Ventilation Positive Pressure Intervention Effect on Indoor Air Quality in a School Building with Moisture Problems
Abstract
1. Introduction
2. Materials and Methods
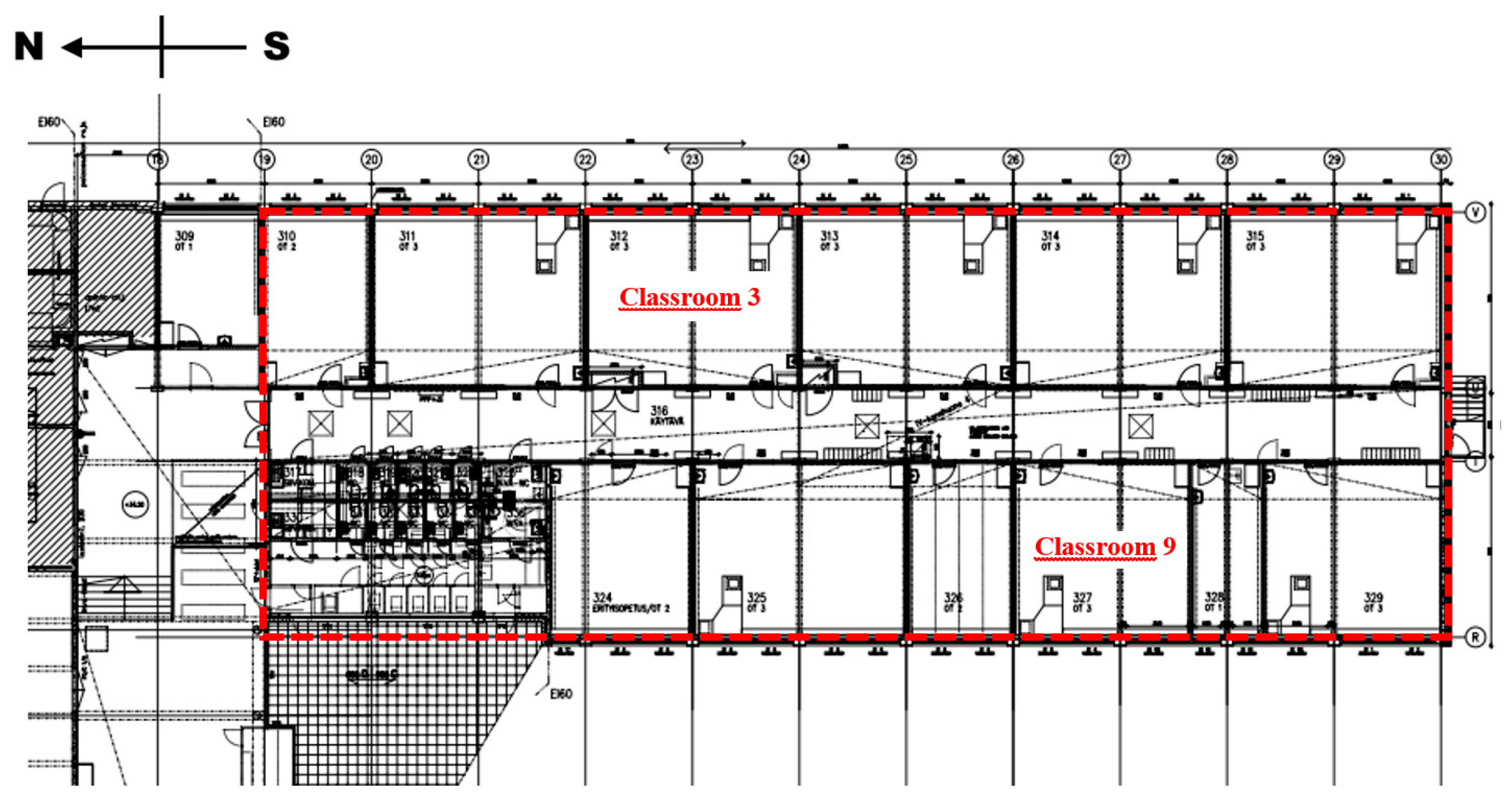
2.1. Building Characteristics
2.2. Previous Investigations
2.3. Set-Up for Ventilation Intervention
2.4. Measurement Methods
2.4.1. Airflow Rate Measurements and Pressure Differences across the Building Envelope
2.4.2. Temperature, Relative Humidity, and CO2 Concentration of Indoor Air
2.4.3. Indoor Air Quality (IAQ) Measurements
2.4.4. Characterization of the Mycobiota in the Indoor Dust
2.4.5. Indoor Air Questionnaire
3. Results and Discussion
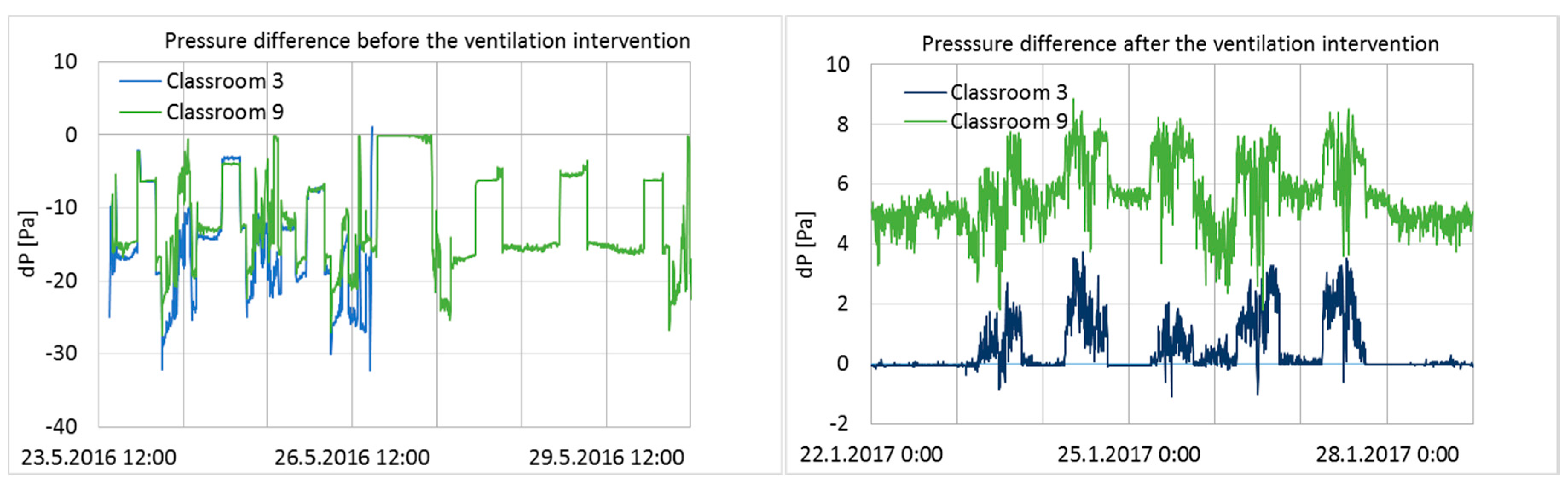
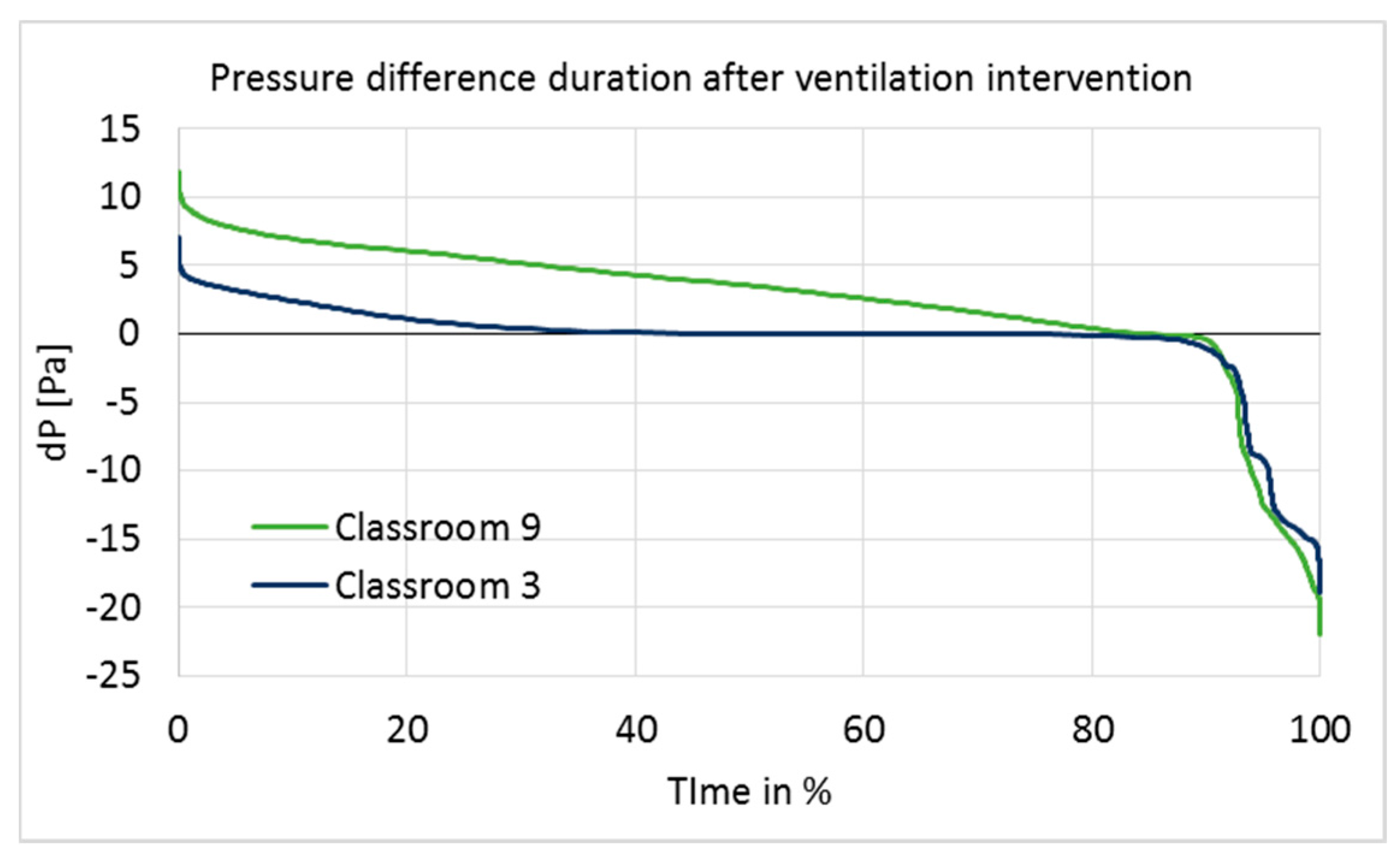
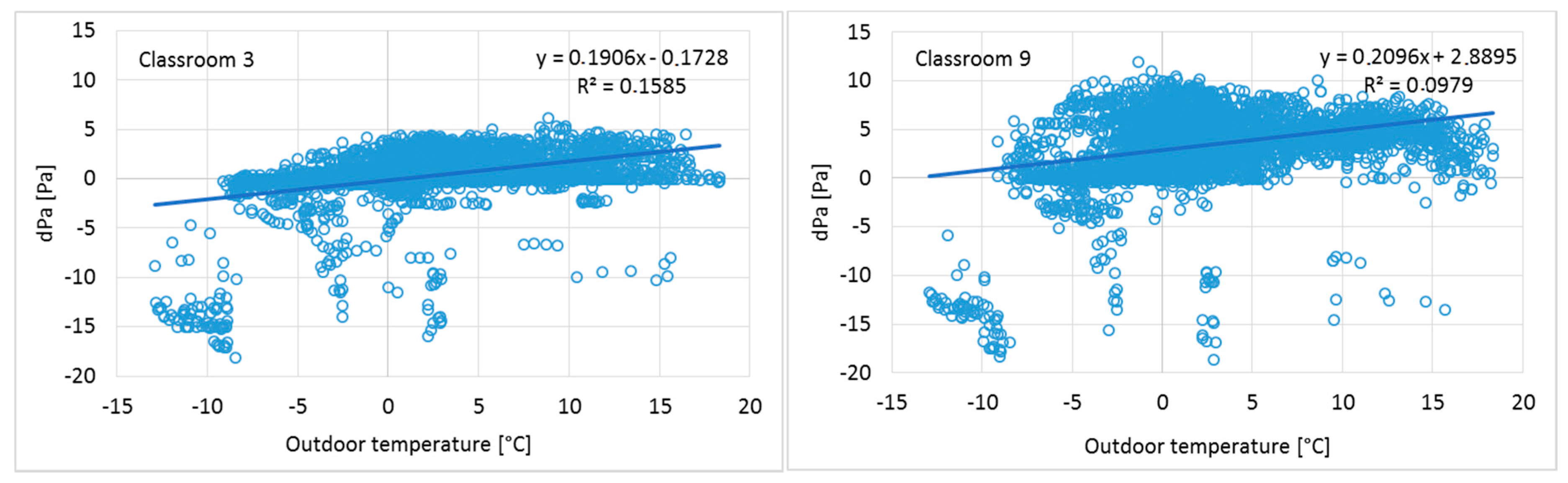
3.1. Airflow Rate Measurements and Pressure Differences across the Building Envelope
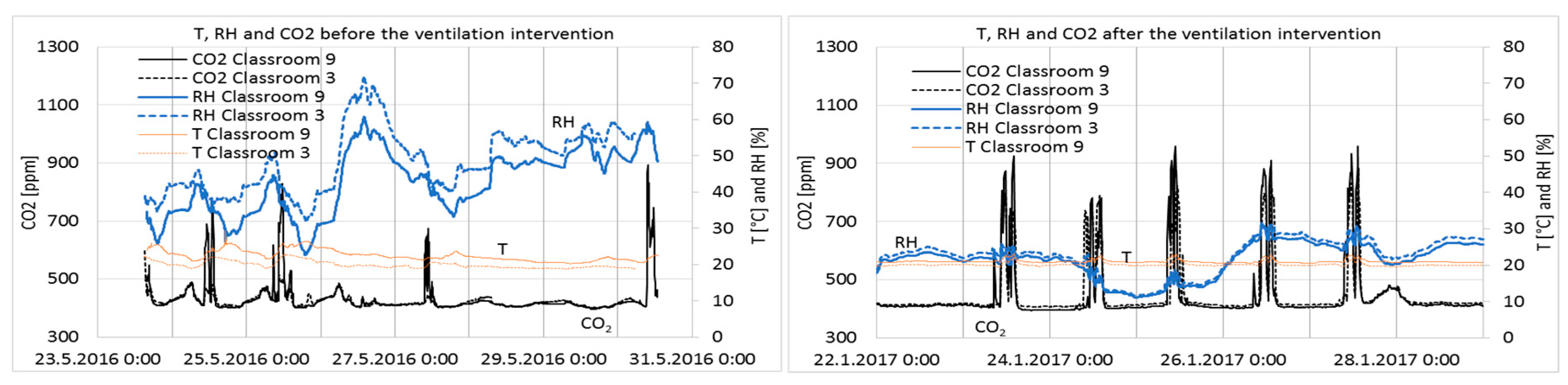
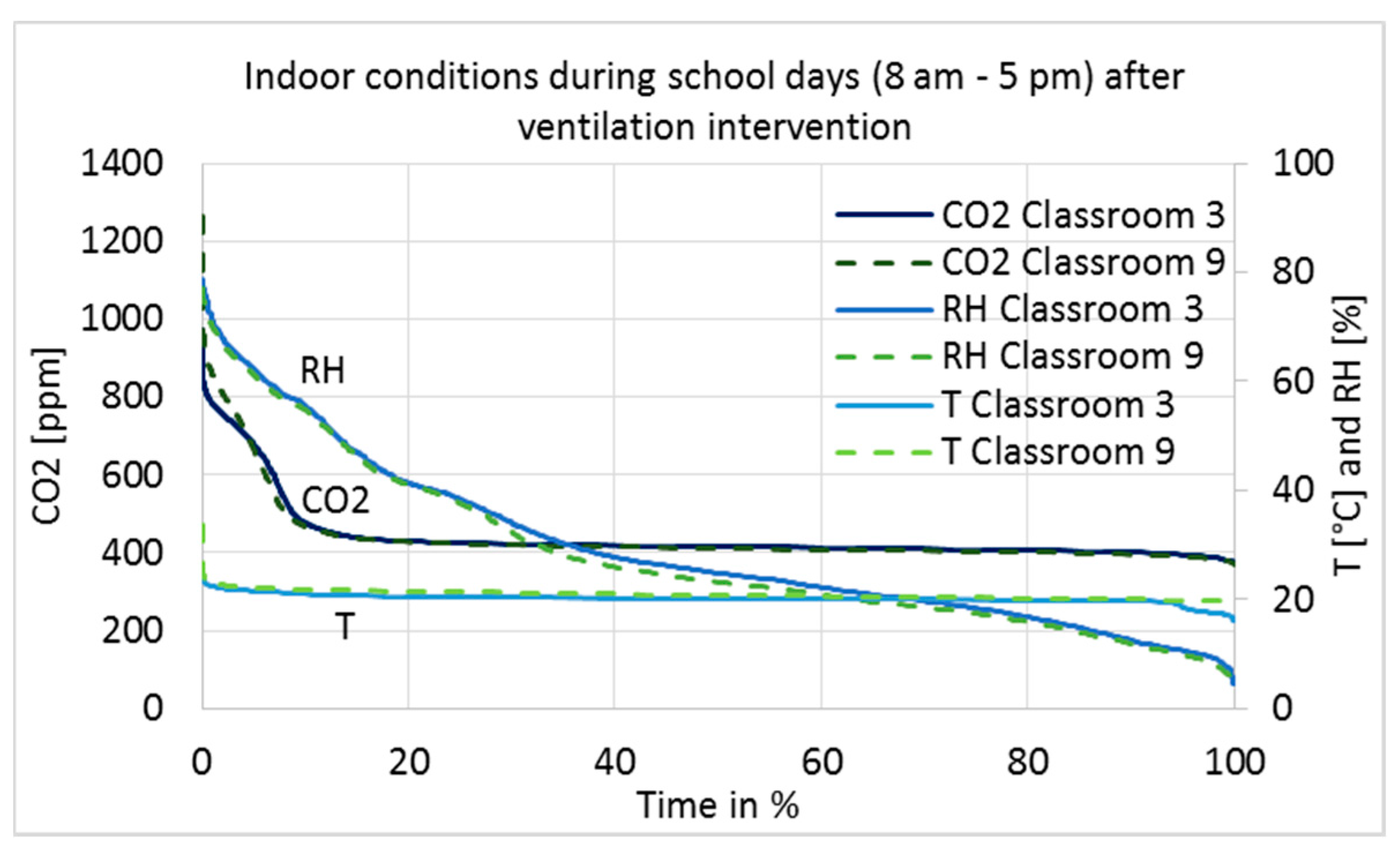
3.2. Temperature, Relative Humidity, and CO2 Concentration of Indoor Air
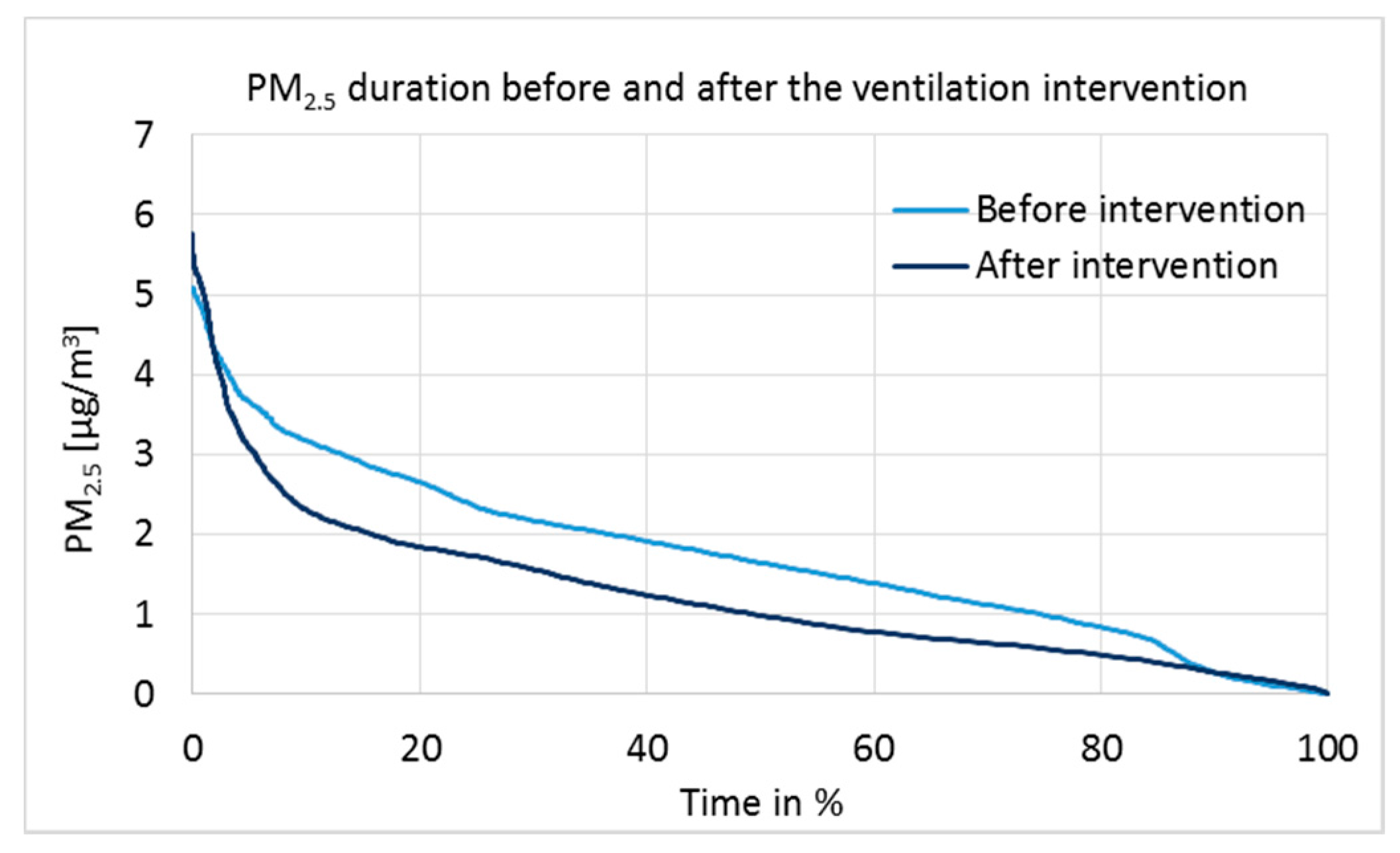
3.3. Indoor Air Quality (IAQ) Measurements
3.4. Characterization of the Mycobiota in the Indoor Dust
3.5. Indoor Air Questionnaire
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| IAQ | Indoor air quality |
| TVOC | Total volatile organic compounds |
| PM2.5 | Fine particulate matter, particle size 2.5 μm |
| RH | Relative humidity |
| VOC | Volatile organic compounds |
| T | Temperature |
| CO2 | Carbon dioxide |
| CO | Carbon monoxide |
| FIOH | Finnish Institute of Occupational Health |
| MEA | Malt extract agar |
| 2E1H | 2-ethyl-1-hexanol |
References
- Haverinen-Shaughnessy, U.; Hyvärinen, A.; Putus, T.; Nevalainen, A. Monitoring success of remediation: seven case studies of moisture and mold damaged buildings. Sci. Total Environ. 2008, 399, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Seppänen, O.A.; Fisk, W.J. Summary of human responses to ventilation. Indoor Air 2004, 14, 102–118. [Google Scholar] [CrossRef] [PubMed]
- Airaksinen, M.; Kurnitski, J.; Pasanen, P.; Seppänen, O. Fungal spore transport through a building structure. Indoor Air 2004, 14, 92–104. [Google Scholar] [CrossRef] [PubMed]
- Airaksinen, M.; Pasanen, P.; Kurnitski, J.; Seppänen, O. Microbial contamination of indoor air due to leakages from crawl space: A field study. Indoor Air 2004, 14, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Meklin, T.; Putus, T.; Pekkanen, J.; Hyvärinen, A.; Hirvonen, M.-R.; Nevalainen, A. Effects of moisture-damage repairs on microbial exposure and symptoms in schoolchildren. Indoor Air 2005, 15, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Wargocki, P.; Sundell, J.; Bischof, W.; Brundrett, G.; Fanger, P.O.; Gyntelberg, F.; Hanssen, S.O.; Harrison, P.; Pickering, A.; Seppänen, O.; et al. Ventilation and health in non-industrial indoor environments: report from a european multidisciplinary scientific consensus meeting (EUROVEN). Indoor Air 2002, 12, 113–128. [Google Scholar] [CrossRef] [PubMed]
- Bayer, C.W.; Grimes, C. The indoor environmental microbiome. Indoor Built Environ. 2015, 24, 1035–1037. [Google Scholar] [CrossRef]
- Cabral, J.P.S. Can we use indoor fungi as bioindicators of indoor air quality? Historical perspectives and open questions. Sci. Total Environ. 2010, 408, 4285–4295. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Wyon, D.P.; Clausen, G.; Fanger, P.O. Impact of indoor air temperature and humidity in an office on perceived air quality, SBS symptoms and performance. Indoor Air 2004, 14, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Wargocki, P.; Wyon, D.P.; Sundell, J.; Clausen, G.; Fanger, P.O. The effects of outdoor air supply rate in an office on perceived air quality, sick building syndrome (SBS) symptoms and productivity. Indoor Air 2000, 10, 222–236. [Google Scholar] [CrossRef] [PubMed]
- Carrer, P.; Wargocki, P.; Fanetti, A.; Bischof, W.; Fernandes, E.D.O.; Hartmann, T.; Kephalopoulos, S.; Palkonen, S.; Seppänen, O. What does the scientific literature tell us about the ventilation–health relationship in public and residential buildings? Build. Environ. 2015, 94, 273–286. [Google Scholar] [CrossRef]
- Kalamees, T. Measured and simulated air pressure conditions in finnish residential buildings. Build. Serv. Eng. Res. Technol. 2010, 31, 177. [Google Scholar] [CrossRef]
- Leivo, V.; Kiviste, M.; Aaltonen, A.; Turunen, M.; Haverinen-Shaughnessy, U. Air pressure difference between indoor and outdoor or staircase in multi-family buildings with exhaust ventilation system in Finland. Energy Procedia 2015, 78, 1218–1223. [Google Scholar] [CrossRef]
- Finnish Ministry of the Environment, Department of Built Environment. D2 National Building Code of Finland. Indoor Climate and Ventilation of the Buildings. Regulations and Guidelines; Finnish Ministry of the Environment, Department of Built Environment: Helsinki, Finland, 2012.
- Sweco Finland Oy. An Indoor Climate and Structural Investigation, Investigation Report); Sweco Asiantuntijapalvelut Oy (Sweco Finland Oy): Helsinki, Finland, 2016. [Google Scholar]
- The Finnish Institute of Occupational Health. Summary of the Levels of Indoor Air Pollutants and Conditions that can Refer to Indoor Environment Problems; The Finnish Institute of Occupational Health: Helsinki, Finland, 2017. [Google Scholar]
- ISO 16000-6 Standard. Indoor Air—Part 6: Determination of Volatile Organic Compounds in Indoor and Test Chamber Air by Active Sampling on Tenax TA Sorbent, Thermal Desorption and Gas Chromatography Using MS or MS-FID; International Organization for Standardization (ISO): Geneva, Switzerland, 2011. [Google Scholar]
- Andersson, M.A.; Jääskeläinen, E.L.; Shaheen, R.; Pirhonen, T.; Wijnands, L.M.; Salkinoja-Salonen, M.S. Sperm bioassay for rapid detection of cereulide-producing bacillus cereus in food and related environments. Int. J. Food Microbiol. 2004, 94, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Bencsik, O.; Papp, T.; Berta, M.; Zana, A.; Forgó, P.; Dombi, G.; Andersson, M.A.; Salkinoja-Salonen, M.; Vágvölgyi, C.; Szekeres, A. Ophiobolin a from bipolaris oryzae perturbs motility and membrane integrities of porcine sperm and induces cell death on mammalian somatic cell lines. Toxins 2014, 6, 2857–2871. [Google Scholar] [CrossRef] [PubMed]
- Samson, R.A.; Hoekstra, E.S.; Frisvad, J.C. Introduction to Food-and Airborne Fungi; Centraalbureau voor Schimmelcultures (CBS): Utrecht, The Netherlands, 2004. [Google Scholar]
- Andersson, M.A.; Mikkola, R.; Raulio, M.; Kredics, L.; Maijala, P.; Salkinoja-Salonen, M.S. Acrebol, a novel toxic peptaibol produced by an acremonium exuviarum indoor isolate. J. Appl. Microbiol. 2009, 106, 909–923. [Google Scholar] [CrossRef] [PubMed]
- Andersson, K. Epidemiological approach to indoor air problems. Indoor Air 1998, 8, 32. [Google Scholar] [CrossRef]
- Mattila, M.; Vornanen-Winqvist, C.; Jerkku, I.; Kurnitski, J. The effect of positive pressure and air tighntess on the moistural behavior of buildings. In The Building Physics Conference 24 October 2017–26 October 2017, Conference Publication 4; Tampere University of Technology: Tampere, Finland, 2017; pp. 91–93. [Google Scholar]
- Finnish Society of Indoor Air Quality and Climate (FiSIAQ). Classification of Indoor Air Environment 2008. Target Values, Design Guidance and Product Requirements; FiSIAQ: Espoo, Finland, 2008. [Google Scholar]
- Azuma, K. Evaluating prevalence and risk factors of building-related symptoms among office workers: Seasonal characteristics of symptoms and psychosocial and physical environmental factors. Environ. Health 2017, 22, 38. [Google Scholar] [CrossRef] [PubMed]
- Haghighat, F.; De Bellis, L. Material emission rates: Literature review, and the impact of indoor air temperature and relative humidity. Build. Environ. 1998, 33, 261–277. [Google Scholar] [CrossRef]
- Wolkoff, P. Impact of air velocity, temperature, humidity, and air on long-term voc emissions from building products. Atmos. Environ. 1998, 32, 2659–2668. [Google Scholar] [CrossRef]
- Fang, L. Impact of temperature and humidity on the perception of indoor air quality. Indoor Air 1998, 8, 80. [Google Scholar] [CrossRef]
- Ministry of Social Affairs and Health. Decree of the Ministry of Social Affairs and Health on Health-Related Conditions of Housing and Other Residential Buildings and Qualification Requirements for Third-Party Experts (545/2015); Ministry of Social Affairs and Health: Helsinki, Finland, 2015.
- Reijula, K.; Sundman Digert, C. Assessment of indoor air problems at work with a questionnaire. Occup. Environ. Med. 2004, 61, 33–38. [Google Scholar] [PubMed]
- Lahtinen, M.; Sundman Digert, C.; Reijula, K. Psychosocial work environment and indoor air problems: A questionnaire as a means of problem diagnosis. Occup. Environ. Med. 2004, 61, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Hellgren, U.M. Complaints and symptoms among hospital staff in relation to indoor air and the condition and need for repairs in hospital buildings. Scand. J. Work Environ. Health 2008, 34, 58. [Google Scholar]
- Jaakkola, J.J.; Heinoneon, O.P.; Seppänen, O. Mechanical ventilation in office buildings and the sick building syndrome: An experimental and epidemiological study. Indoor Air 1991, 1, 111–121. [Google Scholar] [CrossRef]
- Paulussen, C.; Hallsworth, J.E.; Álvarez-Pérez, S.; Nierman, W.C.; Hamill, P.G.; Blain, D.; Rediers, H.; Lievens, B. Ecology of Aspergillosis: Insights into the pathogenic potency of Aspergillus fumigatus and some other Aspergillus species. Microb. Biotechnol. 2017, 10, 296–322. [Google Scholar] [CrossRef] [PubMed]


| Room | Place | Date | Fungi (cfu/g) | Bacteria (cfu/g) | Actinobacteria (cfu/g) |
|---|---|---|---|---|---|
| Classroom 3 | Outer wall, window frame fastening wood | April 2016 | 170,000 Aureobasidium sp., Mucor sp., Penicillium sp., yeast, sterile | 1,400,000 | <100 ** |
| Classroom 3 | Outer wall, mineral wool in the plinth cut | April 2016 | 8000 Exophiala * sp. | 100 | 29,000 * |
| Limit value [16] | 10,000 | 100,000 | 3000 |
| Measured Factor | Device | Accuracy | Place | Time |
|---|---|---|---|---|
| Supply/extract airflow rate | Swemaflow 125D | Air flow: ±3.5%, not better than 0.4 L/s. With backpressure: ±10%, lowest 1 L/s. T ±0.5 °C. Barometer ±3.5 hPa. | Corridor, toilets | Instant |
| SWEMA 3000md | ±0.3% read value, lowest ±0.3 Pa | Classrooms | 60 s average (used to calculate air flow rate) | |
| Pressure difference across the envelope | KIMO CP101, logger Grant 1000 | 1.5% of reading ±3 Pa | Classrooms 3 and 9 | Continuous |
| Temperature (T) | Rotronic CL11 | ±0.3 °C | Classrooms 3 and 9 | Continuous |
| Relative humidity (RH) | Rotronic CL11 | ±3% (10 ... 95%) | Classrooms 3 and 9 | Continuous |
| Carbon dioxide (CO2) | Rotronic CL11 | ±(30 ppm + 5% of reading) | Classrooms 3 and 9 | Continuous |
| T and RH | ThermaData | ±0.5 °C (−10 … 85 °C) | Outdoors | Continuous |
| Formaldehyde | FM-801 | ±10 ppb at 40, 80, 160 ppb | Classroom 3 | Continuous |
| Carbon monoxide (CO) | TG-501 probe | <4%/year | Classroom 3 | Continuous |
| Particulate matter 2.5 μm (PM2.5) | MIE pDR-1500 | ±5% | Classroom 3 | Continuous |
| Volatile organic compounds (VOC) | Tenax TA, TD-GC-MS | ±20% (average) | Classrooms 3 and 9, corridor | 40 min |
| Mycobiota of settled and filter dust | Extract air filter, Classrooms 3 and 9, outdoors | Cultivated for 4 weeks | ||
| Perceived indoor air quality | Örebro (MM40)—questionnaire (Finnish Institute of Occupational Health (FIOH)) | The occupants of the whole building | 2 weeks’ response time | |
| Air Flow Rates (L/s) | Designed | Measured before Balancing 30 May 2016 | Measured after Balancing 22 July 2016 | Measured after Overpressure 2 September 2016 | |||
|---|---|---|---|---|---|---|---|
| Room | Supply/Extract | Supply | Extract | Supply | Extract | Supply | Extract |
| 1 | 135/135 | 146 | 189 | 134 | 135 | 139 | 125 |
| 2 | 270/270 | 313 | 359 | 273 | 273 | 287 | 255 |
| 3 | 270/270 | 273 | 348 | 268 | 265 | 278 | 247 |
| 4 | 270/270 | 301 | 368 | 271 | 271 | 291 | 305 |
| 5 | 270/270 | 283 | 341 | 263 | 264 | 273 | 269 |
| 6 | 270/270 | 259 | 298 | 271 | 271 | 276 | 259 |
| 7 | 220/220 | 141 | 287 | 204 | 202 | 203 | 188 |
| 8 | 80/80 | 126 | 118 | 65 | 62 | 65 | 65 |
| 9 | 215/215 | 215 | 262 | 213 | 216 | 219 | 214 |
| 10 | 135/135 | 133 | 149 | 136 | 134 | 143 | 139 |
| 11 | 270/270 | 289 | 279 | 265 | 264 | 284 | 277 |
| 12 | 180/180 | 175 | 232 | 175 | 175 | 177 | 167 |
| Classroom total | 2585/2585 | 2655 | 3230 | 2538 | 2532 | 2636 | 2510 |
| Cleaning storage | -/30 | 13 | 30 | 31 | |||
| Toilet 1 | -/30 | 10 | 30 | 32 | |||
| Toilet 2 | -/30 | 11 | 30 | 30 | |||
| Toilet 3 | -/30 | 13 | 30 | 36 | |||
| Toilet 4 | -/30 | 15 | 30 | 34 | |||
| Toilet 5 | -/30 | 17 | 30 | 30 | |||
| Toilet 6 | -/30 | 17 | 30 | 30 | |||
| Corridor | 320/110 | 330 | 90 | 329 | 114 | 303 | 79 |
| TOTAL | 2905/2905 | 2984 | 3415 | 2867 | 2856 | 2938 | 2812 |
| Classroom 3 | Classroom 9 | |||||
|---|---|---|---|---|---|---|
| RH (%) | T (°C) | CO2 (ppm) | RH (%) | T (°C) | CO2 (ppm) | |
| Average | 31 | 21 | 518 | 30 | 21 | 520 |
| Min | 5 | 17 | 391 | 4 | 19 | 382 |
| Max | 75 | 24 | 972 | 77 | 34 | 1264 |
| Chemical Compounds of Indoor Air | Classroom 3 | Classroom 9 | Corridor | Limit Value [29] | |||
|---|---|---|---|---|---|---|---|
| (µg/m3) | a | b | a | b | a | b | |
| TVOC | 31 | 19 | 42 | 19 | 34 | 27 | 400 |
| Acetic acid | 2 | 1 | 1 | 2 | 50 | ||
| Acetic acid, butyl ester | 2 | 50 | |||||
| Octamethylcyclotetrasiloxane | 4 | 2 | 4 | 50 | |||
| Decamethylcyclopentasiloxane | 4 | 1 | 2 | 3 | 50 | ||
| 2-ethyl-1-hexanol (2E1H) | 2 | 1 | 3 | 3 | 10 | ||
| Phenoxyethanol | 2 | 2 | 50 | ||||
| 1-Butanol | 1 | 50 | |||||
| Nonanal | 3 | 1 | 3 | 1 | 3 | 2 | 50 |
| Decanal | 2 | 1 | 2 | 1 | 2 | 2 | 50 |
| Acetone | 1 | 50 | |||||
| Formaldehyde | * | * | - | - | - | - | 50/100 |
| Carbon monoxide (CO) | - | 300–1300 | - | - | - | - | 7000 |
| PM2.5 | Before | After |
|---|---|---|
| Measurement points | 9572 | 12,569 |
| Mean | 1.80 | 1.27 |
| Standard deviation | 1.06 | 0.97 |
| Min | 0.10 | 0.10 |
| Max | 5.90 | 5.90 |
| HAMBI Code | Growth at 37 °C | Toxicity | Colony Color | Size of Conidia/Spores | Morphology in Light Microscope | ||
|---|---|---|---|---|---|---|---|
| BMSI | ICP | MEA | (µm) | ||||
| 1. Aspergillus section Flavi | 834 | + | − | − | green | 5–8 |  |
| 2 strains | + | − | − | green | 5–8 | ||
| 2. Aspergillus section Nigri | 379 | + | − | + | black | 3.5–5 |  |
| 10 strains | + | − | + | black | 3.5–5 | ||
| 3. Asp. versicolor | 3670 | − | − | + | yellow | 2–3 | 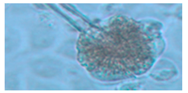 |
| 2 strains | − | − | + | yellow | 2–3 | ||
| 4. Asp. westerdijkiae | 3333 | − | + | + | yellow | 2.5–3 |  |
| 5 strains | − | + | + | yellow | 2.5–3 | ||
| 5. Curvularia sp. | + | + | black-grey | 15 × 5 | 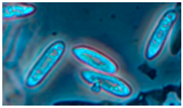 | ||
| 10 strains | + | + | grey | Conidio-phore 15 × 5 | |||
| 6. Paecilomyces variotii | 3342 | + | + | − | yellow | 3–5 | 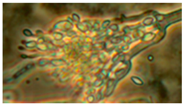 |
| 2 strains | + | + | − | yellow | 3–5 | ||
| 7. Penicillium expansum | 3610 | − | + | + | grey | 3 |  |
| 1 strain | − | + | + | grey | 3 | ||
| 8. Penicillium chrysogenum P. commune | 3631 | − | − | + | green | 3–4 |  |
| 10 strains | − | − | + | green | 3–4 | ||
| 9. Penicillium sp. | − | − | − | yellow | 3 | 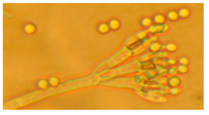 | |
| 10 strains | − | − | − | yellow | 3 | ||
| 10. Trichoderma longibrachiatum | 3643 | + | + | + | green | 1.5 × 3 | 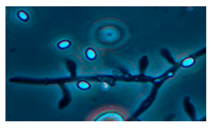 |
| 1 strain | + | + | + | green | 1.5 × 3 | ||
| 11. Trichoderma atroviride T. trixiae | 3369 | − | + | + | green | 3 | 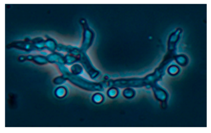 |
| 6 strains | − | + | + | green | 3 | ||
| 12. Rizopus sp. | − | − | − | black | Spores 7–10 |  | |
| 10 strains | − | − | − | black | Spores 7–10 | ||
| Microbial Genera by Sampling Month | Toxic Colonies (%) | Number of Plates Containing Colony Morphotype/All Plates | ||
|---|---|---|---|---|
| School samples | Settled dust | Extract air filter | Settled dust + Extract air filter | |
| Before ventilation intervention | 23 May 2016 | 28 August 2016 | 80 | |
| Rhizopus sp. | Rhizopus sp. | 3/10 | ||
| T. atroviride * | T. atroviride * | 3/10 | ||
| Penicillium chrysogenum * P.commune * | 7/10 | |||
| Aspergillus niger *,1 | 7/10 | |||
| Aspergillus flavus 1 | 2/10 | |||
| (Paecilomyces sp.) *,1 | 1/10 | |||
| Asp. Versicolor * | 2/10 | |||
| After ventilation intervention | 2 September 2016 | 6 February 2017 | 40 | |
| Rhizopus sp. | Rhizopus sp. | 6/12 | ||
| Penicillium sp. | Penicillium sp. | 10/12 | ||
| Asp. Westerdijkiae * | 3/12 | |||
| (Penicillium expansum) * | 1/12 | |||
| (T. trixiae *) | 1/12 | |||
| (T. longibrachiatum) *,1 | 1/12 | |||
| Reference samples (outdoors) | Fall out plate | Settled dust | ||
| Sampling date | 28 August 2016 | 28 August 2016 | ||
| School yard | Curvularia sp.* | Curvularia sp.* | 90 | 4/6 |
| Barn dust | Penicillium sp. | Penicillium sp. | <10 | 6/6 |
| Comparison Values | Studied Part | p-Value | Other Parts | p-Value | |||
|---|---|---|---|---|---|---|---|
| [30,31,32] | 2016 | 2017 | 2016 | 2017 | |||
| Background information | |||||||
| Number of answers | 10 | 8 | 28 | 29 | |||
| Answer (%) | 71 | 58 | 62 | 65 | 72 | ||
| Females (%) | 21 | 60 | 75 | 75 | 86 | ||
| Daily smokers (%) | 10 | 0 | 0 | 0 | |||
| Average age (years) | 46 | 45 | 44 | 46 | |||
| Average employment in this work place (years) | 8 | 6 | 10 | 10 | |||
| Work environment (%) | |||||||
| Draught | 22 | 10 | 38 | 0.275 * | 7 | 21 | 0.253 * |
| Room temperature too high | 17 | 20 | 0 | 0.477 * | 7 | 7 | 1.000 * |
| Varying temperature | 16 | 20 | 57 | 0.162 * | 11 | 22 | 0.295 * |
| Room temperature too low | 13 | 30 | 25 | 1.000 * | 15 | 25 | 0.345 |
| Stuffy air | 34 | 30 | 38 | 1.000 * | 46 | 69 | 0.085 |
| Dry air | 35 | 40 | 50 | 1.000 * | 22 | 43 | 0.103 |
| Insufficient ventilation | 32 | 30 | 63 | 0.342 * | 43 | 55 | 0.352 |
| Smell of mold | 9 | 20 | 25 | 1.000 * | 14 | 21 | 0.730 * |
| Unpleasant odor | 17 | 40 | 25 | 0.638 * | 26 | 28 | 0.889 |
| Environmental tobacco smoke | 4 | 0 | 0 | - | 0 | 0 | - |
| Noise | 17 | 20 | 25 | 1.000 * | 39 | 45 | 0.672 |
| Dim light or reflections | 14 | 10 | 0 | 1.000 * | 7 | 10 | 1.000 * |
| Dust or dirt | 25 | 40 | 38 | 1.000 * | 32 | 43 | 0.408 |
| Work regarded as interesting and stimulating (%) | |||||||
| Often | 75 | 100 | 100 | 89 | 86 | ||
| Sometimes | 20 | 0 | 0 | 11 | 14 | ||
| Seldom or never | 4 | 0 | 0 | - | 0 | 0 | 1.000 * |
| Too much work to do (%) | |||||||
| Often | 20 | 10 | 0 | 14 | 21 | ||
| Sometimes | 59 | 60 | 75 | 61 | 72 | ||
| Seldom or never | 21 | 30 | 25 | 1.000 * | 25 | 7 | 0.185 * |
| Opportunity to influence work conditions (%) | |||||||
| Often | 35 | 60 | 50 | 46 | 38 | ||
| Sometimes | 44 | 30 | 50 | 46 | 55 | ||
| Seldom or never | 21 | 10 | 0 | 0.798 * | 7 | 7 | 0.839 * |
| Fellow workers help with problems in the work (%) | |||||||
| Often | 72 | 80 | 100 | 82 | 83 | ||
| Sometimes | 22 | 20 | 0 | 14 | 17 | ||
| Seldom or never | 6 | 0 | 0 | 0.477 * | 4 | 0 | 1.000 * |
| Allergic diseases (%) | |||||||
| Asthma | 8 | 10 | 13 | 1.000 * | 18 | 7 | 0.253 * |
| Hay fever | 38 | 40 | 75 | 0.188 * | 46 | 45 | 0.903 |
| Atopic eczema | 28 | 40 | 38 | 1.000 * | 14 | 17 | 1.000 * |
| Stress (%) | |||||||
| Quite or very much | 10 | 20 | 0 | 18 | 28 | ||
| Some | 28 | 20 | 63 | 29 | 41 | ||
| No/only little | 63 | 60 | 38 | 0.214 * | 54 | 31 | 0.226 |
| Symptoms (%) | |||||||
| Fatigue | 16 | 10 | 13 | 1.000 * | 14 | 34 | 0.077 |
| Heavy-feeling head | 9 | 0 | 25 | 0.183 * | 11 | 17 | 0.706 * |
| Headache | 7 | 0 | 13 | 0.444 * | 11 | 18 | 0.705 * |
| Concentrating difficulties | 3 | 0 | 13 | 0.444 * | 4 | 14 | 0.352 * |
| Eye irritation | 17 | 20 | 13 | 1.000 * | 37 | 41 | 0.740 * |
| Irritated, stuffy or running nose | 20 | 20 | 25 | 1.000 * | 39 | 62 | 0.085 |
| Hoarse/dry throat | 14 | 10 | 50 | 0.118 * | 36 | 45 | 0.483 |
| Cough | 5 | 20 | 50 | 0.321 * | 11 | 17 | 0.706 |
| Cough disturbing sleep | 1 | 10 | 13 | 1.000 * | 0 | 0 | - |
| Dry of flushed facial skin | 11 | 10 | 13 | 1.000 * | 7 | 29 | 0.036 |
| Hands: dry, itching, red skin | 15 | 10 | 0 | 1.000 * | 4 | 14 | 0.352 * |
| Shortness of breath | 3 | 10 | 13 | 1.000 * | 0 | 7 | 0.491 * |
| Wheezing | 1 | 10 | 13 | 1.000 * | 0 | 0 | - |
| Fever or chills | 2 | 0 | 0 | - | 4 | 7 | 1.000 * |
| Joint pain | 3 | 10 | 0 | 1.000 * | 4 | 0 | 0.491 * |
| Muscular pain | 4 | 0 | 0 | - | 0 | 0 | - |
| Other | 20 | 25 | 1.000 * | 4 | 3 | 1.000 * | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vornanen-Winqvist, C.; Järvi, K.; Toomla, S.; Ahmed, K.; Andersson, M.A.; Mikkola, R.; Marik, T.; Kredics, L.; Salonen, H.; Kurnitski, J. Ventilation Positive Pressure Intervention Effect on Indoor Air Quality in a School Building with Moisture Problems. Int. J. Environ. Res. Public Health 2018, 15, 230. https://doi.org/10.3390/ijerph15020230
Vornanen-Winqvist C, Järvi K, Toomla S, Ahmed K, Andersson MA, Mikkola R, Marik T, Kredics L, Salonen H, Kurnitski J. Ventilation Positive Pressure Intervention Effect on Indoor Air Quality in a School Building with Moisture Problems. International Journal of Environmental Research and Public Health. 2018; 15(2):230. https://doi.org/10.3390/ijerph15020230
Chicago/Turabian StyleVornanen-Winqvist, Camilla, Kati Järvi, Sander Toomla, Kaiser Ahmed, Maria A. Andersson, Raimo Mikkola, Tamás Marik, László Kredics, Heidi Salonen, and Jarek Kurnitski. 2018. "Ventilation Positive Pressure Intervention Effect on Indoor Air Quality in a School Building with Moisture Problems" International Journal of Environmental Research and Public Health 15, no. 2: 230. https://doi.org/10.3390/ijerph15020230
APA StyleVornanen-Winqvist, C., Järvi, K., Toomla, S., Ahmed, K., Andersson, M. A., Mikkola, R., Marik, T., Kredics, L., Salonen, H., & Kurnitski, J. (2018). Ventilation Positive Pressure Intervention Effect on Indoor Air Quality in a School Building with Moisture Problems. International Journal of Environmental Research and Public Health, 15(2), 230. https://doi.org/10.3390/ijerph15020230






