large effects from small exposures. I. Mechanisms for endocrine-disrupting chemicals with estrogenic activity. Environ. Health Perspect. 2003, 111, 994–1006. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-H.; Porta, M.; Jacobs, D.R.; Vandenberg, L.N. Chlorinated persistent organic pollutants, obesity, and type 2 diabetes. Endocr. Rev. 2014, 35, 557–601. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.F.; Laird, B.D.; Chan, H.M. Mercury diminishes the cardiovascular protective effect of omega-3 polyunsaturated fatty acids in the modern diet of Inuit in Canada. Environ. Res. 2017, 152, 470–477. [Google Scholar] [CrossRef] [PubMed]
- Laird, B.D.; Goncharov, A.B.; Egeland, G.M.; Chan, H.M. Dietary advice on Inuit traditional food use needs to balance benefits and risks of mercury, selenium, and n3 fatty acids. J. Nutr. 2013, 143, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Loring, P.A.; Duffy, L.K.; Murray, M.S. A risk-benefit analysis of wild fish consumption for various species in Alaska reveals shortcomings in data and monitoring needs. Sci. Total Environ. 2010, 408, 4532–4541. [Google Scholar] [CrossRef] [PubMed]
- Wennberg, M.; Strömberg, U.; Bergdahl, I.A.; Jansson, J.H.; Kauhanen, J.; Norberg, M.; Salonen, J.T.; Skerfving, S.; Tuomainen, T.P.; Vessby, B.; et al. Myocardial infarction in relation to mercury and fatty acids from fish: A risk-benefit analysis based on pooled Finnish and Swedish data in men. Am. J. Clin. Nutr. 2012, 96, 706–713. [Google Scholar] [CrossRef] [PubMed]
- Foran, J.A.; Good, D.H.; Carpenter, D.O.; Hamilton, M.C.; Knuth, B.A.; Schwager, S.J. Quantitative analysis of the benefits and risks of consuming farmed and wild salmon. J. Nutr. 2005, 135, 2639–2643. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, K.S. Health benefits and potential risks related to consumption of fish or fish oil. Regul. Toxicol. Pharmacol. 2003, 38, 336–344. [Google Scholar] [CrossRef] [PubMed]
- Sirot, V.; Leblanc, J.-C.; Margaritis, I. A risk–benefit analysis approach to seafood intake to determine optimal consumption. Br. J. Nutr. 2012, 107, 1812–1822. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D. Fish, mercury, selenium and cardiovascular risk: Current evidence and unanswered questions. Int. J. Environ. Res. Public Health 2009, 6, 1894–1916. [Google Scholar] [CrossRef] [PubMed]
- Christensen, K.Y.; Raymond, M.R.; Thompson, B.A.; Anderson, H.A. Fish Consumption, Levels of Nutrients and Contaminants, and Endocrine-Related Health Outcomes Among Older Male Anglers in Wisconsin. J. Occup. Environ. Med. 2016, 58, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Rylander, L.; Rignell-hydbom, A.; Hagmar, L. Environmental Health : A Global A cross-sectional study of the association between persistent organochlorine pollutants and diabetes. Environ. Health 2005, 6, 2–7. [Google Scholar]
- Wang, S.L.; Tsai, P.C.; Yang, C.Y.; Guo, Y.L. Increased risk of diabetes and polychlorinated biphenyls and dioxins A 24-year follow-up study of the yucheng cohort. Diabetes Care 2008, 31, 1574–1579. [Google Scholar] [CrossRef] [PubMed]
- Neff, M.R.; Bhavsar, S.P.; Ni, F.J.; Carpenter, D.O.; Drouillard, K.; Fisk, A.T.; Arts, M.T. Risk-benefit of consuming Lake Erie fish. Environ. Res. 2014, 134, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.J. Vitamin D deficiency and diabetes. Biochem. J. 2017, 474, 1321–1332. [Google Scholar] [CrossRef] [PubMed]
- Mitri, J.; Muraru, M.D.; Pittas, A.G. Vitamin D and type 2 diabetes: A systematic review. Eur. J. Clin. Nutr. 2011, 65, 1005–1015. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P.; Stranges, S. Epidemiology of selenium and type 2 diabetes: Can we make sense of it? Free Radic. Biol. Med. 2013, 65, 1557–1564. [Google Scholar] [CrossRef] [PubMed]
- Mansuri, S. Vitamin D and Type 2 Diabetes Mellitus. Master’s Theses, School of Graduate Studies, University of Toronto, Toronto, ON, Canada, 2014. Available online: https://tspace.library.utoronto.ca/handle/1807/67979 (accessed on 10 August 2017).
- Nielsen, N.O.; Bjerregaard, P.; Rønn, P.F.; Friis, H.; Andersen, S.; Melbye, M.; Lundqvist, M.; Cohen, A.S.; Hougaard, D.M.; Jørgensen, M.E. Associations between vitamin D status and type 2 diabetes measures among inuit in greenland may be affected by other factors. PLoS ONE 2016, 11, e0152763. [Google Scholar] [CrossRef] [PubMed]

| Variables | Ontario | Manitoba | ||||
|---|---|---|---|---|---|---|
| Total | Male | Female | Total | Male | Female | |
| Sample size | 1426 | 533 | 893 | 706 | 229 | 477 |
| Type 2 diabetes | 327 | 110 | 217 | 123 | 47 | 76 |
| Type-2 diabetes weighted (%) | 24.4 | 23.5 | 24.6 | 22.0 | 26.0 | 20.0 |
| Type-2 diabetes standardized (%) | 25.0 | 23.7 | 25.7 | 28.4 | 32.1 | 26.5 |
| Age | 46.5 (15.8) | 47.3 (16.0) | 45.9 (15.6) | 42.3 (14.4) | 43.1 (14.3) | 42.0 (14.5) |
| BMI (kg/m2) | 30.9 (5.9) | 30.4 (5.4) | 31.1(6.1) | 30.3 (6.4) | 29.0 (5.8) | 30.9 (6.6) |
| Moderate to vigorous physical activity | 498 (34.9) | 241 (45.2) | 257 (28.8) | 189 (26.8) | 85 (37.1) | 104 (21.8) |
| Smoking (%) | 723 (50.7) | 276 (51.8) | 447 (50.1) | 444 (62.9) | 136 (59.4) | 308 (64.6) |
| Years of education | 11.1 (3.8) | 10.5 (3.5) | 11.5 (3.9) | 9.8 (2.5) | 9.6 (2.7) | 9.9 (2.4) |
| Total energy (kcal/day) | 2042.1 (1026.8) | 2344.5 (1222.1) | 1861.6 (840.4) | 1979.0 (1056.0) | 2315.8 (1219.5) | 1817.3 (926.5) |
| Fruit and vegetable intake (g/day) | 157.6 (234.6) | 141.7 (219.7) | 167.1 (242.7) | 113.1 (242.8) | 88.8 (161.1) | 124.8 (272.9) |
| Household size | 3.4 (2.0) | 3.0 (2.0) | 3.6 (2.0) | 4.4 (2.6) | 3.9 (2.8) | 4.6 (2.5) |
| Fish Species | Ontario | Manitoba | ||||||
|---|---|---|---|---|---|---|---|---|
| Fish Intake | EPA + DHA | DDE | PCBs | Fish Intake | EPA + DHA | DDE | PCBs | |
| g/day | g/100 g | ng/g | ng/g | g/day | g/100 g | ng/g | ng/g | |
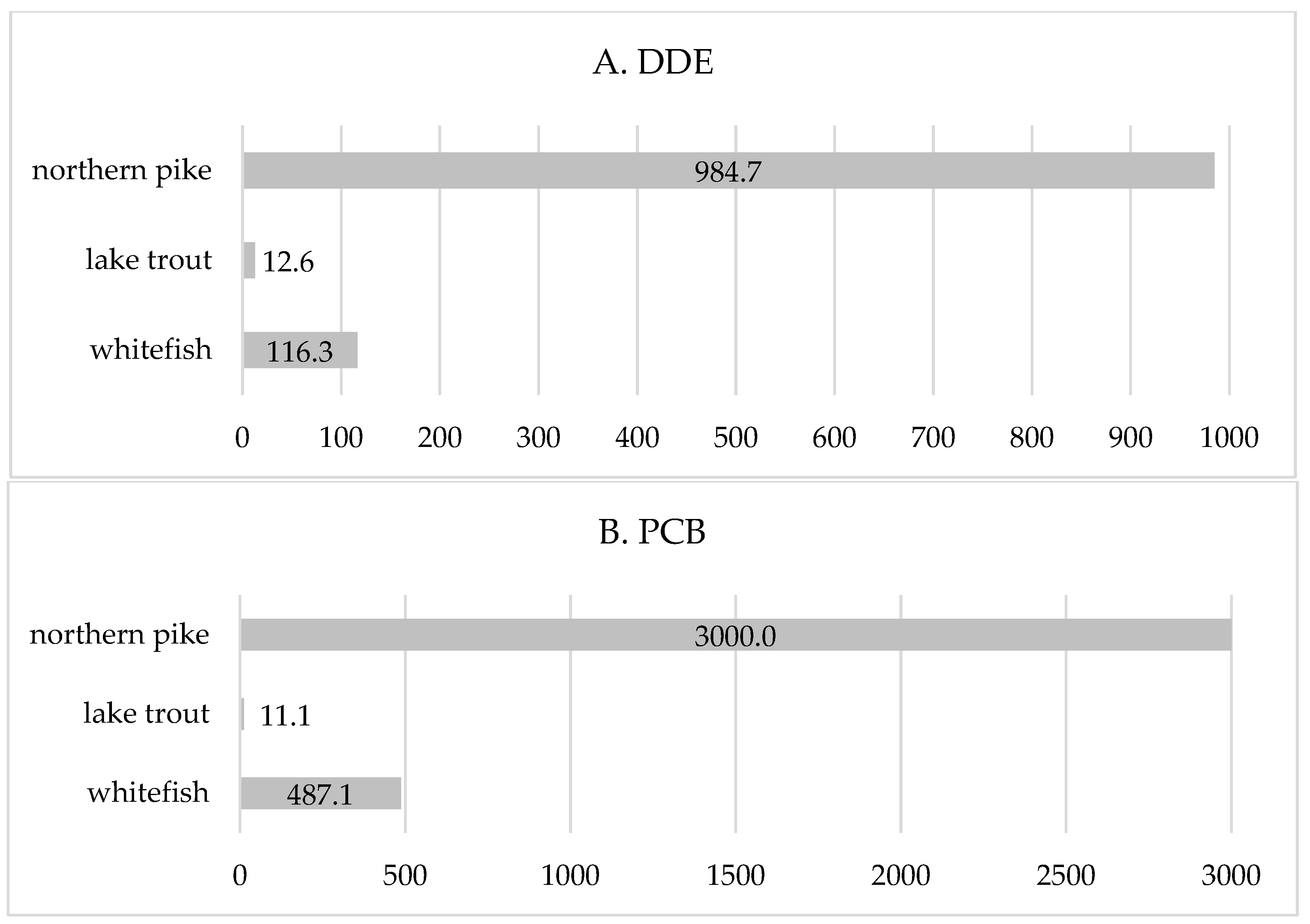
| walleye | 5.6 (13.5) | 0.31 (0.05) | 2.69 (3.36) | 14.75 (19.44) | 3.7 (9.1) | 0.31 (0.05) | - | - |
| whitefish | 2.5 (9.6) | 1.24 (0.56) | 5.89 (7.21) | 14.56 (24.27) | 2.0 (8.1) | 1.24 (0.56) | 1.28 (0.79) | 0.21 (0.26) |
| lake trout | 1.1 (5.6) | 0.73 (0.14) | 26.65 (24.32) | 63.69 (83.54) | 1.4 (5.9) | 0.73 (0.14) | 11.73 (5.76) | 9.24 (2.58) |
| northern pike | 1.7 (7.5) | 0.27 (0.07) | 1.85 (1.94) | 8.98 (11.65) | 1.0 (4.0) | 0.27 (0.07) | 0.15 (0.31) | 0.03 (0.10) |
| yellow perch | 0.5 (2.8) | 0.25 (0.04) | 3.11 (4.18) | 33.18 (62.47) | 0.2 (1.7) | 0.25 (0.04) | - | - |
| subtotal | 11.5 (28.0) | 0.56 (0.42) | 6.28 (11.82) | 22.01 (40.49) | 8.4 (18.4) | 0.56 (0.42) | 1.06 (2.92) | 0.59 (2.21) |
| total | 14.7 (34.1) | 0.67 (0.48) | 10.08 (19.62) | 35.21 (68.06) | 10.7 (24.5) | 0.53 (0.28) | 2.05 (4.37) | 2.00 (5.40) |
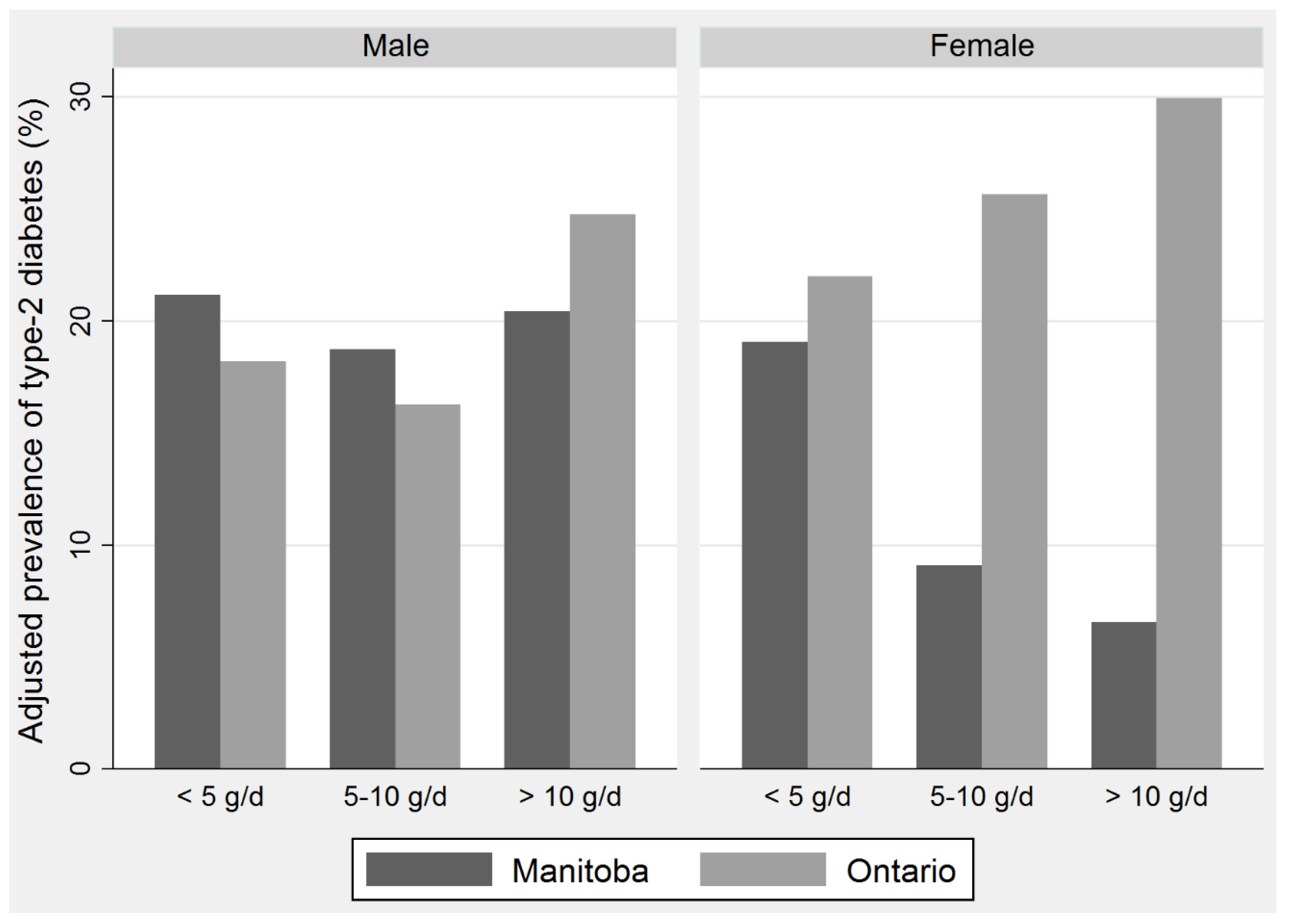
| Variables | <5 g/day | 5–10 g/day | >10 g/day | |||
|---|---|---|---|---|---|---|
| Ontario | Mean | 95% CI | Mean | 95% CI | Mean | 95% CI |
| Male | ||||||
| n | 225 | 86 | 222 | |||
| Total fish intake (g/day) | 1.28 | 0.93–1.64 | 7.11 | 6.58–7.63 | 62.19 | 41.48–82.89 |
| EPA + DHA (mg/day) | 22.04 | 12.50–31.59 | 119.87 | 90.14–149.60 | 935.09 | 636.61–1235.36 |
| DDE (ng/kg/day) | 0.08 | 0.04–0.12 | 0.31 | 0.13–0.50 | 3.19 | 1.60–4.76 |
| PCBs (ng/kg/day) | 0.37 | 0.25–0.49 | 1.41 | 0.69–2.12 | 11.28 | 7.20–15.37 |
| Female | ||||||
| n | 573 | 113 | 207 | |||
| Total fish intake (g/day) | 0.97 | 0.83–1.11 | 6.94 | 6.78–7.09 | 39.21 | 27.49–50.93 |
| EPA + DHA (mg/day) | 14.65 | 12.00–17.31 | 115.24 | 100.57–129.90 | 550.63 | 398.00–703.28 |
| DDE (ng/kg/day) | 0.06 | 0.04–0.09 | 0.5 | 0.26–0.65 | 3.61 | 1.39–5.84 |
| PCBs (ng/kg/day) | 0.249 | 0.17–0.32 | 1.723 | 1.05–2.39 | 9.86 | 4.47–15.24 |
| Manitoba | ||||||
| Male | ||||||
| n | 104 | 32 | 93 | |||
| Total fish intake (g/day) | 1.61 | 0.66–2.57 | 6.9 | 6.29–7.50 | 34.4 | 19.74–49.07 |
| EPA + DHA (mg/day) | 6.27 | 2.58–9.97 | 28.6 | 21.45–35.74 | 195.4 | 72.73–318.07 |
| DDE (ng/kg/day) | 0.012 | 0.001–0.02 | 0.02 | 0.003–0.71 | 0.63 | 0.07–1.21 |
| PCBs (ng/kg/day) | 0.012 | 0.002–0.03 | 0.02 | 0.005–0.09 | 0.45 | 0.09–0.81 |
| Female | ||||||
| n | 346 | 55 | 76 | |||
| Total fish intake (g/day) | 1.31 | 1.08–1.54 | 7.02 | 6.75–7.29 | 30.85 | 26.92–34.78 |
| EPA + DHA (mg/day) | 5.22 | 4.00–6.44 | 31.71 | 27.65–35.76 | 183.12 | 144.41–221.85 |
| DDE (ng/kg/day) | 0.004 | 0.001–0.007 | 0.06 | 0.03–0.09 | 0.34 | 0.26–0.41 |
| PCBs (ng/kg/day) | 0.003 | 0.0004–0.006 | 0.07 | 0.003–0.14 | 0.26 | 0.17–0.35 |
| Variables | Total Population | Female | Male | ||
|---|---|---|---|---|---|
| Model 1 | Model 2 | Model 3 | Model 3 | Model 3 | |
| T2D in Ontario First Nations | 0.53 ** (0.33–0.87) | 0.52 ** (0.30–0.91) | 0.53 * (0.27–1.03) | 0.64 (0.29–1.44) | 0.32 ** (0.12–0.82) |
| Medium fish consumers | 0.43 ** (0.22–0.84) | 0.58 * (0.31–1.09) | 0.59 (0.29–1.18) | 0.29 *** (0.13–0.62) | 1.45 (0.46–4.56) |
| Medium fish consumers in Ontario | 3.05 *** (1.32–7.08) | 2.12 * (0.94–4.77) | 2.22 * (0.86–5.68) | 3.08 ** (1.13–8.42) | 1.79 (0.27–11.67) |
| High fish consumers in Ontario | 2.76 ** (1.25–6.09) | 3.39 *** (1.49–7.68) | 3.53 *** (1.47–8.45) | 14.96 *** (372–60.11) | 2.85 ** (1.14–8.04) |
| n | 2080 | 2080 | 2080 | 1329 | 751 |
| DDE Intake | PCB Intake | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Slope 1 (<BP) | BP | Slope 2 (>BP) | Slope 1 (<BP) | BP | Slope 2 (>BP) | ||||||
| OR | 95% CI | ng/kg/day | SE | OR | 95% CI | OR | 95% CI | ng/kg/day | SE | OR | 95% CI |
| 1.03 | 0.99–1.07 | 2.11 | 1.53 | 2.29 | 1.26–4.17 | 1.00 | 0.96–1.03 | 1.47 | 1.95 | 1.44 | 1.09–1.89 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marushka, L.; Hu, X.; Batal, M.; Sadik, T.; Schwartz, H.; Ing, A.; Fediuk, K.; Tikhonov, C.; Chan, H.M. The Relationship between Persistent Organic Pollutants Exposure and Type 2 Diabetes among First Nations in Ontario and Manitoba, Canada: A Difference in Difference Analysis. Int. J. Environ. Res. Public Health 2018, 15, 539. https://doi.org/10.3390/ijerph15030539
Marushka L, Hu X, Batal M, Sadik T, Schwartz H, Ing A, Fediuk K, Tikhonov C, Chan HM. The Relationship between Persistent Organic Pollutants Exposure and Type 2 Diabetes among First Nations in Ontario and Manitoba, Canada: A Difference in Difference Analysis. International Journal of Environmental Research and Public Health. 2018; 15(3):539. https://doi.org/10.3390/ijerph15030539
Chicago/Turabian StyleMarushka, Lesya, Xuefeng Hu, Malek Batal, Tonio Sadik, Harold Schwartz, Amy Ing, Karen Fediuk, Constantine Tikhonov, and Hing Man Chan. 2018. "The Relationship between Persistent Organic Pollutants Exposure and Type 2 Diabetes among First Nations in Ontario and Manitoba, Canada: A Difference in Difference Analysis" International Journal of Environmental Research and Public Health 15, no. 3: 539. https://doi.org/10.3390/ijerph15030539
APA StyleMarushka, L., Hu, X., Batal, M., Sadik, T., Schwartz, H., Ing, A., Fediuk, K., Tikhonov, C., & Chan, H. M. (2018). The Relationship between Persistent Organic Pollutants Exposure and Type 2 Diabetes among First Nations in Ontario and Manitoba, Canada: A Difference in Difference Analysis. International Journal of Environmental Research and Public Health, 15(3), 539. https://doi.org/10.3390/ijerph15030539