The Effect of a Leisure Time Physical Activity Intervention Delivered via a Workplace: 15-Month Follow-Up Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Setting and Design
2.2. Blood Analyses
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Paffenbarger, R.S., Jr.; Hyde, R.T.; Wing, A.L.; Hsieh, C.C. Physical activity, all-cause mortality, and longevity of college alumni. N. Engl. J. Med. 1986, 314, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Nathan, D.M.; Krolewski, A.S.; Stampfer, M.J.; Willett, W.C.; Hennekens, C.H. A prospective study of exercise and incidence of diabetes among US male physicians. JAMA 1992, 268, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.N.; Heady, J.A.; Raffle, P.A.; Roberts, C.G.; Parks, J.W. Coronary heart-disease and physical activity of work. Lancet 1953, 265, 1111–1120. [Google Scholar] [CrossRef]
- Hu, G.; Tuomilehto, J.; Silventoinen, K.; Barengo, N.C.; Peltonen, M.; Jousilahti, P. The effects of physical activity and body mass index on cardiovascular, cancer and all-cause mortality among 47 212 middle-aged Finnish men and women. Int. J. Obes. 2005, 29, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Borgundvaag, E.; Janssen, I. Objectively Measured Physical Activity and Mortality Risk Among American Adults. Am. J. Prev. Med. 2017, 52, 25–31. [Google Scholar]
- Semlitsch, T.; Jeitler, K.; Hemkens, L.G.; Horvath, K.; Nagele, E.; Schuermann, C.; Pignitter, N.; Herrmann, K.H.; Waffenschmidt, S.; Siebenhofer, A. Increasing physical activity for the treatment of hypertension: A systematic review and meta-analysis. Sports Med. 2013, 43, 1009–1023. [Google Scholar] [CrossRef] [PubMed]
- Hagner-Derengowska, M.; Kaluzny, K.; Kochanski, B.; Hagner, W.; Borkowska, A.; Czamara, A.; Budzynski, J. Effects of Nordic Walking and Pilates exercise programs on blood glucose and lipid profile in overweight and obese postmenopausal women in an experimental, nonrandomized, open-label, prospective controlled trial. Menopause 2015. [Google Scholar] [CrossRef] [PubMed]
- Lieber, B.A.; Taylor, B.; Appelboom, G.; Prasad, K.; Bruce, S.; Yang, A.; Bruce, E.; Christophe, B.; Connolly, E.S., Jr. Meta-analysis of telemonitoring to improve HbA1c levels: Promise for stroke survivors. J. Clin. Neurosci. 2015, 22, 807–811. [Google Scholar] [PubMed]
- Beavers, K.M.; Brinkley, T.E.; Nicklas, B.J. Effect of exercise training on chronic inflammation. Clin. Chim. Acta 2010, 411, 785–793. [Google Scholar] [CrossRef] [PubMed]
- Ding, D.; Rogers, K.; van der Ploeg, H.; Stamatakis, E.; Bauman, A.E. Traditional and emerging lifestyle risk behaviors and all-cause mortelity in middle-aged and older adults: Evidence from a large population-based Australian cohort. PLoS Med. 2015, 12, e1001917. [Google Scholar] [CrossRef]
- Lidegaard, M.; Søgaard, K.; Krustrup, P.; Holtermann, A.; Korshøj, M. Effects of 12 months aerobic exercise intervention on work ability, need for recovery, productivity and rating of exertion among cleaners: A worksite RCT. Int. Arch. Occup. Environ. Health 2017. [Google Scholar] [CrossRef] [PubMed]
- Rutanen, R.; Luoto, R.; Raitanen, J.; Mansikkamäki, K.; Tomás, E.; Nygård, C.H. Short-and long-term effects of a physical exercise intervention on work ability and work strain in symptomatic menopausal women. Saf. Health Work 2014, 5, 186–190. [Google Scholar] [CrossRef] [PubMed]
- To, Q.G.; Chen, T.T.L.; Magnussen, C.G.; McCluskey, S.; Long, A.F. Workplace physical activity interventions: A systematic review. Am. J. Health Promot. 2013, 27, e113–e123. [Google Scholar] [CrossRef] [PubMed]
- Groeneveld, I.F.; Proper, K.I.; van der Beek, A.J.; Hildebrandt, V.H.; van Mechelen, W. Lifestyle-focused interventions at the workplace to reduce the risk of cardiovascular disease-a systemetic review. SJWEH 2010, 36, 202–215. [Google Scholar]
- Malik, S.H.; Blake, H.; Suggs, L.S. A systematic review of workplace health promotion interventions for increasing physical activity. Br. J. Health Psychol. 2014, 19, 149–180. [Google Scholar] [CrossRef] [PubMed]
- Dugdill, L.; Brettle, A.; Hulme, C.; McCluskey, S.; Long, A.F. Workplace physical activity interventions: A systematic review. Int. J. Workplace Health Manag. 2008, 1, 20–40. [Google Scholar] [CrossRef]
- Schröer, S.; Haupt, J.; Pieper, C. Evidence-based lifestyle interventions in the workplace-an overview. Occup. Med. 2014, 64, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Neuhaus, M.; Eakin, E.G.; Straker, L.; Owen, N.; Dunstan, D.W.; Reid, N.; Healy, G.N. Reducing occupational sedentary time: A systematic review and meta-analysis of evidence on activity-permissive workstations. Obes. Rev. 2014, 15, 822–838. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Available online: www.who.int/occupational_health/topics/ workplace/en/hweot-eh (assessed on 6 February 2017).
- Skogstad, M.; Lunde, L.K.; Skare, O.; Mamen, A.; Alfonso, J.H.; Øvstebø, R.; Ulvestad, B. Physical activity initiated by employer and its health effects; an eight week follow-up study. BMC Public Health 2016, 16, 377. [Google Scholar] [CrossRef] [PubMed]
- Lunde, L.K.; Skare, Ø.; Aass, H.C.D.; Mamen, A.; Einarsdóttir, E.; Ulvestad, B.; Skogstad, M. Physical activity initiated by employer induce improvements in a novel set of biomarkers of inflammation. An eight-week follow-up study. Eur. J. Appl. Physiol. 2017, 117, 521–523. [Google Scholar] [CrossRef] [PubMed]
- Holtermann, A.; Marott, J.L.; Gyntelberg, F.; Søgaard, K.; Suadicani, P.; Mortensen, O.S.; Prescott, E.; Schnohr, P. Occupational and leisure time physical activity: Risk of all-cause mortality and myocardial infarction in the Copenhagen City Heart Study. A prospective cohort study. BMJ Open 2012, 2, e000556. [Google Scholar] [CrossRef] [PubMed]
- Harari, G.; Green, M.S.; Zelber-Sagi, S. Combined association of occupational and leisure-time physical activity with all-cause and coronary heart disease mortality among a cohort of men followed-up for 22 years. Occup. Environ. Med. 2015, 72, 617–624. [Google Scholar] [CrossRef] [PubMed]
- Ryan, R.M.; Deci, E.L. Self-determination theory and the facilitation of intrinsic motivation, social development, and well-being. Am. Psychol. 2000, 55, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.Y.; Ntoumanis, N.; Thøgersen-Ntoumani, C.; Deci, E.L.; Ryan, R.M.; Duda, J.L.; Williams, G.C. Self-determination theory applied to helath contexts: A meta-analysis. Perspect. Psychol. Sci. 2012, 7, 325–340. [Google Scholar] [CrossRef] [PubMed]
- Moholdt, T.; Wisloff, U.; Lydersen, S.; Nauman, J. Current physical activity guidelines for health are insufficient to mitigate long-term weight gain: More data in the fitness versus fatness debate (The HUNT study, Norway). Br. J. Sports Med. 2014, 48, 1489–1496. [Google Scholar] [CrossRef] [PubMed]
- Dalager, T.; Justesen, J.B.; Murray, M.; Boyle, E.; Sjøgaard, G. Implementing intelligent physical exercise training at the workplace: Health effects among office workers-a randomized controlled trial. Eur. J. Appl. Physiol. 2016, 116, 1433–1442. [Google Scholar] [CrossRef] [PubMed]
- Tjonna, A.E.; Rognmo, O.; Bye, A.; Stølen, T.O.; Wisløff, U. Time course of endothelial adaptation after acute and chronic exercise in patients with metabolic syndrome. Journal of strength and conditioning research. Nat. Strength Cond. Assoc. 2011, 25, 2552–2558. [Google Scholar] [CrossRef] [PubMed]
- Ramos, J.S.; Dalleck, L.C.; Tjonna, A.E.; Beetham, K.S.; Coombes, J.S. The impact of high-intensity interval training versus moderate-intensity continuous training on vascular function: A systematic review and meta-analysis. Sports Med. 2015, 45, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Perk, J.; De Backer, G.; Gohlke, H.; Graham, I.; Reiner, Z.; Verschuren, M.; Albus, C.; Benlian, P.; Boysen, G.; Cifkova, R.; et al. European Guidelines on cardiovascular disease prevention in clinical practice (version 2012). The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts). Eur. Heart J. 2012, 33, 1635–1701. [Google Scholar] [PubMed]
- Molmen-Hansen, H.E.; Stolen, T.; Tjonna, A.E.; mot, I.L.; Ekeberg, I.S.; Tyldum, G.A.; Stoylen, A. Aerobic interval training reduces blood pressure and improves myocardial function in hypertensive patients. Eur. J. Prev. Cardiol. 2012, 19, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Ridker, P.M.; Maseri, A. Inflammation and atherosclerosis. Circulation 2002, 105, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Liuzzo, G.; Biasucci, L.M.; Gallimore, J.R.; Grillo, R.L.; Rebuzzi, A.G.; Pepys, M.B.; Maseri, A. The prognostic value of C-reactive protein and serum amyloid a protein in severe unstable angina. N. Eng. J. Med. 1994, 331, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Kasapis, C.; Thompson, P.D. The effects of physical activity on serum C-reactive protein and inflammatory markers: A systematic review. J. Am. Coll. Cardiol. 2005, 45, 1563–1569. [Google Scholar] [CrossRef] [PubMed]
- Clays, E.; Lidegaard, M.; De Bacquer, D.; Van Herck, K.; De Backer, G.; Kittel, F.; Holtermann, A. The Combined Relationship of Occupational and Leisure-Time Physical Activity With All-Cause Mortality Among Men, Accounting for Physical Fitness. Am. J. Epidemiol. 2014, 179, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Palmefors, H.; DuttaRoy, S.; Rundqvist, B.; Börjesson, M. The effect of physical activity or exercise on key biomarkers in atherosclerosis—A systematic review. Atherosclerosis 2014, 235, 150–161. [Google Scholar] [CrossRef] [PubMed]
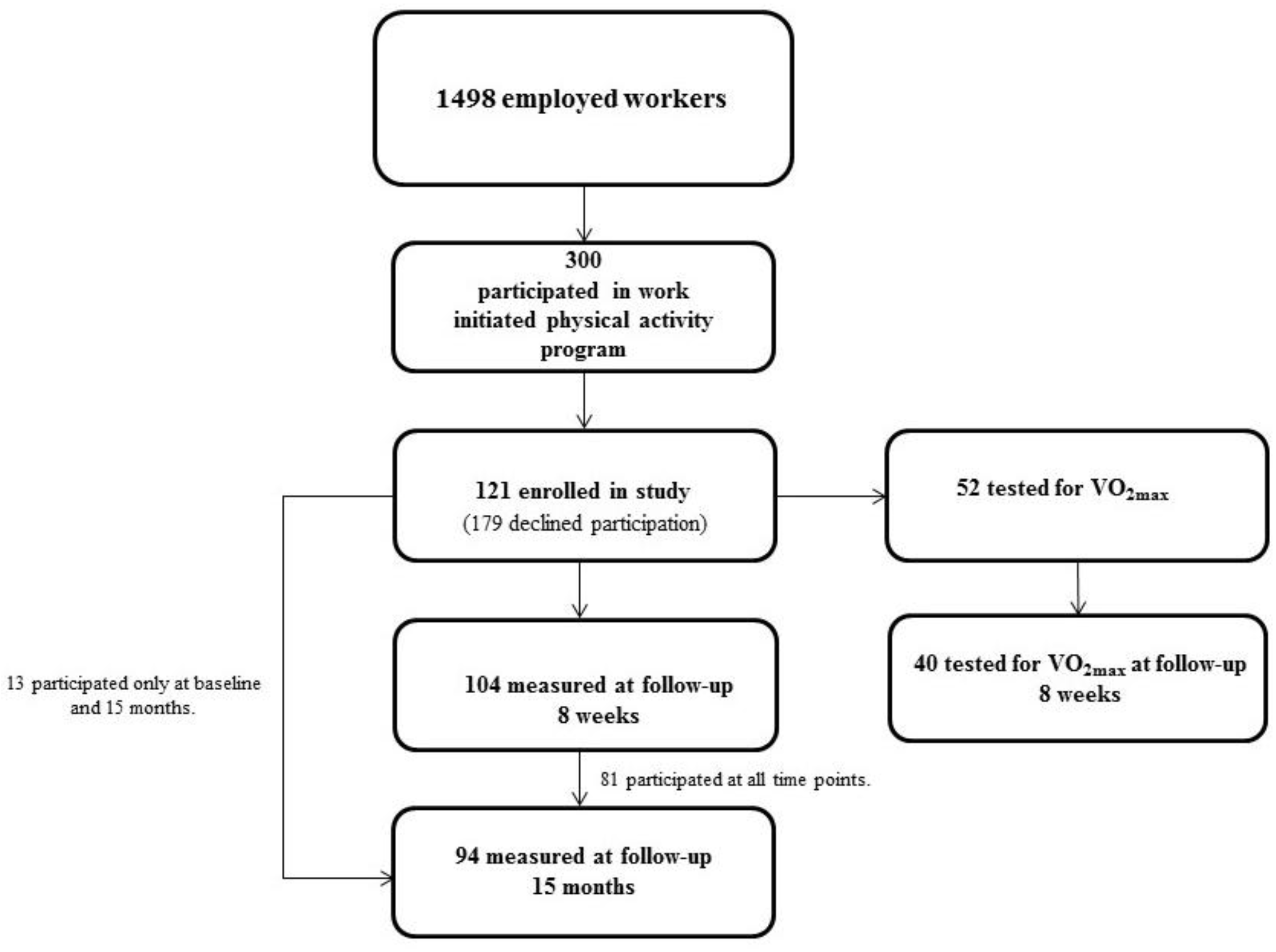
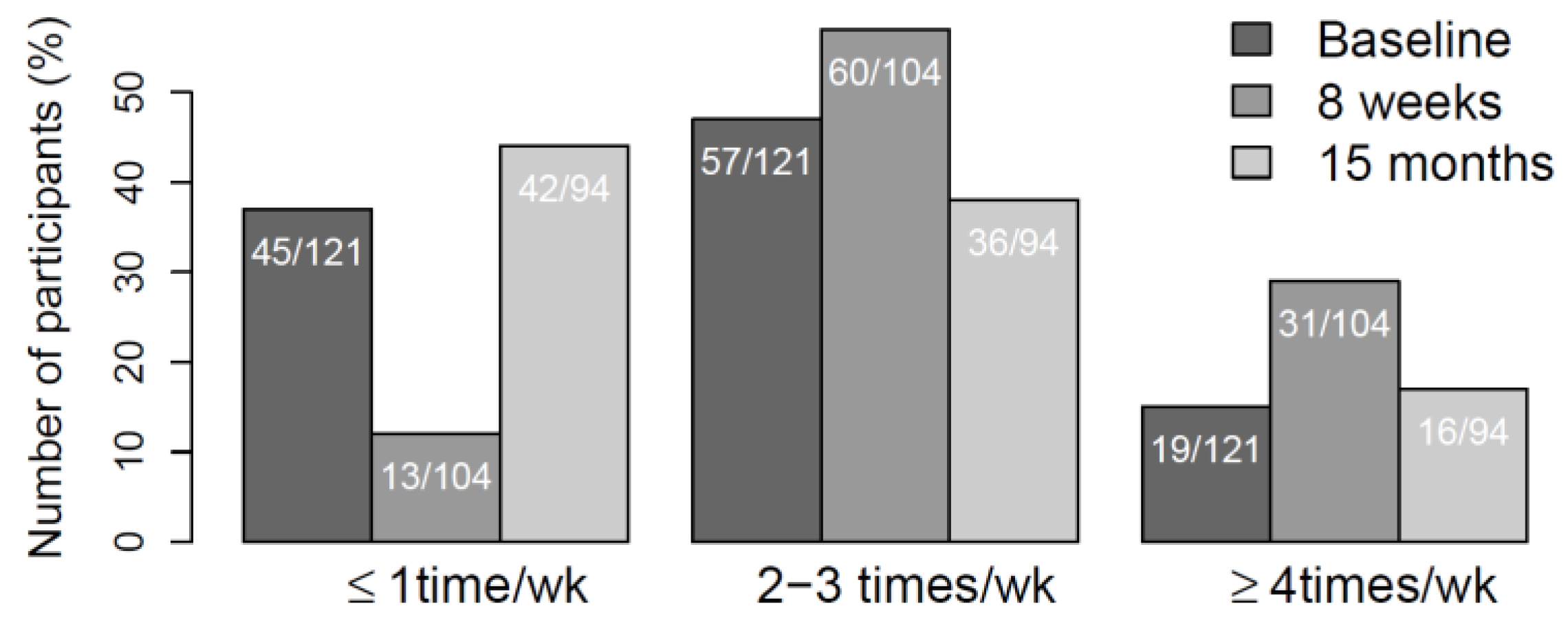
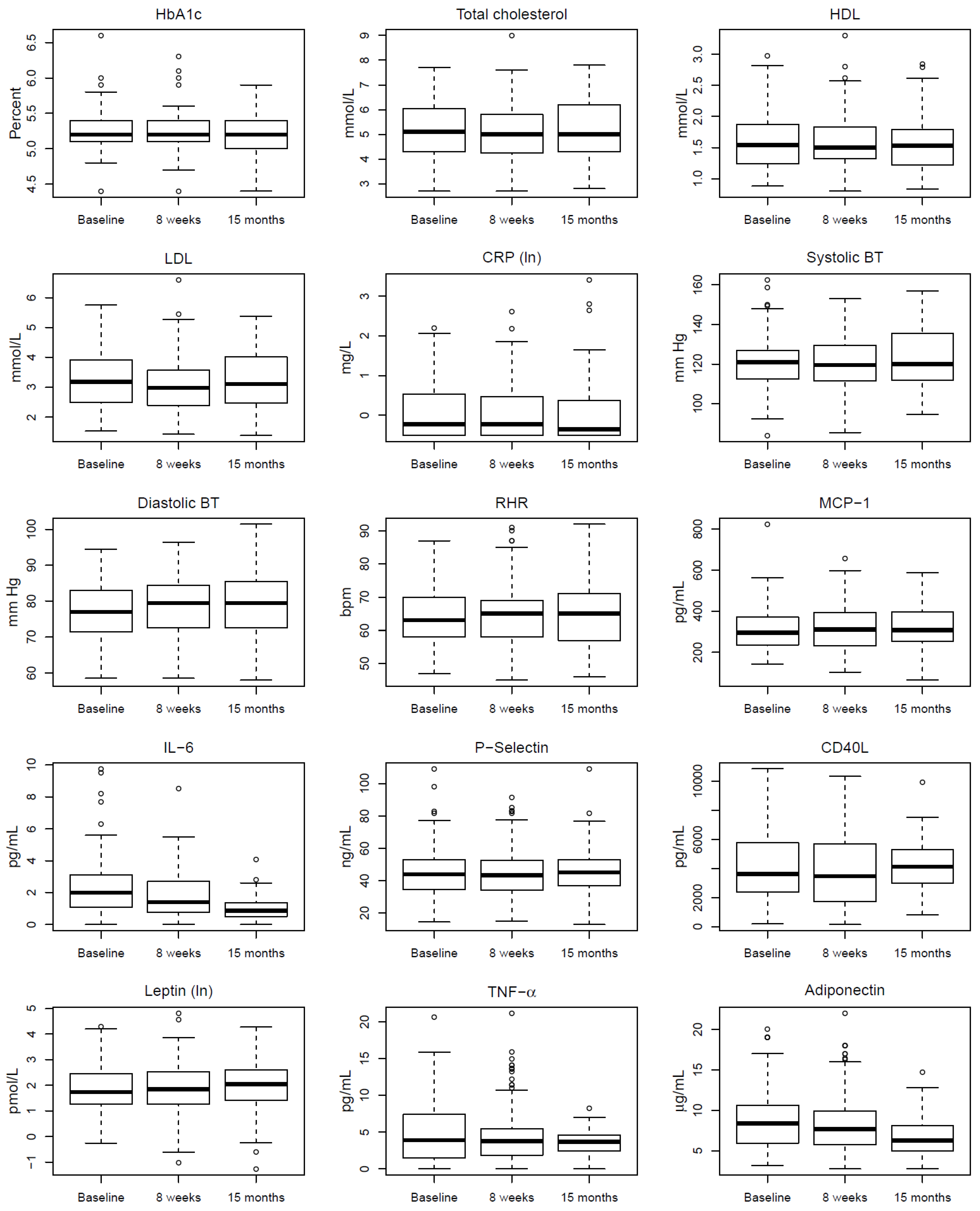
| Outcome | Men Low Education (N = 37) a Mean (SD) | Men High Education (N = 41) e Mean (SD) | Women All (N = 43) b Mean (SD) |
|---|---|---|---|
| Age (years) | 43.0 (12.9) | 40.8 (11.1) | 42.6 (12.5) |
| BMI (kg/m2) | 27.3 (4.8) | 25.4 (3.0) † | 24.4 (3.1) |
| RHR (bpm) Smokers c | 65.7 (9.9) 6 | 61.3 (8.5) † 6 | 67.4 (10.9) * 0 * |
| Systolic BP (mmHg) | 123.3 (13.2) | 122.3 (11.5) | 112 (16) * |
| Diastolic BP (mmHg) | 78.5 (9.5) | 79.2 (87.8) | 74 (8) * |
| CRP (mg/L) | 2.3 (2.3) | 1.5 (1.7) | 1.9 (1.8) |
| Cholesterol (mmol/L) | 5.0 (1.1) | 5.1 (1.1) | 5.4 (1.1) |
| HDL (mmol/L) | 1.3 (0.3) | 1.4 (0.3) † | 1.9 (0.4) * |
| LDL (mmol/L) | 3.2 (0.9) | 3.3 (1.0) | 3.2 (0.9) |
| HbA1c (mmol/L) | 5.4 (0.4) | 5.2 (0.3) † | 5.2 (0.3) |
| VO2max d (mL/kg min) | 35.8 (5.3) | 40.8 (6.7) | 34.1 (9.5) * |
| IL-6 (pg/mL) | 2.9 (2.1) | 2.1 (1.8) | 2.1 (1.4) |
| MCP-1 (pg/mL) | 317.0 (120.7) | 332.6 (126.3) | 288.3 (89.8) * |
| TNF-α (pg/mL) | 7.5 (4.3) | 5.1 (3.6) † | 3.1 (3.1) * |
| P-selectin (ng/mL) | 50.9 (18.7) | 45.9 (16.8) | 41.0 (10.6) * |
| CD40L (pg/mL) | 4864 (2756) | 3336 (2134) † | 4742 (2590) * |
| Leptin (µg/mL) | 8.7 (7.6) | 5.3 (4.4) † | 16.0 (13.6) * |
| Adiponectin (µg/mL) | 6.6 (3.2) | 7.5 (2.6) | 10.5 (3.8) * |
| Females N = 43 | Males N = 78 | All N = 121 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | Unit | Time | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | |||
| Syst. BP | (mmhg) | 15 months vs. BL | −0.81 | −5.88 | 4.27 | 0.752 | 3.92 | 0.42 | 7.42 | 0.029 | 2.27 | −0.59 | 5.13 | 0.119 |
| 15 months vs. 8 weeks | 0.57 | −4.55 | 5.69 | 0.824 | 2.44 | −1.20 | 6.07 | 0.187 | 1.82 | −1.12 | 4.76 | 0.223 | ||
| Diast. BP | (mmhg) | 15 months vs. BL | −0.22 | −3.77 | 3.34 | 0.904 | 2.99 | 0.61 | 5.37 | 0.014 | 1.86 | −0.11 | 3.83 | 0.064 |
| 15 months vs. 8 weeks | −0.56 | −4.15 | 3.03 | 0.756 | 0.59 | −1.89 | 3.06 | 0.640 | 0.19 | −1.83 | 2.21 | 0.851 | ||
| RHR | (bmp) | 15 months vs. BL | 4.03 | 0.49 | 7.58 | 0.026 | 0.17 | −2.64 | 2.99 | 0.903 | 1.18 | −1.07 | 3.43 | 0.303 |
| 15 months vs. 8 weeks | 5.04 | 1.47 | 8.62 | 0.006 | −1.24 | −4.16 | 1.68 | 0.402 | 0.70 | −1.61 | 3.01 | 0.553 | ||
| Females N = 42 | Males N = 77 | All N = 119 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | Unit | Time | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | |||
| Cholest. | (mmol/L) | 15 months vs. BL | −0.04 | −0.31 | 0.22 | 0.741 | 0.11 | −0.08 | 0.31 | 0.256 | 0.05 | −0.10 | 0.21 | 0.500 |
| 15 months vs. 8 weeks | 0.25 | −0.02 | 0.52 | 0.072 | 0.11 | −0.09 | 0.32 | 0.270 | 0.17 | 0.01 | 0.33 | 0.043 | ||
| LDL | (mmol/L) | 15 months vs. BL | −0.05 | −0.24 | 0.15 | 0.645 | 0.10 | −0.06 | 0.27 | 0.226 | 0.05 | −0.08 | 0.18 | 0.440 |
| 15 months vs. 8 weeks | 0.23 | 0.03 | 0.43 | 0.024 | 0.14 | −0.03 | 0.32 | 0.114 | 0.18 | 0.05 | 0.31 | 0.009 | ||
| HDL | (mmol/L) | 15 months vs. BL | 0.01 | −0.09 | 0.11 | 0.856 | −0.04 | −0.09 | 0.01 | 0.151 | −0.02 | −0.07 | 0.02 | 0.330 |
| 15 months vs. 8 weeks | 0.06 | −0.04 | 0.15 | 0.266 | −0.04 | −0.09 | 0.01 | 0.134 | −0.01 | −0.06 | 0.04 | 0.750 | ||
| HbA1c | (mmol/L) | 15 months vs. BL | −0.05 | −0.14 | 0.04 | 0.301 | −0.06 | −0.11 | −0.01 | 0.028 | −0.06 | −0.11 | −0.01 | 0.014 |
| 15 months vs. 8 weeks | −0.02 | −0.12 | 0.07 | 0.617 | −0.05 | −0.10 | 0.01 | 0.080 | −0.04 | −0.09 | 0.01 | 0.088 | ||
| Females N = 42 | Males N = 77 a | All N = 119 b | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | Unit | Time | β | 95% CI | p | β | 95% CI | p | β | 95% CI | p | |||
| logCRP | (mg/L) | 15 months vs. BL | −0.18 | −0.56 | 0.19 | 0.338 | −0.28 | −0.53 | −0.03 | 0.030 | −0.25 | −0.46 | −0.04 | 0.019 |
| 15 months vs. 8 weeks | −0.06 | −0.44 | 0.32 | 0.737 | −0.16 | −0.42 | 0.10 | 0.234 | −0.13 | −0.34 | 0.08 | 0.232 | ||
| IL-6 | (pg/mL) | 15 months vs. BL | 0.00 | −0.51 | 0.52 | 0.994 | −0.62 | −1.1 | −0.13 | 0.013 | −0.39 | −0.75 | −0.04 | 0.032 |
| 15 months vs. 8 weeks | 0.14 | −0.34 | 0.62 | 0.572 | −0.34 | −0.8 | 0.12 | 0.154 | −0.18 | −0.51 | 0.16 | 0.310 | ||
| MCP-1 | (pg/mL) | 15 months vs. BL | 41.36 | 7.10 | 75.61 | 0.020 | 6.82 | −24.40 | 38.04 | 0.669 | 18.63 | −5.23 | 42.50 | 0.127 |
| 15 months vs. 8 weeks | 20.75 | −11.69 | 53.20 | 0.212 | 19.31 | −10.86 | 49.48 | 0.211 | 18.80 | −4.02 | 41.61 | 0.107 | ||
| TNF-α | (pg/mL) | 15 months vs. BL | 0.17 | −0.94 | 1.28 | 0.761 | −0.31 | −1.27 | 0.65 | 0.523 | −0.15 | −0.93 | 0.62 | 0.697 |
| 15 months vs. 8 weeks | −0.06 | −1.11 | 0.99 | 0.907 | 0.01 | −0.91 | 0.93 | 0.983 | −0.06 | −0.80 | 0.68 | 0.877 | ||
| Females N = 42 | Males N = 77 | All N = 119 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Outcome | Unit | Time | β | 95% CI | p | β | 95% CI | p | Β | 95% CI | p | |||
| P-selec. | (ng/mL) | 15 months vs. BL | 1.55 | −2.94 | 6.04 | 0.501 | 1.71 | −2.41 | 5.83 | 0.417 | 1.75 | −1.31 | 4.82 | 0.263 |
| 15 months vs. 8 weeks | 2.73 | −1.53 | 6.98 | 0.211 | 3.30 | −0.67 | 7.28 | 0.105 | 3.06 | 0.13 | 6.00 | 0.041 | ||
| CD40L | (pg/mL) | 15 months vs. BL | 267.82 | −903.72 | 1439.35 | 0.655 | 1149.37 | 316.46 | 1982.28 | 0.007 | 851.70 | 169.57 | 1533.83 | 0.015 |
| 15 months vs. 8 weeks | 803.57 | −299.04 | 1906.19 | 0.155 | 1418.38 | 611.60 | 2225.16 | 0.001 | 1204.99 | 551.66 | 1858.32 | 0.000 | ||
| Leptin ln | (µg/mL) | 15 months vs. BL | 0.10 | −0.17 | 0.37 | 0.482 | 0.08 | −0.11 | 0.28 | 0.408 | 0.09 | −0.08 | 0.25 | 0.311 |
| 15 months vs. 8 weeks | 0.04 | −0.22 | 0.31 | 0.744 | 0.10 | −0.09 | 0.29 | 0.308 | 0.07 | −0.09 | 0.23 | 0.369 | ||
| Adipon. | (µg/mL) | 15 months vs. BL | −0.45 | −1.34 | 0.44 | 0.324 | −0.01 | −0.67 | 0.66 | 0.985 | −0.20 | −0.73 | 0.33 | 0.457 |
| 15 months vs. 8 weeks | −0.49 | −1.33 | 0.35 | 0.252 | −0.11 | −0.75 | 0.53 | 0.727 | −0.28 | −0.78 | 0.23 | 0.283 | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skogstad, M.; Lunde, L.-K.; Ulvestad, B.; Aass, H.C.D.; Clemm, T.; Mamen, A.; Skare, Ø. The Effect of a Leisure Time Physical Activity Intervention Delivered via a Workplace: 15-Month Follow-Up Study. Int. J. Environ. Res. Public Health 2018, 15, 264. https://doi.org/10.3390/ijerph15020264
Skogstad M, Lunde L-K, Ulvestad B, Aass HCD, Clemm T, Mamen A, Skare Ø. The Effect of a Leisure Time Physical Activity Intervention Delivered via a Workplace: 15-Month Follow-Up Study. International Journal of Environmental Research and Public Health. 2018; 15(2):264. https://doi.org/10.3390/ijerph15020264
Chicago/Turabian StyleSkogstad, Marit, Lars-Kristian Lunde, Bente Ulvestad, Hans Christian D. Aass, Thomas Clemm, Asgeir Mamen, and Øivind Skare. 2018. "The Effect of a Leisure Time Physical Activity Intervention Delivered via a Workplace: 15-Month Follow-Up Study" International Journal of Environmental Research and Public Health 15, no. 2: 264. https://doi.org/10.3390/ijerph15020264
APA StyleSkogstad, M., Lunde, L.-K., Ulvestad, B., Aass, H. C. D., Clemm, T., Mamen, A., & Skare, Ø. (2018). The Effect of a Leisure Time Physical Activity Intervention Delivered via a Workplace: 15-Month Follow-Up Study. International Journal of Environmental Research and Public Health, 15(2), 264. https://doi.org/10.3390/ijerph15020264





