Toxicology of Engineered Nanoparticles: Focus on Poly(amidoamine) Dendrimers
Abstract
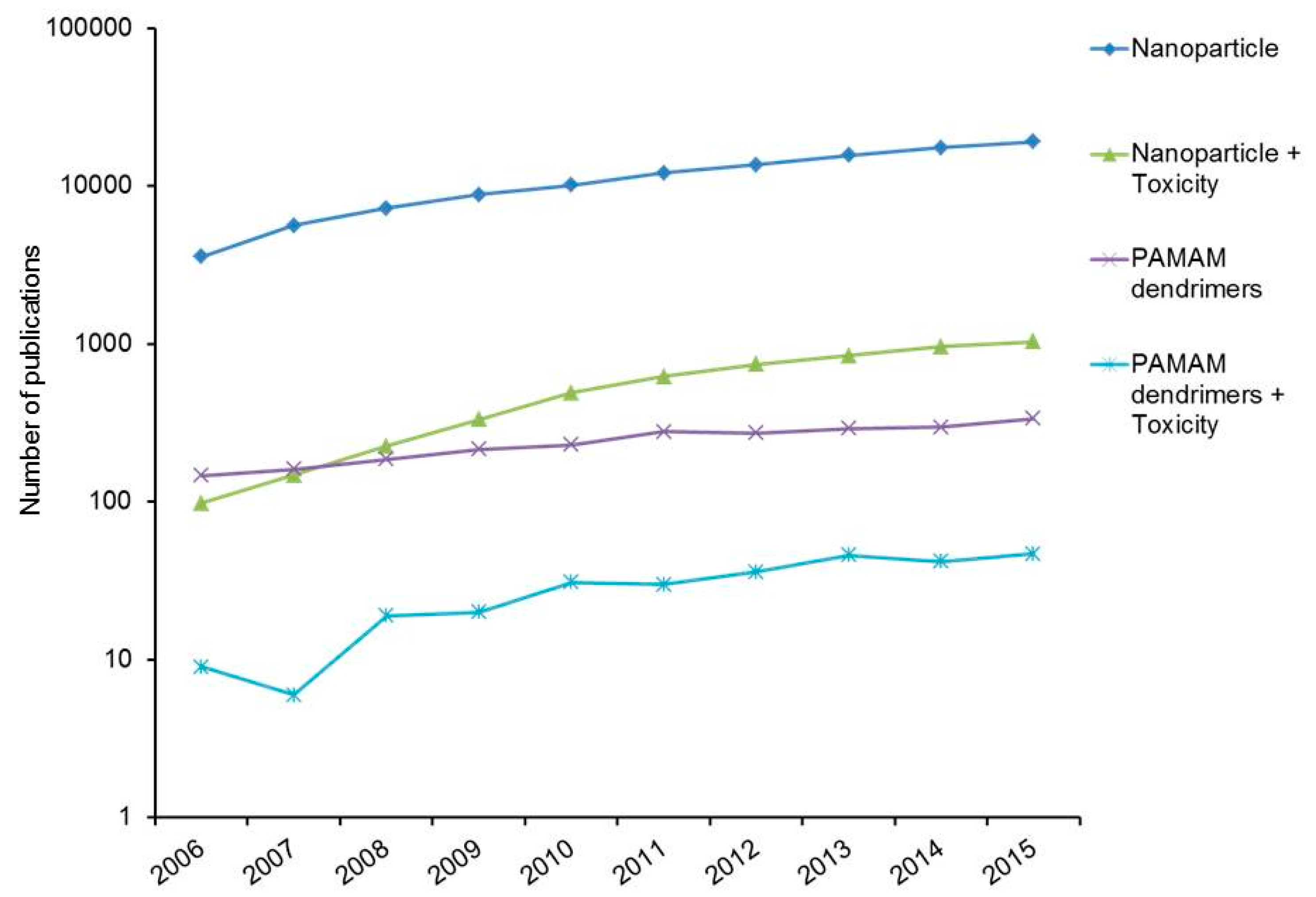
:1. Introduction
2. Toxicology of Engineered Nanoparticles
3. PAMAM Dendrimers
3.1. In Vitro Toxicological Studies of PAMAM Dendrimers
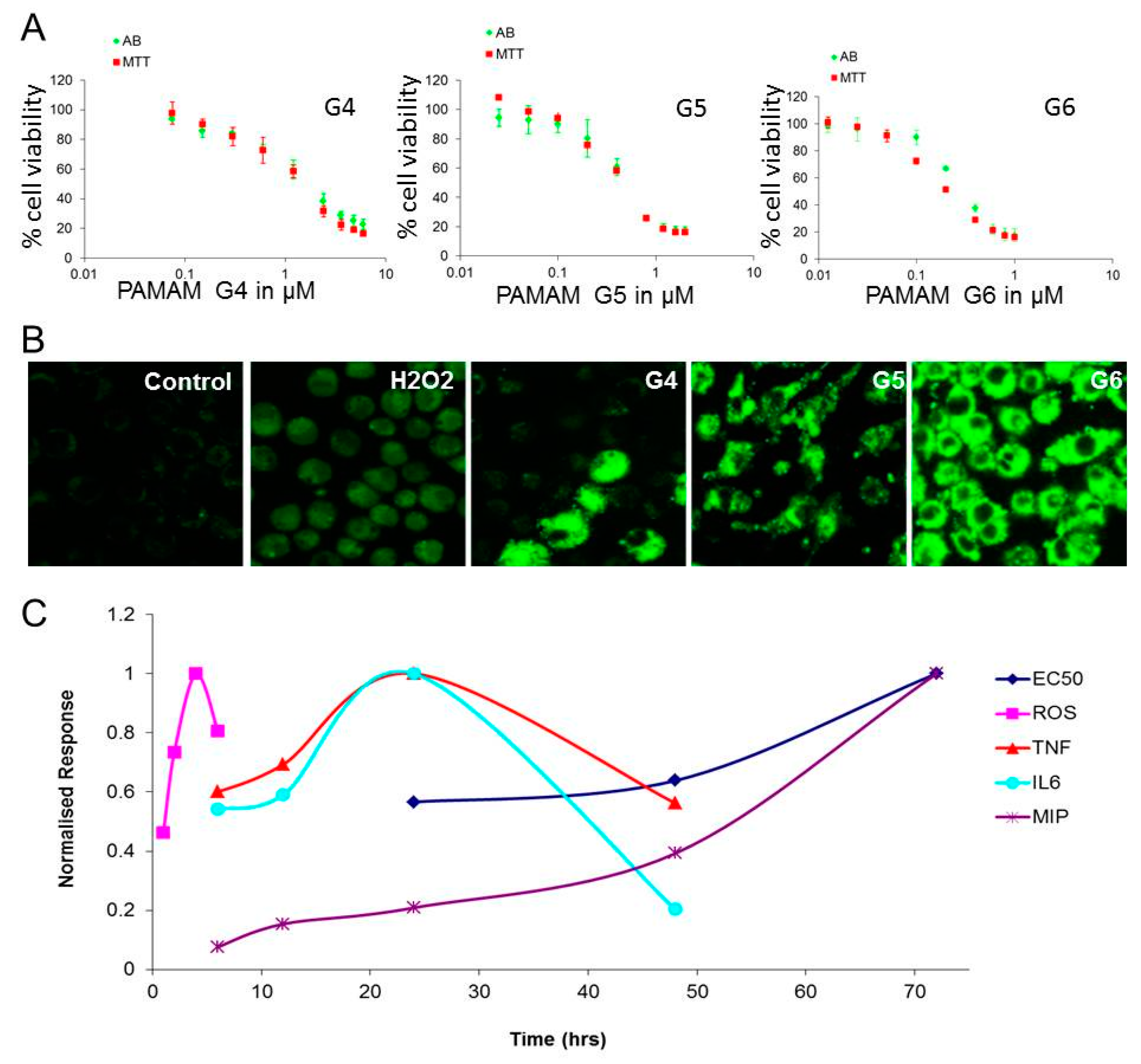
3.1.1. In Vitro Mammalian Toxicological Studies
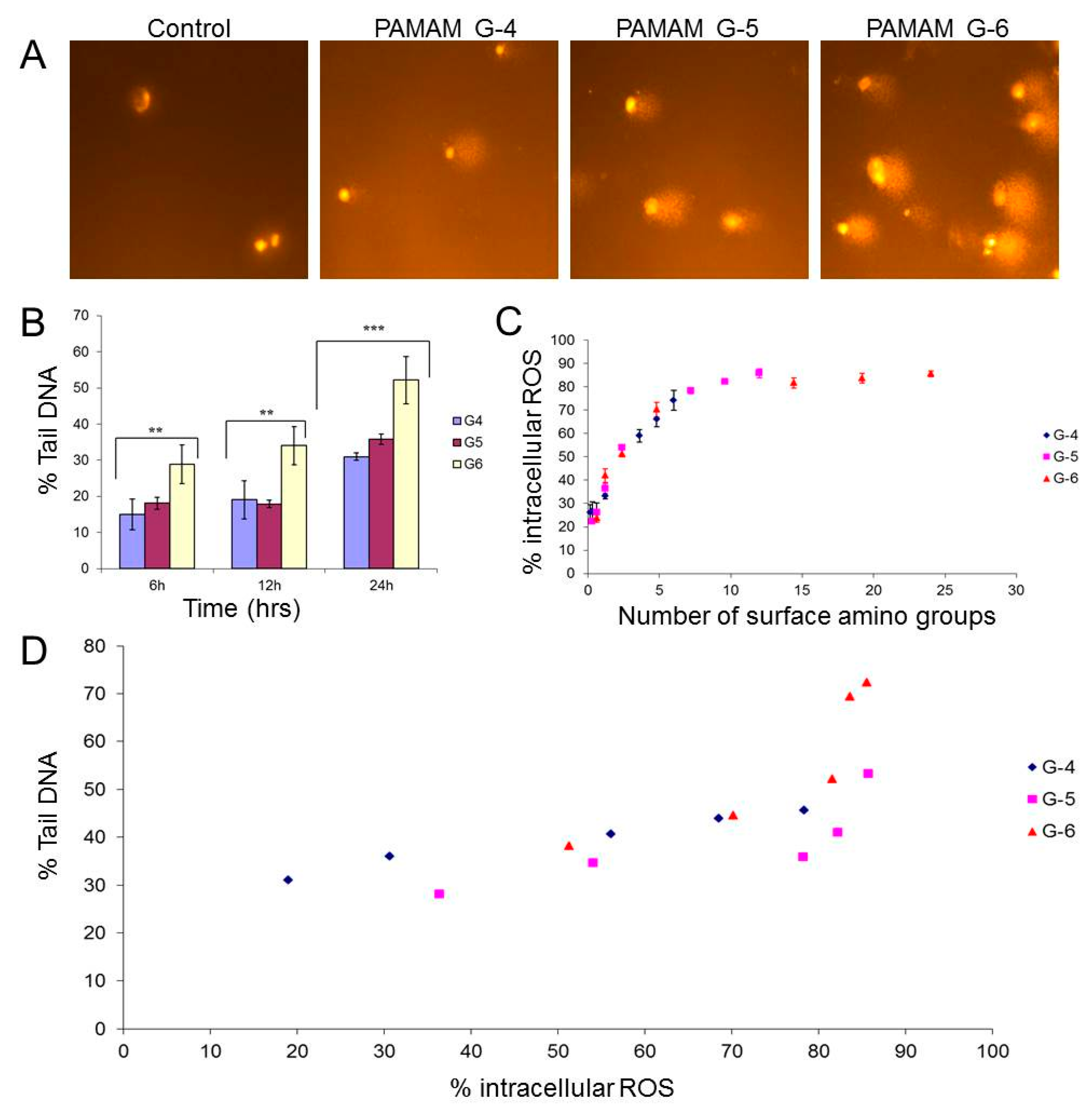
3.1.2. Physicochemical Properties of PAMAM Dendrimers versus Toxicity
3.1.3 Cellular Uptake and Biodegradation
3.1.4. In Vitro Eco Toxicological Studies
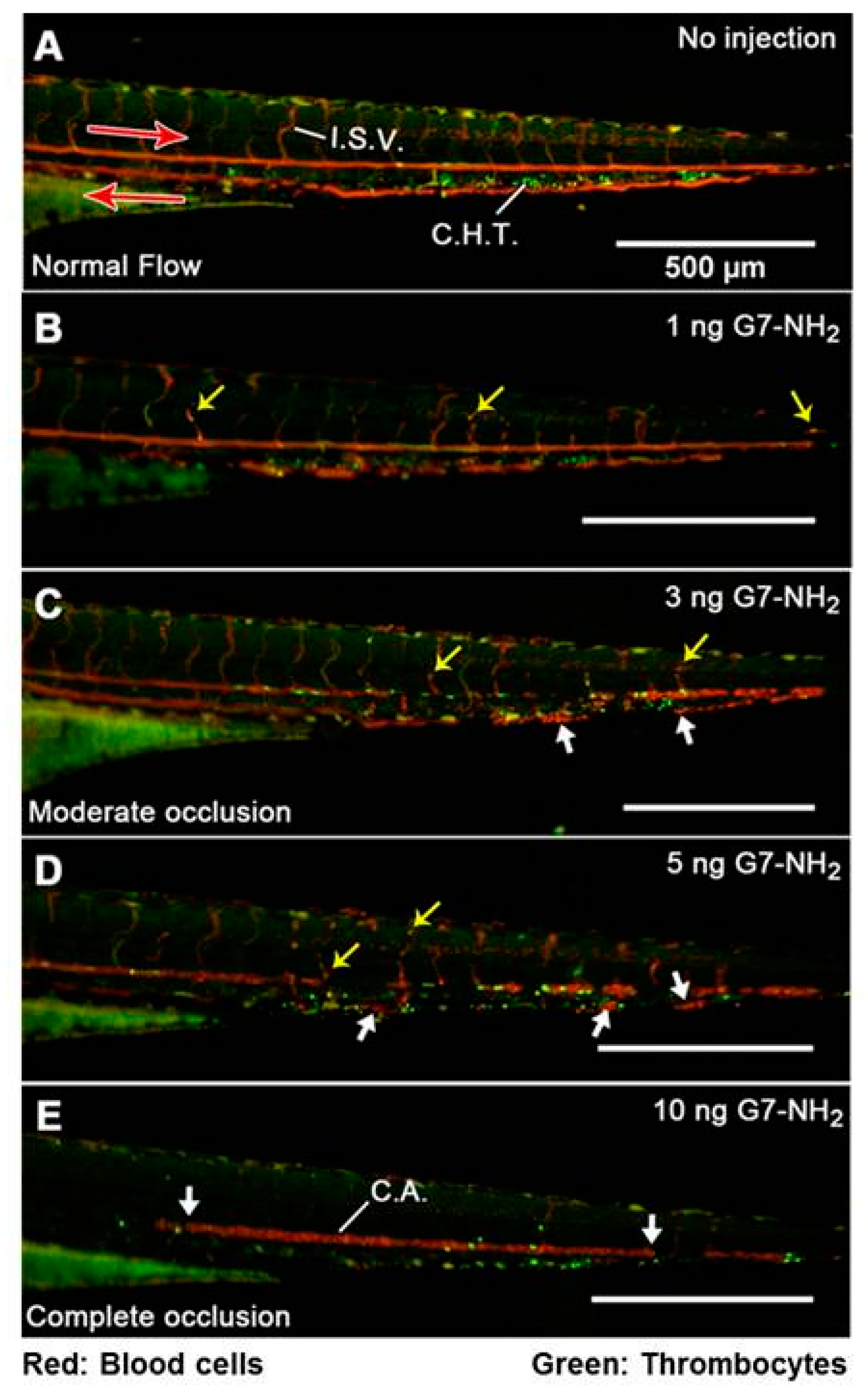
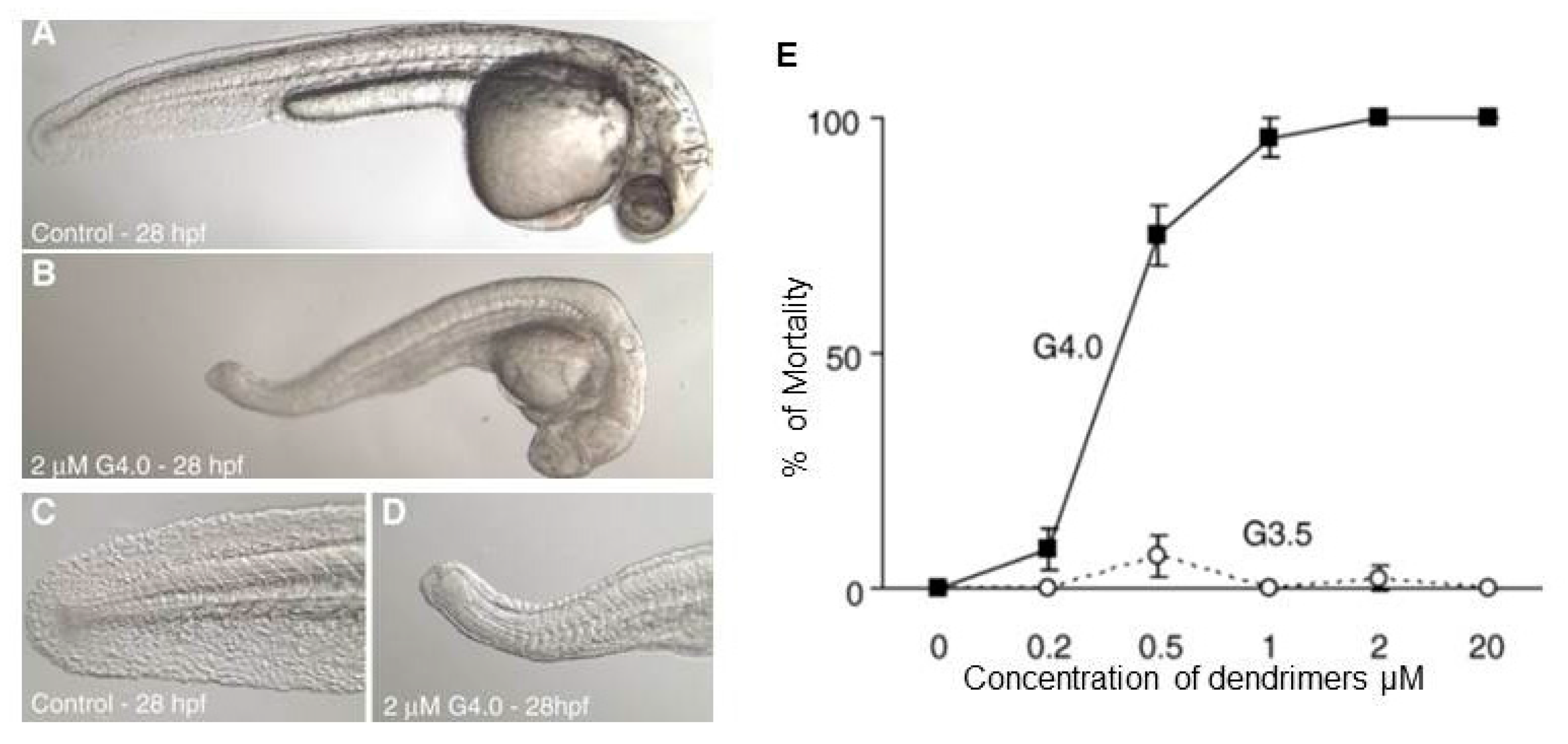
3.2. In Vivo Toxicological Studies of PAMAM Dendrimers
4. Conclusions
Conflicts of Interest
References
- European Commission. Available online: http://ec.europa.eu/environment/chemicals/nanotech/faq/definition_en.htm (accessed on 5 February 2018).
- Luxresearch. Available online: https://portal.luxresearchinc.com/research/report_excerpt/16215 (accessed on 5 February 2018).
- Garland, A. The Global Market for Carbon Nanotubes to 2015. 2009. Available online: http://www.nanoposts.com/indx.php?mod=nanotube (accessed on 20 March 2011).
- Pattni, B.S.; Chupin, V.V.; Torchilin, V.P. New Developments in Liposomal Drug Delivery. Chem. Rev. 2015, 115, 10938–10966. [Google Scholar] [CrossRef] [PubMed]
- Nanotechnology. Available online: http://www.nanotechproject.org/cpi/ (accessed on 5 February 2018).
- Ito, F.; Fujimori, H.; Honnami, H.; Kawakami, H.; Kanamura, K.; Makino, K. Effect of polyethylene glycol on preparation of rifampicin-loaded PLGA microspheres with membrane emulsification technique. Colloids Surf. B Biointerfaces 2008, 66, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Kanchan, V.; Manna, P.K.; Panda, A.K. Improved bioavailability of orally delivered insulin using Eudragit-L30D coated PLGA microparticles. J. Microencapsul. 2008, 25, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Kanchan, V.; Panda, A.K. Evaluation of parenteral depot insulin formulation using PLGA and PLA microparticles. J. Biomater. Appl. 2009, 24, 309–325. [Google Scholar] [CrossRef] [PubMed]
- Storrie, H.; Mooney, D.J. Sustained delivery of plasmid DNA from polymeric scaffolds for tissue engineering. Adv. Drug Deliv. Rev. 2006, 58, 500–514. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Byrne, H.J.; Panda, A.K. Role of Polymeric Excipients on Controlled Release Profile of Glipizide from PLGA and Eudragit RS 100 Nanoparticles. J. Nanopharm. Drug Deliv. 2013, 1, 74–81. [Google Scholar] [CrossRef]
- Gao, L.; Liu, Y.; Kim, D.; Li, Y.; Hwang, G.; Naha, P.C.; Cormode, D.P.; Koo, H. Nanocatalysts promote Streptococcus mutans biofilm matrix degradation and enhance bacterial killing to suppress dental caries in vivo. Biomaterials 2016, 101, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Thaxton, C.S.; Rink, J.S.; Naha, P.C.; Cormode, D.P. Lipoproteins and lipoprotein mimetics for imaging and drug delivery. Adv. Drug Deliv. Rev. 2016, 106, 116–131. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Al Zaki, A.; Hui, J.Z.; Muzykantov, V.R.; Tsourkas, A. Multifunctional nanoparticles: Cost versus benefit of adding targeting and imaging capabilities. Science 2012, 338, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Zaki, A.A.; Hecht, E.; Chorny, M.; Chhour, P.; Blankemeyer, E.; Yates, D.M.; Witschey, W.R.; Litt, H.I.; Tsourkas, A.; et al. Dextran coated bismuth-iron oxide nanohybrid contrast agents for computed tomography and magnetic resonance imaging. J. Mater. Chem. B Mater. Biol. Med. 2014, 2, 8239–8248. [Google Scholar] [CrossRef] [PubMed]
- Pierre, V.C.; Harris, S.M.; Pailloux, S.L. Comparing Strategies in the Design of Responsive Contrast Agents for Magnetic Resonance Imaging: A Case Study with Copper and Zinc. Acc. Chem. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, A.L.; Dhanantwari, A.; Jurcova, M.; Cheheltani, R.; Naha, P.C.; Ivanc, T.; Shefer, E.; Cormode, D.P. Improved sensitivity of computed tomography towards iodine and gold nanoparticle contrast agents via iterative reconstruction methods. Sci. Rep. 2016, 6, 26177. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Lee, N.; Arifin, D.R.; Shats, I.; Janowski, M.; Walczak, P.; Hyeon, T.; Bulte, J.W.M. In Vivo Micro-CT Imaging of Human Mesenchymal Stem Cells Labeled with Gold-Poly-l-Lysine Nanocomplexes. Adv. Funct. Mater. 2017, 27. [Google Scholar] [CrossRef] [PubMed]
- Karunamuni, R.; Naha, P.C.; Lau, K.C.; Al-Zaki, A.; Popov, A.V.; Delikatny, E.J.; Tsourkas, A.; Cormode, D.P.; Maidment, A.D. Development of silica-encapsulated silver nanoparticles as contrast agents intended for dual-energy mammography. Eur. Radiol. 2016, 26, 3301–3309. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Lau, K.C.; Hsu, J.C.; Hajfathalian, M.; Mian, S.; Chhour, P.; Uppuluri, L.; McDonald, E.S.; Maidment, A.D.; Cormode, D.P. Gold silver alloy nanoparticles (GSAN): An imaging probe for breast cancer screening with dual-energy mammography or computed tomography. Nanoscale 2016, 8, 13740–13754. [Google Scholar] [CrossRef] [PubMed]
- Anish, C.; Goswami, D.G.; Kanchan, V.; Mathew, S.; Panda, A.K. The immunogenic characteristics associated with multivalent display of Vi polysaccharide antigen using biodegradable polymer particles. Biomaterials 2012, 33, 6843–6857. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Goswami, D.G.; Singh, R.R.; Surolia, A.; Panda, A.K. Soybean agglutinin coated PLA particles entrapping candidate vaccines induces enhanced primary and sustained secondary antibody response from single point immunization. Eur. J. Pharm. Sci. 2012, 45, 282–295. [Google Scholar] [CrossRef] [PubMed]
- Kanchan, V.; Katare, Y.K.; Panda, A.K. Memory antibody response from antigen loaded polymer particles and the effect of antigen release kinetics. Biomaterials 2009, 30, 4763–4776. [Google Scholar] [CrossRef] [PubMed]
- Kanchan, V.; Katare, Y.K.; Panda, A.K. Role of alum in improving the immunogenicity of biodegradable polymer particle entrapped antigens. Eur. J. Pharm. Sci. 2009, 38, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Rajmohan, G.; Admane, P.; Anish, C.; Panda, A.K. Fusion and self-assembly of biodegradable polymer particles into scaffoldlike and membranelike structures at room temperature for regenerative medicine. Mol. Pharm. 2014, 11, 2190–2202. [Google Scholar] [CrossRef] [PubMed]
- Yoon, A.R.; Kasala, D.; Li, Y.; Hong, J.; Lee, W.; Jung, S.J.; Yun, C.O. Antitumor effect and safety profile of systemically delivered oncolytic adenovirus complexed with EGFR-targeted PAMAM-based dendrimer in orthotopic lung tumor model. J. Controll. Release 2016, 231, 2–16. [Google Scholar] [CrossRef] [PubMed]
- Yuan, A.; Yang, B.; Wu, J.; Hu, Y.; Ming, X. Dendritic nanoconjugates of photosensitizer for targeted photodynamic therapy. Acta Biomater. 2015, 21, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Mohammadpour, R.; Safarian, S.; Buckway, B.; Ghandehari, H. Comparative Endocytosis Mechanisms and Anticancer Effect of HPMA Copolymer- and PAMAM Dendrimer-MTCP Conjugates for Photodynamic Therapy. Macromol. Biosci. 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Heyder, R.S.; Zhong, Q.; Bazito, R.C.; da Rocha, S.R.P. Cellular internalization and transport of biodegradable polyester dendrimers on a model of the pulmonary epithelium and their formulation in pressurized metered-dose inhalers. Int. J. Pharm. 2017, 520, 181–194. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, L.; Liu, N.; Li, S.; Hao, Y.; Zhang, X. EGF-coated nano-dendriplexes for tumor-targeted nucleic acid delivery in vivo. Drug Deliv. 2016, 23, 1718–1725. [Google Scholar] [PubMed]
- Venuganti, V.V.; Saraswathy, M.; Dwivedi, C.; Kaushik, R.S.; Perumal, O.P. Topical gene silencing by iontophoretic delivery of an antisense oligonucleotide-dendrimer nanocomplex: The proof of concept in a skin cancer mouse model. Nanoscale 2015, 7, 3903–3914. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Cao, C.; Liu, Y.; Yu, Q.; Zheng, C.; Sun, D.; Ren, X.; Liu, J. Multifunctional polyamidoamine-modified selenium nanoparticles dual-delivering siRNA and cisplatin to A549/DDP cells for reversal multidrug resistance. Acta Biomater. 2015, 11, 368–380. [Google Scholar] [CrossRef] [PubMed]
- Boni, A.; Bardi, G.; Bertero, A.; Cappello, V.; Emdin, M.; Flori, A.; Gemmi, M.; Innocenti, C.; Menichetti, L.; Sangregorio, C.; et al. Design and optimization of lipid-modified poly(amidoamine) dendrimer coated iron oxide nanoparticles as probes for biomedical applications. Nanoscale 2015, 7, 7307–7317. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Brechbiel, M.W. Nano-sized MRI contrast agents with dendrimer cores. Adv. Drug Deliv. Rev. 2005, 57, 2271–2286. [Google Scholar] [CrossRef] [PubMed]
- Opina, A.C.; Wong, K.J.; Griffiths, G.L.; Turkbey, B.I.; Bernardo, M.; Nakajima, T.; Kobayashi, H.; Choyke, P.L.; Vasalatiy, O. Preparation and long-term biodistribution studies of a PAMAM dendrimer G5-Gd-BnDOTA conjugate for lymphatic imaging. Nanomedicine 2015, 10, 1423–1437. [Google Scholar] [CrossRef] [PubMed]
- Wen, S.; Li, K.; Cai, H.; Chen, Q.; Shen, M.; Huang, Y.; Peng, C.; Hou, W.; Zhu, M.; Zhang, G.; et al. Multifunctional dendrimer-entrapped gold nanoparticles for dual mode CT/MR imaging applications. Biomaterials 2013, 34, 1570–1580. [Google Scholar] [CrossRef] [PubMed]
- Sheng, K.-C.; Kalkanidis, M.; Pouniotis, D.S.; Esparon, S.; Tang, C.K.; Apostolopoulos, V.; Pietersz, G.A. Delivery of antigen using a novel mannosylated dendrimer potentiates immunogenicity in vitro and in vivo. Eur. J. Immunol. 2008, 38, 424–436. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, K.; Naha, P.C.; Naydenova, I.; Mintova, S.; Byrne, H.J. Reactive oxygen species mediated DNA damage in human lung alveolar epithelial (A549) cells from exposure to non-cytotoxic MFI-type zeolite nanoparticles. Toxicol. Lett. 2012, 215, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.M.; Donaldson, K.; Borm, P.J.; Schins, R.P.; Dehnhardt, M.; Gilmour, P.; Jimenez, L.A.; Stone, V. Calcium and ROS-mediated activation of transcription factors and TNF-alpha cytokine gene expression in macrophages exposed to ultrafine particles. Am. J. Physiol. Lung Cell. Mol. Physiol. 2004, 286, L344–L353. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.M.; Wilson, M.R.; MacNee, W.; Stone, V.; Donaldson, K. Size-dependent proinflammatory effects of ultrafine polystyrene particles: A role for surface area and oxidative stress in the enhanced activity of ultrafines. Toxicol. Appl. Pharmacol. 2001, 175, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Colvin, V.L. The potential environmental impact of engineered nanomaterials. Nat. Biotechnol. 2003, 21, 1166–1170. [Google Scholar] [CrossRef] [PubMed]
- Davoren, M.; Herzog, E.; Casey, A.; Cottineau, B.; Chambers, G.; Byrne, H.J.; Lyng, F.M. In vitro toxicity evaluation of single walled carbon nanotubes on human A549 lung cells. Toxicol. In Vitro 2007, 21, 438–448. [Google Scholar] [CrossRef] [PubMed]
- Herzog, E.; Casey, A.; Lyng, F.M.; Chambers, G.; Byrne, H.J.; Davoren, M. A new approach to the toxicity testing of carbon-based nanomaterials—The clonogenic assay. Toxicol. Lett. 2007, 174, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Jeng, H.A.; Swanson, J. Toxicity of metal oxide nanoparticles in mammalian cells. J. Environ. Sci. Health Part A 2006, 41, 2699–2711. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Bhattacharya, K.; Tenuta, T.; Dawson, K.A.; Lynch, I.; Gracia, A.; Lyng, F.M.; Byrne, H.J. Intracellular localisation, geno- and cytotoxic response of polyN-isopropylacrylamide (PNIPAM) nanoparticles to human keratinocyte (HaCaT) and colon cells (SW 480). Toxicol. Lett. 2010, 198, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Davoren, M.; Lyng, F.M.; Byrne, H.J. Reactive oxygen species (ROS) induced cytokine production and cytotoxicity of PAMAM dendrimers in J774A.1 cells. Toxicol. Appl. Pharmacol. 2010, 246, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Vevers, W.F.; Jha, A.N. Genotoxic and cytotoxic potential of titanium dioxide (TiO2) nanoparticles on fish cells in vitro. Ecotoxicology 2008, 17, 410–420. [Google Scholar] [CrossRef] [PubMed]
- Gottardo, S.; Quiros-Pesudo, L.; Totaro, S.; Riego-Sintes, J.; Crutzen, H. NANoREG Harmonised Terminology for Environmental Health and Safety Assessment of Nanomaterials; Publications Office of the European Union: Rue Mercier, Luxembourg, 2016. [Google Scholar]
- Naha, P.C.; Casey, A.; Tenuta, T.; Lynch, I.; Dawson, K.A.; Byrne, H.J.; Davoren, M. Preparation, characterization of NIPAM and NIPAM/BAM copolymer nanoparticles and their acute toxicity testing using an aquatic test battery. Aquat. Toxicol. 2009, 92, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Davoren, M.; Casey, A.; Byrne, H.J. An ecotoxicological study of poly(amidoamine) dendrimers-toward quantitative structure activity relationships. Environ. Sci. Technol. 2009, 43, 6864–6869. [Google Scholar] [CrossRef] [PubMed]
- Stoeger, T.; Takenaka, S.; Frankenberger, B.; Ritter, B.; Karg, E.; Maier, K.; Schulz, H.; Schmid, O. Deducing in vivo toxicity of combustion-derived nanoparticles from a cell-free oxidative potency assay and metabolic activation of organic compounds. Environ. Health Perspect. 2009, 117, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Bouwmeester, H.; Lynch, I.; Marvin, H.J.; Dawson, K.A.; Berges, M.; Braguer, D.; Byrne, H.J.; Casey, A.; Chambers, G.; Clift, M.J.; et al. Minimal analytical characterization of engineered nanomaterials needed for hazard assessment in biological matrices. Nanotoxicology 2011, 5, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Nel, A.; Xia, T.; Madler, L.; Li, N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Xia, T.; Kovochich, M.; Brant, J.; Hotze, M.; Sempf, J.; Oberley, T.; Sioutas, C.; Yeh, J.I.; Wiesner, M.R.; Nel, A.E. Comparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigm. Nano Lett. 2006, 6, 1794–1807. [Google Scholar] [CrossRef] [PubMed]
- Rahman, I. Regulation of nuclear factor-kappa B, activator protein-1, and glutathione levels by tumor necrosis factor-alpha and dexamethasone in alveolar epithelial cells. Biochem. Pharmacol. 2000, 60, 1041–1049. [Google Scholar] [CrossRef]
- Lanone, S.; Boczkowski, J. Biomedical applications and potential health risks of nanomaterials: Molecular mechanisms. Curr. Mol. Med. 2006, 6, 651–663. [Google Scholar] [CrossRef] [PubMed]
- Park, E.J.; Park, K. Oxidative stress and pro-inflammatory responses induced by silica nanoparticles in vivo and in vitro. Toxicol. Lett. 2009, 184, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Dobrovolskaia, M.A.; McNeil, S.E. Immunological properties of engineered nanomaterials. Nat. Nanotechnol. 2007, 2, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, Y.; Jiao, F.; Lao, F.; Li, W.; Gu, Y.; Li, Y.; Ge, C.; Zhou, G.; Li, B.; et al. Time-dependent translocation and potential impairment on central nervous system by intranasally instilled TiO(2) nanoparticles. Toxicology 2008, 254, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Srinageshwar, B.; Peruzzaro, S.; Andrews, M.; Johnson, K.; Hietpas, A.; Clark, B.; McGuire, C.; Petersen, E.; Kippe, J.; Stewart, A.; et al. PAMAM Dendrimers Cross the Blood-Brain Barrier When Administered through the Carotid Artery in C57BL/6J Mice. Int. J. Mol. Sci. 2017, 18, 628. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Chhour, P.; Cormode, D.P. Systematic in vitro toxicological screening of gold nanoparticles designed for nanomedicine applications. Toxicol. In Vitro 2015, 29, 1445–1453. [Google Scholar] [CrossRef] [PubMed]
- Nowack, B.; Bucheli, T.D. Occurrence, behavior and effects of nanoparticles in the environment. Environ. Pollut. 2007, 150, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Baun, A.; Sorensen, S.N.; Rasmussen, R.F.; Hartmann, N.B.; Koch, C.B. Toxicity and bioaccumulation of xenobiotic organic compounds in the presence of aqueous suspensions of aggregates of nano-C(60). Aquat. Toxicol. 2008, 86, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Flahaut, E.; Cheng, S.H. Effect of carbon nanotubes on developing zebrafish (Danio rerio) embryos. Environ. Toxicol. Chem. 2007, 26, 708–716. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Xing, B. Phytotoxicity of nanoparticles: Inhibition of seed germination and root growth. Environ. Pollut. 2007, 150, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Lovern, S.B.; Klaper, R. Daphnia magna mortality when exposed to titanium dioxide and fullerene (C60) nanoparticles. Environ. Toxicol. Chem. 2006, 25, 1132–1137. [Google Scholar] [CrossRef] [PubMed]
- Lovern, S.B.; Strickler, J.R.; Klaper, R. Behavioral and physiological changes in Daphnia magna when exposed to nanoparticle suspensions (titanium dioxide, nano-C60, and C60HxC70Hx). Environ. Sci. Technol. 2007, 41, 4465–4470. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.J.; Shaw, B.J.; Handy, R.D. Toxicity of single walled carbon nanotubes to rainbow trout, (Oncorhynchus mykiss): Respiratory toxicity, organ pathologies, and other physiological effects. Aquat. Toxicol. 2007, 82, 94–109. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhao, Q.; Li, Y.; Cai, X.; Li, W. The interaction and toxicity of multi-walled carbon nanotubes with Stylonychia mytilus. J. Nanosci. Nanotechnol. 2006, 6, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Handy, R.D.; van den Brink, N.; Chappell, M.; Muhling, M.; Behra, R.; Dusinska, M.; Simpson, P.; Ahtiainen, J.; Jha, A.N.; Seiter, J.; et al. Practical considerations for conducting ecotoxicity test methods with manufactured nanomaterials: What have we learnt so far? Ecotoxicology 2012, 21, 933–972. [Google Scholar] [CrossRef] [PubMed]
- Heinlaan, M.; Ivask, A.; Blinova, I.; Dubourguier, H.C.; Kahru, A. Toxicity of nanosized and bulk ZnO, CuO and TiO2 to bacteria Vibrio fischeri and crustaceans Daphnia magna and Thamnocephalus platyurus. Chemosphere 2008, 71, 1308–1316. [Google Scholar] [CrossRef] [PubMed]
- Mortimer, M.; Kasemets, K.; Heinlaan, M.; Kurvet, I.; Kahru, A. High throughput kinetic Vibrio fischeri bioluminescence inhibition assay for study of toxic effects of nanoparticles. Toxicol. In Vitro 2008, 22, 1412–1417. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Byrne, H.J. Generation of intracellular reactive oxygen species and genotoxicity effect to exposure of nanosized polyamidoamine (PAMAM) dendrimers in PLHC-1 cells in vitro. Aquat. Toxicol. 2013, 132–133, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Glomstad, B.; Altin, D.; Sorensen, L.; Liu, J.; Jenssen, B.M.; Booth, A.M. Carbon Nanotube Properties Influence Adsorption of Phenanthrene and Subsequent Bioavailability and Toxicity to Pseudokirchneriella subcapitata. Environ. Sci. Technol. 2016, 50, 2660–2668. [Google Scholar] [CrossRef] [PubMed]
- Sanchis, J.; Olmos, M.; Vincent, P.; Farre, M.; Barcelo, D. New Insights on the Influence of Organic Co-Contaminants on the Aquatic Toxicology of Carbon Nanomaterials. Environ. Sci. Technol. 2016, 50, 961–969. [Google Scholar] [CrossRef] [PubMed]
- Fraser, T.W.; Reinardy, H.C.; Shaw, B.J.; Henry, T.B.; Handy, R.D. Dietary toxicity of single-walled carbon nanotubes and fullerenes (C60) in rainbow trout (Oncorhynchus mykiss). Nanotoxicology 2011, 5, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Huk, A.; Izak-Nau, E.; Reidy, B.; Boyles, M.; Duschl, A.; Lynch, I.; Dusinska, M. Is the toxic potential of nanosilver dependent on its size? Part. Fibre Toxicol. 2014, 11, 65. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, S.P.; Strauss, M.; Delite, F.S.; Clemente, Z.; Castro, V.L.; Martinez, D.S. Activated carbon from pyrolysed sugarcane bagasse: Silver nanoparticle modification and ecotoxicity assessment. Sci. Total Environ. 2016, 565, 833. [Google Scholar] [CrossRef] [PubMed]
- Ramskov, T.; Forbes, V.E.; Gilliland, D.; Selck, H. Accumulation and effects of sediment-associated silver nanoparticles to sediment-dwelling invertebrates. Aquat. Toxicol. 2015, 166, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Oprsal, J.; Blaha, L.; Pouzar, M.; Knotek, P.; Vlcek, M.; Hrda, K. Assessment of silver nanoparticle toxicity for common carp (Cyprinus carpio) fish embryos using a novel method controlling the agglomeration in the aquatic media. Environ. Sci. Pollut. Res. Int. 2015, 22, 19124–19132. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.C.; MacKay, J.A.; Frechet, J.M.; Szoka, F.C. Designing dendrimers for biological applications. Nat. Biotechnol. 2005, 23, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Sevenson, S.; Tomalia, D.A. Dendrimers in biomedical applications-reflections on the field. Adv. Drug Deliv. Rev. 2012, 64, 102–115. [Google Scholar] [CrossRef]
- Boas, U.; Karlsson, A.J.; de Waal, B.F.; Meijer, E.W. Synthesis and properties of new thiourea-functionalized poly(propylene imine) dendrimers and their role as hosts for urea functionalized guests. J. Org. Chem. 2001, 66, 2136–2145. [Google Scholar] [CrossRef] [PubMed]
- Jansen, J.; Meijer, E.W.; de Brabander-van den Berg, E.M. The Dendritic Box: Shape-Selective Liberation of Encapsulated Guests. J. Am. Chem. Soc. 1995, 117, 4417–4418. [Google Scholar] [CrossRef]
- Jansen, J.F.; de Brabander-van den Berg, E.M.; Meijer, E.W. Encapsulation of guest molecules into a dendritic box. Science 1994, 266, 1226–1229. [Google Scholar] [CrossRef] [PubMed]
- D'Emanuele, A.; Attwood, D. Dendrimer-drug interactions. Adv. Drug Deliv. Rev. 2005, 57, 2147–2162. [Google Scholar] [CrossRef] [PubMed]
- Duncan, R.; Izzo, L. Dendrimer biocompatibility and toxicity. Adv. Drug Deliv. Rev. 2005, 57, 2215–2237. [Google Scholar] [CrossRef] [PubMed]
- Svenson, S.; Tomalia, D.A. Dendrimers in biomedical applications—Reflections on the field. Adv. Drug Deliv. Rev. 2005, 57, 2106–2129. [Google Scholar] [CrossRef] [PubMed]
- Venuganti, V.V.; Perumal, O.P. Effect of poly(amidoamine) (PAMAM) dendrimer on skin permeation of 5-fluorouracil. Int. J. Pharm. 2008, 361, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Kannan, R.M.; Nance, E.; Kannan, S.; Tomalia, D.A. Emerging concepts in dendrimer-based nanomedicine: From design principles to clinical applications. J. Intern. Med. 2014, 276, 579–617. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, Y.; Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: Mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986, 46, 6387–6392. [Google Scholar] [PubMed]
- Gillies, E.R.; Frechet, J.M. Dendrimers and dendritic polymers in drug delivery. Drug Discov. Today 2005, 10, 35–43. [Google Scholar] [CrossRef]
- Miyano, T.; Wijagkanalan, W.; Kawakami, S.; Yamashita, F.; Hashida, M. Anionic amino acid dendrimer-trastuzumab conjugates for specific internalization in HER2-positive cancer cells. Mol. Pharm. 2010, 7, 1318–1327. [Google Scholar] [CrossRef] [PubMed]
- Shukla, R.; Thomas, T.P.; Desai, A.M.; Kotlyar, A.; Park, S.J.; Baker, J.R. HER2 specific delivery of methotrexate by dendrimer conjugated anti-HER2 mAb. Nanotechnology 2008, 19, 295102. [Google Scholar] [CrossRef] [PubMed]
- Shukla, R.; Thomas, T.P.; Peters, J.L.; Desai, A.M.; Kukowska-Latallo, J.; Patri, A.K.; Kotlyar, A.; Baker, J.R., Jr. HER2 specific tumor targeting with dendrimer conjugated anti-HER2 mAb. Bioconjug. Chem. 2006, 17, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Barth, R.F.; Yang, W.; Kawabata, S.; Zhang, L.; Green-Church, K. Targeted delivery of methotrexate to epidermal growth factor receptor-positive brain tumors by means of cetuximab (IMC-C225) dendrimer bioconjugates. Mol. Cancer Ther. 2006, 5, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Wu, G.; Barth, R.F.; Swindall, M.R.; Bandyopadhyaya, A.K.; Tjarks, W.; Tordoff, K.; Moeschberger, M.; Sferra, T.J.; Binns, P.J.; et al. Molecular targeting and treatment of composite EGFR and EGFRvIII-positive gliomas using boronated monoclonal antibodies. Clin. Cancer Res. 2008, 14, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Rajananthanan, P.; Attard, G.S.; Sheikh, N.A.; Morrow, W.J. Evaluation of novel aggregate structures as adjuvants: Composition, toxicity studies and humoral responses. Vaccine 1999, 17, 715–730. [Google Scholar] [CrossRef]
- Hulikova, K.; Benson, V.; Svoboda, J.; Sima, P.; Fiserova, A. N-Acetyl-d-glucosamine-coated polyamidoamine dendrimer modulates antibody formation via natural killer cell activation. Int. Immunopharmacol. 2009, 9, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Wiener, E.C.; Brechbiel, M.W.; Brothers, H.; Magin, R.L.; Gansow, O.A.; Tomalia, D.A.; Lauterbur, P.C. Dendrimer-based metal chelates: A new class of magnetic resonance imaging contrast agents. Magn. Reson. Med. 1994, 31, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Konda, S.D.; Aref, M.; Brechbiel, M.; Wiener, E.C. Development of a tumor-targeting MR contrast agent using the high-affinity folate receptor: Work in progress. Investig. Radiol. 2000, 35, 50–57. [Google Scholar] [CrossRef]
- OECD. Series on the Safety of Manufactured Nanomaterials. No. 27 List of Manufactured Nanomaterials and List of Endpoints for Phase One of the Sponsorship Programme for the Testing of Manufactured Nanomaterials: Revision. Available online: http://www.oecd.org/officialdocuments/publicdisplaydocumentpdf/?cote=env/jm/mono%282010%2946&doclanguage=en (accessed on 12 Ferubuary 2018).
- Mukherjee, S.P.; Byrne, H.J. Polyamidoamine dendrimer nanoparticle cytotoxicity, oxidative stress, caspase activation and inflammatory response: Experimental observation and numerical simulation. Nanomedicine 2013, 9, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.P.; Davoren, M.; Byrne, H.J. In vitro mammalian cytotoxicological study of PAMAM dendrimers—Towards quantitative structure activity relationships. Toxicol. In Vitro 2010, 24, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.P.; Lyng, F.M.; Garcia, A.; Davoren, M.; Byrne, H.J. Mechanistic studies of in vitro cytotoxicity of poly(amidoamine) dendrimers in mammalian cells. Toxicol. Appl. Pharmacol. 2010, 248, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Cha, K.E.; Kim, M.S.; Hong, H.W.; Chung, D.J.; Ryu, G.; Myung, H. Nanosized polyamidoamine (PAMAM) dendrimer-induced apoptosis mediated by mitochondrial dysfunction. Toxicol. Lett. 2009, 190, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Labieniec, M.; Gabryelak, T. Preliminary biological evaluation of poli(amidoamine) (PAMAM) dendrimer G3.5 on selected parameters of rat liver mitochondria. Mitochondrion 2008, 8, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Bielinska, A.U.; Mecke, A.; Keszler, B.; Beals, J.L.; Shi, X.; Balogh, L.; Orr, B.G.; Baker, J.R., Jr.; Banaszak Holl, M.M. Interaction of poly(amidoamine) dendrimers with supported lipid bilayers and cells: Hole formation and the relation to transport. Bioconjug. Chem. 2004, 15, 774–782. [Google Scholar] [CrossRef] [PubMed]
- Leroueil, P.R.; Berry, S.A.; Duthie, K.; Han, G.; Rotello, V.M.; McNerny, D.Q.; Baker, J.R., Jr.; Orr, B.G.; Holl, M.M. Wide varieties of cationic nanoparticles induce defects in supported lipid bilayers. Nano Lett. 2008, 8, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Leroueil, P.R.; Hong, S.; Mecke, A.; Baker, J.R., Jr.; Orr, B.G.; Banaszak Holl, M.M. Nanoparticle interaction with biological membranes: Does nanotechnology present a Janus face? Acc. Chem. Res. 2007, 40, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Malik, N.; Wiwattanapatapee, R.; Klopsch, R.; Lorenz, K.; Frey, H.; Weener, J.W.; Meijer, E.W.; Paulus, W.; Duncan, R. Dendrimers: Relationship between structure and biocompatibility in vitro, and preliminary studies on the biodistribution of 125I-labelled polyamidoamine dendrimers in vivo. J. Controll. Release 2000, 65, 133–148. [Google Scholar] [CrossRef]
- Roberts, J.C.; Bhalgat, M.K.; Zera, R.T. Preliminary biological evaluation of polyamidoamine (PAMAM) Starburst dendrimers. J. Biomed. Mater. Res. 1996, 30, 53–65. [Google Scholar] [CrossRef]
- Bodewein, L.; Schmelter, F.; Di Fiore, S.; Hollert, H.; Fischer, R.; Fenske, M. Differences in toxicity of anionic and cationic PAMAM and PPI dendrimers in zebrafish embryos and cancer cell lines. Toxicol. Appl. Pharmacol. 2016, 305, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Xiong, W.; Zhu, Y.; Xu, H.; Yang, X. Protective effect of PEGylation against poly(amidoamine) dendrimer-induced hemolysis of human red blood cells. J. Biomed. Mater. Res. Part B Appl. Biomater. 2010, 93, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Khalid, H.; Mukherjee, S.P.; O'Neill, L.; Byrne, H.J. Structural dependence of in vitro cytotoxicity, oxidative stress and uptake mechanisms of poly(propylene imine) dendritic nanoparticles. J. Appl. Toxicol. 2016, 36, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Maher, M.A.; Naha, P.C.; Mukherjee, S.P.; Byrne, H.J. Numerical simulations of in vitro nanoparticle toxicity—The case of poly(amido amine) dendrimers. Toxicol. In Vitro 2014, 28, 1449–1460. [Google Scholar] [CrossRef] [PubMed]
- Bertero, A.; Boni, A.; Gemmi, M.; Gagliardi, M.; Bifone, A.; Bardi, G. Surface functionalisation regulates polyamidoamine dendrimer toxicity on blood-brain barrier cells and the modulation of key inflammatory receptors on microglia. Nanotoxicology 2014, 8, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Albertazzi, L.; Gherardini, L.; Brondi, M.; Sulis Sato, S.; Bifone, A.; Pizzorusso, T.; Ratto, G.M.; Bardi, G. In vivo distribution and toxicity of PAMAM dendrimers in the central nervous system depend on their surface chemistry. Mol. Pharm. 2013, 10, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Feliu, N.; Kohonen, P.; Ji, J.; Zhang, Y.; Karlsson, H.L.; Palmberg, L.; Nystrom, A.; Fadeel, B. Next-generation sequencing reveals low-dose effects of cationic dendrimers in primary human bronchial epithelial cells. ACS Nano 2015, 9, 146–163. [Google Scholar] [CrossRef] [PubMed]
- Ryman-Rasmussen, J.P.; Riviere, J.E.; Monteiro-Riviere, N.A. Surface coatings determine cytotoxicity and irritation potential of quantum dot nanoparticles in epidermal keratinocytes. J. Investig. Dermatol. 2007, 127, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Lazniewska, J.; Milowska, K.; Zablocka, M.; Mignani, S.; Caminade, A.M.; Majoral, J.P.; Bryszewska, M.; Gabryelak, T. Mechanism of Cationic Phosphorus Dendrimer Toxicity against Murine Neural Cell Lines. Mol. Pharm. 2013, 10, 3484–3496. [Google Scholar] [CrossRef] [PubMed]
- Ciolkowski, M.; Rozanek, M.; Bryszewska, M.; Klajnert, B. The influence of PAMAM dendrimers surface groups on their interaction with porcine pepsin. Biochim. Biophys. Acta 2013, 1834, 1982–1987. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, Z.; Ma, G.; Lin, W.; Chen, S. Reducing the cytotoxity of poly(amidoamine) dendrimers by modification of a single layer of carboxybetaine. Langmuir 2013, 29, 8914–8921. [Google Scholar] [CrossRef] [PubMed]
- Janaszewska, A.; Ciolkowski, M.; Wrobel, D.; Petersen, J.F.; Ficker, M.; Christensen, J.B.; Bryszewska, M.; Klajnert, B. Modified PAMAM dendrimer with 4-carbomethoxypyrrolidone surface groups reveals negligible toxicity against three rodent cell-lines. Nanomedicine 2013, 9, 461–464. [Google Scholar] [CrossRef] [PubMed]
- Ciolkowski, M.; Petersen, J.F.; Ficker, M.; Janaszewska, A.; Christensen, J.B.; Klajnert, B.; Bryszewska, M. Surface modification of PAMAM dendrimer improves its biocompatibility. Nanomedicine 2012, 8, 815–817. [Google Scholar] [CrossRef] [PubMed]
- Kolhatkar, R.B.; Kitchens, K.M.; Swaan, P.W.; Ghandehari, H. Surface acetylation of polyamidoamine (PAMAM) dendrimers decreases cytotoxicity while maintaining membrane permeability. Bioconjug. Chem. 2007, 18, 2054–2060. [Google Scholar] [CrossRef] [PubMed]
- Demaurex, N. pH Homeostasis of cellular organelles. News Physiol. Sci. 2002, 17, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Seib, F.P.; Jones, A.T.; Duncan, R. Comparison of the endocytic properties of linear and branched PEIs, and cationic PAMAM dendrimers in B16f10 melanoma cells. J. Controll. Release 2007, 117, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.; Athey, B.D.; Wetzel, A.W.; Meixner, W.; Baker, J.R. Structural molecular dynamics studies on polyamidoamine dendrimers for a therapeutic application: Effects of pH and generation. Macromolecules 2002, 35, 4510–4520. [Google Scholar] [CrossRef]
- Perumal, O.P.; Inapagolla, R.; Kannan, S.; Kannan, R.M. The effect of surface functionality on cellular trafficking of dendrimers. Biomaterials 2008, 29, 3469–3476. [Google Scholar] [CrossRef] [PubMed]
- Albertazzi, L.; Serresi, M.; Albanese, A.; Beltram, F. Dendrimer Internalization and Intracellular Trafficking in Living Cells. Mol. Pharm. 2010, 7, 680–688. [Google Scholar] [CrossRef] [PubMed]
- Feliu, N.; Walter, M.V.; Montanez, M.I.; Kunzmann, A.; Hult, A.; Nystrom, A.; Malkoch, M.; Fadeel, B. Stability and biocompatibility of a library of polyester dendrimers in comparison to polyamidoamine dendrimers. Biomaterials 2012, 33, 1970–1981. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Zhao, L.; Li, Y.; Xu, T. Design of biocompatible dendrimers for cancer diagnosis and therapy: Current status and future perspectives. Chem. Soc. Rev. 2011, 40, 2673–2703. [Google Scholar] [CrossRef] [PubMed]
- Maher, M.A.; Byrne, H.J. Modification of the in vitro uptake mechanism and antioxidant levels in HaCaT cells and resultant changes to toxicity and oxidative stress of G4 and G6 poly(amidoamine) dendrimer nanoparticles. Anal. Bioanal. Chem. 2016, 408, 5295–5307. [Google Scholar] [CrossRef] [PubMed]
- Girard, P.M.; Graindorge, D.; Smirnova, V.; Rigolet, P.; Francesconi, S.; Scanlon, S.; Sage, E. Oxidative stress in mammalian cells impinges on the cysteines redox state of human XRCC3 protein and on its cellular localization. PLoS ONE 2013, 8, e75751. [Google Scholar] [CrossRef] [PubMed]
- He, Y.Y.; Huang, J.L.; Ramirez, D.C.; Chignell, C.F. Role of reduced glutathione efflux in apoptosis of immortalized human keratinocytes induced by UVA. J. Biol. Chem. 2003, 278, 8058–8064. [Google Scholar] [CrossRef] [PubMed]
- Madesh, M.; Benard, O.; Balasubramanian, K.A. Glutathione modulates lipid composition of human colon derived HT-29 cells. Int. J. Biochem. Cell Biol. 1998, 30, 1345–1352. [Google Scholar] [CrossRef]
- Tobi, S.E.; Paul, N.; McMillan, T.J. Glutathione modulates the level of free radicals produced in UVA-irradiated cells. J. Photochem. Photobiol. B 2000, 57, 102–112. [Google Scholar] [CrossRef]
- Hipkiss, A.R. Carnosine and its possible roles in nutrition and health. Adv. Food Nutr. Res. 2009, 57, 87–154. [Google Scholar] [PubMed]
- Guarnieri, D.; Sabella, S.; Muscetti, O.; Belli, V.; Malvindi, M.A.; Fusco, S.; De Luca, E.; Pompa, P.P.; Netti, P.A. Transport across the cell-membrane dictates nanoparticle fate and toxicity: A new paradigm in nanotoxicology. Nanoscale 2014, 6, 10264–10273. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.F.; Campbell, R.A.; Brooks, A.E.; Assemi, S.; Tadjiki, S.; Thiagarajan, G.; Mulcock, C.; Weyrich, A.S.; Brooks, B.D.; Ghandehari, H.; et al. Cationic PAMAM dendrimers aggressively initiate blood clot formation. ACS Nano 2012, 6, 9900–9910. [Google Scholar] [CrossRef] [PubMed]
- Greish, K.; Thiagarajan, G.; Herd, H.; Price, R.; Bauer, H.; Hubbard, D.; Burckle, A.; Sadekar, S.; Yu, T.; Anwar, A.; et al. Size and surface charge significantly influence the toxicity of silica and dendritic nanoparticles. Nanotoxicology 2012, 6, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Cai, X.; Su, Y.; Hu, J.; Wu, Q.; Zhang, H.; Xiao, J.; Cheng, Y. Reducing cytotoxicity while improving anti-cancer drug loading capacity of polypropylenimine dendrimers by surface acetylation. Acta Biomater. 2012, 8, 4304–4313. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Guo, F.; Zou, Z.; Li, C.; Hong, X.; Zhao, Y.; Wang, C.; Wang, H.; Liu, H.; Yang, P.; et al. Cationic nanoparticles directly bind angiotensin-converting enzyme 2 and induce acute lung injury in mice. Part. Fibre Toxicol. 2015, 12, 4. [Google Scholar] [CrossRef] [PubMed]
- Durocher, I.; Girard, D. In vivo proinflammatory activity of generations 0-3 (G0-G3) polyamidoamine (PAMAM) nanoparticles. Inflamm. Res. 2016, 65, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Heiden, T.C.; Dengler, E.; Kao, W.J.; Heideman, W.; Peterson, R.E. Developmental toxicity of low generation PAMAM dendrimers in zebrafish. Toxicol. Appl. Pharmacol. 2007, 225, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Lesniak, W.G.; Mishra, M.K.; Jyoti, A.; Balakrishnan, B.; Zhang, F.; Nance, E.; Romero, R.; Kannan, S.; Kannan, R.M. Biodistribution of fluorescently labeled PAMAM dendrimers in neonatal rabbits: Effect of neuroinflammation. Mol. Pharm. 2013, 10, 4560–4571. [Google Scholar] [CrossRef] [PubMed]
- Thiagarajan, G.; Sadekar, S.; Greish, K.; Ray, A.; Ghandehari, H. Evidence of oral translocation of anionic G6.5 dendrimers in mice. Mol. Pharm. 2013, 10, 988–998. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naha, P.C.; Mukherjee, S.P.; Byrne, H.J. Toxicology of Engineered Nanoparticles: Focus on Poly(amidoamine) Dendrimers. Int. J. Environ. Res. Public Health 2018, 15, 338. https://doi.org/10.3390/ijerph15020338
Naha PC, Mukherjee SP, Byrne HJ. Toxicology of Engineered Nanoparticles: Focus on Poly(amidoamine) Dendrimers. International Journal of Environmental Research and Public Health. 2018; 15(2):338. https://doi.org/10.3390/ijerph15020338
Chicago/Turabian StyleNaha, Pratap C., Sourav P. Mukherjee, and Hugh J. Byrne. 2018. "Toxicology of Engineered Nanoparticles: Focus on Poly(amidoamine) Dendrimers" International Journal of Environmental Research and Public Health 15, no. 2: 338. https://doi.org/10.3390/ijerph15020338
APA StyleNaha, P. C., Mukherjee, S. P., & Byrne, H. J. (2018). Toxicology of Engineered Nanoparticles: Focus on Poly(amidoamine) Dendrimers. International Journal of Environmental Research and Public Health, 15(2), 338. https://doi.org/10.3390/ijerph15020338





