Individual and Neighborhood Stressors, Air Pollution and Cardiovascular Disease
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Adverse Childhood Experiences (ACEs)
2.3. Neighborhood Deprivation Index (NDI)
2.4. Air Pollution
+ 2009 annual average air pollution * (214/365 days)
2.5. Cardiovascular Disease (CVD) Measures and Risk Factors
2.6. Covariates
2.7. Statistical Analysis
3. Results
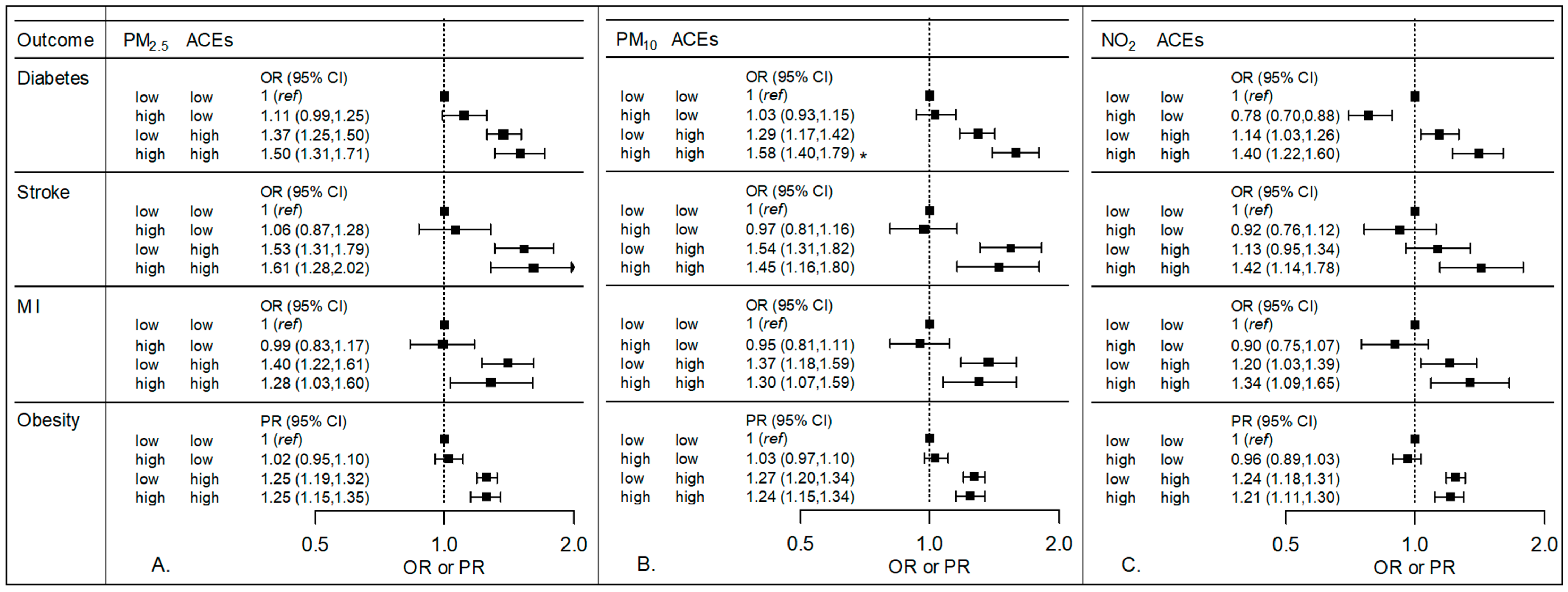
3.1. Air Pollution and ACEs
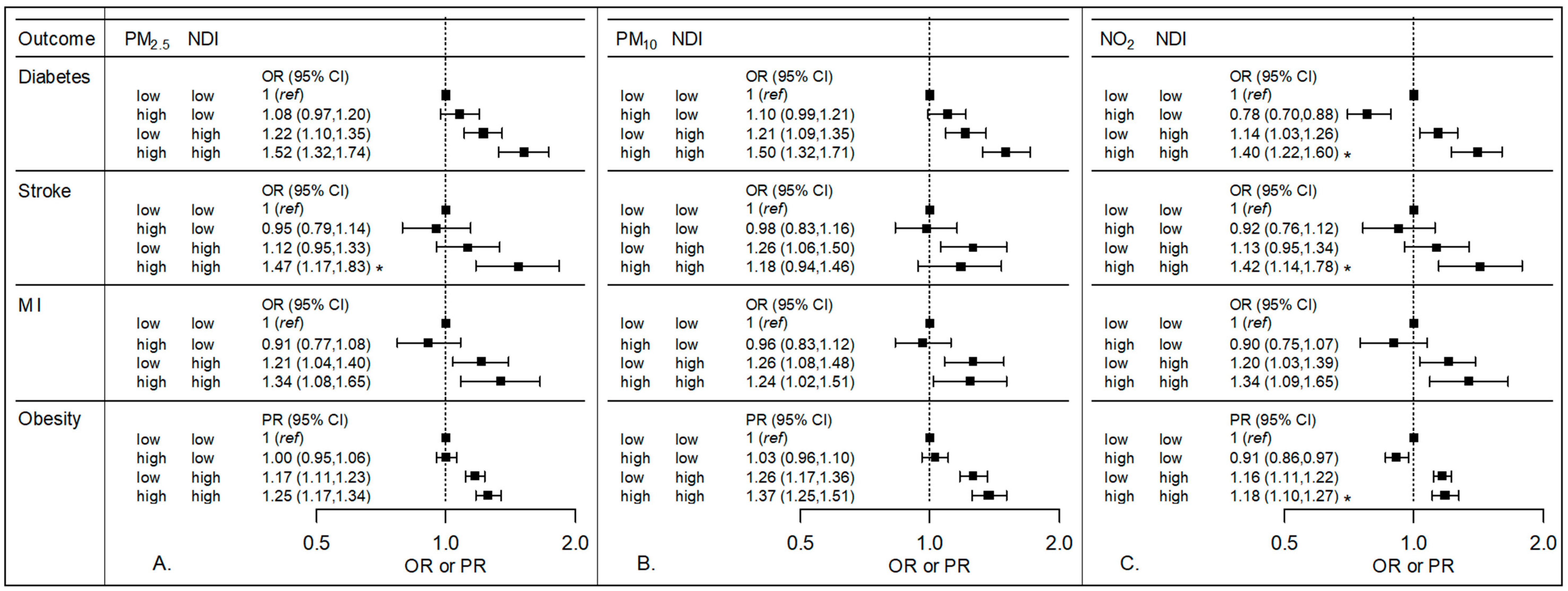
3.2. Air Pollution and NDI
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nurius, P.S.; Green, S.; Logan-Greene, P.; Longhi, D.; Song, C. Stress pathways to health inequalities: Embedding ACEs within social and behavioral contexts. Int. Public Health J. 2016, 8, 241–256. [Google Scholar] [PubMed]
- Theall, K.P.; Drury, S.S.; Shirtcliff, E.A. Cumulative neighborhood risk of psychosocial stress and allostatic load in adolescents. Am. J. Epidemiol. 2012, 176, S164–S174. [Google Scholar] [CrossRef] [PubMed]
- Walsemann, K.; Goosby, B.; Farr, D. Life course SES and cardiovascular risk: Heterogeneity across race/ethnicity and gender. Soc. Sci. Med. 2016, 28, 1304–1314. [Google Scholar] [CrossRef] [PubMed]
- Cosselman, K.E.; Navas-Acien, A.; Kaufman, J.D. Environmental factors in cardiovascular disease. Nat. Rev. Cardiol. 2015, 12, 627–642. [Google Scholar] [CrossRef] [PubMed]
- Havranek, E.P.; Mujahid, M.S.; Barr, D.A.; Blair, I.; Cohen, M.S.; Cruz-Flores, S.; Davey-Smith, G.; Dennison-Himmelfarb, C.R.; Lauer, M.S.; Lockwood, D.W.; et al. Social determinants of risk and outcomes for cardiovascular disease: A scientific statement from the American Heart Association. Circulation 2015, 132, 873–898. [Google Scholar] [CrossRef] [PubMed]
- Halonen, J.I.; Stenholm, S.; Pentti, J.; Kawachi, I.; Subramanian, S.; Kivimäki, M.; Vahtera, J. Childhood psychosocial adversity and adult neighborhood disadvantage as predictors of cardiovascular disease: A cohort study. Circulation 2015, 132, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Hicken, M.; Adar, S.; Hajat, A.; Kershaw, K.; Do, D.; Barr, R.; Kaufman, J.; Diez Roux, A. Air Pollution, Cardiovascular Outcomes, and Social Disadvantage: The Multi-ethnic Study of Atherosclerosis. Epidemiology 2016, 27, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Chi, G.C.; Hajat, A.; Bird, C.E.; Cullen, M.R.; Griffin, B.A.; Miller, K.A.; Shih, R.A. Individual and Neighborhood Socioeconomic Status and the Association between Air Pollution and Cardiovascular Disease. Environ. Health Perspect. 2016, 1840, 1840–1847. [Google Scholar] [CrossRef] [PubMed]
- Hajat, A.; Diez-Roux, A.V.; Adar, S.D.; Auchincloss, A.H.; Lovasi, G.S.; O’Neill, M.; Sheppard, L.; Kaufman, J. Air pollution and individual and neighborhood socioeconomic status: Evidence from the Multi-Ethnic Study of Atherosclerosis (MESA). Environ. Health Perspect. 2013, 121, 1325–1333. [Google Scholar] [CrossRef] [PubMed]
- Shmool, J.L.C.; Kubzansky, L.D.; Newman, O.D.; Spengler, J.; Shepard, P.; Clougherty, J.E. Social stressors and air pollution across New York City communities: A spatial approach for assessing correlations among multiple exposures. Environ. Health 2014, 13, 91. [Google Scholar] [CrossRef] [PubMed]
- Slopen, N.; Lewis, T.T.; Gruenewald, T.L.; Mujahid, M.S.; Ryff, C.D.; Albert, M.A.; Williams, D.R. Early Life Adversity and Inflammation in African Americans and Whites in the Midlife in the United States Survey. Psychosom. Med. 2010, 72, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Nazmi, A.; Oliveira, I.O.; Horta, B.L.; Gigante, D.P.; Victora, C.G. Lifecourse socioeconomic trajectories and C-reactive protein levels in young adults: Findings from a Brazilian birth cohort. Soc. Sci. Med. 2010, 70, 1229–1236. [Google Scholar] [CrossRef] [PubMed]
- Nazmi, A.; Diez Roux, A.; Ranjit, N.; Seeman, T.E.; Jenny, N.S. Cross-sectional and longitudinal associations of neighborhood characteristics with inflammatory markers: Findings from the multi-ethnic study of atherosclerosis. Health Place 2010, 16, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Nurius, P.S.; Uehara, E.; Zatzick, D.F. Intersection of Stress, Social Disadvantage, and Life Course Processes: Reframing Trauma and Mental Health. Am. J. Psychiatr. Rehabil. 2013, 16, 91–114. [Google Scholar] [CrossRef] [PubMed]
- Hajat, A.; Allison, M.; Diez-Roux, A.V.; Jenny, N.S.; Jorgensen, N.W.; Szpiro, A.A.; Vedal, S.; Kaufman, J.D. Long-term exposure to air pollution and markers of inflammation, coagulation, and endothelial activation: A repeat-measures analysis in the Multi-Ethnic Study of Atherosclerosis (MESA). Epidemiology 2015, 26, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Peretz, A.; Sullivan, J.H.; Leotta, D.F.; Trenga, C.A.; Sands, F.N.; Allen, J.; Carlsten, C.; Wilkinson, C.W.; Gill, E.A.; Kaufman, J.D. Diesel exhaust inhalation elicits acute vasoconstriction in vivo. Environ. Health Perspect. 2008, 116, 937–942. [Google Scholar] [CrossRef] [PubMed]
- McEwen, C.A.; McEwen, B.S. Social Structure, Adversity, Toxic Stress, and Intergenerational Poverty: An Early Childhood Model. Annu. Rev. Sociol. 2017, 43, 445–472. [Google Scholar] [CrossRef]
- Schwartz, J.; Bellinger, D.; Glass, T. Exploring potential sources of differential vulnerability and susceptibility in risk from environmental hazards to expand the scope of risk assessment. Am. J. Public Health 2011, 101, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Diez-Roux, A.; Merkin, S.S.; Arnett, D.; Chambless, L.; Massing, M.; Nieto, F.J.; Sorlie, P.; Szklo, M.; Tyroler, H.A.; Watson, R.L. Neighborhood of residence and incidence of coronary heart disease. N. Engl. J. Med. 2001, 345, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Felitti, V.J.; Anda, R.F.; Nordenberg, D.; Williamson, D.F.; Spitz, A.M.; Edwards, V.; Koss, M.P.; Marks, J.S. Relationship of childhood abuse and household dysfunction to many of the leading causes of death in adults. The Adverse Childhood Experiences (ACE) Study. Am. J. Prev. Med. 1998, 14, 245–258. [Google Scholar] [CrossRef]
- Korkeila, J.; Vahtera, J.; Korkeila, K.; Kivimäki, M.; Sumanen, M.; Koskenvuo, K.; Koskenvuo, M. Childhood adversities as predictors of incident coronary heart disease and cerebrovascular disease. Heart 2010, 96, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Jimenez, M.P.; Roberts, C.T.F.; Loucks, E.B. The Role of Adverse Childhood Experiences in Cardiovascular Disease Risk : A Review with Emphasis on Plausible Mechanisms. Curr. Cardiol. Rep. 2015, 17, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Rich-Edwards, J.W.; Mason, S.; Rexrode, K.; Spiegelman, D.; Hibert, E.; Kawachi, I.; Jun, H.J.; Wright, R.J. Physical and sexual abuse in childhood as predictors of early-onset cardiovascular events in women. Circulation 2012, 126, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Washington State Department of Health Behavioral Risk Factor Surveillance System (BRFSS). Available online: http://www.doh.wa.gov/DataandStatisticalReports/DataSystems/BehavioralRiskFactorSurveillanceSystemBRFSS (accessed on 28 June 2017).
- Centers for Disease Control and Prevention (CDC) Behavioral Risk Factor Surveillance System. Available online: https://www.cdc.gov/brfss/index.html (accessed on 28 June 2017).
- US Census Bureau American Community Survey (ACS). Available online: https://www.census.gov/programs-surveys/acs/ (accessed on 28 June 2017).
- Christine, P.J.; Auchincloss, A.H.; Bertoni, A.G.; Carnethon, M.R.; Sánchez, B.N.; Moore, K.; Adar, S.D.; Horwich, T.B.; Watson, K.E.; Diez Roux, A.V. Longitudinal associations between neighborhood physical and social environments and incident type 2 diabetes mellitus: The Multi-Ethnic Study of Atherosclerosis (MESA). JAMA Intern. Med. 2015, 175, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.; Diez Roux, A.V.; Auchincloss, A.; Evenson, K.R.; Kaufman, J.; Mujahid, M.; Williams, K. Home and work neighbourhood environments in relation to body mass index: The Multi-Ethnic Study of Atherosclerosis (MESA). J. Epidemiol. Community Health 2013, 67, 846–853. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.K. Area Deprivation and Widening Inequalities in US Mortality, 1969–1998. Am. J. Public Health 2003, 93, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Laraia, B.A.; Karter, A.J.; Warton, E.M.; Schillinger, D.; Moffet, H.H.; Adler, N. Place matters: Neighborhood deprivation and cardiometabolic risk factors in the Diabetes Study of Northern California (DISTANCE). Soc. Sci. Med. 2012, 74, 1082–1090. [Google Scholar] [CrossRef] [PubMed]
- Turner, E.L.; Dobson, J.E.; Pocock, S.J. Categorisation of continuous risk factors in epidemiological publications: A survey of current practice. Epidemiol. Perspect. Innov. 2010, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Sampson, P.D.; Richards, M.; Szpiro, A.A.; Bergen, S.; Sheppard, L.; Larson, T.V.; Kaufman, J.D. A regionalized national universal kriging model using Partial Least Squares regression for estimating annual PM2.5 concentrations in epidemiology. Atmos. Environ. 2013, 75, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Young, M.T.; Bechle, M.J.; Sampson, P.D.; Szpiro, A.A.; Marshall, J.D.; Sheppard, L.; Kaufman, J.D. Satellite-Based NO2 and Model Validation in a National Prediction Model Based on Universal Kriging and Land-Use Regression. Environ. Sci. Technol. 2016, 28, 3686–3694. [Google Scholar] [CrossRef] [PubMed]
- Thurston, G.D.; Ahn, J.; Cromar, K.R.; Shao, Y.; Reynolds, H.R.; Jerrett, M.; Lim, C.C.; Shanley, R.; Park, Y.; Hayes, R.B. Ambient particulate matter air pollution exposure and mortality in the NIH-AARP diet and health cohort. Environ. Health Perspect. 2016, 124, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Jones, D.M.; Leip, E.P.; Larson, M.G.; D’Agostino, R.B.; Beiser, A.; Wilson, P.W.F.; Wolf, P.A.; Levy, D. Prediction of lifetime risk for cardiovascular disease by risk factor burden at 50 years of age. Circulation 2006, 113, 791–798. [Google Scholar] [CrossRef] [PubMed]
- Hall, S.A.; Kaufman, J.S.; Ricketts, T.C. Defining urban and rural areas in U.S. epidemiologic studies. J. Urban Health 2006, 83, 162–175. [Google Scholar] [CrossRef] [PubMed]
- VanderWeele, T.J.; Knol, M.J. A Tutorial on Interaction. Epidemiol. Methods 2014, 3, 33–72. [Google Scholar] [CrossRef]
- Logan-Greene, P.; Green, S.; Nurius, P.S.; Longhi, D. Distinct Contributions of Adverse Childhood Experiences and Resilience Resources: A Cohort Analysis of Adult Physical and Mental Health. Soc. Work Health Care 2014, 53, 776–797. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Giles, W.H.; Felitti, V.J.; Dube, S.R.; Williams, J.E.; Chapman, D.P.; Anda, R.F. Insights into causal pathways for ischemic heart disease: Adverse childhood experiences study. Circulation 2004, 110, 1761–1766. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.E.; Chen, E.; Parker, K.J. Psychological Stress in Childhood and Susceptibility to the Chronic Disease of Aging: Moving towards a Model of Behavioral and Biological Mechanisms. Psychol. Bull. 2011, 137, 959–997. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, J.D.; Adar, S.D.; Barr, R.G.; Budoff, M.; Burke, G.L.; Curl, C.L.; Daviglus, M.L.; Roux, A.V.D.; Gassett, A.J.; Jacobs, D.R.; et al. Association between air pollution and coronary artery calcification within six metropolitan areas in the USA (the Multi-Ethnic Study of Atherosclerosis and Air Pollution): A longitudinal cohort study. Lancet 2016, 6736, 1–9. [Google Scholar]
- Nurius, P.S.; Fleming, C.M.; Brindle, E. Life Course Pathways From Adverse Childhood Experiences to Adult Physical Health: A Structural Equation Model. J. Aging Health 2017, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Evans, G.W.; Exner-Cortens, D.; Kim, P.; Bartholomew, D. Childhood Poverty and Late Adolescents’ Blood Pressure Reactivity to and Recovery From an Acute Stressor: The Mediating Role of Family Conflict. Psychosom. Med. 2013, 75, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Danese, A.; McEwen, B.S. Adverse childhood experiences, allostasis, allostatic load, and age-related disease. Physiol. Behav. 2012, 106, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Hajat, A.; Hsia, C.; O’Neill, M.S. Socioeconomic Disparities and Air Pollution Exposure: A Global Review. Curr. Environ. Health Rep. 2015, 2, 440–450. [Google Scholar] [CrossRef] [PubMed]
- Morello-Frosch, R.; Jesdale, B.M. Separate and unequal: Residential segregation and estimated cancer risks associated with ambient air toxins in U.S. metropolitan areas. Environ. Health Perspect. 2006, 114, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Gee, G.C.; Payne-Sturges, D.C. Environmental health disparities: A framework integrating psychosocial and environmental concepts. Environ. Health Perspect. 2004, 112, 1645–1653. [Google Scholar] [CrossRef] [PubMed]
- Clougherty, J.E.; Kubzansky, L.D. A Framework for Examining Social Stress and Susceptibility to Air Pollution in Respiratory Health. Environ. Health Perspect. 2009, 117, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Fuller, C.H.; Feeser, K.R.; Sarnat, J.A.; O’Neill, M.S. Air pollution, cardiovascular endpoints and susceptibility by stress and material resources: A systematic review of the evidence. Environ. Health 2017, 16, 58. [Google Scholar] [CrossRef] [PubMed]
- Hicken, M.T.; Adar, S.D.; Roux, A.V.D.; O’Neill, M.S.; Magzamen, S.; Auchincloss, A.H.; Kaufman, J.D. Do psychosocial stress and social disadvantage modify the association between air pollution and blood pressure: The multi-ethnic study of atherosclerosis. Am. J. Epidemiol. 2013, 178, 1550–1562. [Google Scholar] [CrossRef] [PubMed]
- Hicken, M.T.; Dvonch, J.T.; Schulz, A.J.; Mentz, G.; Max, P. Fine particulate matter air pollution and blood pressure: The modifying role of psychosocial stress. Environ. Res. 2014, 133, 195–203. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S. Neurobiological and Systemic Effects of Chronic Stress. Chronic Stress 2017. [Google Scholar] [CrossRef] [PubMed]
- Pearlin, L.I.; Schieman, S.; Fazio, E.M.; Meersman, S.C. Stress, Health, and the Life Course: Some Conceptual Perspectives. J. Health Soc. Behav. 2005, 46, 205–219. [Google Scholar] [CrossRef] [PubMed]
- Nurius, P.; Green, S.; Logan-Greene, P.; Borja, S. Life Course Pathways of Adverse Childhood Experiences Toward Adult Psychological Well-Being: A Stress Process Analysis. Child Abuse Negl. 2015, 45, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Pollitt, R.A.; Daniel, M.; Kaufman, J.S.; Lynch, J.W.; Salonen, J.T.; Kaplan, G.A. Mediation and modification of the association between hopelessness, hostility, and progression of carotid atherosclerosis. J. Behav. Med. 2005, 28, 53–64. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chan, S.H.; Van Hee, V.C.; Bergen, S.; Szpiro, A.A.; DeRoo, L.A.; London, S.J.; Marshall, J.D.; Kaufman, J.D.; Sandler, D.P. Long-term air pollution exposure and blood pressure in the Sister Study. Environ. Health Perspect. 2015, 123, 951–958. [Google Scholar] [CrossRef] [PubMed]
| Participant Characteristic | Overall (n = 32,151) | Low PM2.5 b (n = 24,115) | High PM2.5 b (n = 8036) | ≤1 ACE (n = 19,941) | 2+ ACEs (n = 12,210) | Low NDI c (n = 24,193) | High NDI c (n = 7958) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age d | 57 | (16) | 57 | (16) | 56 | (16) | 59 | (16) | 53 | (15) | 57 | (16) | 56 | (17) |
| Female | 19,420 | (60) | 14,536 | (60) | 4884 | (61) | 11,684 | (59) | 7736 | (63) | 14,593 | (60) | 4827 | (61) |
| Race/ethnicity | ||||||||||||||
| White | 27,878 | (87) | 20,989 | (87) | 6889 | (86) | 17,495 | (88) | 10,383 | (85) | 21,470 | (89) | 6408 | (81) |
| Hispanic | 1534 | (5) | 1078 | (4) | 456 | (6) | 907 | (5) | 627 | (5) | 743 | (3) | 791 | (10) |
| Black | 383 | (1) | 260 | (1) | 123 | (2) | 214 | (1) | 169 | (1) | 224 | (0.9) | 159 | (2) |
| Asian | 691 | (2) | 505 | (2) | 186 | (2) | 574 | (3) | 117 | (1) | 582 | (2) | 109 | (1) |
| American Indian, Alaska Native | 314 | (1) | 250 | (1) | 64 | (0.8) | 118 | (0.6) | 196 | (2) | 201 | (0.8) | 113 | (1) |
| Native Hawaiian, Pacific Islander | 90 | (0.3) | 60 | (0.3) | 30 | (0.4) | 53 | (0.3) | 37 | (0.3) | 60 | (0.3) | 30 | (0.4) |
| Multiracial | 769 | (2) | 588 | (2) | 181 | (2) | 292 | (1) | 477 | (4) | 552 | (2) | 217 | (3) |
| Other | 191 | (0.6) | 155 | (0.6) | 36 | (0.5) | 100 | (0.5) | 91 | (0.8) | 135 | (0.6) | 56 | (0.7) |
| Education | ||||||||||||||
| Less than HS | 1719 | (5) | 1287 | (5) | 432 | (5) | 955 | (5) | 764 | (6) | 912 | (4) | 807 | (10) |
| HS | 7270 | (23) | 5495 | (23) | 1775 | (22) | 4548 | (23) | 2722 | (22) | 4928 | (20) | 2342 | (29) |
| Some college | 23,123 | (72) | 17,304 | (72) | 5819 | (73) | 14,409 | (72) | 8714 | (71) | 18,327 | (76) | 4796 | (60) |
| Annual income | ||||||||||||||
| <$35,000 | 9648 | (34) | 7189 | (34) | 2459 | (35) | 5752 | (33) | 3896 | (36) | 6599 | (31) | 3049 | (44) |
| $35,000–$49,999 | 4756 | (17) | 3567 | (17) | 1189 | (17) | 2964 | (17) | 1792 | (17) | 3511 | (16) | 1245 | (18) |
| Employed | 15,793 | (49) | 11,739 | (49) | 4054 | (51) | 9350 | (47) | 6443 | (53) | 12,068 | (50) | 3725 | (47) |
| Smoking | ||||||||||||||
| Current | 4282 | (13) | 2181 | (13) | 1101 | (14) | 1942 | (9) | 2340 | (19) | 2930 | (12) | 1352 | (17) |
| Former | 10,300 | (32) | 7821 | (33) | 2479 | (31) | 6075 | (31) | 4225 | (35) | 7788 | (32) | 2512 | (32) |
| Binge Drinking | 3809 | (12) | 2908 | (12) | 901 | (11) | 1989 | (10) | 1820 | (15) | 2907 | (12) | 902 | (11) |
| BMI (kg/m2) d | 28 | (6) | 28 | (6) | 28 | (6) | 27 | (6) | 28 | (6) | 27 | (6) | 28 | (6) |
| CVD and CVD risk factors | ||||||||||||||
| Diabetes | 3685 | (11) | 2705 | (11) | 980 | (12) | 2238 | (11) | 1447 | (12) | 2590 | (11) | 1095 | (14) |
| Stroke | 1178 | (4) | 865 | (4) | 313 | (4) | 688 | (3) | 490 | (4) | 817 | (3) | 361 | (5) |
| MI | 1573 | (5) | 1201 | (5) | 372 | (5) | 986 | (5) | 587 | (5) | 1089 | (5) | 484 | (6) |
| CHD | 1699 | (5) | 1309 | (5) | 390 | (5) | 1057 | (5) | 642 | (5) | 1225 | (5) | 474 | (6) |
| Obesity | 8460 | (28) | 6001 | (40) | 1745 | (37) | 4711 | (25) | 3749 | (32) | 6028 | (26) | 2432 | (32) |
| Hypertension e | 7746 | (39) | 6298 | (27) | 2162 | (28) | 4931 | (40) | 2815 | (38) | 5765 | (39) | 1981 | (41) |
| Overall | PM2.5 (µg/m3) | PM10 (µg/m3) | NO2 (ppb) | |||
|---|---|---|---|---|---|---|
| 6.54 | (1.41) | 13.95 | (2.97) | 7.50 | (2.86) | |
| ACEs a | ||||||
| Low | 6.56 | (1.40) | 13.43 | (2.48) | 7.48 | (2.82) |
| High | 6.53 | (1.44) | 13.42 | (2.47) | 7.52 | (2.91) |
| p | 0.20 | 0.71 | 0.25 | |||
| NDI b | ||||||
| Low | 6.57 | (1.35) | 13.33 | (2.36) | 7.48 | (2.71) |
| High | 6.45 | (1.58) | 13.70 | (2.78) | 7.56 | (3.26) |
| p | <0.01 | <0.01 | 0.03 | |||
| Outcome | PM2.5 and ACEs | PM10 and ACEs | NO2 and ACEs | |||
|---|---|---|---|---|---|---|
| Diabetes | 0.02 | (−0.22, 0.25) | 0.26 | (0.05, 0.48) * | 0.09 | (−0.13, 0.30) |
| Stroke | 0.02 | (−0.40, 0.44) | −0.06 | (−0.44, 0.31) | 0.14 | (−0.27, 0.55) |
| MI | −0.10 | (−0.44, 0.24) | −0.02 | (−0.33, 0.30) | −0.10 | (−0.44, 0.25) |
| Obesity | −0.03 | (−0.18, 0.13) | −0.08 | (−0.23, 0.07) | −0.01 | (−0.16, 0.14) |
| Outcome | PM2.5 and NDI | PM10 and NDI | NO2 and NDI | |||
|---|---|---|---|---|---|---|
| Diabetes | 0.22 | (−0.02, 0.46) | 0.20 | (−0.03, 0.42) | 0.47 | (0.26, 0.69) * |
| Stroke | 0.39 | (0.02, 0.77) * | −0.06 | (−0.41, 0.28) | 0.37 | (0.01, 0.74) * |
| MI | 0.22 | (−0.12, 0.55) | 0.02 | (−0.29, 0.33) | 0.24 | (−0.09, 0.58) |
| Obesity | 0.13 | (−0.04, 0.30) | 0.08 | (−0.08, 0.24) | 0.15 | (−0.01, 0.31) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hazlehurst, M.F.; Nurius, P.S.; Hajat, A. Individual and Neighborhood Stressors, Air Pollution and Cardiovascular Disease. Int. J. Environ. Res. Public Health 2018, 15, 472. https://doi.org/10.3390/ijerph15030472
Hazlehurst MF, Nurius PS, Hajat A. Individual and Neighborhood Stressors, Air Pollution and Cardiovascular Disease. International Journal of Environmental Research and Public Health. 2018; 15(3):472. https://doi.org/10.3390/ijerph15030472
Chicago/Turabian StyleHazlehurst, Marnie F., Paula S. Nurius, and Anjum Hajat. 2018. "Individual and Neighborhood Stressors, Air Pollution and Cardiovascular Disease" International Journal of Environmental Research and Public Health 15, no. 3: 472. https://doi.org/10.3390/ijerph15030472
APA StyleHazlehurst, M. F., Nurius, P. S., & Hajat, A. (2018). Individual and Neighborhood Stressors, Air Pollution and Cardiovascular Disease. International Journal of Environmental Research and Public Health, 15(3), 472. https://doi.org/10.3390/ijerph15030472





