Estimated Residential Exposure to Agricultural Chemicals and Premature Mortality by Parkinson’s Disease in Washington State
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources
2.2. Measures
2.2.1. Outcome Variable
2.2.2. Exposure Variables
2.3. Demographic Variables
2.4. Statistical Analysis
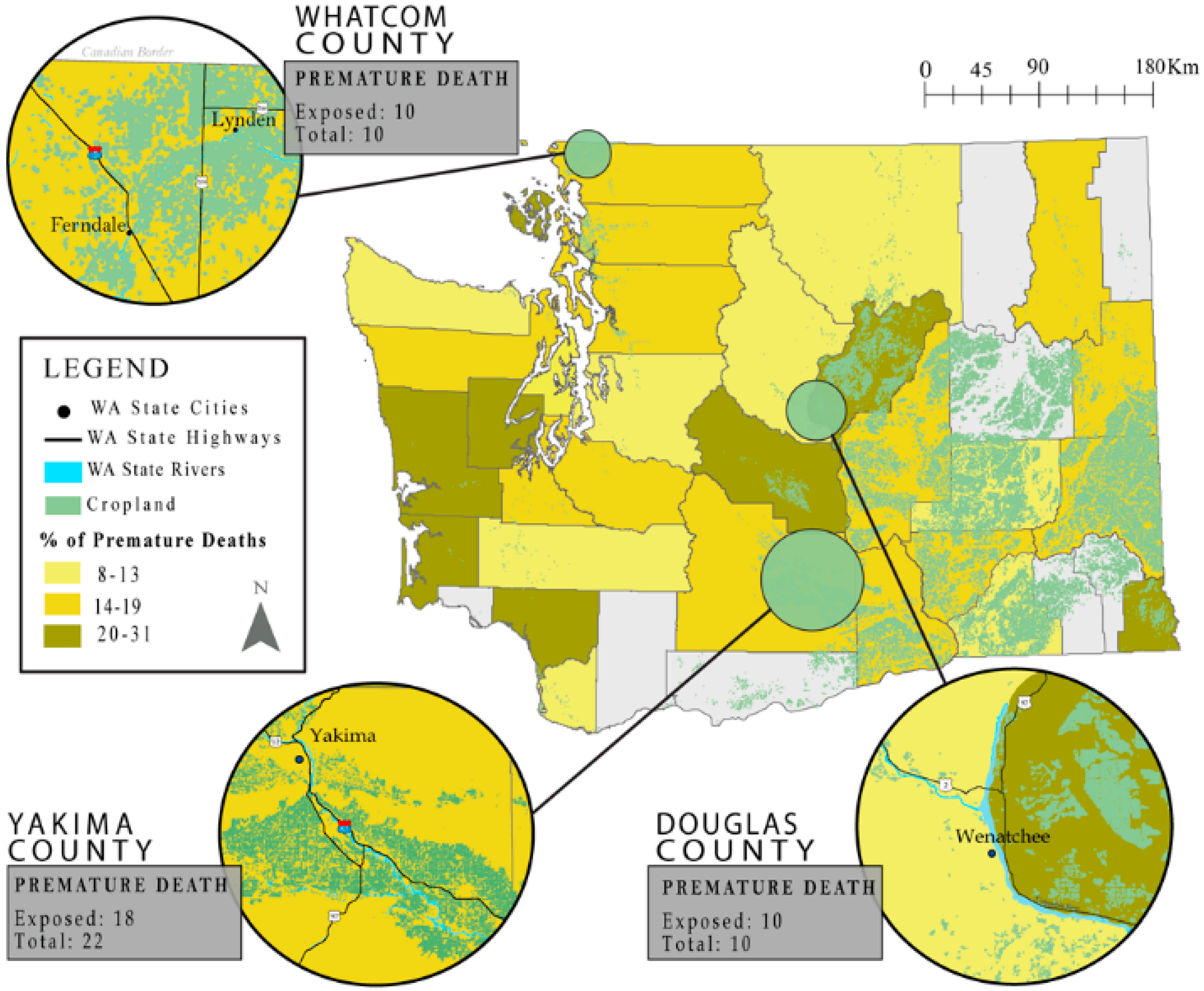
2.5. Mapping
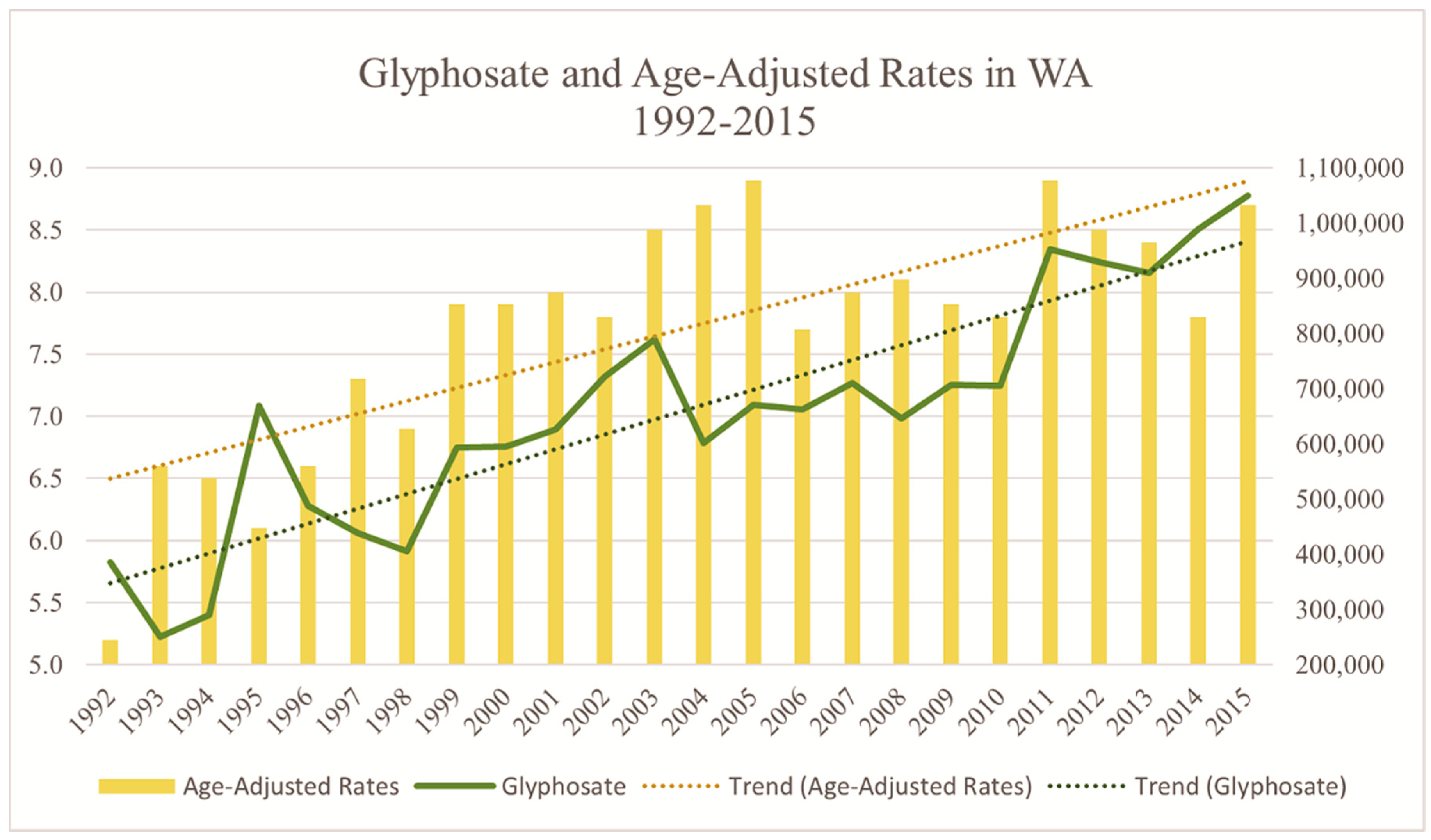
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baker, M.; Gershanik, O. Parkinson’s disease. In Neurological Disorders: Public Health Challenges; Aarli, J.A., Dua, T., Janca, A., Muscetta, A., Eds.; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Bower, J.H.; Maraganore, D.M.; McDonnell, S.K.; Rocca, W.A. Influence of strict, intermediate, and broad diagnostic criteria on the age- and sex-specific incidence of Parkinson’s disease. Mov. Disord. 2000, 15, 819–825. [Google Scholar] [CrossRef]
- Kowal, S.L.; Dall, T.M.; Chakrabarti, R.; Storm, M.V.; Jain, A. The current and projected economic burden of Parkinson’s disease in the United States. Mov. Disord. 2013, 28, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, L.S.; Cheesbrough, A.; Brayne, C.; Schrag, A. Estimated life expectancy of Parkinson’s patients compared with the UK population. J. Neurol. Neurosurg. Psychiatry 2007, 78, 1304–1309. [Google Scholar] [CrossRef] [PubMed]
- Delamarre, A.; Meissner, W.G. Epidemiology, environmental risk factors and genetics of Parkinson’s disease. La Presse Médicale 2017, 46, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.H.; Lo, S.K.; See, L.C.; Chen, H.Z.; Chen, R.S.; Weng, Y.H.; Chang, F.C.; Lu, C.S. Environmental risk factors of young onset Parkinson’s disease: A case-control study. Clin. Neurol. Neurosurg. 2002, 104, 328–333. [Google Scholar] [CrossRef]
- Ritz, B.; Costello, S. Geographic model and biomarker-derived measures of pesticide exposure and Parkinson’s disease. Ann. N. Y. Acad. Sci. 2006, 1076, 378–387. [Google Scholar] [CrossRef]
- Wang, A.; Costello, S.; Cockburn, M.; Zhang, X.; Bronstein, J.; Ritz, B. Parkinson’s disease risk from ambient exposure to pesticides. Eur. J. Epidemiol. 2011, 26, 547–555. [Google Scholar] [CrossRef]
- van der Mark, M.; Brouwer, M.; Kromhout, H.; Nijssen, P.; Huss, A.; Vermeulen, R. Is pesticide use related to Parkinson disease? Some clues to heterogeneity in study results. Environ. Health Perspect. 2012, 120, 340–347. [Google Scholar] [CrossRef]
- Engel, L.S.; Checkoway, H.; Keifer, M.C.; Seixas, N.S.; Longstreth, W.T.; Scott, K.C.; Hudnell, K.; Anger, W.K.; Camicioli, R. Parkinsonism and occupational exposure to pesticides. Occup. Environ. Med. 2001, 58, 582. [Google Scholar] [CrossRef]
- Firestone, J.A.; Smith-Weller, T.; Franklin, G.; Swanson, P.; Longstreth, W.T., Jr.; Checkoway, H. Pesticides and risk of Parkinson disease: A population-based case-control study. Arch. Neurol. 2005, 62, 91–95. [Google Scholar] [CrossRef]
- Washington State Department of Agriculture. Washington Agriculture: A Global Impact. Available online: https://agr.wa.gov/aginwa/default.aspx (accessed on 11 July 2018).
- Centers for Disease Control and Prevention. Parkinson’s Disease Mortality by State. Available online: https://www.cdc.gov/nchs/pressroom/sosmap/parkinsons_disease_mortality/parkinsons_disease.htm (accessed on 20 July 2018).
- Firestone, J.A.; Lundin, J.I.; Powers, K.M.; Smith-Weller, T.; Franklin, G.M.; Swanson, P.D.; Longstreth, W.T., Jr.; Checkoway, H. Occupational factors and risk of Parkinson’s disease: A population-based case-control study. Am. J. Ind. Med. 2010, 53, 217–223. [Google Scholar] [CrossRef]
- Gatto, N.M.; Cockburn, M.; Bronstein, J.; Manthripragada, A.D.; Ritz, B. Well-water consumption and Parkinson’s disease in rural California. Environ. Health Perspect. 2009, 117, 1912–1918. [Google Scholar] [CrossRef] [PubMed]
- Baker, N.T.; Stone, W.W. Estimated Annual Agricultural Pesticide Use for Counties of the Conterminous United States, 2008–2012; U.S. Geological Survey: Reston, VA, USA, 2015.
- Filipov, N.M.; Stewart, M.A.; Carr, R.L.; Sistrunk, S.C. Dopaminergic toxicity of the herbicide atrazine in rat striatal slices. Toxicology 2007, 232, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Cattani, D.; de Liz Oliveira Cavalli, V.L.; Heinz Rieg, C.E.; Domingues, J.T.; Dal-Cim, T.; Tasca, C.I.; Mena Barreto Silva, F.R.; Zamoner, A. Mechanisms underlying the neurotoxicity induced by glyphosate-based herbicide in immature rat hippocampus: Involvement of glutamate excitotoxicity. Toxicology 2014, 320, 34–45. [Google Scholar] [CrossRef]
- Costello, S.; Cockburn, M.; Bronstein, J.; Zhang, X.; Ritz, B. Parkinson’s disease and residential exposure to maneb and paraquat from agricultural applications in the central valley of California. Am. J. Epidemiol. 2009, 169, 919–926. [Google Scholar] [CrossRef]
- United States Department of Agriculture. Washington Cropland Data Layer. Available online: https://www.nass.usda.gov/Research_and_Science/Cropland/SARS1a.php (accessed on 6 June 2018).
- Washington State Department of Ecology. Well Reports. Available online: https://fortress.wa.gov/ecy/wellconstruction/map/wclswebMap/default.aspx (accessed on 21 June 2018).
- Chen, J.T.; Rehkopf, D.H.; Waterman, P.D.; Subramanian, S.V.; Coull, B.A.; Cohen, B.; Ostrem, M.; Krieger, N. Mapping and measuring social disparities in premature mortality: The impact of census tract poverty within and across Boston neighborhoods, 1999–2001. J. Urban Health 2006, 83, 1063–1084. [Google Scholar] [CrossRef] [PubMed]
- Mansfield, C.J.; Wilson, J.L.; Kobrinski, E.J.; Mitchell, J. Premature mortality in the United States: The roles of geographic area, socioeconomic status, household type, and availability of medical care. Am. J. Public Health 1999, 89, 893–898. [Google Scholar] [CrossRef]
- ESRI. ArcGIS Desktop; Environmental Systems Research Institute: Redlands, CA, USA, 2018. [Google Scholar]
- Yan, L.; Roy, D.P. Conterminous United States crop field size quantification from multi-temporal Landsat data. Remote Sens. Environ. 2016, 172, 67–86. [Google Scholar] [CrossRef]
- Ward, M.H.; Lubin, J.; Giglierano, J.; Colt, J.S.; Wolter, C.; Bekiroglu, N.; Camann, D.; Hartge, P.; Nuckols, J.R. Proximity to crops and residential exposure to agricultural herbicides in Iowa. Environ. Health Perspect. 2006, 114, 893–897. [Google Scholar] [CrossRef]
- Warlow, C.P. Epidemiology of stroke. Lancet 1998, 352, SIII1–SIII4. [Google Scholar] [CrossRef]
- Myers, J.P.; Antoniou, M.N.; Blumberg, B.; Carroll, L.; Colborn, T.; Everett, L.G.; Hansen, M.; Landrigan, P.J.; Lanphear, B.P.; Mesnage, R.; et al. Concerns over use of glyphosate-based herbicides and risks associated with exposures: A consensus statement. Environ. Health 2016, 15, 19. [Google Scholar] [CrossRef] [PubMed]
- Washington State Department of Health Services Center for Health Statistic. Death Data. Available online: https://www.doh.wa.gov/DataandStatisticalReports/HealthStatistics/Death (accessed on 5 June 2018).
- Snyder, H.S.; D’amato, J.R. MPTP: A neurotoxin relevant to the pathophysiology of Parkinson’s disease: The 1985 George C. Cotzias Lecture. Neurology 1986, 36, 250–258. [Google Scholar] [CrossRef]
- McCormack, A.L.; Thiruchelvam, M.; Manning-Bog, A.B.; Thiffault, C.; Langston, J.W.; Cory-Slechta, D.A.; Di Monte, D.A. Environmental risk factors and Parkinson’s disease: Selective degeneration of nigral dopaminergic neurons caused by the herbicide paraquat. Neurobiol. Dis. 2002, 10, 119–127. [Google Scholar] [CrossRef]
- Brown, D.G.; Johnson, K.M.; Loveland, T.R.; Theobald, D.M. Rural Land-Use Trends in the Conterminous United States, 1950–2000. Ecol. Appl. 2005, 15, 1851–1863. [Google Scholar] [CrossRef]
- Gunier, R.B.; Bradman, A.; Harley, K.G.; Kogut, K.; Eskenazi, B. Prenatal Residential Proximity to Agricultural Pesticide Use and IQ in 7-Year-Old Children. Environ. Health Perspect. 2017, 125, 057002. [Google Scholar] [CrossRef] [PubMed]
- Harnly, M.; McLaughlin, R.; Bradman, A.; Anderson, M.; Gunier, R. Correlating agricultural use of organophosphates with outdoor air concentrations: A particular concern for children. Environ. Health Perspect. 2005, 113, 1184–1189. [Google Scholar] [CrossRef]
- Harnly, M.E.; Bradman, A.; Nishioka, M.; McKone, T.E.; Smith, D.; McLaughlin, R.; Kavanagh-Baird, G.; Castorina, R.; Eskenazi, B. Pesticides in dust from homes in an agricultural area. Environ. Sci. Technol. 2009, 43, 8767–8774. [Google Scholar] [CrossRef]
- Szekacs, A.; Darvas, B. Forty years with Glyphosate. In Herbicide Properties, Synthesis and Control of Weeds; TechOpen: London, ON, Canada, 2012. [Google Scholar]
- Bureau of Labor Statistics. Washington State Occupational Employment Statistics. Available online: https://esd.wa.gov/labormarketinfo/occupations (accessed on 18 July 2018).
- EPA. Reregistration Eligibility Decision (RED) for Glyphosate; EPA 738-R-93-014; Environmental Protection Agency: Washington, DC, USA, 1993.
- Vandenberg, L.N.; Blumberg, B.; Antoniou, M.N.; Benbrook, C.M.; Carroll, L.; Colborn, T.; Everett, L.G.; Hansen, M.; Landrigan, P.J.; Lanphear, B.P.; et al. Is it time to reassess current safety standards for glyphosate-based herbicides? J. Epidemiol. Community Health 2017, 71, 613–618. [Google Scholar] [CrossRef]
- EPA. Federal Insecticide, Fungicide, and Rodenticide Act; Environmental Protection Agency: Washington, DC, USA, 1996.
| Characteristics | Total | Premature Mortality (Age ≤ 74 Years) | Non-Premature Mortality (Age > 75 Years) | p-Value |
|---|---|---|---|---|
| (n = 4591) | (n = 659) | (n = 3932) | ||
| Age (median, IQR) | 83 (78–88) | 71 (67–72) | 85 (81–89) | <0.001 |
| Sex (no. (%)) | ||||
| Female | 1744 | 189 (28.7) | 1555 (39.5) | <0.001 |
| Male | 2847 | 470 (71.3) | 2377 (60.5) | |
| Race/ethnicity (no. (%)) | ||||
| Other | 274 | 45 (6.8) | 229 (5.8) | 0.31 |
| Non-Hispanic white | 4311 | 612 (92.9) | 3699 (94.1) | |
| Unknown | 6 | 2 (0.3) | 4 (0.1) | |
| Education (no. (%)) | ||||
| Less than High School Diploma | 587 | 66 (10.0) | 521 (13.3) | <0.001 |
| High School Diploma or equivalent | 1573 | 191 (29.0) | 1382 (35.1) | |
| Some college | 1025 | 185 (28.1) | 840 (21.4) | |
| Associate’s Degree or higher | 1399 | 216 (32.8) | 1183 (30.1) | |
| Unknown | 7 | 1 (0.1) | 6 (0.1) | |
| Marital status (no. (%)) | ||||
| Married or living with domestic partner | 2243 | 399 (60.5) | 1844 (46.9) | <0.001 |
| Never married, divorced, separated, or widowed | 2348 | 260 (39.5) | 2088 (53.1) | |
| Likelihood of occupational exposure (no. (%)) | ||||
| Likely | 112 | 11 (1.7) | 101 (2.6) | 0.17 |
| Unlikely | 4479 | 648 (98.3) | 3831 (97.4) | |
| Likelihood of well-water intake (no. (%)) | ||||
| Likely | 295 | 52 (18.1) | 243 (81.9) | 0.10 |
| Unlikely | 4296 | 607 (14.2) | 3689 (85.8) |
| Agricultural Chemical | N | Unadjusted OR (95%CI) | p-Value | N | Adjusted OR (95%CI) | p-Value |
|---|---|---|---|---|---|---|
| All Pesticide | ||||||
| No Exposure | 3526 | Reference | 3516 | Reference | ||
| Exposed | 1065 | 1.16 (0.96–1.40) | 0.13 | 1062 | 1.19 (0.98–1.44) | 0.09 |
| Atrazine | ||||||
| No Exposure | 4357 | Reference | 4344 | Reference | ||
| Exposed | 234 | 1.17 (0.82–1.67) | 0.40 | 231 | 1.21 (0.84–1.74) | 0.31 |
| Diazinon | ||||||
| No Exposure | 3833 | Reference | 3821 | Reference | ||
| Exposed | 758 | 1.06 (0.85–1.31) | 0.63 | 757 | 1.07 (0.85–1.34) | 0.63 |
| Glyphosate | ||||||
| No Exposure | 3932 | Reference | 3914 | Reference | ||
| Exposed | 659 | 1.30 (1.04–1.62) | 0.02 | 664 | 1.33 (1.06–1.67) | 0.01 |
| Paraquat | ||||||
| No Exposure | 3780 | Reference | 3769 | Reference | ||
| Exposed | 811 | 1.20 (0.98–1.48) | 0.09 | 809 | 1.22 (0.99–1.51) | 0.07 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caballero, M.; Amiri, S.; Denney, J.T.; Monsivais, P.; Hystad, P.; Amram, O. Estimated Residential Exposure to Agricultural Chemicals and Premature Mortality by Parkinson’s Disease in Washington State. Int. J. Environ. Res. Public Health 2018, 15, 2885. https://doi.org/10.3390/ijerph15122885
Caballero M, Amiri S, Denney JT, Monsivais P, Hystad P, Amram O. Estimated Residential Exposure to Agricultural Chemicals and Premature Mortality by Parkinson’s Disease in Washington State. International Journal of Environmental Research and Public Health. 2018; 15(12):2885. https://doi.org/10.3390/ijerph15122885
Chicago/Turabian StyleCaballero, Mariah, Solmaz Amiri, Justin T. Denney, Pablo Monsivais, Perry Hystad, and Ofer Amram. 2018. "Estimated Residential Exposure to Agricultural Chemicals and Premature Mortality by Parkinson’s Disease in Washington State" International Journal of Environmental Research and Public Health 15, no. 12: 2885. https://doi.org/10.3390/ijerph15122885
APA StyleCaballero, M., Amiri, S., Denney, J. T., Monsivais, P., Hystad, P., & Amram, O. (2018). Estimated Residential Exposure to Agricultural Chemicals and Premature Mortality by Parkinson’s Disease in Washington State. International Journal of Environmental Research and Public Health, 15(12), 2885. https://doi.org/10.3390/ijerph15122885






