Biocapture of CO2 by Different Microalgal-Based Technologies for Biogas Upgrading and Simultaneous Biogas Slurry Purification under Various Light Intensities and Photoperiods
Abstract
1. Introduction
2. Methods and Materials
2.1. Collection of Algal Strains and Culturing Conditions for the Selected Microalgal-Based Technologies
2.1.1. Culture 1: Mono-Cultivation of Microalgae Strain
2.1.2. Culture 2: Co-Cultivation of Microalgae with Fungi
2.1.3. Culture 3: Co-Cultivation of Microalgae with Activated Sludge
2.2. Biogas Slurry and Biogas
2.3. Photobioreactor
2.4. Experimental Procedure
2.5. Sampling and Analyses
2.6. Statistical Analyses
3. Results and Discussion
3.1. The Strains Growth for the Selected Three Microalgae-Based Technologies at Different Photoperiod Treatments
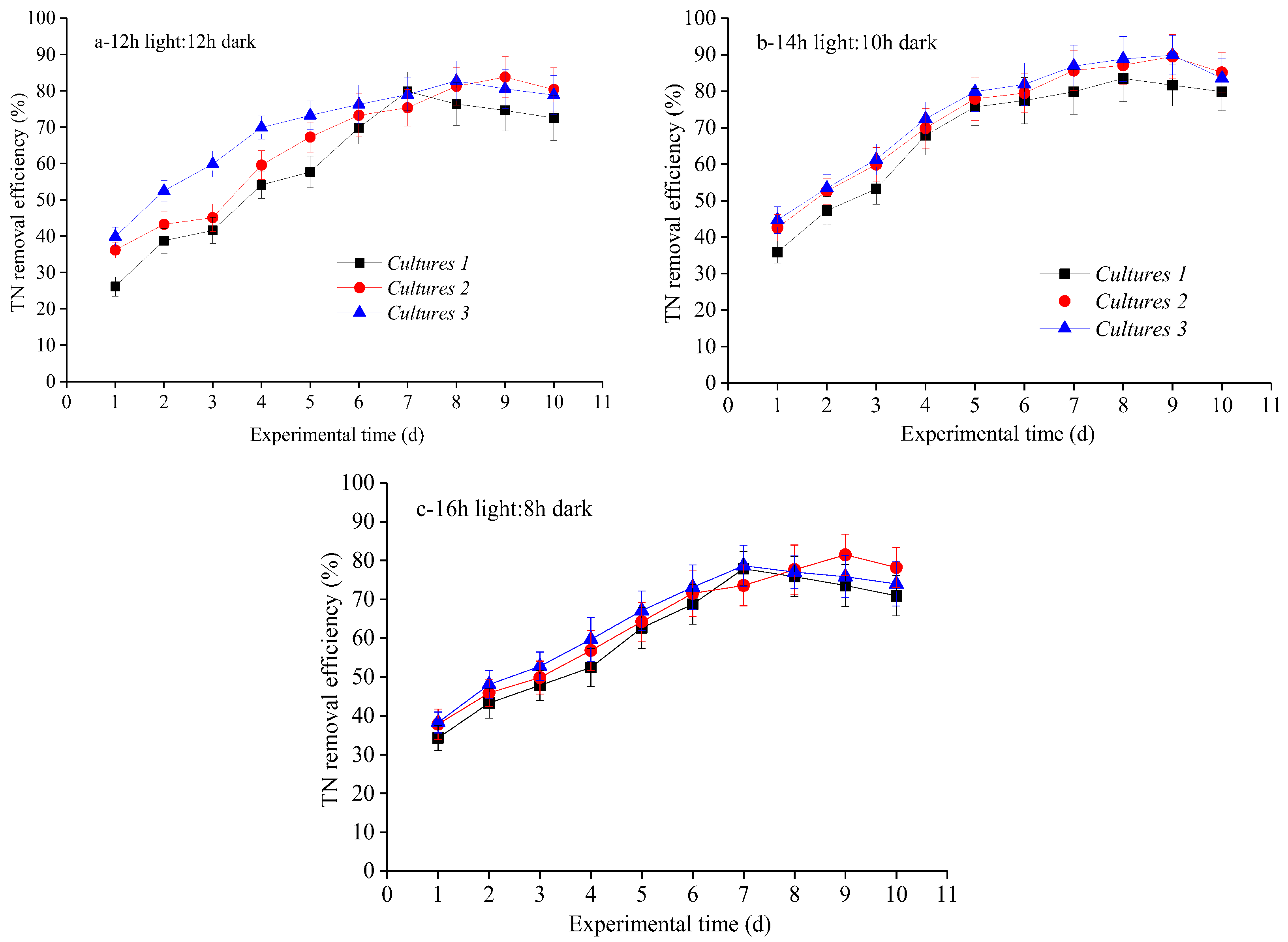
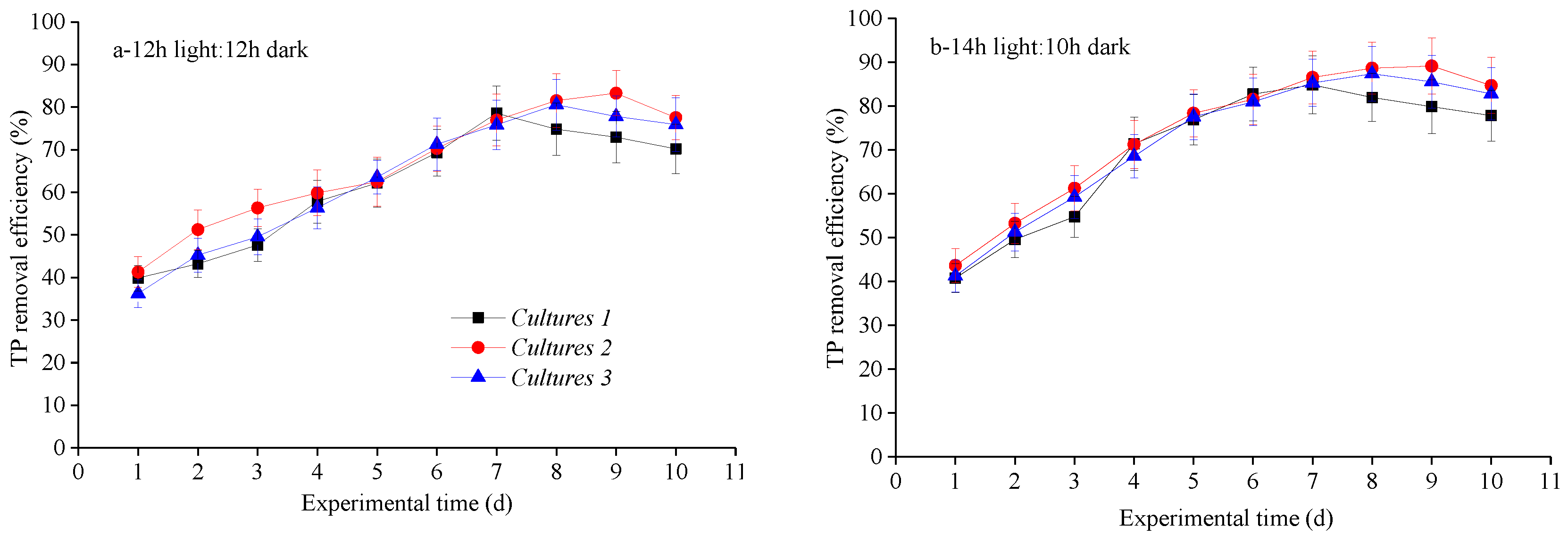
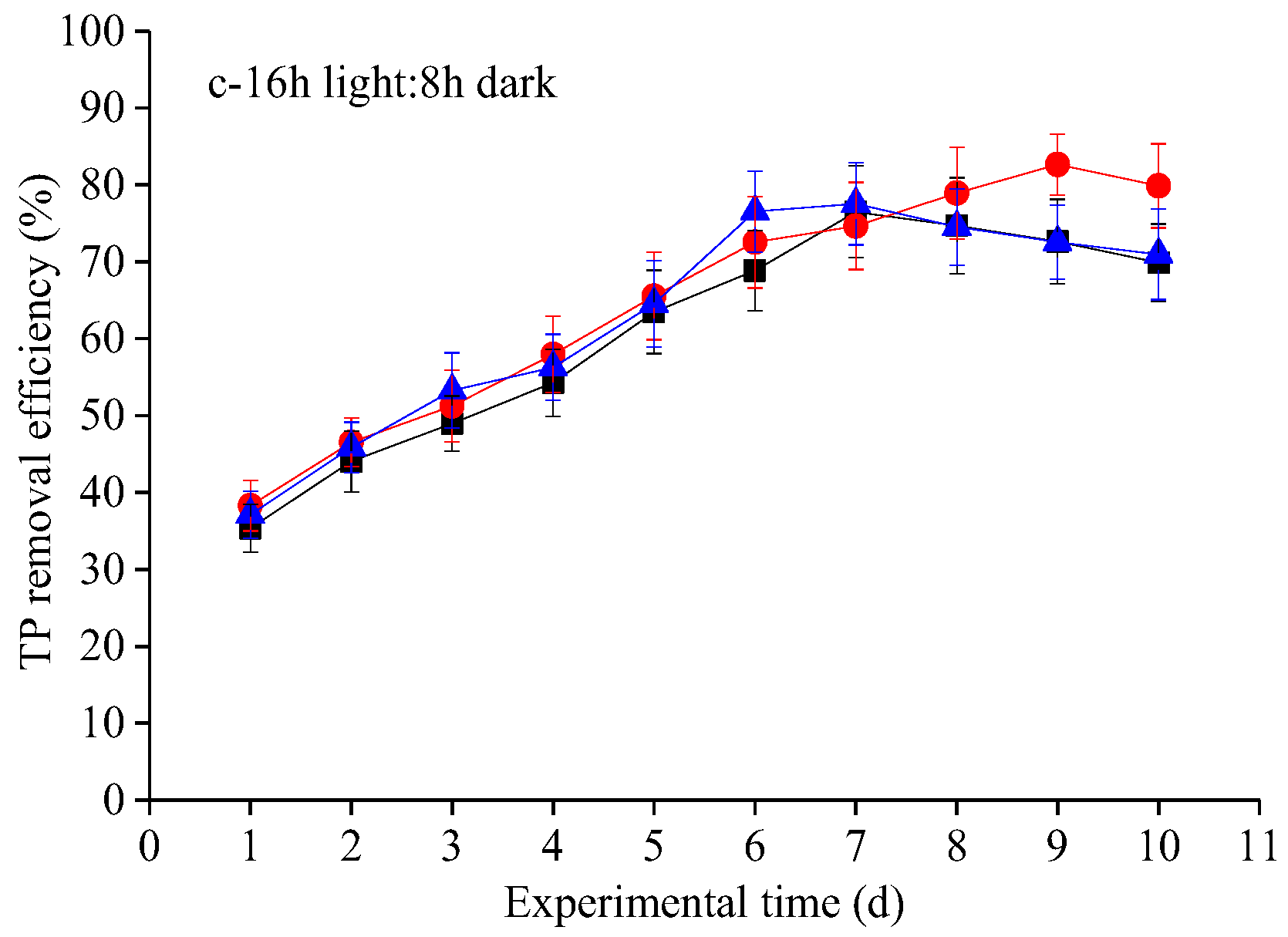
3.2. Nutrient Removal Efficiencies at Different Photoperiod Treatments
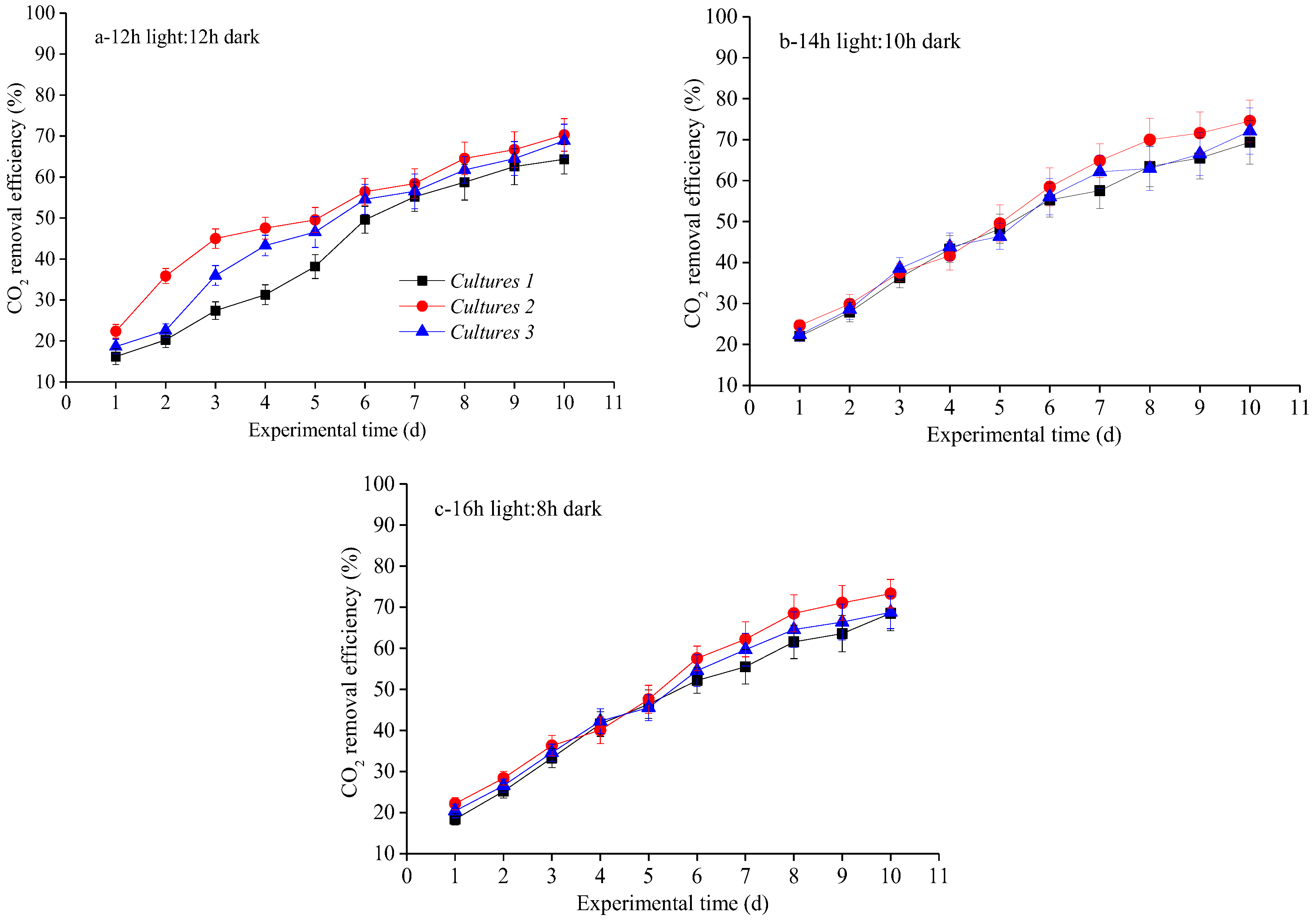
3.3. Biogas Upgrading
3.4. Economic Efficiency of the Energy Consumption
3.5. Optimization of LED Light Intensities Treatments for the Selected Two Cultures
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhu, L.; Yan, C.; Li, Z. Microalgal cultivation with biogas slurry for biofuel production. Bioresour. Technol. 2016, 220, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Zhu, L.; Wang, Y. Photosynthetic CO2 uptake by microalgae for biogas upgrading and simultaneously biogas slurry decontamination by using of microalgae photobioreactor under various light wavelengths, light intensities, and photoperiods. Appl. Energy 2016, 178, 9–18. [Google Scholar] [CrossRef]
- Zhang, Y.; Bao, K.; Wang, J.; Zhao, Y.; Hu, C. Performance of mixed LED light wavelengths on nutrient removal and biogas upgrading by different microalgal-based treatment technologies. Energy 2017, 130, 392–401. [Google Scholar] [CrossRef]
- Sigarchian, S.G.; Paleta, R.; Malmquist, A.; Pina, A. Feasibility study of using a biogas engine as backup in a decentralized hybrid (PV/wind/battery) power generation system—Case study Kenya. Energy 2015, 90, 1830–1841. [Google Scholar] [CrossRef]
- Bacsik, Z.; Vasiliev, P.; Hedin, N. Selective separation of CO2 and CH4 for biogas upgrading on zeolite NaKA and SAPO-56. Appl. Energy 2016, 162, 613–621. [Google Scholar] [CrossRef]
- Luo, G.; Wang, W.; Angelidaki, I. A new degassing membrane coupled upflow anaerobic sludge blanket (UASB) reactor to achieve in-situ biogas upgrading and recovery of dissolved CH4 from the anaerobic effluent. Appl. Energy 2014, 132, 536–542. [Google Scholar] [CrossRef]
- Sun, Q.; Li, H.; Yan, J.; Liu, L.; Yu, Z.; Yu, X. Selection of appropriate biogas upgrading technology—A review of biogas cleaning, upgrading and utilisation. Renew. Sustain. Energy Rev. 2015, 51, 521–532. [Google Scholar] [CrossRef]
- Xu, H.; Wang, K.; Holmes, D.E. Bioelectrochemical removal of carbon dioxide (CO2): An innovative method for biogas upgrading. Bioresour. Technol. 2014, 173, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Wei, H.; Li, P.; Miao, X.; Zhong, J. CO2 biofixation and fatty acid composition of Scenedesmus obliquus and Chlorella pyrenoidosa in response to different CO2 levels. Bioresour. Technol. 2010, 102, 3071–3076. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhao, Y.; Ge, Z.; Zhang, H.; Sun, S. Selection of microalgae for simultaneous biogas upgrading and biogas slurry nutrient reduction under various photoperiods. J. Chem. Technol. Biotechnol. 2016, 91, 1982–1989. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, J.; Zhang, H.; Yan, C.; Zhang, Y. Effects of various LED light wavelengths and intensities on microalgae-based simultaneous biogas upgrading and digestate nutrient reduction process. Bioresour. Technol. 2013, 136C, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Sun, S.; Hu, C.; Zhang, H.; Xu, J.; Ping, L. Performance of three microalgal strains in biogas slurry purification and biogas upgrade in response to various mixed light-emitting diode light wavelengths. Bioresour. Technol. 2015, 187, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Zheng, Z. Performance of mixed LED light wavelengths on biogas upgrade and biogas fluid removal by microalga Chlorella sp. Appl. Energy 2014, 113, 1008–1014. [Google Scholar] [CrossRef]
- Leite, G.B.; Abdelaziz, A.E.M.; Hallenbeck, P.C. Algal biofuels: Challenges and opportunities. Bioresour. Technol. 2013, 145, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Pragya, N.; Pandey, K.K.; Sahoo, P.K. A review on harvesting, oil extraction and biofuels production technologies from microalgae. Renew. Sustain. Energy Rev. 2013, 24, 159–171. [Google Scholar] [CrossRef]
- Salim, S.; Vermuë, M.H.; Wijffels, R.H. Ratio between autoflocculating and target microalgae affects the energy-efficient harvesting by bio-flocculation. Bioresour. Technol. 2012, 118, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Hu, B. A novel method to harvest microalgae via co-culture of filamentous fungi to form cell pellets. Bioresour. Technol. 2012, 114, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Marazzi, F.; Ficara, E.; Fornaroli, R.; Mezzanotte, V. Factors Affecting the Growth of Microalgae on Blackwater from Biosolid Dewatering. Water Air Soil Pollut. 2017, 228, 68. [Google Scholar] [CrossRef]
- Marazzi, F.; Sambusiti, C.; Monlau, F.; Cecere, S.E.; Scaglione, D.; Barakat, A.; Mezzanotte, V.; Ficara, E. A novel option for reducing the optical density of liquid digestate to achieve a more productive microalgal culturing. Algal Res. 2017, 24, 19–28. [Google Scholar] [CrossRef]
- Sun, S.; Ge, Z.; Zhao, Y.; Hu, C.; Zhang, H.; Ping, L. Performance of CO2 concentrations on nutrient removal and biogas upgrading by integrating microalgal strains cultivation with activated sludge. Energy 2016, 97, 229–237. [Google Scholar] [CrossRef]
- Hakgeun, J.; Junghoon, L.; Misun, C. Energy efficient growth control of microalgae using photobiological methods. Renew. Energy 2013, 54, 161–165. [Google Scholar]
- Cheirsilp, B.; Torpee, S. Enhanced growth and lipid production of microalgae under mixotrophic culture condition: Effect of light intensity, glucose concentration and fed-batch cultivation. Bioresour. Technol. 2012, 110, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Luo, X.; Zheng, Z. Effects of various LED light qualities and light intensity supply strategies on purification of slurry from anaerobic digestion process by Chlorella vulgaris. Int. Biodeterior. Biodegrad. 2013, 79, 81–87. [Google Scholar] [CrossRef]
- Yan, C.; Zhang, L.; Luo, X.; Zheng, Z. Effects of various LED light wavelengths and intensities on the performance of purifying synthetic domestic sewage by microalgae at different influent C/N ratios. Ecol. Eng. 2013, 51, 24–32. [Google Scholar] [CrossRef]
- Yan, C.; Zhao, Y.; Zheng, Z.; Luo, X. Effects of various LED light wavelengths and light intensity supply strategies on synthetic high-strength wastewater purification by Chlorella vulgaris. Biodegradation 2013, 24, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Zheng, Z. Performance of photoperiod and light intensity on biogas upgrade and biogas effluent nutrient reduction by the microalgae Chlorella sp. Bioresour. Technol. 2013, 139, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.G.; La, H.J.; Ahn, C.Y.; Park, Y.H.; Oh, H.M. Harvest of Scenedesmus sp. with bioflocculant and reuse of culture medium for subsequent high-density cultures. Bioresour. Technol. 2011, 102, 3163–3168. [Google Scholar] [CrossRef] [PubMed]
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energy Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef]
- Ugwu, C.U.; Aoyagi, H.; Uchiyama, H. Influence of irradiance, dissolved oxygen concentration, and temperature on the growth of Chlorella sorokiniana. Photosynthetica 2007, 45, 309–311. [Google Scholar] [CrossRef]
- Muradov, N.; Taha, M.; Miranda, A.F.; Wrede, D.; Kadali, K.; Gujar, A.; Stevenson, T.; Ball, A.S.; Mouradov, A. Fungal-assisted algal flocculation: Application in wastewater treatment and biofuel production. Biotechnol. Biofuels 2015, 8, 24. [Google Scholar] [CrossRef] [PubMed]
- Gultom, S.O.; Hu, B. Review of microalgae harvesting via co-pelletization with filamentous fungus. Energies 2013, 6, 5921–5939. [Google Scholar] [CrossRef]
- Wrede, D.; Taha, M.; Miranda, A.F.; Kadali, K.; Stevenson, T.; Ball, A.S.; Mouradov, A. Co-cultivation of fungal and microalgal cells as an efficient system for harvesting microalgal cells, lipid production and wastewater treatment. PLoS ONE 2014, 9, e113497. [Google Scholar] [CrossRef] [PubMed]
- De-Bashan, L.E.; Hernandez, J.P.; Morey, T.; Bashan, Y. Microalgae growth-promoting bacteria as “helpers” for microalgae: A novel approach for removing ammonium and phosphorus from municipal wastewater. Water Res. 2004, 38, 466–474. [Google Scholar] [CrossRef] [PubMed]
- De-Bashan, L.E.; Moreno, M.; Hernandez, J.P.; Bashan, Y. Removal of ammonium and phosphorus ions from synthetic wastewater by the microalgae Chlorella vulgaris coimmobilized in alginate beads with the microalgae growth-promoting bacterium Azospirillum brasilense. Water Res. 2002, 36, 2941–2948. [Google Scholar] [CrossRef]
- Perezgarcia, O.; Escalante, F.M.; Debashan, L.E.; Bashan, Y. Heterotrophic cultures of microalgae: Metabolism and potential products. Water Res. 2011, 45, 11–36. [Google Scholar] [CrossRef] [PubMed]
- Rachlin, J.W.; Grosso, A. The effects of pH on the growth of Chlorella vulgaris and its interactions with cadmium toxicity. Arch. Environ. Contam. Toxicol. 1991, 20, 505–508. [Google Scholar] [CrossRef] [PubMed]
- Zwietering, M.H.; Jongenburger, I.; Rombouts, F.M.; Van’t Riet, K. Modeling of the bacterial growth curve. Appl. Environ. Microbiol. 1990, 56, 1875–1881. [Google Scholar] [PubMed]
- Serejo, M.L.; Posadas, E.; Boncz, M.A.; Blanco, S.; Garcíaencina, P.; Muñoz, R. Influence of biogas flow rate on biomass composition during the optimization of biogas upgrading in microalgal-bacterial processes. Environ. Sci. Technol. 2015, 49, 3228–3236. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Wang, Z.; Shu, Q.; Takala, J.; Hiltunen, E.; Feng, P.; Yuan, Z. Nutrient removal and biodiesel production by integration of freshwater algae cultivation with piggery wastewater treatment. Water Res. 2013, 47, 4294–4302. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Ge, Z.; Zhang, H.; Bao, J.; Sun, S. Nutrient removal from biogas slurry and biogas upgrading of crude biogas at high CO2 concentrations using marine microalgae. J. Chem. Technol. Biotechnol. 2015, 91, 1113–1118. [Google Scholar] [CrossRef]
- Ouyang, Y.; Zhao, Y.; Sun, S.; Hu, C.; Ping, L. Effect of light intensity on the capability of different microalgae species for simultaneous biogas upgrading and biogas slurry nutrient reduction. Int. Biodeterior. Biodegrad. 2015, 104, 157–163. [Google Scholar] [CrossRef]
- Ho, S.H.; Chen, C.Y.; Chang, J.S. Effect of light intensity and nitrogen starvation on CO2 fixation and lipid/carbohydrate production of an indigenous microalga Scenedesmus obliquus CNW-N. Bioresour. Technol. 2012, 113, 244–252. [Google Scholar] [CrossRef] [PubMed]

| Parameter | Before Pretreatment | After Pretreatment |
|---|---|---|
| pH | 6.78 ± 0.18 | 6.97 ± 0.17 |
| Dissolved oxygen (mg L−1) | 5.67 ± 0.44 | 5.43 ± 0.39 |
| Dissolved inorganic carbon (mg L−1) | 982.14 ± 32.73 | 971.08 ± 41.39 |
| COD (mg L−1) | 1521.75 ± 47.19 | 1495.62 ± 59.94 |
| TN (mg L−1) | 289.78 ± 29.37 | 278.46 ± 30.13 |
| TP (mg L−1) | 29.82 ± 3.26 | 27.98 ± 2.87 |
| Culture 1 | Culture 2 | Culture 3 | |
|---|---|---|---|
| Specific Growth Rate (d−1) | |||
| 12 h light:12 h dark | 0.279 ± 0.07 | 0.362 ± 0.07 | 0.325 ± 0.06 |
| 14 h light:10 h dark | 0.308 ± 0.08 | 0.381 ± 0.09 | 0.337 ± 0.07 |
| 16 h light:8 h dark | 0.257 ± 0.05 | 0.353 ± 0.08 | 0.296 ± 0.05 |
| Mean daily productivity (g L−1 d−1) | |||
| 12 h light:12 h dark | 0.085 ± 0.007 | 0.143 ± 0.011 | 0.129 ± 0.011 |
| 14 h light:10 h dark | 0.098 ± 0.006 | 0.162 ± 0.013 | 0.147 ± 0.010 |
| 16 h light:8 h dark | 0.081 ± 0.007 | 0.134 ± 0.012 | 0.113 ± 0.009 |
| Cultivation Approaches/Photoperiods | Time (h) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 24 | 48 | 72 | 96 | 120 | 144 | 168 | 192 | 216 | 240 | |
| Culture 1 | |||||||||||
| 12 h light:12 h dark | 6.87 ± 0.16 | 6.91 ± 0.21 | 6.94 ± 0.18 | 6.98 ± 0.26 | 7.03 ± 0.24 | 7.05 ± 0.31 | 7.07 ± 0.33 | 7.09 ± 0.29 | 7.13 ± 0.29 | 7.16 ± 0.34 | 7.19 ± 0.34 |
| 14 h light:10 h dark | 6.83 ± 0.14 | 6.93 ± 0.27 | 6.96 ± 0.19 | 7.01 ± 0.21 | 7.05 ± 0.25 | 7.07 ± 0.24 | 7.13 ± 0.34 | 7.16 ± 0.21 | 7.19 ± 0.31 | 7.21 ± 0.36 | 7.17 ± 0.29 |
| 16 h light:8 h dark | 6.81 ± 0.19 | 6.92 ± 0.23 | 6.97 ± 0.22 | 7.05 ± 0.29 | 7.08 ± 0.27 | 7.06 ± 0.27 | 7.14 ± 0.35 | 7.18 ± 0.25 | 7.21 ± 0.32 | 7.15 ± 0.27 | 7.19 ± 0.41 |
| Culture 2 | |||||||||||
| 12 h light:12 h dark | 6.79 ± 0.17 | 6.82 ± 0.21 | 6.86 ± 0.25 | 6.92 ± 0.27 | 7.06 ± 0.22 | 7.08 ± 0.31 | 7.13 ± 0.24 | 7.15 ± 0.32 | 7.17 ± 0.33 | 7.14 ± 0.21 | 7.16 ± 0.33 |
| 14 h light:10 h dark | 6.83 ± 0.12 | 6.87 ± 0.22 | 6.91 ± 0.19 | 6.97 ± 0.23 | 7.07 ± 0.29 | 7.09 ± 0.33 | 7.14 ± 0.28 | 7.16 ± 0.31 | 7.19 ± 0.31 | 7.11 ± 0.23 | 7.15 ± 0.35 |
| 16 h light:8 h dark | 6.86 ± 0.15 | 6.91 ± 0.29 | 6.94 ± 0.26 | 6.99 ± 0.24 | 7.02 ± 0.21 | 7.05 ± 0.29 | 7.08 ± 0.23 | 7.12 ± 0.34 | 7.15 ± 0.37 | 7.19 ± 0.34 | 7.17 ± 0.37 |
| Culture 3 | |||||||||||
| 12 h light:12 h dark | 6.77 ± 0.23 | 6.83 ± 0.24 | 6.87 ± 0.21 | 6.93 ± 0.21 | 7.05 ± 0.21 | 7.08 ± 0.32 | 7.12 ± 0.35 | 7.13 ± 0.32 | 7.14 ± 0.37 | 7.18 ± 0.38 | 7.14 ± 0.21 |
| 14 h light:10 h dark | 6.85 ± 0.15 | 6.88 ± 0.21 | 6.92 ± 0.23 | 6.98 ± 0.25 | 7.01 ± 0.23 | 7.04 ± 0.23 | 7.08 ± 0.31 | 7.11 ± 0.36 | 7.13 ± 0.32 | 7.11 ± 0.35 | 7.16 ± 0.32 |
| 16 h light:8 h dark | 6.82 ± 0.18 | 6.86 ± 0.19 | 6.89 ± 0.22 | 6.95 ± 0.22 | 7.03 ± 0.28 | 7.09 ± 0.29 | 7.12 ± 0.27 | 7.17 ± 0.34 | 7.19 ± 0.35 | 7.13 ± 0.31 | 7.18 ± 0.34 |
| Cultivation Approaches/Photoperiods | Removal Efficiency (%) | Economic Efficiency (USD−1) | ||||||
|---|---|---|---|---|---|---|---|---|
| COD | TN | TP | CO2 | COD | TN | TP | CO2 | |
| Culture 1 | ||||||||
| 12 h light:12 h dark | 56.32 b ± 5.24 | 59.16 b ± 5.35 | 61.66 b ± 5.26 | 42.36 b ± 3.13 | 25.71 b ± 2.04 | 29.14 b ± 2.53 | 29.98 b ± 2.77 | 20.14 b ± 1.65 |
| 14 h light:10 h dark | 63.43 a ± 5.76 | 68.22 a ± 5.26 | 70.06 a ± 5.24 | 48.87 a ± 3.96 | 30.05 a ± 2.28 | 32.25 a ± 3.02 | 33.63 a ± 2.61 | 24.38 a ± 2.19 |
| 16 h light:8 h dark | 54.96 b ± 5.19 | 60.75 b ± 5.72 | 60.87 b ± 5.76 | 46.59 a ± 4.19 | 24.08 b ± 2.17 | 29.63 b ± 2.77 | 29.92 b ± 2.24 | 23.09 a ± 1.95 |
| Culture 2 | ||||||||
| 12 h light:12 h dark | 62.84 b ± 5.77 | 64.55 b ± 5.91 | 66.09 b ± 6.11 | 51.66 a ± 4.65 | 29.78 b ± 2.72 | 30.97 a ± 2.84 | 32.45 b ± 2.96 | 26.23 a ± 2.37 |
| 14 h light:10 h dark | 70.24 a ± 6.86 | 72.99 a ± 6.12 | 73.83 a ± 5.81 | 52.26 a ± 5.37 | 34.13 a ± 3.01 | 36.42 a ± 3.25 | 37.16 a ± 3.07 | 27.37 a ± 2.71 |
| 16 h light:8 h dark | 59.47 b ± 5.86 | 63.71 b ± 5.93 | 64.82 b ± 6.03 | 50.68 a ± 4.75 | 28.43 b ± 2.84 | 30.09 a ± 2.38 | 30.95 b ± 2.54 | 25.62 a ± 2.62 |
| Culture 3 | ||||||||
| 12 h light:12 h dark | 57.46 b ± 5.13 | 69.27 b ± 4.97 | 63.24 b ± 5.78 | 47.32 a ± 3.79 | 26.86 b ± 2.78 | 32.98 b ± 2.95 | 29.08 b ± 2.68 | 23.85 a ± 2.14 |
| 14 h light:10 h dark | 68.54 a ± 5.96 | 74.25 a ± 5.95 | 71.98 a ± 6.04 | 49.94 a ± 4.92 | 32.63 a ± 2.35 | 37.83 a ± 3.16 | 35.68 a ± 2.64 | 24.59 a ± 2.77 |
| 16 h light:8 h dark | 56.09 b ± 5.38 | 64.45 c ± 6.12 | 62.92 b ± 5.09 | 48.31 a ± 4.84 | 25.02 b ± 2.52 | 31.03 b ± 2.83 | 30.43 b ± 2.17 | 24.14 a ± 1.95 |
| Cultivation Approaches/Light Intensities | Mean Daily Productivity (g L−1 d−1) | COD Removal Efficiency (%) | TN Removal Efficiency (%) | TP Removal Efficiency (%) | CO2 Removal (%) |
|---|---|---|---|---|---|
| Culture 2 | |||||
| 250 μmol m−2 s−1 | 0.349 ± 0.021 | 79.12 b ± 4.38 | 80.13 a ± 6.25 | 82.06 a ± 6.35 | 62.83 a ± 4.11 |
| 450 μmol m−2 s−1 | 0.382 ± 0.024 | 83.43 a ± 5.74 | 81.27 a ± 6.77 | 83.52 a ± 6.61 | 59.37 b ± 5.07 |
| 650 μmol m−2 s−1 | 0.321 ± 0.018 | 72.86 c ± 5.87 | 76.48 b ± 5.91 | 81.57 a ± 5.97 | 57.49 b ± 5.79 |
| Culture 3 | |||||
| 250 μmol m−2 s−1 | 0.336 ± 0.025 | 75.08 b ± 5.93 | 79.98 b ± 6.95 | 77.13 b ± 5.71 | 54.72 a ± 4.27 |
| 450 μmol m−2 s−1 | 0.368 ± 0.022 | 80.55 a ± 6.24 | 82.64 a ± 6.24 | 82.79 a ± 5.83 | 50.34 b ± 4.39 |
| 650 μmol m−2 s−1 | 0.314 ± 0.019 | 73.97 b ± 6.81 | 78.59 b ± 6.76 | 74.24 b ± 4.92 | 48.97 b ± 4.86 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, P.; Zhang, Y.; Zhao, Y. Biocapture of CO2 by Different Microalgal-Based Technologies for Biogas Upgrading and Simultaneous Biogas Slurry Purification under Various Light Intensities and Photoperiods. Int. J. Environ. Res. Public Health 2018, 15, 528. https://doi.org/10.3390/ijerph15030528
Guo P, Zhang Y, Zhao Y. Biocapture of CO2 by Different Microalgal-Based Technologies for Biogas Upgrading and Simultaneous Biogas Slurry Purification under Various Light Intensities and Photoperiods. International Journal of Environmental Research and Public Health. 2018; 15(3):528. https://doi.org/10.3390/ijerph15030528
Chicago/Turabian StyleGuo, Pengfei, Yuejin Zhang, and Yongjun Zhao. 2018. "Biocapture of CO2 by Different Microalgal-Based Technologies for Biogas Upgrading and Simultaneous Biogas Slurry Purification under Various Light Intensities and Photoperiods" International Journal of Environmental Research and Public Health 15, no. 3: 528. https://doi.org/10.3390/ijerph15030528
APA StyleGuo, P., Zhang, Y., & Zhao, Y. (2018). Biocapture of CO2 by Different Microalgal-Based Technologies for Biogas Upgrading and Simultaneous Biogas Slurry Purification under Various Light Intensities and Photoperiods. International Journal of Environmental Research and Public Health, 15(3), 528. https://doi.org/10.3390/ijerph15030528




